Abstract
In the last decade, several studies have proven that when at low concentration reactive oxygen species (ROS) show an adaptive beneficial effect and posited the idea that they can be utilized as inexpensive and convenient inducers of tissue regeneration. On the other hand, the recent discovery that cancer cells are more sensitive to oxidative damage paved the way for their use in the selective killing of tumor cells, and sensors to monitor ROS production during cancer treatment are under extensive investigation. Nevertheless, although ROS-activated signaling pathways are well established, less is known about the mechanisms underlying the switch from an anabolic to a cytotoxic response. Furthermore, a high variability in biological response is observed between different modalities of administration, cell types, donor ages, eventual concomitant diseases, and external microenvironment. On the other hand, available preclinical studies are scarce, whereas the quest for the most suitable systems for in vivo delivery is still elusive. Furthermore, new strategies to control the temporal pattern of ROS release need to be developed, if considering their tumorigenic potential. This review initially discusses ROS mechanisms of action and their potential application in stem cell biology, tissue engineering, and cancer therapy. It then outlines the state of art of ROS-based drugs and identifies challenges faced in translating ROS research into clinical practice.
Introduction
Since its first discovery in 1818 1 and its first use as an in vivo intravenous drug to treat broncho-pneumonia in 1920, 2 hydrogen peroxide became a known cytotoxic agent and a popular antiseptic. Only recently has a growing body of evidence shown that when at low concentration, it also exerts a biological effect. The cell response to oxidative stress appears indeed to be biphasic, with low doses possessing an adaptive stimulatory effect and high doses showing a toxic effect, according to a process known in toxicology as “hormesis.” For instance, it was observed that, in the nanomolar order, hydrogen peroxide increased the proliferation of rabbit epithelial cells; at 100 μM it reduced cell growth, whereas at concentrations higher than 1 mM it induced cell death. 3 Similarly, low (10 µM) hydrogen peroxide doses increased the proliferation of murine mesenchymal stem cells (MSCs) and hematopoietic stem cells, whereas higher ones inhibited their growth. 4 Low oxidative stress was furthermore proven to activate pathways leading to migration 5 and collagen synthesis,6,7 and this capability has been widely attractive for tissue regeneration purposes. In this context, extensive research was done with respect to fibroblasts and epithelial cells. A few proangiogenic and wound healing products have recently been developed (e.g., topical creams such as Mycosinate from Nektr Technologies, LHP from Bioglan, and Crystacide from Mipharm SpA; hydrogen peroxide-releasing hydrogels; 8 soaked gauzes; 9 and enriched honeys 10 ), and some of them have already undergone clinical investigation ( Table 1 ). On the other hand, the application of hydrogen peroxide in other target tissues seems far from being realizable, because its biological effect has still not been fully unraveled. Furthermore, different reactive oxygen species (ROS) concentrations, modalities of administration, and species may even result in opposing biological outcomes. Additionally, not only does each cell type respond differently, but also the microenvironment appears to be a determinant factor affecting biological outcome. Furthermore, moving to in vivo experimentation, the quest for the optimal delivery system is still elusive. This paper proposes to critically discuss the research progress and applications of ROS-based products. ROS physiological production and catabolism, differences in ROS response depending on cell type and experimental conditions, and signaling and mechanisms regulating the switch between anabolic and cytotoxic effects are initially discussed. The application of ROS-based drugs in stem cell biology and cancer therapy, the modality of ROS administration, and associated drawbacks are later analyzed.
Clinical Trials Investigating ROS-Based Drugs for the Treatment of Wound Healing Disorders.
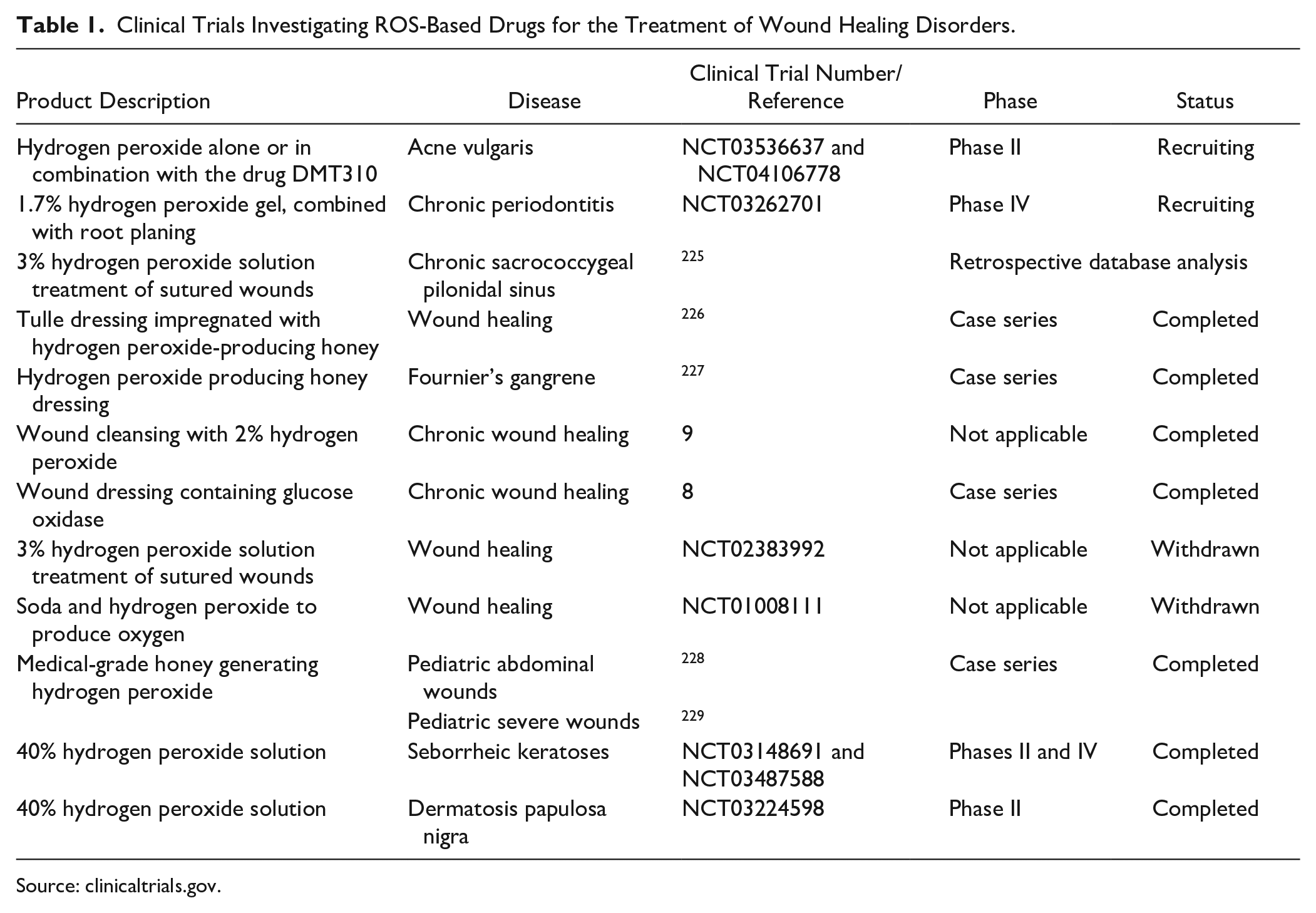
Source: clinicaltrials.gov.
