Abstract
The best-known rapid test using gold nanoparticles (AuNPs) is the human chorionic gonadotropin pregnancy test. AuNPs are a powerful tool in point-of-care testing because of their flexibility, modifiability, and visibility. Here, we report a method to detect impurities for at-line process control in water-for-injection (WFI) manufacturing through the example of endotoxins. If a distinct concentration of these amphipathic molecules, originated from gram-negative bacteria, enters the human body, it will result in septic shock, followed by organ failure and possibly death. Every fluid given parenterally is subject to strict regulatory requirements and therefore endotoxin testing. Through use of traditional methods like the limulus amebocyte lysate (LAL) test, it takes more than 2 h to complete. With the presented method, one-fifth of the sample volume is sufficient compared with the LAL test. Once the assay components have been mixed, the result can be interpreted visually within 2 min without the use of further instruments.
Introduction
Water forms the basis of all life. In pharmaceuticals, water for injection (WFI) is the basis of almost every liquid that enters the human body parenterally. WFI is produced by reverse osmosis or distillation. 1 Stringent requirements within the process are defined in ISO 22519. 2 To assess the WFI quality, among others, total organic carbon, conductivity, free chlorine, and endotoxin levels need to stay below a distinct threshold. Endotoxins, which are also called lipopolysaccharides (LPSs) from a chemical point of view, present a major hazard, because they are known to cause sepsis in humans.3,4 It is estimated that sepsis potentially leads to about 6 million deaths per year worldwide. 5
LPSs are located in the outer membrane of gram-negative bacteria such as Escherichia coli, Salmonella, or Pseudomonas, 6 and belong to the group of pyrogens. As a consequence, they induce fever and a strong immune response, which might lead to multiple organ failure and possibly to death. The threshold for developing sepsis is around 4 ng of endotoxin/kg of bodyweight. 7 Endotoxins are very heat stable and cannot be inactivated by simply autoclaving or sterile filtering (0.2 µm) the contaminated goods, due to the small size of monomers (10–20 kDa).8–10
However, LPSs enter the organism not only by direct bacterial infection, but also by contaminated implants, tubing, syringes, or fluids like vaccines or dialysis water. Endotoxin limits are given in the United States Pharmacopeia (USP) 11 and are divided into parenteral and intrathecal drugs. Every batch needs to be tested before release. The limit of maximum endotoxin burden ranges from 20 endotoxin units (EU) per parenteral implanted medical device to 0.2 EU/kg of bodyweight for intrathecal applied injections.12–15 One EU can be converted to an interval from 0.1 to 0.2 ng of endotoxin. 16
Food and Drug Administration (FDA)-approved test systems rely on animal components (blood of the horseshoe crab), or even in vivo animal experiments with rabbits are performed. As early as 1912, pharmaceuticals were injected in rabbits, and they were tested for body temperature increase. 8 This so-called rabbit pyrogen test (RPT) is still used, 11 but because of animal welfare and inaccuracy, alternative testing methods were developed or are still under development.
Since the 1960s, the LAL assay is one of the most commonly used tests for endotoxin detection in vitro. 17 A specific blood component circulating in the horseshoe crab called amoebocyte initiates serine protease cascade, which results in coagulation after contact with bacterial endotoxin. The LAL test has been the standard endotoxin test to the present day. There are synthetic alternatives available, but acceptance in the industry progresses slowly. The recombinant factor C assay has been proven to be as sensitive as the original LAL test,18–20 but due to the highly regulated structures that are dependent on different institutions, manufacturers stick to their traditional approval procedures, even if there are promising novel products available. 21 Furthermore, the testing procedure, necessary equipment, expenses, and time are comparable to those for the conventional LAL assay.
Here we report a fast and easy “do it yourself” (DIY) method that could be used to monitor samples, for instance, in WFI systems, in an at-line control manner.
The test is based on gold nanoparticles (AuNPs) and their characteristics and interaction with different charged molecules. AuNPs are an integral part of rapid diagnostic tests like lateral flow assays. The pregnancy test might be a well-known representative. 22
In this study, we exploited the AuNP color change during aggregation 23 for detecting the absence of the model target endotoxin. During the citrate reduction synthesis, a citrate shell covers the spheres and prevents immediate aggregation. This shell becomes unstable with increasing ionic strength, for example, by adding chloride ions. Aggregation can be quantified by spectrophotometry and detected due to the AuNP plasmon resonance through a color shift from wine red to blue-gray.
