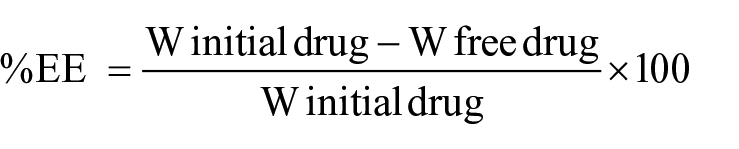Abstract
Anastrozole, an aromatase inhibitor drug, is used for the treatment of breast cancer in pre- and postmenopausal women. Anastrozole’s incorporation into nanoparticulate carriers would enhance its therapeutic performance. To perceive the exact loaded amount of drug in nanocarriers, a valid analytical method is required. The reversed-phase high-performance liquid chromatography (RP-HPLC) method was developed and validated by using the C18 column, 150 × 4.6 mm, 5 µm particle size, in isocratic mobile phase composed of 50:50 V/V (volume/volume) acetonitrile–phosphate buffer (pH 3) flowing at a rate of 1.0 mL/min, and a diode array detector (DAD) set at λmax = 215 nm. The validation parameters such as linearity, accuracy, specificity, precision, and robustness have proven the accuracy of the method, with the relative standard deviation percentage (% RSD) values < 2. The limit of detection of the method was found equal to 0.0150 µg/mL, and the limit of quantitation was 0.0607 µg/mL. The percent recovery of sample was in the range of 98.04–99.25%. The method has the advantage of being rapid with a drug retention time of 2.767 min, specific in terms of resolution of peaks void of interference with any of the excipients, and high reproducibility. This makes it highly applicable for quality control purposes.
Introduction
Breast cancer is the major concern of death among women worldwide. Approximately 252,710 cases occur each year and cause a great number of cancer-related deaths among women. It is expected that there will be 3 million cases at the end of 2020. 1 Among all subtypes, estrogen receptor–positive breast cancer is the most common type, comprising approximately 80% of breast cancer among all female cancer patients. Estrogen hormone produced naturally in the body is mainly responsible for the growth and spread of estrogen receptor–positive breast cancer cells. 2
The aromatase cytochrome P450 enzyme stimulates catalyzing androgens to estrogens and is responsible for the production of hormone.3–7 Aromatase inhibitors (AIs) are preferred to treat hormone receptor–positive breast cancer as compared to chemotherapy due to the former’s fewer side effects. Anastrazole (ANZ), a well-studied nonsteroidal AI medication that interferes with estrogen, is used to treat breast cancer in women after menopause. It has been reported that ANZ is a potent, selective, and orally active AI that significantly reduces the amount of estrogen available for the body to use in postmenopausal women with breast cancer.8,9 The pharmacokinetic profile of ANZ showed that the majority of the drug is eliminated by liver metabolism, while only 10% is eliminated unchanged via renal excretion. 10 Nanopharmaceutical technology has introduced a number of rational alternatives for enhancing the therapeutic efficacy of drugs with poor bioavailability and pharmacokinetic profiles. Several types of nanoparticles (NPs)—including liposomes, emulsions, nanocrystals, micelles, solid lipid NPs, and polymeric NPs—have been used to deliver drugs efficiently to specific targets.11–16 Enhancement of the therapeutic efficacy of drugs by incorporating them into NPs can be attributed to extension of drug release, prolongation of drug circulation time in the body, and/or potentiation of drug cellular uptake, resulting in a reduction of dose-dependent adverse effects.
Various ANZ NPs have been successfully synthesized and showed superior in vitro cytotoxicity on the MCF7 breast cancer cell line.17–23 In another study, a comparative examination of the efficacy and toxicity of ANZ NPs versus conventional tablets in vivo revealed that ANZ NPs are more protective and a better alternative for long-term treatment of breast cancer. 24 Valid standard analytical methods are urgently needed to evaluate drug entrapment efficiency, pharmacokinetics, and toxicity for newly developed ANZ NPs. As revealed in our literature survey, reversed-phase high-performance liquid chromatography (RP-HPLC) is a widely acceptable method to separate and accurately quantify many drugs from various nanoformulations, including letrozole, curcumin, isoniazid, and haloperidol.25–30 To carry out accurate quantitation, the role of separation and resolution of the component is highly important. The selection of column, mobile phase, pH of the mobile phase, detector, and λmax comprises the important parameters for the validation of the method. No single HPLC method has been used in the literature reports on the quantitation of ANZ incorporated into NPs. In the current study, we developed and validated a simple, sensitive, and specific RP-HPLC method to determine the presence of ANZ in hybrid polymer–lipid nanoparticles (PLNs). Subsequently, this validated method was used successfully to quantify the amount of ANZ encapsulated in the prepared PLN systems and to evaluate drug content. The efficiency of entrapped drug in PLNs was precisely determined.
