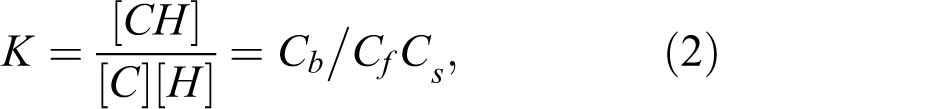Abstract
Endocrine-disrupting chemicals are compounds that alter the normal functioning of the endocrine system. Organophosphorus insecticides, as chlorpyrifos (CPS), receive an increasing consideration as potential endocrine disrupters. Physiological estrogens, including estrone (E1), 17β-estradiol (E2), and diethylstilbestrol (DES) fluctuate with life stage, suggesting specific roles for them in biological and disease processes. There has been great interest in whether certain organophosphorus pesticides can affect the risk of breast cancer. An understanding of the interaction processes is the key to describe the fate of CPS in biological media. The objectives of this study were to evaluate total, bound, and freely dissolved amount of CPS in the presence of three estrogenic sex hormones (ESHs). In vitro experiments were conducted utilizing a headspace solid phase microextraction (HS-SPME) combined with high-performance liquid chromatography (HPLC) method. The obtained Scatchard plot based on the proposed SPME-HPLC method was employed to determine CPS-ESHs binding constant and the number of binding sites as well as binding percentage of each hormone to CPS. The number of binding sites per studied hormone molecule was 1.10, 1, and 0.81 for E1, E2, and DES, respectively. The obtained results confirmed that CPS bound to one class of binding sites on sex hormones.
Introduction
Pesticide toxicity has been clearly demonstrated to alter a variety of physiological functions. In addition, evidences suggest that pesticide exposure increases the risk of cancer. Recent evidence also demonstrates the ability of pesticides to act as endocrine disruptors. 1 –3
Chlorpyrifos (CPS; O, O-diethyl-O-(3,5,6-trichloro-2-pyridyl)-phosphorothioate) is a chlorinated organophosphate (Figure 1(a)) insecticide with a broad spectrum of activity against arthropod pests of plants, animals, and humans and has been widely used in both agricultural and nonagricultural settings. 4 Due to their low price and effective ability to control pests, weeds and diseases, organophosphorus insecticides, including CPS, still account for up to 50% of global insecticidal use. 5 European Union has established the maximum residue limits for vegetables, fruits, and animal tissues intended for human consumption among the range from 20 ng/g to 500 ng/g. 6 The exposure of laboratory animals to organophosphorus pesticides, in particular to CPS, elicits a number of effects including hepatic dysfunction, 7,8 ciliotoxicity 9 immunological abnormalities, 10 genotoxicity, 11 and testicular damage. 12
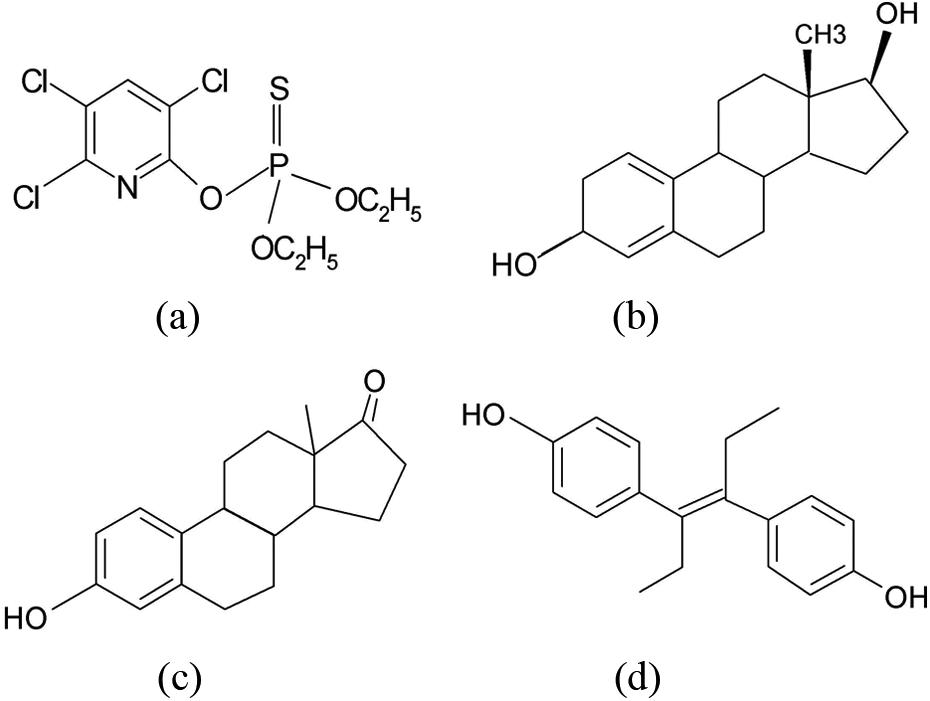
The structural formula of CPS (a), E2 (b), E1 (c), and DES (d). CPS: chlorpyrifos; DES: diethylstilbestrol; E1: estrone; E2: 17 β-stradiol.