Materials and Methods
Materials
Gold (III) chloride trihydrate (HAuCl4), trisodium citrate, bidistilled water, and sodium citrate were procured from Merck KGaA (Darmstadt, Germany). The tested samples were produced using LAL reagent water (LRW; Lonza) and spiked with Control Standard Endotoxin (CSE; Lonza, Cologne, Germany) from Escherichia coli strain O55:B5. The PYROGENT-5000 kinetic turbidimetric LAL assay (Lonza) was used as a reference test and was performed according to the manufacturer’s instructions.
AuNP Synthesis
AuNPs with a diameter of 15 nm were synthesized using the citrate reduction method, developed by Turkevich et al. 24 and Frens 25 and modified by Lakatos et al. 26 Each batch was characterized by scanning electron microscopy and Vis spectroscopy.
Glassware was cleaned using aqua regia (concentrated hydrochloric acid and nitric acid at a mixing ratio of 3:1) and purified water. A 101.5 mM tetrachloro-auric acid stock solution was prepared by dissolving the gold salt in ultrapure water. Fifty milliliters of a 0.5 mM tetrachloro-auric acid working solution was heated to 75 °C in a water bath with a magnetic stirrer. While stirring, 7 mL of a 34 mM sodium citrate solution was added quickly. A color change from pale yellow to colorless, followed by violet and a wine red, had been observed, indicating a successful nanoparticle formation. The solution was kept stirring for 1 h while cooling down to room temperature. With this ratio of tetrachloro-gold (III) acid and sodium citrate, a AuNP diameter of 15 nm was reached. Moderate heat ensured monodispersed particles. AuNPs were stored protected from light, at 4 °C, and used within 4 weeks (pH = 6.4).
AuNP Characterization
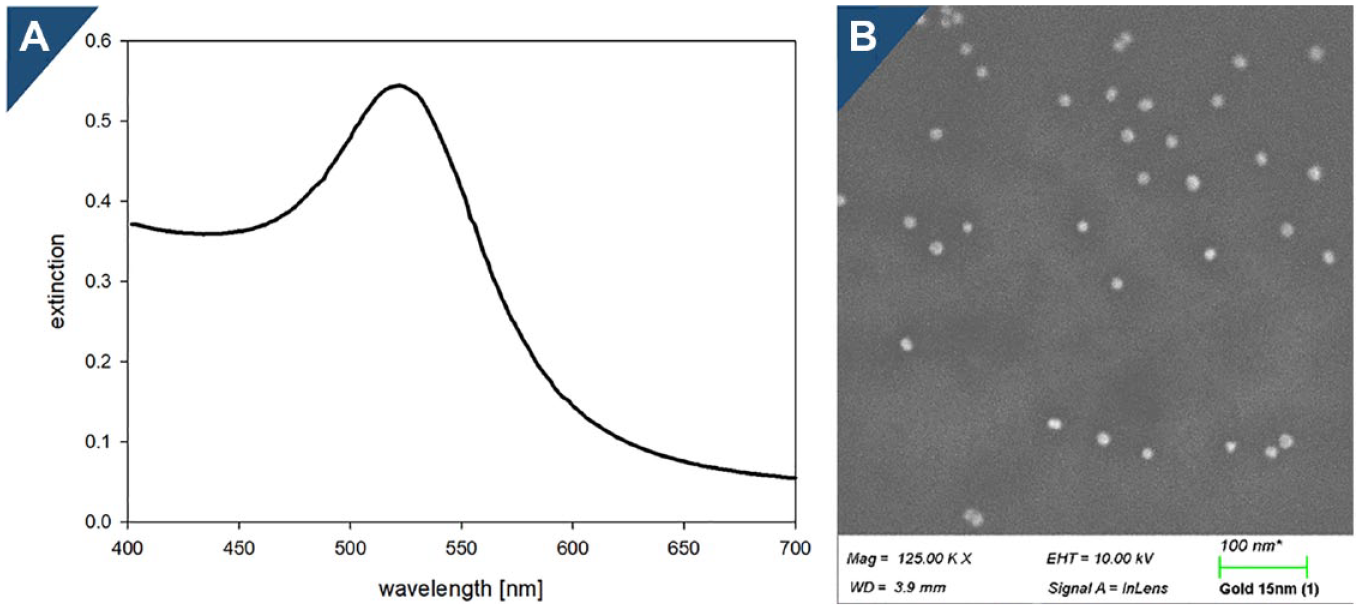
Spectrophotometry was performed to determine the AuNP size and size distribution. The absorption peak of 15 nm diameter AuNPs is expected at 520 nm. The maximum wavelength shifts to blue with increasing particle size. A shoulder in the spectrum indicates particles with different sizes or particle agglomeration. The spectrum of the AuNPs was monitored using the TECAN (Mannendorf, Switzerland) infinite 200 plate reader (400–700 nm) (
(
Preparation of the Solutions and Spiked Model Samples
CSE was dissolved in LRW following the certificate of analysis to obtain a concentration of 100 EU/mL. The solution was votexed by room temperature for 15 min (Vortex Genie 2). Serial dilutions were prepared from 10 to 0.01 EU/mL. Each dilution was vortexed for 1 min.
A sodium chloride stock solution (1 M) was prepared in LRW. The concentration of the working solutions ranged from 0.3 to 1 M.
Sodium Chloride Titration and Colorimetric Assay