ROS Production and Catabolism
ROS are chemically reactive radical and nonradical oxygen species, and they include hydrogen peroxide (H2O2), superoxide anion (O2−), and hydroxyl radical (*HO) (
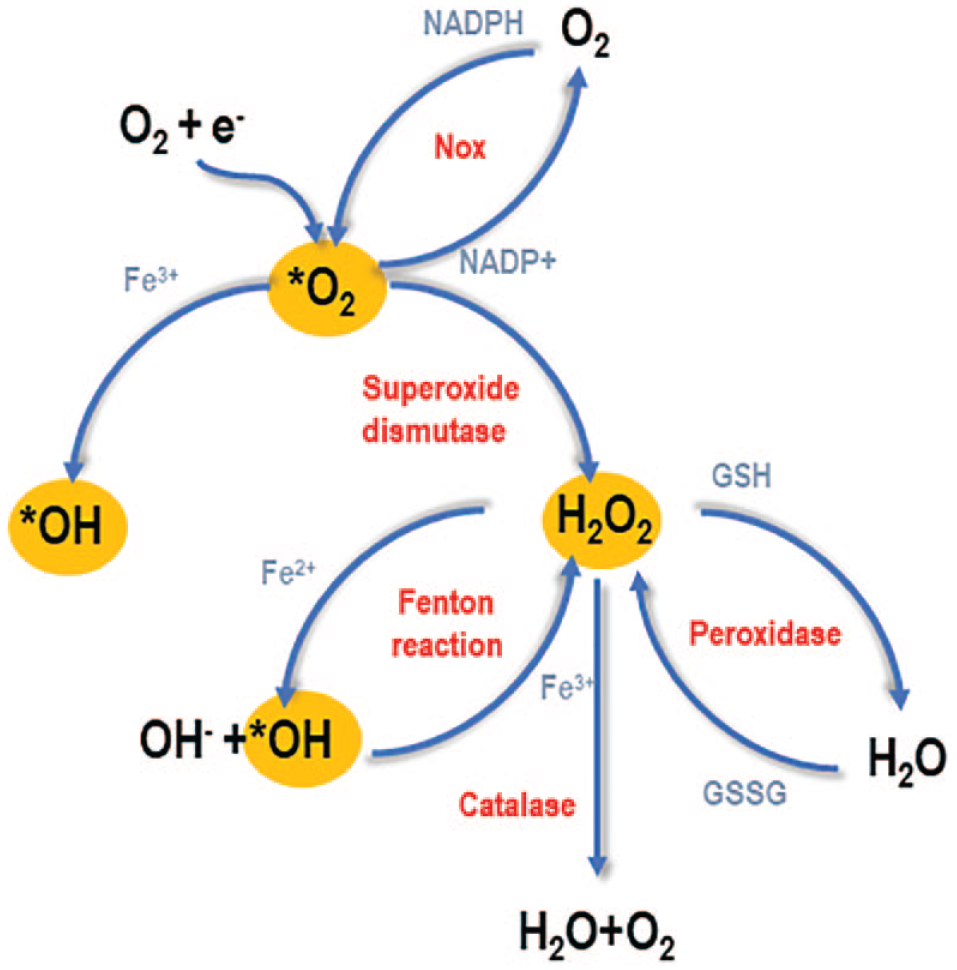
Schematic representation of ROS production and catabolism. ROS mainly include hydrogen peroxide (H2O2), superoxide (*O2), and hydroxyl radical (*HO). Superoxide is produced either by spontaneous addition of an electron into atmospheric oxygen (mainly in the mitochondrial respiratory chain) or through action of the enzyme Nox. Superoxide can be converted into hydroxyl radical or hydrogen peroxide, by SOD. Hydrogen peroxide can be converted into hydroxyl radical (via the Fenton reaction) or oxidized into H2O by either the CAT or the peroxidase.
ROS Biological Effect: The Effect of Cell Type and External Environment
The discovery that, at low concentration, hydrogen peroxide increases cell proliferation dates back to 1974.
18
Its proliferative effect was initially attributed to its capability to mimic insulin, as shown by the fact that it activates 3-O-methylglucose transport (which is physiologically stimulated by insulin) in rat brown fat cells
18
and that insulin itself stimulates H2O2 production in rat epididymal fat cells.
19
ROS are furthermore involved in cell proliferation, as suggested by the fact that their production is higher when cells are proliferating and gradually decreases when they are close to confluence,
20
and that treatment with SOD and/or CAT decreases cell proliferation and increases cell death.
21
For all these reasons, ROS were traditionally considered insulinomimetic.
22
In reality, in recent decades it has become evident that the cell response to oxidative stress is extremely complex; it not only involves insulin signaling, but also is influenced by several external and internal factors. For instance, hydrogen peroxide hormetic concentrations are variable depending on cell types and range from a few nanomoles (0.1–10 nM) for human bone marrow mesenchymal stem cells (BMSCs),
23
5 µM for synoviocytes,
24
and 25 µM for the murine prechondrogenic cell line ATDC5,
25
to concentrations as high as 200–300 µM for human uterosacral ligament fibroblasts.
6
On the contrary, cytotoxic effects were already observed with 25 µM in rat BMSCs,
26
with concentrations even as low as 100 nM inducing telomere instability and a reduced population doubling rate in primary chondrocyte cultures.
27
Also, the donor’s age affected the cell response, with older donors showing higher propensity to cytotoxic effects.
28
Furthermore, cell culture conditions affected basal ROS production and catabolism. For instance, in murine tenocytes, Nox1 and Nox4 expression and H2O2 production increased when cells where grown in high-glucose medium,
29
and similarly, in bovine aortic smooth muscle cells,
30
endothelial cells,
30
and keratinocytes,
31
high glucose levels stimulated Akt-induced Nox-mediated H2O2 production. High-glucose culture media also stimulated 50% more ROS production in bovine chondrocytes,
32
HUVEC cells,
33
human adipose-derived stem cells (ADSCs),
34
and Caco-2 cells.
35
Similarly, the addition of serum increased ROS production in bovine pulmonary artery endothelial cells.
36
Also, low oxygen tension increased superoxide and hydrogen peroxide production by about 25%–30%,37–39 probably by increasing the abundance of the partially reduced ubisemiquinone anion, which in turn stimulated superoxide production.
39
Due to different basal levels, distinct behavior was observed when further exogenous ROS were added to cell cultures. For instance, human tenocytes, when grown at high glucose levels, become more sensitive to H2O2-induced apoptosis.
40
Under low glucose (5 mM), hydrogen peroxide exposure increased their collagen I expression, while under a high-glucose environment (17.5 mM), it only resulted in increased caspase activation and p53 expression.