Endocrine-disrupting chemicals are compounds that have the ability to mimic or modulate hormones in the endocrine systems of humans and animals. 13 Hormones play a crucial role in guiding normal cell differentiation in early life forms, and so exposure to endocrine-disrupting substances in the egg or in the womb (mammals) can alter the normal process of development. 14 Sex hormones play a critical role in the development and functions of many organs and tissues. 15
The major estrogenic components that have been identified as the natural estrogens include estrone (E1) and 17β-estradiol (E2; Figure 1(b) and (c)), which are either produced endogenously by animals or used as pharmaceutical products in both human and veterinary medicine. 16,17 Diethylstilbestrol (DES; Figure 1(d)) is a kind of synthetic nonsteroidal estrogen. 18 It has been widely used not only in livestock production to promote growth but also as a treatment for estrogen deficiency. Recent prospective studies have provided strong evidence that the risk of developing breast cancer in postmenopausal women is increased by high serum levels of estrogens, low levels of sex hormone-binding globulin, and, hence, high circulating levels of free steroid sex hormones. 19 –23 Animal studies show, in addition, that injections of some pesticides can stimulate the development of breast cancers in male mice normally resistant to breast cancer. 24
Solid-phase microextraction (SPME) is a successful solvent-free extraction technique often used for the determination of a high number of volatile and semivolatile compounds. 25 –27 In fact, SPME has been applied to the determination of the freely available concentration of different analytes in the presence of complex matrixes. 28 –30 In headspace (HS) extraction mode, the analytes are extracted in a three-phase system, namely, sample (liquid or solid), HS, and fiber coating. 31 The advantage of HS-SPME sampling is that the matrix in the sample cannot interfere with the fiber.
Because of the recently increased interest in changes in hormone levels due to the cancer risk, in this study, we studied in vitro interaction of CPS with three sex hormones using HS-SPME, to measure the freely available concentration of pesticide and the lowering of the concentration due to binding to hormones. Hormones were nonvolatile compounds, so HS-SPME was straightforward and recommended. CPS was incubated with E1, E2, and DES solutions, and the free amount of CPS was extracted by HS-SPME and determined by high-performance liquid chromatography (HPLC). Desorption of CPS was performed off-line, in especial vial with the desorption solvent.
Materials and methods
Chemicals
Sodium acetate, acetonitrile, glacial acetic acid, sodium phosphate, and methanol were purchased from Merck (Darmstadt, Germany). CPS technical grade was obtained from Shandong Dacheng Pesticide Co. Ltd (Zibo, China). CPS stock solution was prepared in methanol and stored in a refrigerator. It was stable for several months. More dilute solutions were prepared in phosphate buffer (pH 7.4) just before use. Hormones (E1, E2, and DES; 99% purity) were obtained from Sigma-Aldrich (St Louis, Missouri, USA) dissolved in phosphate buffer (0.1 M, pH 7.4) at a concentration of 100 µM and stored in the dark. All organic solvents were HPLC grade.