The starting point for the test is to define the relationship between ionic strength and AuNP agglomeration. This is called sodium chloride titration. In practice, this means that 10 µL of sodium chloride dilution series is mixed with 100 µL of AuNP solution in order to identify the ionic strength needed for AuNP agglomeration and therefore color change. Slight AuNP batch-to-batch variations were detected.
This titration is then repeated including spiked samples with defined endotoxin concentrations. One colorimetric sensor reaction contained 20 µL of a spiked sample, 10 µL of sodium chloride solution, and 100 µL of self-synthesized AuNPs (8.8 × 1011, 2.6 nM). The AuNPs were added after sample and saline were mixed in a 96-well plate. In negative control samples, 20 µL of LRW was used instead of the spiked sample volume. The concentration of sodium chloride ranged from 0.3 to 1 M for NaCl titration series.
Quantification of LPS Concentration with the Naked Eye
The bare AuNP solution appears wine red. This is attributed to the fact that colloidal metal particles, and AuNPs in particular, absorb green light in the range of 497–550 nm. 28 The citrate shell, which forms during AuNP synthesis, prevents immediate particle agglomeration. If ionic strength increases, the shell is destabilized and the particles agglomerate, which results in a visible color change from ruby red to gray. LPSs provide additional protection to the citrate shell. Given the same ionic strength, the AuNPs remain in their ruby red monodispersed state with protective molecules present.
Results
Even with a simple DIY test setup, achieving reproducible and conclusive results is possible. Here we present our preliminary data on the development of a colorimetric sensor, which could be used in WFI at-line testing or in medical device productions where closed-mesh bacterial endotoxin tests need to be performed.
Quality and Reproducibility of Synthesized Particles
AuNPs with a diameter of 15 nm were synthesized by performing a citrate reduction reaction. This is an easy and quick way to produce fresh AuNPs on demand. Besides clean glassware, only three ingredients are required: bidistilled water, tetrachloro-gold (III) acid, and sodium citrate. The particle size was controlled by the gold-to-reductant ratio and set to a diameter of 15 nm for sufficient optical properties and stability. The size and monodispersity of the particles were reviewed by electron microscopy and Vis spectrometry (
where A is absorption at the maximum absorption wavelength, l is the optical path lengh measured (3.12 mm; volume of 100 µL in a 96-well plate), and ε is the extinction coefficient for 15 nm diameter gold spheres (6.86∙108 M−1 cm−1). 29 The absorbance at the maximum absorbance wavelength ranged from 0.5 to 0.6 in every batch (n = 4).