41
Yet, it was observed that a mild preexposure to ROS activated an antioxidant machinery, which protected cells from subsequent exposures. For instance, when hamster fibroblasts were exposed to UV radiation, hydrogen peroxide production and nuclear fragmentation increased, concomitant with a significative decrease in cell viability. However, if cells were pretreated with 30 µM hydrogen peroxide for 5 days per week for 15 days, despite no change in their lipid peroxidation status, nor in the GSH/GSSG ratio or their GPx levels, they appeared more resistant to UV-mediated damage, due to increased basal CAT levels.
42
Similarly, when a mouse-derived myoblast cell line
43
and human ADSCs
44
were pretreated with sublethal doses of hydrogen peroxide for 1 h/day for 1 week, they became more resistant to lethal doses. Differences in cell type and microenvironment may give rise to a high variability in the observed biological outcome. While the microenvironment can be easily controlled in in vitro applications, this is more difficult in the case of in vivo studies. Several diseases are characterized by perturbations in redox or glucose homeostasis, and diseased cells may respond differently to exogenous ROS exposure. For instance, fibroblasts from Alzheimer’s patients,
45
as well as pancreatic β-cells from diabetic murine models,
46
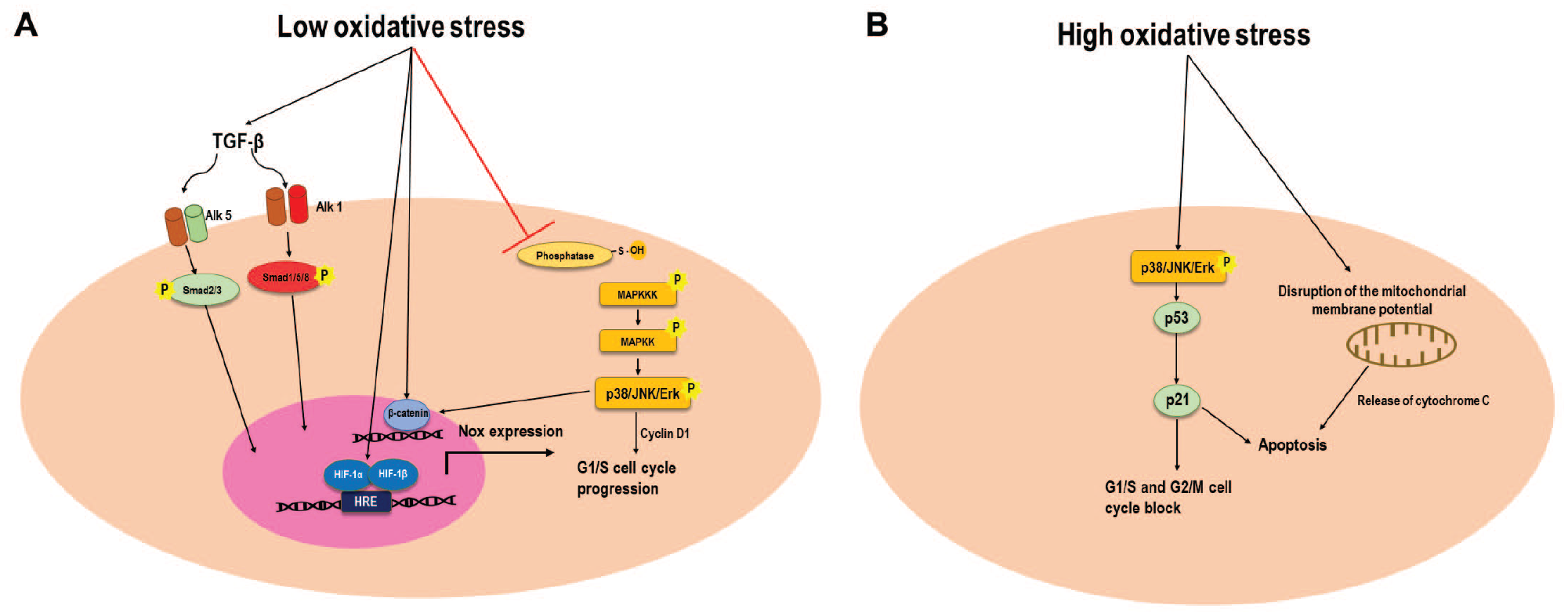
which in vivo are exposed to high—but not lethal—oxidative stress, showed an altered response and a higher resistance to lethal hydrogen peroxide doses. Variability in ROS response was generally attributed to activation of distinct signaling routes. These generally include mitogen-activated protein kinases (MAPKs), hypoxia-inducible factors (HIFs), Wnt signaling, and transforming growth factor β (TGF-β) (
(
Cell Signaling Activated by ROS
MAPKs
The activity of tyrosine phosphatases is regulated by a conserved Cys residue in their active site: when the thiol group is reduced (S–H), they are active, while when it is oxidized into sulfenic acid (S–OH), they are inactive. ROS are able to oxidize the Cys, thereby inactivating the phosphatases and eventually increasing phosphorylation of target tyrosines (Tyr). Several signaling pathways (e.g., EGF 47 or VEGF 48 ) are triggered from the increased Tyr phosphorylation. Yet the most investigated are the MAPKs, a family of serine–threonine kinases traditionally including three members: the canonical ERKs (extracellular signal-regulated kinases), the JNKs (Jun amino-terminal kinases), and p38/SAPKs (stress-activated protein kinases). While the first one is mainly involved in cell growth, survival, and differentiation, depending on target tissue, the other two also trigger inflammatory and apoptotic processes. 49 Depending on cell type, low oxidative stress activated p38,50,51 JNK,4,50,52,53 canonical MAPK Erk,51,54–56 Erk5 (also known as big MAPK1 [BMK1]),57,58 or even JNK2α2, a rare member of the noncanonical JNK family, which in turn stimulated Erk1/2 signaling. 59 ROS-mediated MAPK activation generally resulted in increased cell migration5,58,60 and proliferation,51,58–62 as it upregulated cyclin D1, thereby facilitating G1/S transition. 51 Nevertheless, it is worth noting that despite the mechanisms still not being well understood, and probably depending on the length of their activation, 63 MAPKs may also show a dual role and trigger apoptotic cascades, 64 for instance, in lens epithelial cells, Erk1/2- and JNK-mediated ROS induction of apoptotic cascade. 55 This indicates that a better characterization of MAPK mechanisms of action is still needed.