Materials and Methods
Chemicals and Reagents
A reference standard of ANZ was purchased from UFC Biotechnology (Amherst, NY). Stearic acid (SA), polycaprolactone (PCL, molecular weight: 42 KDa), polyvinyl alcohol (PVA), and polyethlene glycol (PEG) 6000 were purchased from Sigma Aldrich (St. Louis, MO). Methanol and acetonitrile solvents used in this study were of HPLC grade (Merck, Darmstadt, Germany). Water collected from the PURELAB Option-R (ELGA LabWater, High Wycombe, UK) was used for the preparation of buffer and other aqueous solutions. All the solvents and reagents were of HPLC grade.
Experimental
The mobile phase consisted of acetonitrile and phosphate buffer adjusted to pH 3.0±0.05 [1:1 V/V (volume/volume)]. Freshly prepared mobile phase was filtered through 0.45 mm Whatman filter paper (Millipore, Milford, MA). An Agilent 1200 series HPLC system equipped with a 1260 series Infinity Diode Array Detector (DAD; Agilent, Santa Clara, CA) was used for the analysis of ANZ. A ZORBAX Eclipse XDB column (cat. no. 993967, Agilent) C18, 150 mm × 4.6 mm, with 5 µm particle size was used for the separation and quantification. The preconditioned column was used at laboratory temperature (23±2 °C). The auto-injector was adjusted to an injection volume of 20 µL, and a flow rate of 1 mL/min was maintained throughout the analysis. The detection was attained at λmax = 215 nm. The peak areas were integrated automatically by using HPLC ChemStation software, version B.03 (Agilent).
Preparation of Mobile Buffer Phase
Ten millimolars (10 mM) of phosphate buffer were prepared by mixing 445 mL of double-deionized distilled water containing 6.056 g of potassium dihydrogen orthophosphate with 55 mL of 0.1M phosphoric acid. Then, the pH of the buffer solution was adjusted to 3.0 with glacial acetic acid. The prepared buffer solution was filtered through 0.45 µm Whatman filter paper and degassed. Every time, freshly prepared mobile phase was degassed by sonication.
Preparation of Standard Solution
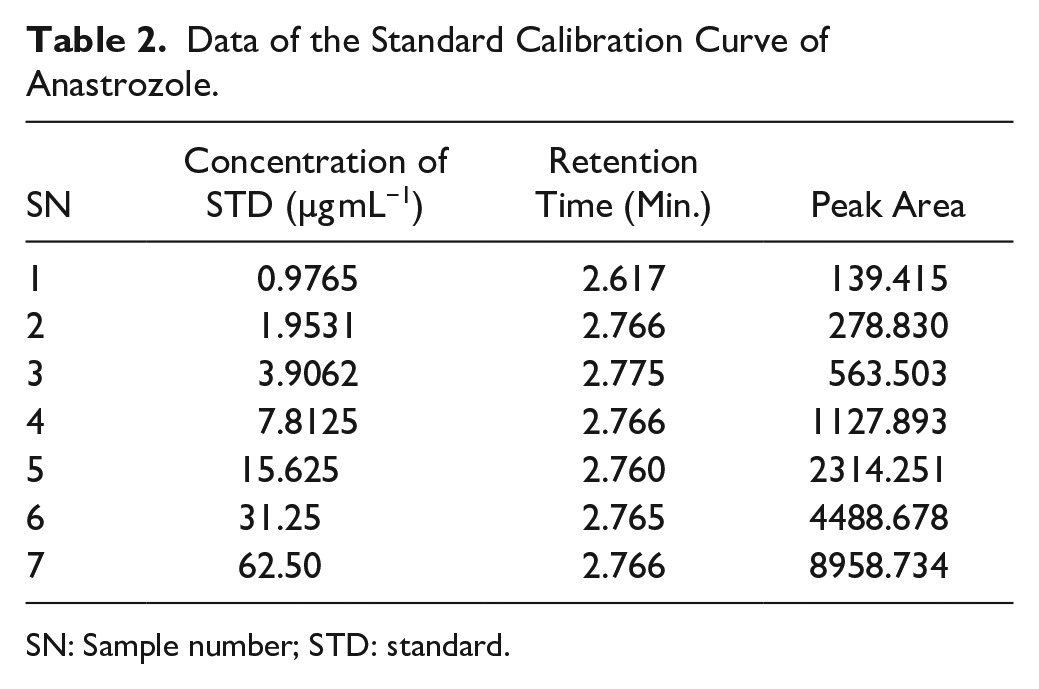
Accurately weighed 50 mg ANZ was dissolved in 50 mL double-distilled water to make 1000 µg/mL stock solution. From this stock solution, we prepared a working solution of different concentrations: 0.49, 0.98, 1.95, 3.91, 7.82, 15.62, 31.25, and 62.25 µg/mL. All working solutions were prepared in mobile phase. From each concentration, 20 µL of standard solution was injected into the HPLC system for the study of suitability and linearity. The chromatogram of each standard was recorded and tabulated to design the linearity curve. A plot of concentration (0.976 to 62.25 µg/mL) versus peak area was drawn.