The endotoxin contamination level of AuNPs was examined through the kinetic turbidimetric LAL assay and was below the limit of detection (<0.01 EU/mL). This test is commonly used and formalized in the European, United States, and Japanese Pharmacopeias as the bacterial endotoxin test.12–15 A previous study reported a shift in the maximum absorbance wavelength of 1 nm. 30 Since the resolution limit of the used plate reader is 2 nm, an aberrance in the spectrum was neither detected nor considered.
Spectroscopic Inspection to Detect the Absence of Endotoxins
The idea or principle behind the colorimentric sensor is shown in
Figure 2
and was inspired by Alsager et al.
31
The plasmon peak (absorption maximum) of the monodispersed 15 nm diameter AuNPs in the Vis spectrum was detected at 522 nm wavelength. The synthesis of AuNPs through the citrate reduction reaction causes a stabilizing citrate shell around the spheres. The absorption maximum decreased in intensity while the concentration of NaCl increased (
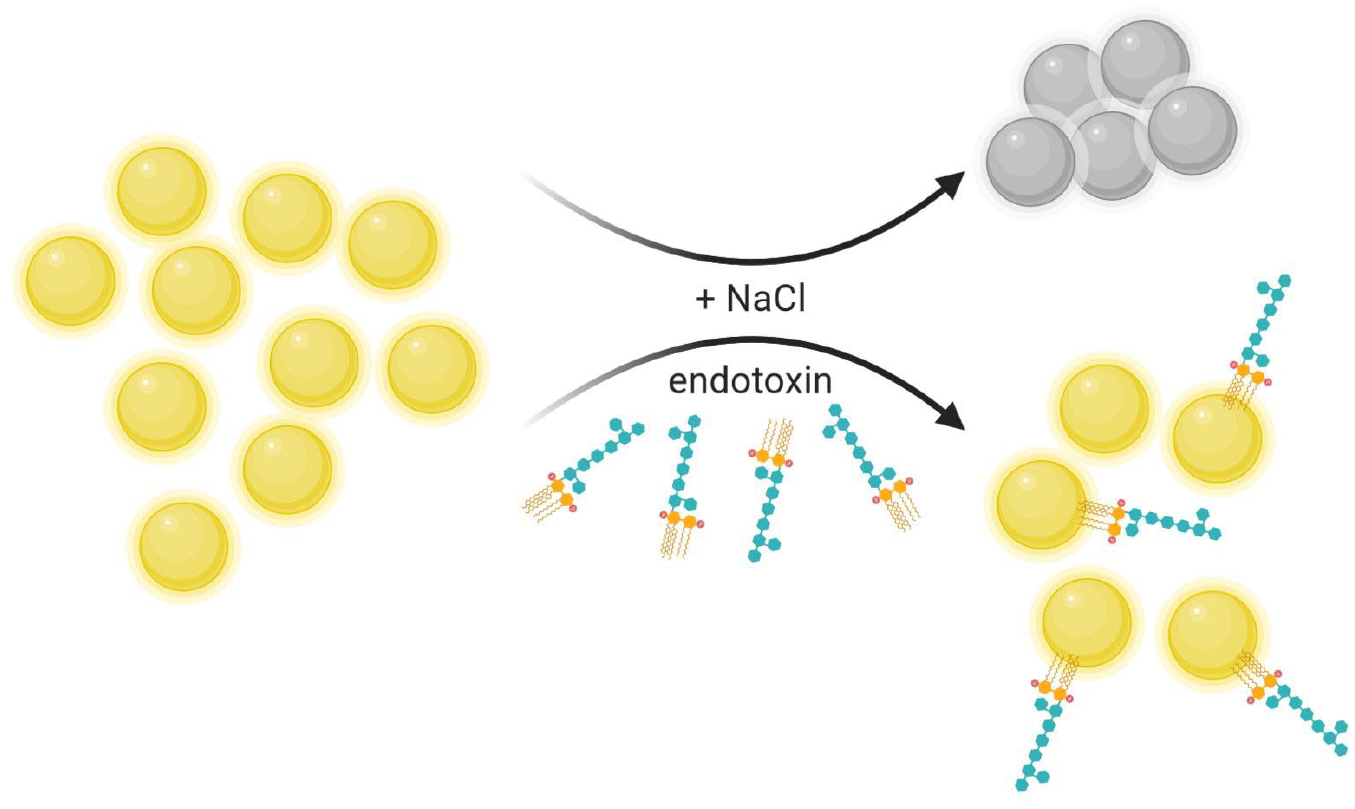
AuNPs in solution will aggregate in the presence of sodium chloride (upper arrow). In the presence of endotoxins, the aggregation is prevented (lower arrow). A color change indicates the state of aggregation of the 15 nm diameter AuNP.