Hypoxia and HIF-1α
HIFs are heterodimeric transcription factors composed by α and β subunits. Under atmospheric oxygen tension (20% O2), HIF-α subunits are rapidly degraded due to hydroxylation of specific prolines, yet at lower oxygen tension (below 5%), they are stable and can heterodimerize with β subunits and translocate into the nucleus, activating the expression of target genes. 65 Crosstalk between hypoxia and oxidative stress has been largely discussed: hypoxia is a known mediator of ROS production,37–39 and vice versa, ROS exposure 66 and Nox overexpression 51 were shown to increase the expression of HIF-1α. While some studies attributed hypoxia-mediated ROS production to an increase in the abundance of the partially reduced ubisemiquinone anion, 39 HIF-1α-induced upregulation of Nox was demonstrated in several cell types,67–69 and the direct binding of HIF-1α to Nox2 mRNA was confirmed by ChIP in HUVEC cells. 70 HIF-1α generally acts as an inducer of cell survival under stressful conditions, although underlying mechanisms are still not clear. In BMSCs, preconditioning with a sublethal concentration (20 µM) of hydrogen peroxide indeed rendered cells more resistant to subsequent exposure to lethal doses (100 µM) via HIF-1α, 66 whereas both HIF-1α overexpression 71 and hypoxia preconditioning 72 protected cells from lethal oxidative damage. On the other hand, a cross-play between hypoxia and ROS regulates the aggressiveness69,73 and survival 74 of solid cancer cells. Very recently, hydrogen peroxide preconditioning has been exploited for tissue engineering applications,4,75 as it can potentially facilitate graft growth and resistance under in vivo adverse conditions/excessive oxidative stress by increasing basal levels of active HIF-1α.
Wnt Signaling
Wnt is a family of secreted glycoproteins involved in cell differentiation, inflammation, and cancer progression. In its canonical route, Wnt binding to the receptor Frizzled activates the Dishevelled protein (Dvl), which in turn inhibits the glycogen synthase kinase 3β (GSK-3β), a kinase responsible for β-catenin phosphorylation. This results in an increase in the amount of nonphosphorylated (active) β-catenin protein, which translocates into the nucleus and acts as a transcription factor. Oxidative stress is believed to inhibit nucleoredoxin (NRX), which is an inhibitor of Dvl,
76
by oxidizing specific Cys (
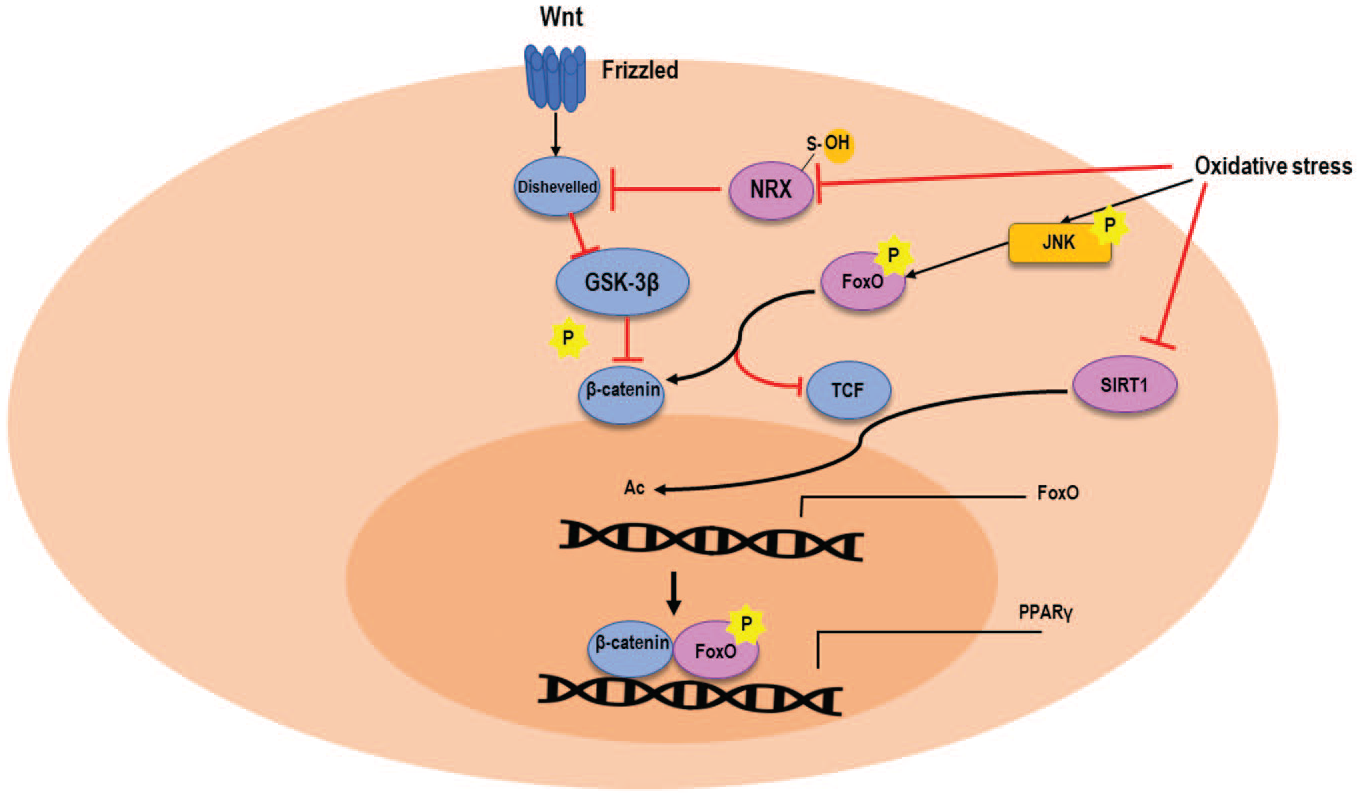
Wnt proteins physiologically act by binding to their receptor Frizzled, which in turn activates the Dvl protein. Dvl further inhibits the GSK-3β, which is responsible for the phosphorylation of β-catenin. This results in an increase in nonphosphorylated β-catenin, which represents the active (most stable) form. Oxidative stress is believed to increase β-catenin signaling, as it inhibits the NRX protein, which is an inhibitor of Dvl, by oxidizing specific Cys residues. This results in an increase in the amount of active Dvl. Nevertheless, a few studies have shown that ROS diverts β-catenin from its canonical target TCF to the FoxO proteins. Particularly, oxidative stress was shown to activate the FoxO proteins (via the JNwK kinases) and to inhibit the deacetylase SIRT1, an inhibitor of their transcription. Thus, under oxidative stress, the β-catenin–FoxO complex activates transcription of the genes involved in adipogenic differentiation (e.g., PPARγ), at the expense of osteogenic differentiation.