Preparation of Anastrozole Polymer–Lipid Hybrid Nanoparticles
The method described by Massadeh et al. 31 was used for the preparation of ANZ PLNs. Briefly, 10 mg of ANZ was dissolved in 20 mL distilled water using an ultrasonic bath set at 35 °C to prepare a standard 0.5 mg/mL ANZ aqueous solution (solution A). In separate container, 20 mg PCL and 5 mg SA were dissolved in 10 mL chloroform (solution B). In a small glass beaker, 2 mL of solution A was mixed with 10 mL of solution B and probe-sonicated for 3 min at 40% intensity under an ice bath using a probe sonicater (Q700, Qsonica, Melville, NY) to allow for the formation of a primary emulsion. Then, the formed emulsion (12 mL) was dispersed in 15 mL of 2% PVA, and a second cycle of 3 min of probe sonication under an ice bath was performed to allow for the formation of the secondary emulsion. The formed nanodispersion was magnetically stirred for 3 h at room temperature under a biological safety cabinet to allow for the complete evaporation of chloroform. The resulted nanosuspension was centrifuged (Heraeus Megafuge 16R, Thermo Fisher Scientific, Dreieich, Germany) at 15,000 rpm for 30 min at 4 °C. Then, the supernatant was withdrawn and collected in a separate container. After applying two washing cycles by dispersion in cold water and centrifugation, the precipitated ANZ PLNs were stored in a −30 °C freezer. After 24 h, they were dried using a freeze dryer (Beta 2-8 LDplus, Martin Christ, Osterode am Harz, Germany). Samples from the supernatant