Abstract
The color of the teeth is an important topic for many people and can be influenced by intrinsic and extrinsic stains. There is an increasing demand for whitening of the teeth year by year. The most popular way of whitening is “bleaching,” which is the result of the breakdown of pigments located in the enamel and/or the dentin, caused by reactive oxygen species (ROS) released from bleaching agents. These bleaching agents could increase matrix metalloproteinase (MMP)-mediated collagen degradation in dentin. The aim of this study was to compare biochemical changes and oxidative stress levels of the human premolar dentin–pulp complex after three different bleaching methods containing hydrogen peroxide (H2O2) bleaching agents. Individuals, whose first four premolars were extracted for orthodontic purposes, included into the study. Group 1—Laser: bleaching gel containing 46% H2O2 (LaserWhite20 whitening gel, Biolase Technology Inc., San Clemente, CA, USA) and a diode laser activation (Ezlase 940 nm system). Group 2: 35% H2O2 containing whitening gel (Whiteness HP Maxx, FGM) and halogen light source activation (Optilux 501, Kerr, Orange, CA, USA). Group 3: 35% H2O2 containing whitening gel (Whiteness HP Maxx, FGM). Group 4—Control: No whitening treatment. According to the test results, there were no significant differences among groups in the values of cathepsin B and MMP proteolytic activities (p > 0.05). The total ROS values released from the dentin tissue were higher than those obtained from the pulp tissue (p < 0.05). There were significant differences among the bleaching groups in the ROS values released from the dentin tissue.
Introduction
Tooth color is influenced by a combination of their intrinsic color and the presence of any extrinsic stains on the tooth surface. 1 Extrinsic tooth stains usually result from the colored components of various foods and beverages, including coffee, tea, red wine, and tobacco products. 2 Also, intrinsic tooth stains such as aging, tetracycline, intrapulpal hemorrhage, calcific metamorphosis, pulp necrosis, and certain diseases or tooth defects can cause to teeth discoloration. 2 Color producing stains within tooth structures are often organic compounds that contain conjugated double bonds. It is known from dye chemistry that decoloration can occur due to the breakup of a chromophore, and that destruction of one or more of the double bonds within the conjugated system is probably involved. 3
There are various methods and techniques in the literature have been used to remove tooth-surface stains to achieve a whitening effect. In general, peroxide-based bleaching products and abrasive materials are used to remove extrinsic and intrinsic tooth stains. 2 Vital tooth bleaching therapies involve home- or office-type treatments. In-office bleaching, the bleaching gel is applied by the dentist during a clinical session and may be activated by a light source. However, in home bleaching, the patients use the trays and gels according to the dentist’s descriptions. 4 The use of hydrogen peroxide (H2O2) solution to remove tooth stains can be traced back more than a century. 5 H2O2 can penetrate dentin, where it releases oxygen and thereby breaks the double bonds of the organic and inorganic compounds inside the dentinal tubules. 6 Dental bleaching depends on the penetration of H2O2-free radicals through the enamel and into the dentin, 7 breaking the dentin’s chromogenic molecules down into smaller structures. 8,9 Tooth bleaching is the result of the breakdown of pigments located in the enamel and/or the dentin, which caused by reactive oxygen species (ROS) released from bleaching agents such as hydroxyl radicals (OH−) and singlet oxygen (1O2). 10 The reaction of free oxygen radicals with the colored organic materials found within tooth structures can lead to a reduction in colour. 11
For many years, heat or light has been used to speed up the breakdown of hydrogen peroxide for a faster whitening result. Various devices, including halogen curing lights, light emitting diodes (LEDs), diode lasers, argon lasers, and plasma arc lamps, have been used. 12 To provide a faster and more effective treatment, office bleaching agents have been used in association with an energy source. Absorbance of the energy in the bleaching gel produces heat that accelerates the oxidation reaction in the office bleaching agent. In terms of the effect of heat on office bleaching, it has been reported that a 10°C increase can double the chemical reaction rate. 13
The release of free radicals from the bleaching agents can be facilitated by laser radiation, which leads to a faster whitening process. Furthermore, it has been demonstrated that the laser irradiation could minimize hypersensitivity of the teeth during the bleaching process. 14,15 Laser is therefore a promising coadjutant treatment that can be used in the photodynamic therapy, 16 stimulating cell differentiation, and reduction of inflammation and tissue repair. 17 The biostimulatory effects of low-level laser therapy (LLLT) on pulp cells such as odontoblasts are important, because these cells are responsible for dentin matrix deposition and its mineralization. In addition, odontoblasts are the first pulp cells to be reached by products released from dental materials capable of diffusing through enamel/dentin. 18