Apparatus
All pH measurements were made with a Metrohm 744 pH meter (Metrohm AG; Herisau, Switzerland) using a combined pH electrode. An Agilent 1100 series HPLC apparatus (Agilent technologies, Palo Alto, California, USA) equipped with quaternary pump, inline degasser, and diode array detector was used. Separations were carried out on a C18-ODS (Octadecylsilane) column from Supelco (Bellefonte, Pennsylvania, USA; 250 × 4.6 mm inner diameter (ID), 5 μm particle size). A personal computer equipped with an Agilent ChemStation program for the LC system was used to process chromatographic data. Microporous polypropylene hollow fiber membrane, ACCUREL 50/280 (280 µm ID, Membrana GmbH, Wuppertal, Germany) was used in the experiments. A 40-kHz and 350 W ultrasonic bath (Eurosonic 4D; Euronda S.P.A., Vicenza, Italy) was used in extraction and washing steps.
Safety considerations
These hormones are powerful, uptake of which can deregulate many processes in the body, including reproduction. They are also suspected carcinogens. Therefore, ingestion of them should be prevented by working hygienically and uptake via the skin can be prevented by wearing gloves.
Chromatographic and detection conditions
The chromatographic analyses were performed under isocratic conditions with a flow of 1 mL/min. The binary mobile phase consisted of acetonitrile/acetate buffer (pH = 4.7, 50 mM; 93:7 v/v). The HPLC mobile phase was filtered through a 0.45-µm membrane (Whatman Limited, Maidstone, UK) before use. The temperature was ambient. The detection wavelength was 230 nm.
SPME protocol
However, in this study the parameters of the SPME analysis were optimized as follows, in order to obtain the best extraction performance: salt 0%, desorption solvent; methanol 500 µL, extraction time 30 min, extraction temperature 42°C, stirring rate 800 r/min, pH 7.4. For SPME of the analytes, a stainless steel wire of 2 cm length coated with narrow bore hollow fiber membrane (280 μm ID) 32 without a specific holder, such as the needle tip of microsyringe, was used. Working solutions were prepared in a 20-mL screw-cap glass vial by dissolving various amounts of CPS into 5 mL phosphate buffer (50 mM, pH 7.4) and coupled with SPME system during optimization. The assembled SPME fiber to screw-cap was exposed to the sample headspace for an extraction time of 30 min and temperature of 42°C. Magnetic stirring was used to agitate the solution. After extraction, the fiber was inserted into the desorption solvent (500 µL methanol) under ultrasonic treatment. Figure 2 shows the schematic of the new device for SPME of headspace sampling and desorption process. Finally, an aliquot (50 µL) of methanol was injected directly into the HPLC system. Analyses were performed at least twice for each sample. Following each analysis, the fiber was washed three times with 2 mL methanol in ultrasound. Then, the fiber was left exposed in the air for 5 min in order to remove methanol remaining on the fiber.

Schematic representation of the HS-SPME and desorption processes with the new SPME sampler. HS-SPME: headspace solid-phase microextraction.
Theory/calculation
This study examines CPS and its interaction with the estrogenic sex hormones (ESHs). CPS hormone binding is a reversible reaction between CPS molecule and a hormone. The binding between CPS (C) and a hormone (H) can be described by the following equilibrium:
where, K is apparent equilibrium constant expressing the affinity of the hormone for CPS and C b, C f, and C s are the concentrations of bound CPS to hormone, free CPS, and free hormone, respectively. 33
Results
Calibration curve
The HS-SPME was performed on 5 mL of 50 mM phosphate buffer (pH 7.4) free hormone solutions of CPS in the concentration range of 0.75 µM to 142.62 µM at selected physical conditions. For this purpose, a CPS standard methanol solution was spiked into the buffer solution to obtain a sample solution of a given concentration. After HS-SPME step, the amount of extracted CPS on fibers was stripped and determined by HPLC to construct calibration curves. A sample chromatogram related to CPS in the absence of hormone is shown in Figure 3(a). The obtained calibration curves were used to calculate the amount of free CPS in solutions containing hormone and CPS.