TGF-β, Fibrosis, and Cancer Progression
TGF-β is a family of multifunctional cytokines, mainly involved in cell growth and differentiation. They bind to a complex formed by type I (Alk1 and Alk5) and type II tyrosine kinase receptors, which subsequently phosphorylate intracellular Smad proteins. TGF-β show different—or even opposing—biological effects, depending on the type of cell receptors and Smad protein activated.65,87,88 ROS activation of TGF-β1 signaling was proven in several cell types,89–93 whereas TGF-β stimulation in turn increased ROS production.91,94,95 Activation of TGF-β signaling and the biological outcome significantly varied depending on the tissue. For instance, hydrogen peroxide even decreased TGF-β expression of human umbilical cord MSCs. 75 On the contrary, ROS-mediated activation of TGF-β was indispensable to stimulating collagen synthesis in fibroblasts,6,93 but led to pathological fibrosis in hepatic cells, 95 renal epithelial cells 96 and lung cells.96,97 On the other hand, it played a pivotal role in cancer aggressiveness and progression.91,94,98,99 In this context, it is noteworthy to mention the recent discovery of the hydrogen peroxide inducible clone-5 (Hiac-5), a member of the paxillin superfamily, located at focal adhesions, which transduces signals in response to changes in cell adhesion. It is activated by both ROS and TGF-β 100 and is associated with the progression of several cancers.101–105
NF-κB and Inflammatory Pathways
Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) is an inducible transcription factor and mediator of the immune response that resides in a latent form in the cytoplasm until activated by specific stimuli and is responsible for increasing the expression of cytokines such as IL-1β. 106 For a long time, hydrogen peroxide was considered a pro-inflammatory molecule acting through NF-κB.107–110 In reality, recent evidence suggests that hydrogen peroxide only acts as a mediator rather than as a direct NF-κB activator, and that it equally induces the synthesis of pro- and anti-inflammatory molecules, possibly acting as a fine-tuning regulator of inflammatory processes. For instance, in human keratinocytes, despite activation of the upstream molecule IKKα, hydrogen peroxide did not increase NF-κB activity, 47 whereas in bovine chondrocytes, it decreased IL-1β-mediated NF-κB activation. 89 Similarly, ROS mediated the human tonsil-derived MSC inhibitory effect on macrophage–IL-1β production. 111 In antigen-presenting cells, ROS production was furthermore associated with tolerance induction, and conversely, ROS scavenging promoted inflammatory T-cell responses and NF-κB activation. 112 Similarly, ROS reduced monocyte inflammatory rolling and extravasation via reduction of the N-glycans on the cell surface, 113 whereas in human PBMCs, it activated the expression of the anti-inflammatory molecules adiponectin and IL-6, while reducing the expression of toll-like receptors (TLRs). 114 Thus, the traditional role of oxidative stress as the inducer of inflammatory processes needs to be revisited and evaluated on a case-by-case basis.
Lethal ROS Concentrations and Triggering of Apoptotic Pathways
When at high concentration or due to long-term incubation, ROS can further oxidize Cys residues into sulfinic (–SO2H) and sulfonic (–SO3H) acids, modifications that are generally irreversible. Furthermore, when two or more cysteines in the same protein are oxidized, they may form intramolecular disulfide bonds that alter both protein structure and activity, triggering cytotoxic effects.115,116 Long-term exposure to ROS can also disrupt the mitochondrial membrane potential, leading to the release of apoptogenic factors.117,118 Yet mechanisms underlying ROS cytotoxicity are still not fully understood, with a high intercellular variability observed. For instance, in human chondrocytes, while sublethal hydrogen peroxide concentrations (0.5 and 1 µM) activated JNK2, on the contrary, lethal concentrations (higher than 10 µM) decreased its activity. 52 Conversely, in hepatocytes, low hydrogen peroxide concentrations (5–10 μM) stimulated cell proliferation through Akt, whereas high concentrations (20 μM) decreased Akt activation and instead activated JNK. 119 In human uterosacral ligament fibroblasts, sublethal doses (200 and 400 µM) induced collagen synthesis through TGF-β, whereas high doses reduced TGF-β signaling and led to tissue degeneration. 6 The triggering of apoptotic cascades generally involved p21, a cyclin-dependent kinase inhibitor, mediator of p53-dependent cell cycle arrest, and inducer of G1/S and G2/M phase arrest. ROS-mediated upregulation of p5328,41,120 and p2128,45,55,121–124 was indeed observed in several cell types, even only 2 h after administration. 122 Interestingly, a study using rat mouse gingival fibroblasts showed that treatment with 20 µM hydrogen peroxide increased cell proliferation and phosphorylation of the p53 at the same time. 28 This may suggest that the activation of p53 may only belong to physiological cellular defense mechanisms against an unwanted risk of oncogenic alterations, which would normally be activated in highly proliferating cells.
ROS in Stem Cell Biology and Tissue Engineering
A growing body of evidence recently pointed out that oxidative stress regulated stem cell multipotency and differentiation. MSCs are physiologically subjected to lower oxidative stress than adult cells, as they possess less mitochondria125,126 and tend to utilize glycolysis instead of mitochondrial oxidative phosphorylation.
127
Even though at a low level, oxidative stress is required to maintain their stemness, in terms of Sox-2, Oct-4, Nanog expression,34,128,129 and colony-forming ability,
130
and probably acts by activating Notch signaling.
131
Physiologically relevant oxidative stress was also required by MSCs to exit from quiescence,
125
whereas hydrogen peroxide preconditioning increased their in vivo regenerative capability.4,75 In mouse and human airway basal stem cells, it was described as a negative feedback loop, in which ROS themselves activated an antioxidant machinery to prevent oxidative damage.
131
Stem cell behavior changed depending on the cells’ origin; for instance, ADSCs were more sensitive to oxidative damage compared with Wharton’s jelly-derived MSCs,
122
while MSCs from the maternal decidua basalis tissue (DBMSCs)—as they normally are open to high levels of inflammation and other stressful conditions—were resistant to concentrations of hydrogen peroxide as high as 200 μM,
130
which are normally toxic for other MSCs.26,122 Low-level oxidative stress was furthermore shown to drive stem cell fate. Not surprisingly, if considering the crosstalk between insulin and H2O2, perturbation in cellular redox homeostasis was observed during physiological adipogenesis, which was characterized by an initial increase in ROS,132–135 followed by an increase in antioxidant enzymes such as SOD2, CAT, and GPx.134,135 Exogenous hydrogen peroxide increased adipogenic differentiation of ADSCs,133,134 BMSCs,
136
and murine precursor cells,
137
via activation of the peroxisome proliferator-activated receptor gamma (PPARγ). On the contrary, the antioxidant molecule N-acetyl cysteine (NAC) inhibited adipocyte differentiation.133,134,136 The ROS mechanism of action involved an increase in cell clonal expansion, as judged by the fact that during adipogenic differentiation, ROS activated the CCAAT enhancer-binding protein beta (C/EBPβ), a transcription factor promoting cell cycle progression from S to G2/M, while antioxidants caused an S-phase arrest.
137
Another mechanism of action involved the FoxO proteins, which are known mediators of adipogenesis and, under oxidative stress, are phosphorylated and activated by the JNK kinase.138,139 Nox4 overexpression and exogenous ROS administration indeed induced adipogenic differentiation of human ADSCs by activating the protein FoxO1.