solution were collected and filtered through a 0.45 µm membrane filter. The drug entrapment efficiency (%EE) was indirectly calculated by the developed RP-HPLC method to determine the amount of ANS in the supernatant obtained after centrifugation of the ANZ PLN dispersion using the following equation:
Method Development
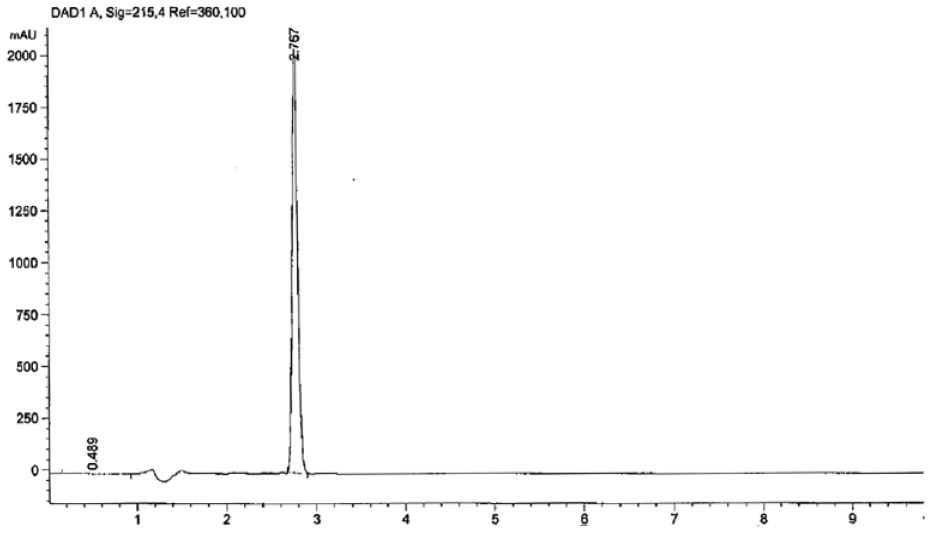
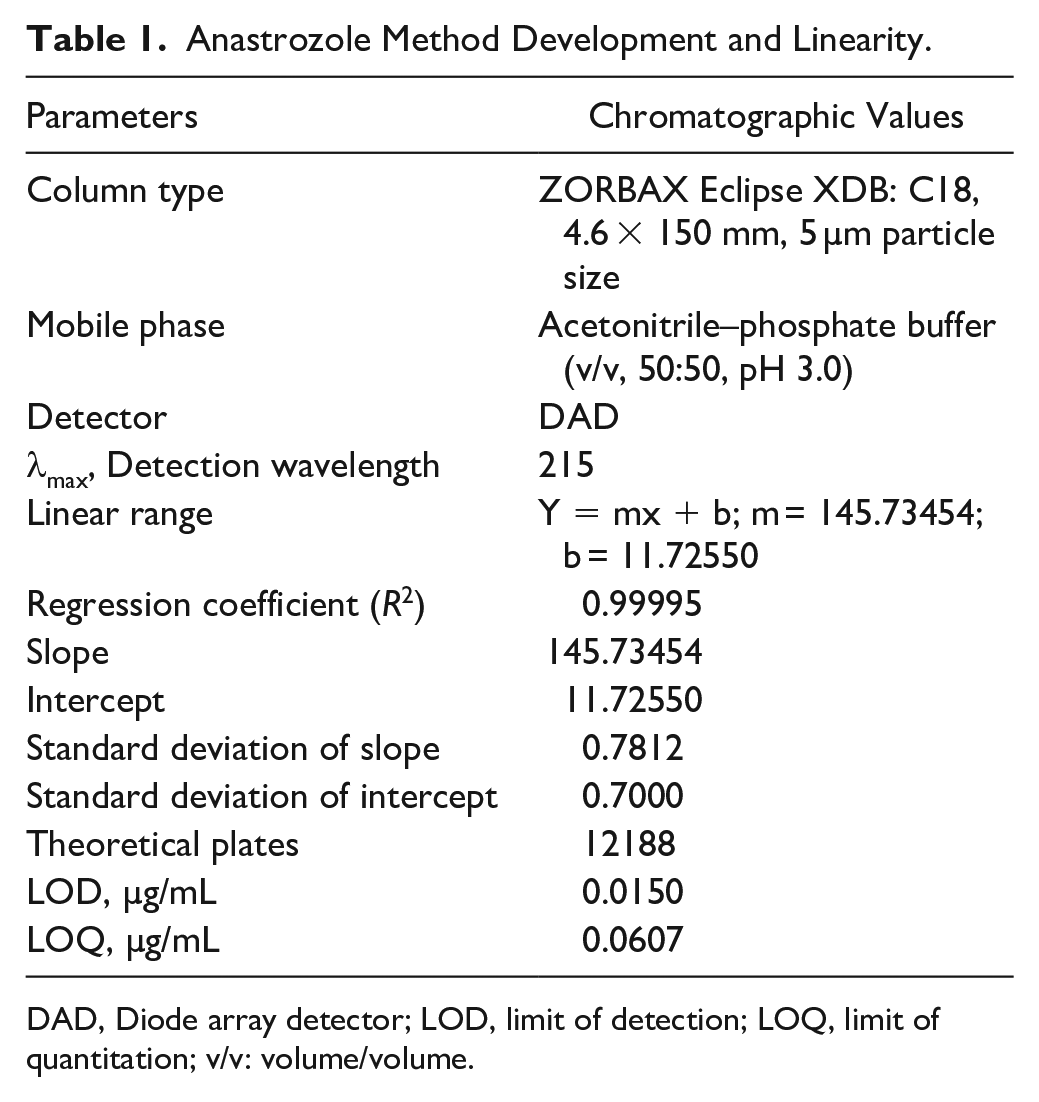
An ideal wavelength is an important tool to get better sensitivity with the HPLC method. The maximum wavelength (λmax) in HPLC was set at 215 nm after spectrometric scanning (200–400 nm) of ANZ. The method was optimized applying the above-described conditions. These conditions were settled after experimenting with various possibilities of different flow rates, column specifications, mobile phase compositions, and pH of mobile phase. Acetonitrile and buffer mixtures at pH 3.0 were found to be appropriate for the separation of ANZ and optimization of the method. Various mixture ratios of acetonitrile or methanol water–phosphate buffer solutions were tried to get the optimum composition providing the best resolution. The flow rate of 1.0 mL/min was found to be the most suitable after trying 0.5, 0.7, 0.8, and 0.9 mL/min. Column selection C18 was also decided based on satisfactory various system suitability parameters. The standard and samples were filtered with a polytetrafluoroethylene (PTFE) disposable filter device (Puradisc 25 TF, GE Healthcare, Chalfont St Giles, UK) before injection into the HPLC system.