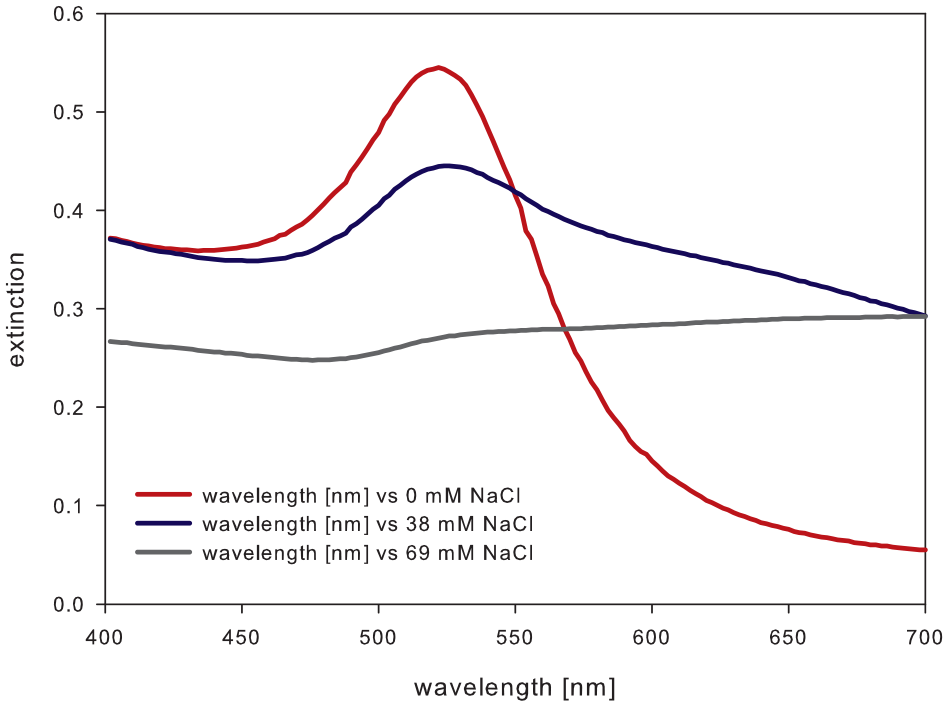
Vis spectrum of AuNP solution with a maximum adsorption at a wavelength of 522 nm. The plasmon peak decreases in intensity with increasing NaCl concentration.
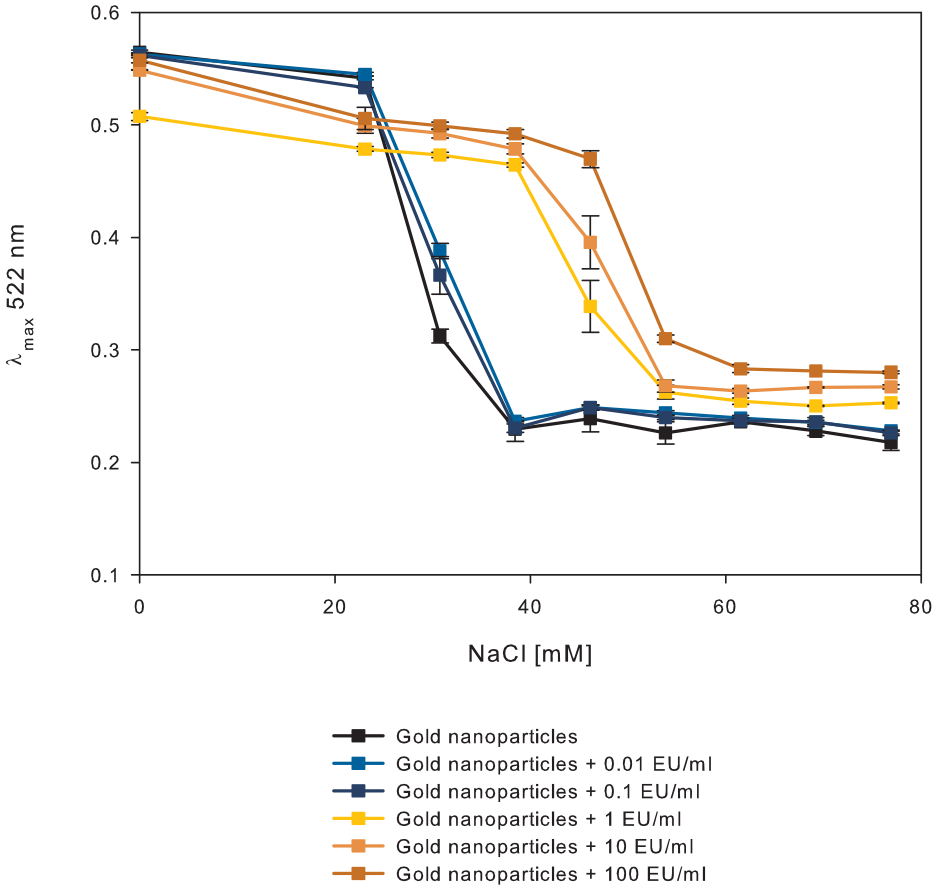
Graphical representation of the course of aggregation of AuNPs in the presence of endotoxins (0.01–100 EU/mL) at different sodium chloride concentrations (n = 3). The graph depicts the maximum absorption wavelength of 522 nm. The measurement was performed after 10 min of incubation of mixed solutions at room temperature.
Here, a total reaction volume is composed of 100 µL of AuNP solution, 10 µL of sodium chloride solution, and 20 µL of spiked sample or LRW as the negative control. Bare AuNPs aggregated in this total reaction volume of 130 µL at a sodium chloride concentration of 38.5 mM. In the presence of endotoxins, the NaCl concentration needed for aggregation of AuNPs was shifted to 54 mM.
Endotoxins, which are amphiphilic molecules, protect the AuNP citrate shell and prevent aggregation at increasing salt concentrations (
Visual Inspection to Detect the Absence of Endotoxins