134
On the other hand, while the deacetylase sirtuin1 (SIRT1) physiologically maintained low expression of FoxO3, the addition of exogenous H2O2 decreased Sirt1 activity, thereby increasing FoxO3 transcription and eventually facilitating the adipogenic differentiation of BMSCs (
The capability of ROS to finely regulate stem cell differentiation is attractive for tissue engineering applications. For instance, cold atmospheric plasma stimulation techniques (which generate ROS) were recently used to stimulate in vitro neurogenesis,159,160 whereas different biomaterials releasing ROS are under development.161,162 Yet, further investigation is certainly needed, especially with respect to their potential side effects. In this context, it is indeed worth mentioning that, for instance, despite at physiologically relevant concentrations, cytotoxic effects have also been reported during ROS-driven chondrogenic differentiation. 152
Modality of ROS Administration and ROS-Releasing Biomaterials
While potential applications of ROS in stem cell biology and tissue engineering appear obvious, more dubious is their modality of administration. While ROS include several members, hydrogen peroxide has been the most (or almost the only one) investigated, due to its ease of availability, lower reactivity, and higher stability compared with the other members. Only a few studies have shown that superoxide also exerts increased proliferation of fibroblasts,163,164 leukemia cells, 165 epithelial cells,166,167 and vascular smooth muscle cells. 168 One could think that the superoxide acts by stimulating hydrogen peroxide production through CAT, yet a study where xanthine and hypoxanthine oxidase were exogenously added into fibroblast cultures to produce superoxide showed that co-incubation with SOD, but not with CAT, inhibited superoxide-induced cell proliferation. This suggests that the mechanism of action was independent of hydrogen peroxide formation. 163 The addition of the nitric oxide (NO) donors (Z)-1[N-(2-aminoethyl)-N-(2-ammonioethyl)amino]diazen-1-ium-1,2-diolate (DETA-NONOate) and sodium nitroprusside110,169–171 was also used to stimulate NO production—which belongs to reactive nitrogen species yet acts similarly to ROS. Nevertheless, while simple and convenient, due to their instability, the exogenous administration of ROS appears infeasible for tissue regeneration purposes. For instance, 100 µM H2O2 was shown to decay during the first 30–60 min after administration to cells,172,173 meaning that it would need to be repeatedly supplied. Furthermore, the administration of a bolus of ROS results in an initial supraphysiological dose, which may possess a cytotoxic effect. Indeed, a study observed that, despite a similar intracellular oxidative level, the number of apoptotic cells and caspase activation rate only increased if Jurkat cells were exposed to 150 µM hydrogen peroxide, but not to an enzymatic system producing 2 µM/min. 174 Among alternative methods, cell transfection with the Nox enzymes has been used (e.g., the long form of Nox5 [NOX5-L] 54 or Nox1 175 ). Nevertheless, although the sole known function of Nox is to produce ROS, a different biological effect was observed when cells were transfected with different Nox isoforms, with Nox2 activating p38 and Nox4 activating JNK and Erk. 176 Thus, a better understanding of the mechanisms of action of different isoforms is needed. The administration of glucose oxidase—which reduces one molecule of atmospheric oxygen into hydrogen peroxide while oxidizing a molecule of glucose—is also under investigation. 142 A steady-state delivery system in which the desired H2O2 concentration was initially reached by the addition of exogenous H2O2 and then maintained by further addition of glucose oxidase was also developed and allowed to better control the final hydrogen peroxide concentration. 177 On the other hand, biomaterial-releasing ROS have been emerging at the forefront of tissue engineering research, with, for instance, solid peroxide scaffolds (e.g., containing sodium percarbonate, magnesium peroxide, or calcium peroxide), 178 hydrogen peroxide-releasing greige cotton nonwoven fibers, 179 and glucose oxidase-conjugated hydrogels 180 being recently developed. Compared with other delivery systems, three-dimensional scaffolds would better suit the needs of tissue engineering, offering support for cells to grow and increasing localization at the transplantation site. Yet, further comparison of the biological effects of different ROS (hydrogen peroxide, superoxide, or even reactive nitrogen species) and their side effects is still needed. Furthermore, the risk of an unwanted tumorigenic potential—associated with systems enabling long-term uncontrolled ROS release—needs to be carefully evaluated.
Oxidative Stress in Cancer
Not surprisingly, if considering the ROS proliferative effect, oxidative stress plays a pivotal role in tumor initiation and progression. Cancer cells are characterized by increased intracellular and extracellular ROS production. 181 The former is attributed to mitochondrial dysfunction and the instability of electron transport reactions, 182 whereas the latter is attributed to the overexpression of ROS-producing enzymes. Upregulation of the Nox was indeed reported in several cancer types, including stomach, 183 colon 184 adenocarcinomas, and gastric cancers; 185 carcinomas of the head and neck, esophagus, bladder, ovary and prostate, and melanoma; 186 and gallbladder cancers, 187 and a gradual increase in extracellular ROS generation was observed during polymorphonuclear leukocyte cancer progression. 188 Furthermore, cancer-associated fibroblasts (CAFs) (i.e., a heterogeneous population of cells responsible for remodeling the extracellular matrix [ECM] while inhibiting both innate and adaptive antitumor immune responses 106 ), compared with matched normal fibroblasts, showed higher hydrogen peroxide production and secretion into the culture media. 