Method Validation
The guidelines described by the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) were used for validation of the established method for the analysis of ANZ PLNs.32,33 To generate trustworthy results and a validation study, a linear graph of standard ANZ at different concentrations versus peak area was computed. The system suitability, precision, specificity, accuracy, limit of detection (LOD), limit of quantitation (LOQ), and robustness were calculated statistically.
Results and Discussion
Optimization and Method Validation
ANZ PLNs were analyzed and quantitated by using the RP-HPLC method developed. Multiple chromatographic columns, ranging from C8 and C18 specifications, were tried to get the best separation in the study. Mobile phase mixtures of acetonitrile–buffer or methanol–buffer solution of various ratios—80:20, 70:30, 60:40, and 50:50—were tried to get the best peak in terms of tailing and sharpness of the peak. Finally, the mobile phase comprising 50:50 V/V (volume/volume) acetonitrile–10 mM phosphate buffer solution at pH 3.0 and column temperature 23 °C was found to have enormous supportive parameters to get sharp peak, and minimum tailing factor with a short runtime. The first exploratory run was set at 40 min to observe excipients in the injected sample. The detector was set at 215 nm with a runtime of 6 min. The adopted flow rate of 1.0 mL/min together with the isocratic mode of mobile phase resulted in a sharp ANZ peak at a short retention time at 2.76 min, as presented in Figure 1 . Figure 3 depicts the chromatogram of placebo PLN (ANZ-free PLN). It clearly shows no peaks in the range from 2.3 to 2.8 min, indicating the suitability of the method for the detection of ANZ in PLNs without any possibility of interference.
A typical anastrazole high-performance liquid chromatography (HPLC) chromatogram.
Linearity
A linear relationship is the ability to evaluate the analytical procedure within the expected range of the concentration and directly proportional to the drug present in the solutions. The runtime with the described chromatographic condition was acknowledged at a short retention time equal to 2.76 min at a 1.0 mL/min flow rate with an isocratic mode of mobile phase. This shorter retention time is highly suitable for the quality control laboratory. The regression line was constructed by plotting peak areas versus concentrations of ANZ, and it resulted in a straight line of working standard solutions of concentrations: 0.98, 1.95, 3.91, 7.84, 15.63, 31.25, and 62.5 µg/mL ( Table 2 ). The data from the regression line were used to calculate the correlation coefficient, as shown in Table 1 . The standard calibration curve of ANZ ( Fig. 2 ) demonstrated high linearity, as indicated from the squared regression coefficient (R2) value (very close to 1). The R2 value as coefficient of determination was found to equal 0.99995 throughout the range of concentration 0.976–62.5 ug/mL. The R2 is significantly less than 5% of the response nominal 100% concentration value.34,35 Considering favorable parameters, this method proved specific without any excipient interference, and cost-effective due to its short retention time (2.76 min) for the quantitative analysis of a larger number of samples.
Anastrozole Method Development and Linearity.
DAD, Diode array detector; LOD, limit of detection; LOQ, limit of quantitation; v/v: volume/volume.
Data of the Standard Calibration Curve of Anastrozole.
SN: Sample number; STD: standard.
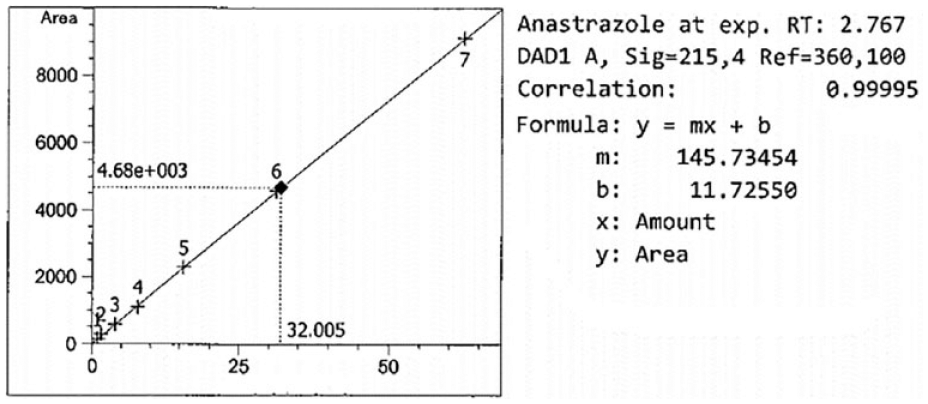
Linearity standard curve of different concentrations of anastrazole.