HPLC-UV chromatograms relevant to standard solution of CPS (HS-SPME) in the absence of hormones (a); binding solution in the presence of 100 μM E1 (b); 100 μM E2 (c); and 100 μM DES (d). HPLC: high-performance liquid chromatography; UV: ultraviolet; CPS: chlorpyrifos; HS-SPME: headspace solid-phase microextraction; DES: diethylstilbestrol; E1: estrone; E2: 17β-stradiol.
Study of the binding behavior using HS-SPME with HPLC
By assuming that only the free analyte (CPS) concentration is available to partition into the SPME fiber, the free CPS concentration in the system with hormone present was calculated from the HPLC response factor and the calibration curve. Working solutions were prepared in 20 mL vials by dissolving proper amounts of CPS into 5 mL of phosphate buffer (50 mM, pH 7.4). Suitable amounts of ESH were added to the working solutions, respectively and submitted to HS-SPME. The amount of CPS molecules bound to hormone was calculated as the difference between the initial concentration and that of the free analyte. The moles of CPS (ν) bound per mole of hormone was calculated using the following equation:
Percentage hormone binding data
The binding of CPS-ESH causes only a decrease in CPS area peaks without any significant shift of the peaks, which clearly indicates the considerable changes in the hormone structure. Typical chromatograms resulted from the solutions containing CPS and various hormones are presented in Figure 3(b), (c) and (d).
The fraction unbound to receptor values, f u, was determined by plotting the free concentration versus the total concentration. The slope of the fitted linear curve is equal to f u (Figure 4). The obtained f u values are presented in Table 1.

Plot of free concentration of CPS in the presence of 100 µM E1 (a), 100 µM E2 (b), 100 µM DES(c) versus the total concentration of CPS. The slope of the fitted linear curve gives f u. CPS: chlorpyrifos; E1: estrone; E2: 17β-stradiol; DES: diethylstilbestrol; f u: unbound fraction.
Fraction bound to receptor of CPS in solutions containing E1, E2, and DES.

CPS: chlorpyrifos; E1: estrone; E2: 17β-stradiol; DES: diethylstilbestrol; f u: unbound fraction; R: correlation coefficient; [X]f: molar free concentration; [X]t: molar total concentration.
aFraction unbound to receptor of CPS was determined by linear regression of [X]t versus [X]f.
bStandard deviation with
Scatchard method
The Scatchard plots for binding analysis between CPS and selected hormones are shown in Figure 5. From the regression equation of the Scatchard plots, the values derived from slopes, y-intercept and x-intercept as follows: Scatchard plot for CPS-E1 (a), CPS-E2 (b), and CPS-DES (c) binding analysis. CPS: chlorpyrifos; E1: estrone; E2: 17β-stradiol; DES: diethylstilbestrol.
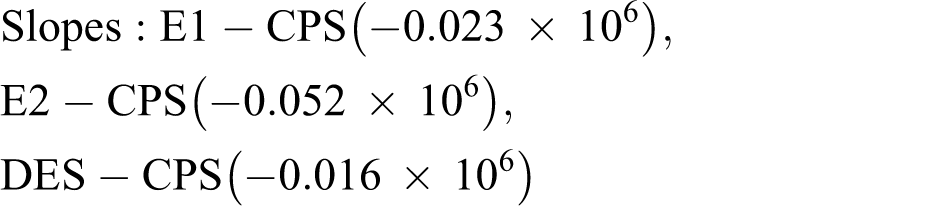


Based on the above results, the binding constants values as log K was calculated as 4.36, 4.72, and 4.20 for E1-CPS, E2-CPS, and DES-CPS, respectively. Also, the number of binding sites per hormone molecule for E1, E2, and DES was 1.10, 1, and 0.81, respectively.