Even without the use of a spectrometer, the WFI quality can be verified with the naked eye. The colloidal AuNPs absorb green light. 28 Therefore, bare AuNP solution appears wine red, which was described in the literature by plasmon resonance.32,33 The citrate shell, which forms during AuNP synthesis, prevents particle agglomeration. If ionic strength increases, the AuNPs will clump together. The reduced distance between the particles leads to overlapping and coupling of the plasmons. 34 Green light wavelength is not absorbed any longer, but a red shift results in a visible color change to gray.
In the case of the colorimetric sensor, we decided to add 20 µL of the solution to be tested to 10 µL of 0.6 mM NaCl solution and mix it with 100 µL of AuNP. This results in a final salt concentration of 46 mM in the total volume of 130 µL. Interestingly, the color change was easily visible within the first 2 min after the chemicals were mixed. If the sample contains 0.1 EU/mL or less, the particles aggregate, and the wine red AuNP solution turns to gray immediately. In the presence of at least 1 EU/mL, the test solution remains wine red (
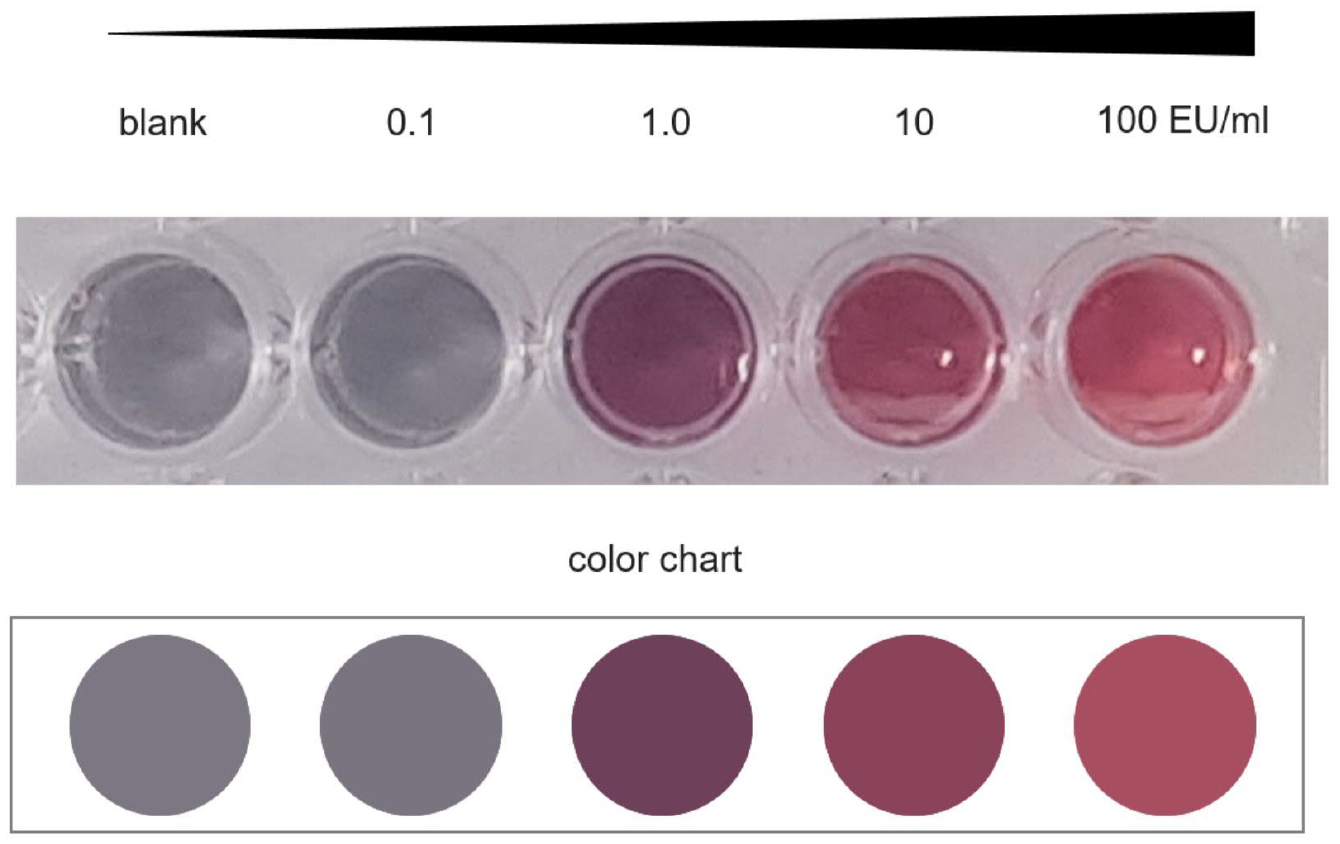
AuNPs (100 µL) in the presence of a final NaCl concentration of 46 mM with increasing endotoxin concentration (sample volume of 130 µL). To classify the sample result semiquantitatively, a color chart (bottom) can be used to compare the color change within 2 min after mixing the test components.
Summary and Discussion
In WFI systems, bacterial contamination might happen through leakages or if water remains in valves, or during a system lockdown the bacterial burden might increase undiscovered until a certain amount of samples or batches are collected. Gram-negative bacteria shed endotoxins during cell lysis and constantly during growing and dividing processes. 35 To allow a much quicker reaction, we present instructions for a DIY at-line test procedure. This method might be used for testing other aqueous samples, which occur, for example, in implant testing too. Another hot topic regarding endotoxin burden is dialysis water, where the endotoxin concentration should be lower than 2 EU/mL. 36 The major problem is the formation of bacterial biofilms inside the medical tubing.