System Suitability
System suitability tests are requisite components to validate any chromatographic method. Suitability parameters such as theoretical plates, tailing factor, and peak area were investigated to verify reproducibility of procedure and chromatographic system for the analysis of ANZ ( Tables 1 – 3 ). To examine the validity of certain parameters, a known concentration of ANZ was injected repeatedly to get the relative standard deviation percentage (% RSD). All the required parameters for suitability were calculated and found to be within the limit (< 2% RSD). Further higher value of theoretical plates (N = 12,188; Table 1 ) is an indication of a better separation, which is higher than 2000 as the requirement for better resolution. 36
Precision (Repeatability and Reproducibility of Anastrozole) and Sample Repeatability.

HPLC: High-performance liquid chromatography; N: number of replicates; % RSD: relative standard deviation percentage; SD: standard deviation; STD: standard.
Precision
The precision of the method is used to examine the closeness of data values. Repeatability and reproducibility were evaluated by multiple injections of prepared homogeneous solution under the same chromatographic conditions and day tested. 37 Precision was also studied to find out the variations when ANZ drug was injected at different times within the same day. The repeatability was tested on three consecutive days as well. All the analysis was carried out in six replicates to assess the system’s suitability. The % RSD for intra- and interday precision was in the range of 0.802 to 1.680 ( Table 3 ). The observed % RSD (< 2%) value indicated that the developed method is acceptable and qualifies for the suitability test. 38
Accuracy and Recovery
Accuracy and recovery are determined by injecting three levels (low, medium, and high) of standard concentrations other than those used in developing the standard curve and calculating the percentage of the amount recovered from each sample. The degree of closeness to 100% is considered an indication of the accuracy of the analytical method. 39 Table 4 represents the mean percent recovery of the three tested samples. They were found to be within the limit of 99.25%, 98.36%, and 98.04%, with very low % RSD, indicating the validity of the proposed method.
Recovery of Anastrozole Drug.
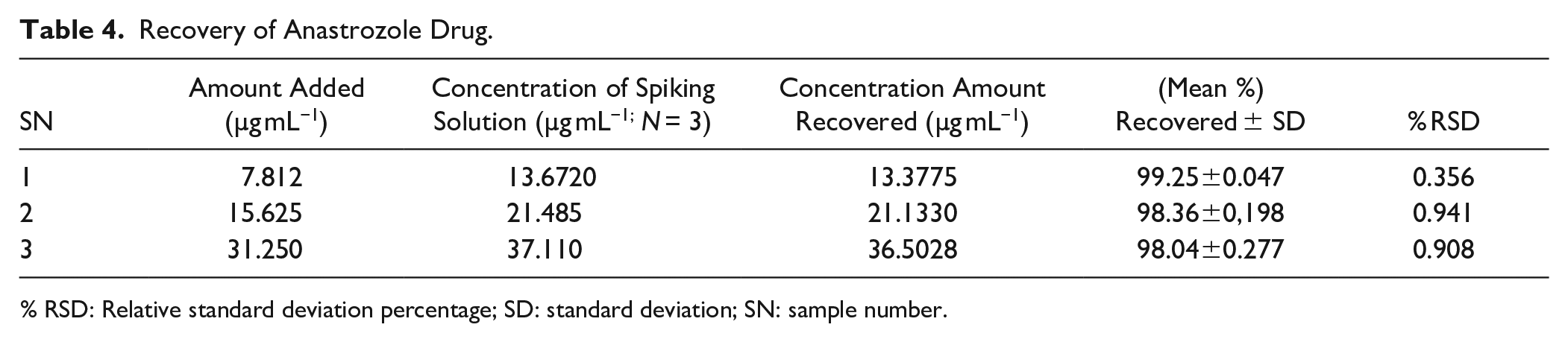
% RSD: Relative standard deviation percentage; SD: standard deviation; SN: sample number.
Specificity
Specificity is the ability to quantitate and ensure the identity of desired analyte in a pharmaceutical sample without any interference from any excipients. The specificity was examined by injecting blank, placebo (drug-free) PLN and standard ANZ PLN samples into HPLC, and the chromatograms were observed as shown ( Figs. 3 – 4 ). The applied method was interpreted and found that there was no interference of diluent or placebo with analyte qualitatively; therefore, the method was observed to be selective and specific. The chromatogram of standard samples of ANZ PLNs showed a symmetric neat peak that appeared at a retention time of 2.76 min free from any interference with excipients. From the chromatogram of these three samples, it can be concluded that the system was specific for the analysis of ANZ from ANZ PLN samples.