189 Much evidence has suggested a role of ROS in tumorigenic transformation; for instance, human immortalized fibroblasts with a deficit in their mitochondrial respiratory chain (generated by silencing mitochondrial transcription factor A) overproduced hydrogen peroxide and showed an increased proliferative rate and tumor-forming capability in xenograft models. Furthermore, they loss the expression of caveolin 1; such a change is typically associated with early breast tumor recurrence, metastasis, and treatment failure. 189 Normal human keratinocytes and fibroblasts, when exposed to exogenous hydrogen peroxide in vitro, acquired characteristics similar to the ones of tumoral cells. 189 Overexpression of Nox1175,190 increased cell growth and tumorigenicity and induced morphological alterations and anchorage-independent growth of the 3T3 murine fibroblasts. Interestingly, such changes were observed when extracellular superoxide concentration doubled, 175 suggesting that even a relatively low oxidative stress is sufficient to achieve cell transformation. CAFs, when co-cultured with human oral cancer cells, enhanced proliferation, migration, and invasion of the latter, by stimulating endogenous ROS production. 191
Possibly due to their higher basal levels, cancer cells appeared more sensitive than normal cells to exogenous oxidative damage. One study using 10 normal human cell lines and 15 human cancer cell lines showed that the former were indeed more capable of metabolizing exogenous hydrogen peroxide, due to a higher CAT activity. Thus, treatment with ascorbate induced cytotoxicity, mediated by DNA damage and ATP depletion, in tumoral cells but not in healthy ones. 192 Similarly, another study comparing 10 cancer and 4 normal cell lines found that the former were more sensitive to ascorbate-induced cell apoptosis. It also described a difference between tissue origins, with the lymphoma cell line, for instance, being more sensitive than breast and lung cancer ones. 193 Furthermore, while overexpression of the enzyme Nox5 induced growth of the normal fibroblast and epithelial cell lines WI-38 and MCF 10A, it inhibited proliferation of the tumorigenic cell lines G- 61 (skin malignant melanoma), SK-BR-3 (breast adenocarcinoma), and HOP-92 (lung carcinoma). 54 In another study, hydrogen peroxide (from 5 to 100 µM) induced the death of HL-60human myeloid leukemia cells but not of normal primary murine bone marrow or human primary human umbilical cord cells. Furthermore, concentrations higher than 5 µM abolished myeloid cell colony formation in HL-60 but not in normal cells. 194 Interestingly, endogenous ROS levels also appeared predictive of cancer cell sensitivity to chemotherapeutic drugs. 182
ROS Therapeutic Delivery into Cancer Cells
The discovery of the high oxidative status of cancer cells paved the way to the development of H2O2-responsive anticancer prodrugs.195,196 On the other hand, exploiting the high susceptibility of cancer cells, various drugs inducing oxidative stress are under development. In this context, ascorbic acid has been largely used197–199 and has even reached clinical experimentation ( Table 2 ). Although it is a well-known antioxidant, when at high concentrations, it generates hydrogen peroxide by autooxidation. 200 Yet, it is generally systematically administered, lacking specificity to cancer cells. Even a simple hydrogen peroxide solution has been proposed as a neoadjuvant in surgical excision, but it is only suitable for skin cancers. 201 Alternative systems to increase drug selectivity and reduce side effects are under development. For instance, nanodrugs producing hydrogen peroxide and releasing Fe3+, to convert it into hydroxyl radicals 202 or Fe nanoparticles that simply accelerate endogenous hydrogen peroxide conversion into hydroxyl radicals via the Fenton reaction, 203 were developed. In particular, the latter decreased the in vitro viability of the HeLa cancer cell line to 22%, whereas it maintained the viability of the healthy cell line NIH-3T3 at levels higher than 80%. Also, silicon dioxide nanoparticles induced in vitro cancer cell apoptosis via ROS generation, yet they lack cancer specificity. 204 One study pointed out that even cold atmospheric plasma stimulation (i.e., ionized gas, which recently found application in cancer therapy) owes its cancer selectivity to ROS. Indeed, this treatment increased ROS synthesis and consequently caspase activation in the cancer cell line A543, but not in the healthy BEAS-2B cell line. 205 Yet, this selective mechanism needs to be confirmed in a larger number of cell lines. Some ROS delivery systems have already reached preclinical investigation. For instance, camptothecin (CPT) is an inhibitor of cellular respiration and increases mitochondrial ROS synthesis. A study developed an ROS-responsive CPT prodrug that, when delivered to cancer cells, was activated by the high endogenous ROS levels. The active CPT in turn further stimulated significant mitochondrial ROS generation, eventually leading to cell death. Cancer specificity was further enhanced by conjugating the CPT prodrug with an integrin-targeting moiety. In vivo drug efficacy and cancer specificity were demonstrated in mammary carcinoma 4T1-bearing mice. 206 Similarly, dihydrolipoamide (DLDH) is an enzyme that produces ROS. A bioengineered DLDH with RGD motifs (targeting the membrane integrins), when subcutaneously injected into tumor-bearing mice, showed a pharmacological safety profile and effectively reduced the tumor size. 207 To further control in vivo localization, nanoparticles targeting the mitochondria through pyruvate and releasing ROS only when exposed to a near-infrared (NIR) laser were developed and tested in a murine xenograft model. Interestingly, the high levels of ROS produced were even able to temporarily inactivate the adenosine triphosphate (ATP)-driven transmembrane efflux pumps, which are responsible for cancer multidrug-resistant phenotypes. 208
Ongoing Clinical Trials Investigating ROS-Based Drugs for Cancer Treatment and Tissue Engineering Purposes.
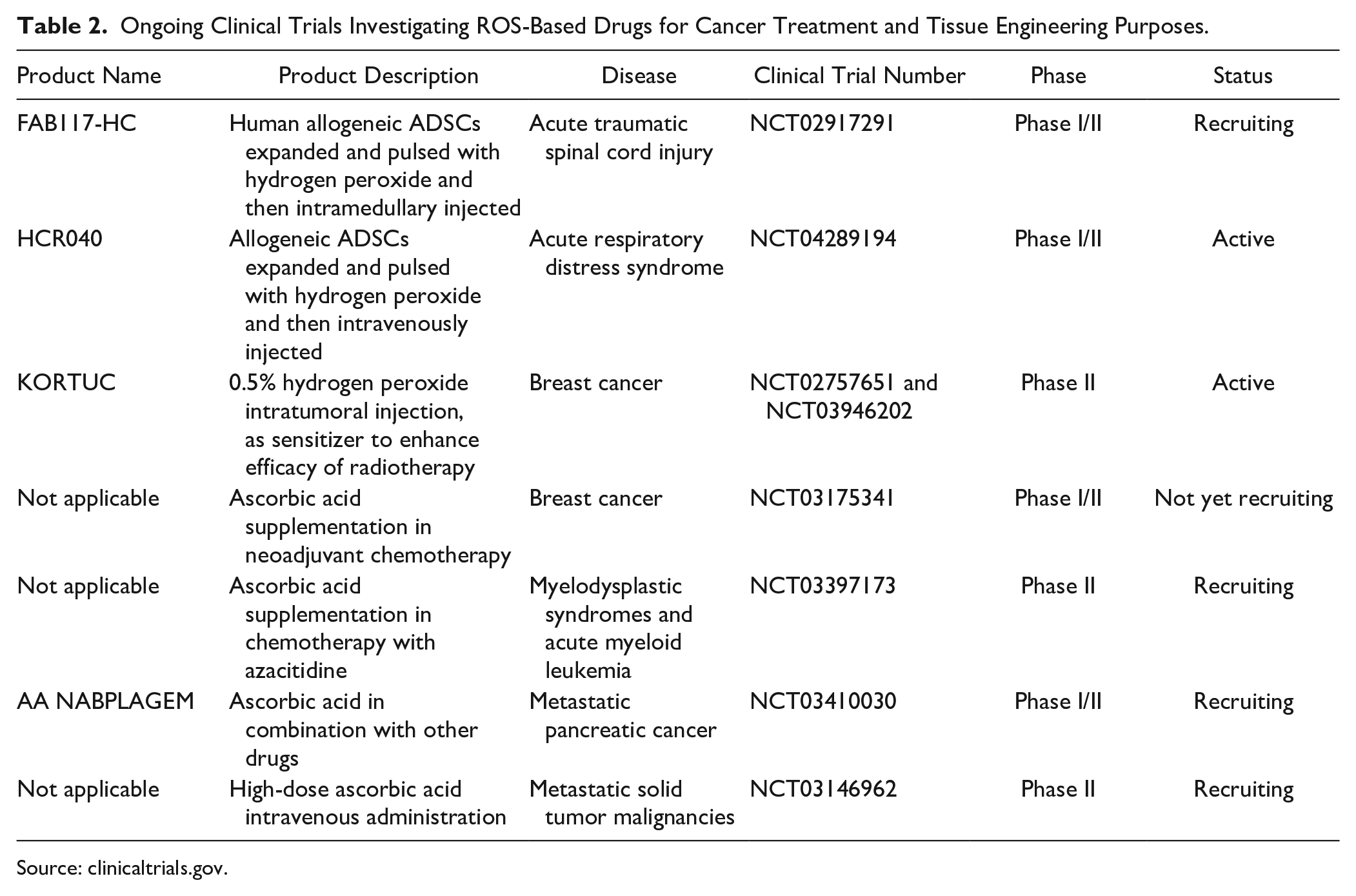
Source: clinicaltrials.gov.