Discussion
A key for environmental sciences is identifying and understanding the basic biological processes that are altered or regulated by environmental factors and that stimulate disease processes to begin, or the course of the disease to be substantially altered. Cancer incidence rates have increased in the last half of the 20th century; since 1990 worldwide incidence rates have increased by 19%. Eleven million people each year are now diagnosed with cancer, and this rate is set to increase to 16 million cases per year by 2020. 34,35 There is mounting evidence that persistent organic chemical contaminants, such as pesticides, are involved in the etiology of cancer and that these chemicals exert their effect during critical periods of development, at low, environmentally relevant levels. Endocrine disruption is a term that has arisen in human health risk assessment and is defined as the action of an external agent, such as a chemical, to interfere with normal activity of circulating hormones. This “interference” would include altering the synthesis, storage, release, or degradation of hormones as well as actions at hormone receptors such as stimulating receptor activity (receptor agonist activity) or inhibiting receptor activity (receptor antagonist activity).
In this study, a new HS-SPME-HPLC technique has been introduced for the study of the binding ability of the organophosphorus insecticide CPS to a series of human sex hormones for the first time. The powerful ability of the proposed method as a fast and reliable procedure in quality recognition of binding between CPS and sex hormones as a suggested model can be provided from Figure 3. As can be seen, the peak area of CPS in the presence of E1 and E2 (chromatograms b and c, Figure 3) shows a significant decrease in comparison with DES (chromatogram d, Figure 3), so that it may be stated that there is a strong interaction between CPS and hormones E1 and E2. This result has been confirmed through calculation of thermodynamic parameters using the proposed method.
In the calibration curve, the amount of the CPS partitioned onto the fiber coating can be calculated from the response factor and the area count of the fiber injection. The calibration curve of the total concentration versus the area count can be used as the calibration curve to determine the free concentration in the hormone binding study.
SPME can be used to determine the free and total concentration of ligand in a solution with a known concentration of receptor. When the concentration of receptor is unknown, it can be determined at the same time with the total and free concentration of ligand. 36