The one-pot AuNP synthesis was reported decades ago.24,25 Nevertheless, it is still a widely used, simple method to prepare monodispersed AuNPs. Changing the relation from gold (III) chloride trihydrate and trisodium citrate influences the size of the AuNPs directly. We synthesized 15 nm diameter AuNPs and proved their monodispersity with scanning electron microscopy. The Vis spectrum showed a clear plasmon peak at 522 nm (
To prove that the absence of endotoxin is as simple as mixing a cocktail, for a quantitative statement, three ingredients are needed: 20 µL of the samples to be tested, 10 µL of a 0.6 M NaCl solution, and 100 µL of AuNPs of 15 nm in diameter. For the direct comparison and as a potential positive control, a second reaction volume using purified water instead of the sample could be used. We wanted the test result to be easily estimated, similar to the pH paper color code. Figure 5 shows that the absence of endotoxins can be detected by immediate nanoparticle aggregation and therefore a color shift from wine red to gray. In the presence of at least 1 EU/mL, the AuNPs are protected from aggregation at a final NaCl concentration of 46 mM. Recently, Zhu et al. described a colorimetric test based on AuNPs too. 37 They functionalized the particles with aptamers and were able to detect 1 µg/mL endotoxins. The big advantage of this study is, of course, the specificity they included with the aptamers, but the colorimetric sensor described here is 105 times more sensitive, if the concentration is converted to EU/mL (1 EU/mL = 0.1 – 0.2 ng/mL). 16
What must be seen critically is the absence of a specific binder in an antibody-dependent manner. The specificity of this method was addressed, among others, by polymyxin B. This cyclic polypeptide was reported to be useful in removing endotoxins from liquids through affinity chromatography.38,39 Since these molecules are polar, they acted similar to sodium chloride and promoted AuNP aggregation.
On the contrary, if AuNPs were incubated with anti-LPS antibody (WN1 222-5) prior to the sodium chloride titration, the AuNPs were protected from aggregation even at higher sodium chloride concentrations. The test aimed for a higher affinity between the endotoxin and its specific antibody compared with the nonspecific interaction between AuNPs and antibodies. Probably, the thiol groups attached to the gold surface but left the paratope uncovered. In this case, the endotoxin molecules might also bind to the antibody and stabilize the monodispersed spheres at higher ionic strength (data not shown).
Since the properties of WFI, ultrapure water, and dialysis water are very well defined and highly regulated, one can state that if the test turns out positive, the batch needs to be further analyzed or discarded anyway. WFI serves only as an example where this DIY test could allow at-line testing. The colorimetric sensor testing procedure needs only a small sample volume of 20 µL, and a small number of samples can be tested within minutes. If compared with the LAL assay, a fivefold sample volume is needed, and the procedure takes approximately 1.5–2 h. Single-sample testing is not sufficient, since a calibration curve, generated by at least three known endotoxin concentrations, is used to calculate the unknown endotoxin concentration.
Bringing the easy test system to the next level and improving simplicity and digitalization, our idea is to use a device acting in the context of the Internet of Things. After mixing the unknown sample, sodium chloride, and AuNP solution, an image is taken with a camera or scanner. The executed inspection protocol, photodocumentation, and consequently test result are then transferred to a laboratory information and management system.
We propose that the presented DIY test can serve as a first indicator comparable to a traffic light system. If the AuNP solution remains red after mixing the components (samples to be tested, salt, and nanoparticles), further tests are required. If the colorimetric sensor switches to gray, no impurities were detected.
Supplemental Material
sj-pdf-1-jla-10.1177_2472630320978187 – Supplemental material for Development of a Gold Nanoparticle-Based Colorimetric Sensor for Water for Injection At-Line Impurity Testing
Supplemental material, sj-pdf-1-jla-10.1177_2472630320978187 for Development of a Gold Nanoparticle-Based Colorimetric Sensor for Water for Injection At-Line Impurity Testing by Saskia Reichelt, Elke Boschke, Olena Reinhardt, Thomas Walther and Felix Lenk in SLAS Technology
Footnotes
Acknowledgements
We thank Dipl.-Ing. Petra Fiala from Technische Universität Dresden, Institute of Process Engineering and Environmental Technology, for her help with characterizing the gold nanoparticles with scanning electron microscopy.
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful for the financial support of this work by the Federal Ministry for Economic Affairs and Energy, Central Innovation Programme for Small and Middle-Sized Enterprises (ZIM), grant number ZF4100923AJ7.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