Yet, ROS-based drugs for cancer therapy are still in their infancy, with limited available preclinical studies and research comparing their efficacy with one of the already established drugs. Furthermore, it is still not clear whether all or only certain types of cancers are sensitive to such drugs, whereas delivery systems still need to be optimized. Thus, only more investigation in this direction and time will tell whether they can represent an ultimate cure.
Sensors to Monitor ROS in Cancer Cells
Redox sensors can potentially be used for a real-time monitoring of cancer progression and chemotherapy efficacy, or even for developing personalized treatments. Traditional methods are based on fluorescence, using probes that are oxidized into their fluorescent forms by ROS. For instance, MitoSOX Red, 2,7-dichlorodihydrofluorescein diacetate (DCFDA), 209 hydroethidine (HET), and Amplex Red probes 210 have been successfully used to monitor extracellular and intracellular ROS production in in vitro cultures of breast and pancreatic cancer cells, using a plate spectrofluorometer or fluorescence-activated cell sorting (FACS) analysis, respectively. Alternatively, modified GFP proteins such as the redox-sensitive green fluorescent proteins (roGFPs) or “HyPer sensor” can also be expressed in transgenic animals. Yet, until now, they have only been used for high-throughput in vitro drug screenings,211–213 as their use in in vivo deep layers of tumors appears technically difficult. 214 The roGFP possesses two cysteine residues forming an intramolecular bond and detects changes not only in ROS concentration, but also in the GSSG/GSH ratio. The HyPer sensor was instead generated by introducing the hydrogen peroxide-sensing domain of the prokaryotic transcription factor OxyR in a modified yellow fluorescent protein, 215 yet it is not ratiometric, rendering quantitative analysis difficult. 216 While the drawbacks of fluorescent techniques are the risk of signal artifacts, the presence of background noise, and the impossibility to obtain single-cell resolution, one study showed that fluorescence lifetime imaging microscopy (FLIM) (i.e., a microscopy technique that measures the lifetime of a fluorophore, rather than its intensity) of CellROX Green fluorescence dye-stained cells could overcome these limitations. 217 Alternative systems are based instead on Förster resonance energy transfer (FRET) and contain an energy donor and an energy acceptor, separated by an ROS-sensitive domain, whose conformational change modulates the emission of fluorescence. 215 A FRET-based sensor combining roGFP as donor and a red fluorescent protein (RFP) as acceptor was used to measure mitochondrial ROS dynamics in neuroblastoma cells exposed to various metabolic stresses. When coupled with a cytosolic roGFP protein, it further enabled simultaneous monitoring of cytosol and mitochondria redox dynamics. 216 On the other hand, few studies used chemiluminescence systems, in which ROS react to probes such as lucigenin, L-012, or luminol, to generate light. While these systems generally show difficulty in quantifying the low level of ROS produced by most healthy cells, 218 they are suitable for cancer cells and can even be used in vivo. For instance, a luciferin/luciferase reporter system was developed to detect endogenous H2O2 production in a murine model of prostate cancer and to assess the effect of testosterone in cancer ROS production. 219
Most recent approaches rely on the use of electrochemical biosensors, which consist of enzymes specific to hydrogen peroxide (such as horseradish peroxidase or SOD) or metallic probes conjugated to different materials; they measure the current produced by the reduction of the hydrogen peroxide, showing high sensitivity. 220 For instance, an electrochemical biosensor measuring zinc oxide production by the human hepatoma cell line HepG2—cultured in media supplemented with reduced zinc—was able to monitor, even from a single-cell level, endogenous ROS levels. 221 Similarly, biosensors formed by poly(ethylene glycol) hydrogels, conjugated with ferrocene and SOD, were used to monitor superoxide production in prostate cancer cells. 222 By using platinized silicon carbide nanowire electrodes, a study determined that an ovarian carcinoma cell line not only had higher ROS basal levels, but also was more sensitive to further paclitaxel-induced ROS generation, compared with the healthy cell line 3T3. 223 Nanoelectrodes already reached preclinical experimentation: when inserted in tumor-bearing mice, they allowed the real-time measurement of cancer ROS production, enabling estimation of the modality of action and efficacy (in terms of ROS production) of different chemotherapy drugs. 214
The possibility of real-time ROS monitoring to estimate chemotherapy efficacy is certainly fascinating, yet the complexity of available methods (especially with respect to electrochemical sensors), their limited biocompatibility, the ability to penetrate tissues, and the difficulty in measuring signal from in vivo deep tissues (especially in the case of fluorescent probes) still render them unsuitable for clinical applications. Furthermore, it still needs to be confirmed if and under what conditions cancer ROS production effectively correlates with chemotherapy efficacy.
Future Perspectives and Conclusion
In the past decades, ROS have been emerging as potential therapeutic products, finding application in a wide variety of areas, which varied from wound healing 4 and hair growth enhancement 224 to cancer treatment, stem cell differentiation, and tissue engineering. Yet despite significant advances, several challenges need to be faced in ROS clinical translation. First, there is a scarce understanding of their cytotoxic mechanisms. While several ROS mechanisms of action have been widely investigated, only few studies have investigated the mechanisms underlying the switch toward a cytotoxic effect, and their results only emphasized a high intercellular variability. For instance, while in chondrocytes, low—but not high—oxidative stress activated JNK kinase; 52 in hepatocytes, JNK activation was instead associated with the response to lethal doses of ROS. 119 Furthermore, while Erk1/2 was generally associated with the triggering of cell growth and proliferation, it mediated ROS induction of an apoptotic cascade in epithelial cells. 55 The effect of the external environment is also widely underestimated. In this context, only few studies exposed diseased cells (e.g., from diabetes, Alzheimer’s disease, or cancer-affected patients) to ROS, and they clearly indicated how results from healthy cells are difficult to generalize to diseased ones. A better understanding of cellular intrinsic and extrinsic variability is needed to develop strategies to drive the cell response toward the desired signaling route.
Another crucial aspect concerns ROS delivery methods. There is a lack of preclinical experimentation, and moving from in vitro to in vivo, the quest for an appropriate delivery system is fundamental. Despite the infeasibility of ROS bolus administration, alternative delivery systems have not been compared yet in terms of length, dosage, and ROS species released or potential side effects. Although their sole known role is to produce superoxide, the overexpression of different Nox isoforms did not result in activation of the same signaling pathway; 176 this indicates that different ROS modalities of administration do not necessarily lead to the same effect. It is also noteworthy that an excessive and long-term ROS release possesses a potential tumorigenic effect. Thus, understanding how long cells can be safely exposed to ROS and developing strategies to control the pattern of temporal production may be beneficial. In the case of Nox overexpression, a transient transfection system or the use of induced promoters may be considered. In the case of solid scaffolds, the kinetic profile of ROS release should be carefully assessed.
In conclusion, ROS research is an extremely active field. By finely controlling stem cell differentiation and tipping the balance of adult cells toward survival or death, it holds the potential to treat several diseases. Nevertheless, only advances in understanding the mechanisms of action, potential side effects, and development of new systems to control the temporal pattern of ROS production will enable their use in several fields of biology, from cancer treatment to tissue engineering applications, wound healing, and developmental biology research studies.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