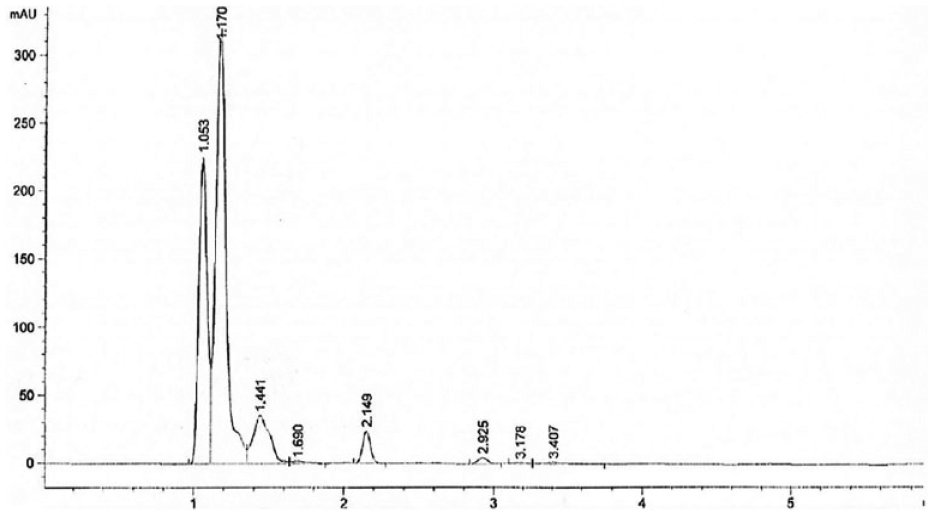
Reversed-phase high-performance liquid chromatography (RP-HPLC) chromatogram of placebo mixture used in a nanoparticle lipid carrier.
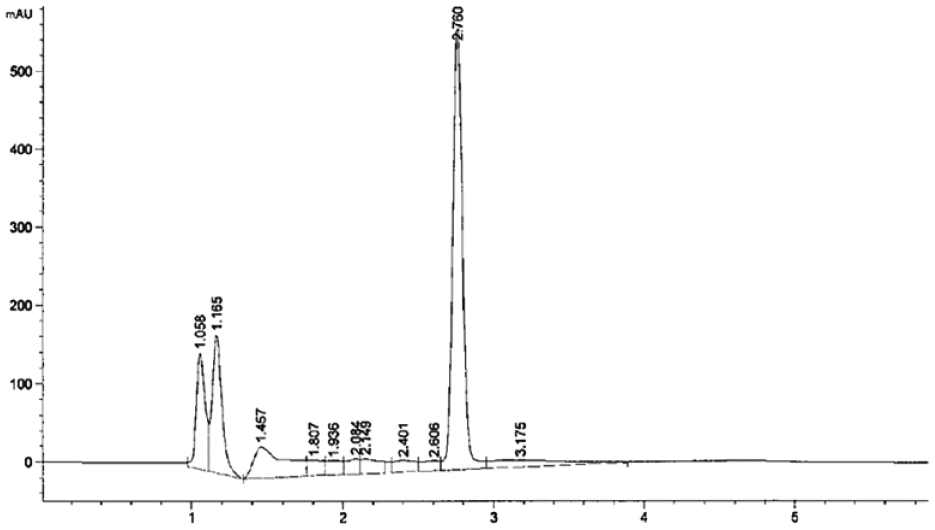
Chromatogram: anastrazole recovery from polymer-lipid hybrid nanoparticles formulation.
Limit of Detection and Limit of Auantitation
The LOD is the lowest concentration of analyte in the sample that can be detected from signals of a sample. A signal-to-noise ratio of, generally, 3:1 is considered for the determination of LOD. The LOQ is the lowest concentration that can be determined with acceptable precision under the optimized conditions. The LOD and LOQ were calculated to validate the developed RP-HPLC method. The LOD and LOQ were found to equal 0.0150 and 0.0607 µg/mL, respectively. Such low values of LOD and LOQ indicate that the method is sensitive and suitable enough to detect low concentrations of ANZ.
Robustness
Robustness is the ability of a method to remain unaffected by minor, planned changes in process variables; it is a measure of the consistency of a method. 40 By changing the flow rate by ±2 ml/min, mobile phase composition ratio by ±5%, and column temperature by ±2 °C, it has been observed that there were no significant changes, and the result was found to be within limits of % RSD < 2%, as shown in Table 5 .