The source for the percentage ligand binding data (f u, fraction unbound to receptor) of ligand was the handbooks of Goodman and Gilmans 37 and of Dollery. 38 f u was determined by plotting the free concentration versus the total concentration (Figure 4). The experimental findings signify a much stronger binding of E1 and E2 to CPS compared to the DES (34% and 46% versus 5%).
The molar free concentration ([X]f), the moles of CPS bound per mole of hormone (ν), and the value of ν/[X]f were calculated using equation (4) and the obtained results are summarized in Table 2.
The Scatchard method was applied to estimate the equilibrium constant.
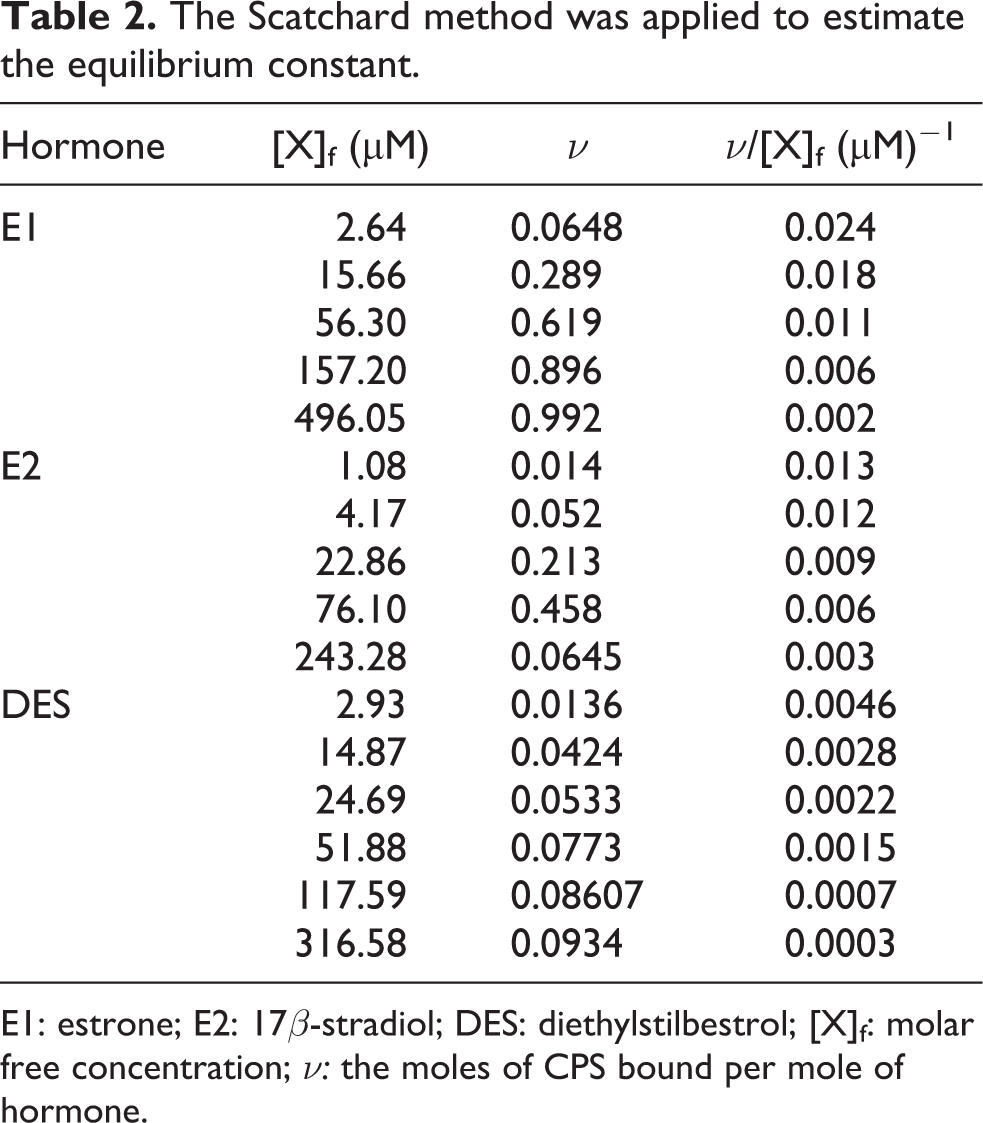
E1: estrone; E2: 17β-stradiol; DES: diethylstilbestrol; [X]f: molar free concentration; ν: the moles of CPS bound per mole of hormone.
In equation (4), ν is the ratio of concentration of the binding CPS to ESH, n is the binding site multiplicity per class of binding site and K is the association binding constant which is given by the intercept on the ordinate of a Scatchard plot. 39 The linear Scatchard plot indicates the identical independent set of sites, whereas the non-linear curves indicate the nonidentical and dependent set of binding sites. 40 All Scatchard plots for binding of CPS to hormones show high linearity, indicating identical and independent binding sites. The value range of K suggested the existence of high-affinity specific binding for E2 and E1 and an intermediate affinity binding site for DES under near physiological conditions. These contradicting results indicate rather weak CPS-DES interactions. The linearity in the plot of ν/[X]f against ν confirms that CPS bound to one class of binding sites on sex hormones.
Conclusions
The results of HS-SPME-HPLC indicate that the concentration levels of three important ESHs were changed in the presence of CPS. The binding study of pesticides with hormones has toxicological importance. This study is expected to provide important insight into the interactions of the physiologically natural and synthetic ESHs with a toxic chlorinated pesticide. The interaction characteristics of sex hormones and CPS in vitro may provide a useful database for future investigations of the molecular mechanisms of toxic pesticides on breast cancer risk. The proposed SPME methods for the study of CPS sex hormones binding proved to be useful for the determination of binding constants and number of binding sites. Moreover, it was shown that the concentration of sex hormones, total, and free CPS could be determined. The methodology is simple, fast; it covers a satisfactory concentration range and overcomes limitations of conventional methods, such as equilibrium dialysis or ultrafiltration, which suffers from the loss of volatile components in the binding or transfer process. The proposed experimental set up facilitates an easy and low cost assay on the hormones binding properties of volatile and semivolatile compounds.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.