Proposed Robustness Studies.
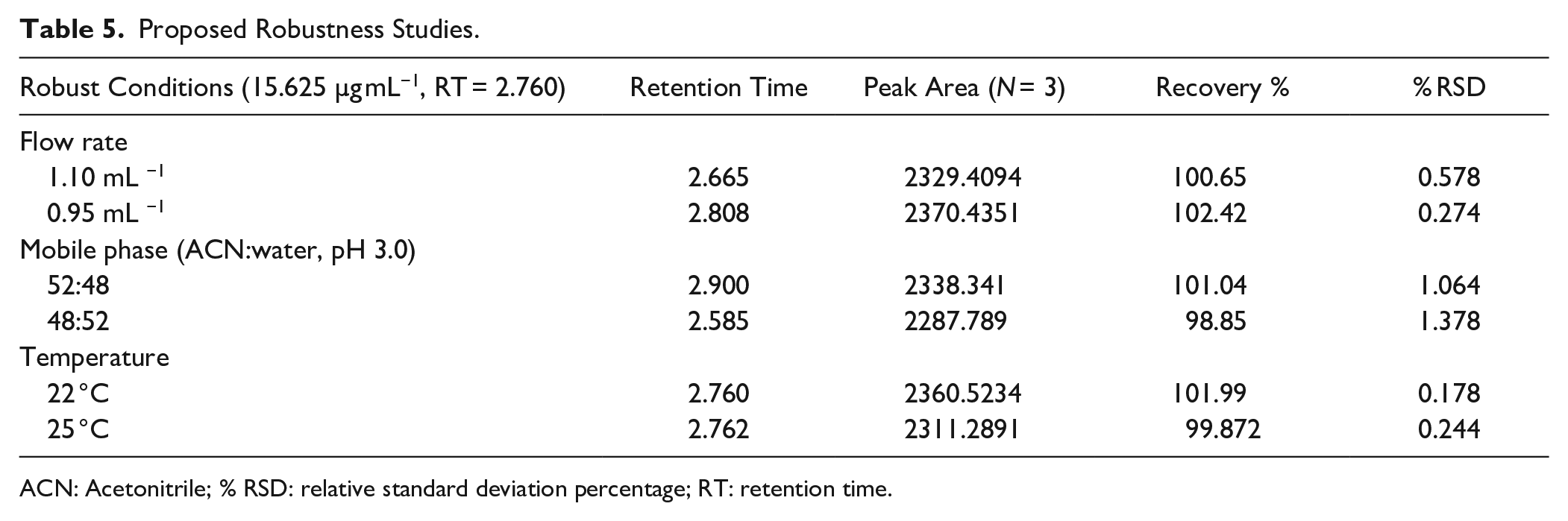
ACN: Acetonitrile; % RSD: relative standard deviation percentage; RT: retention time.
Encapsulation Efficiency
The validated RP-HPLC method was applied for the determination of %EE ANZ in different ANZ PLN formulations. The results showed %EE values in the range between 72.7±0.92% and 81.3±1.13%. The coefficients of variation were found to be between 1.3% and 1.4%, indicating very high reproducibility from batch to batch of the method for %EE determination.
Conclusions
This is the first time in the literature that a RP-HPLC method for the quantitative analysis of ANZ from NP formulation has been described. The method has been assessed for specificity, accuracy, precision, selectivity, and robustness that were found to be convenient. Linearity ranged from 0.976 to 62.50 µg/ml, with a correlation coefficient of 0.99995 and recovery percentages of 98.04%, 98.36%, and 99.25%. The system’s suitability with good resolution of peaks confirms that the method is free from any interference of components used in PLN formulation or any excipients. The advantage of the low value of an LOQ of 0.06070 µg/mL and the remarkably short retention time of analysis, 2.76 min, ensure that the method is distinct. Furthermore, statistical analysis values of % RSD and a low coefficient of variation ensure the method is suitable for the quality control and the determination of ANZ in various settings. The high consistency and reproducibility of %EE values indicated the robustness of the method. This method shows high potential to be modified for the detection of ANZ from any other NP systems, as well as in biological samples and for multiple purposes.
Footnotes
Acknowledgements
The authors would like to thank Ms. Sara Dilshad, Aligarh Muslim University, India, for helping in the statistical analysis of the data.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from King Abdullah International Research Center, National Guard Health Affairs, Riyadh, Saudi Arabia (grant no. SP18/363/R). The funding agency had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.