When bleaching gels with high concentrations of H2O2, such as those proposed for the in-office bleaching technique, are applied to the tooth surface, a large quantity of H2O2 diffuses rapidly through the enamel and the dentin, causing intense pulpal damage and tooth sensitivity. 19 Although H2O2 is effective in whitening darkened/colored teeth, it has been claimed that its oxidative effect is responsible for histochemical 20 and morphological alterations in the structure of dental tissues. 21 This may explain why the mechanical properties of these tissues are significantly reduced after bleaching treatment. 22 Moreover, in certain circumstances, H2O2 can reach the pulp chamber through dentinal tubules, causing reduction of cell proliferation, metabolism, and viability, 23 as well as the reduction of pulp-reparative capacity 24 and tissue necrosis. 19
It has been reported that bleaching agents could increase matrix metalloproteinase (MMP)-mediated collagen degradation in dentin. 9 Several in vitro studies have established the adverse effects of bleaching agents, such as cytotoxicity and DNA modification. 25,26 According to these studies, the human dentin–pulp complex contains, in addition to MMPs, cysteine proteases, which, in conjunction with MMPs, may play a part in the remodeling/degradation process of the dentin matrix in both sound and caries affected teeth. 27,28
Although several in vitro studies have evaluated the cytotoxicity of bleaching agents to the culture of cells, there is little available data concerning the effects of bleaching agents on the human dentin–pulp complex. 7,29,30 Therefore, the aim of this study is to compare the potential effects of the three different bleaching methods on proteolytic activities (cathepsin B, MMPs) and ROS designation responses of the human premolar dentin–pulp complex.
The null hypotheses to be investigated in this study were as follows: There would be no differences between the proteolytic activities induced by different bleaching methods (the bleaching methods would not be effective on proteolytic activities—cathepsin B, MMPs, and ROS). There would be no differences between the proteolytic activities of the dentin and pulp tissues.
Materials and methods
Study design
The study protocol was reviewed and approved by the Ethics Committee of the University of Gaziosmanpasa, Turkey; the protocol number is 14-KAEK-259 and is in accordance with the Declaration of Helsinki on experimentation involving human subjects. Written informed consent was obtained from the participants. Individuals between the ages of 15 and 25, whose first four premolars were to be extracted for orthodontic reasons, were included. The criteria included a plaque index score of ≤20%, first or second premolars with no gingival recession, caries or restorations, and no history of previous whitening treatment or tobacco use. Also, color shade of premolar teeth to be extracted was A2 or darker. It was aimed to decrease color of teeth one color shade for all experimental groups. A total of 40 premolar teeth were whitened in the study, 20 each from the upper and lower premolars of 10 individuals (n = 10 teeth). A split-mouth method was utilized for the study. The initial color of the premolar teeth was determined using a spectrophotometer (Vita Easyshade, Ivoclar Vivadent AG, Schaan, Liechtenstein). The first bleaching cycles of groups were applied according to the manufacturer’ recommendations. The tooth surface was cleaned from the bleaching gel using a saliva ejector with high-power suction. The second bleaching cycle was immediately repeated in the same session if necessary, until one shade color change was achieved in all groups except the control group. After bleaching, color values were measured again.
Group preparation
The premolar teeth to be extracted received the following tooth whitening treatments:
Group 1—laser: The premolar tooth surface was covered with LaserWhite20 whitening gel (Biolase Technology Inc., San Clemente, CA, USA) containing 46% H2O2. Activation of bleaching gel was performed with a diode laser (940 nm; Epic 10, Biolase Technology Inc., San Clemente, CA, USA). The diode laser was applied according to the manufacturer’s recommendations: continuous mode, 200j, 7 W with 30 s of power. Next, the bleaching gel was removed from the enamel surface using a saliva ejector with high-power suction.
Group 2—whitening gel and halogen light (W + HL): The tooth surface was covered with Whiteness HP Maxx (FGM, Joinville, SC, Brazil) containing 35% H2O2. The bleaching gel was activated with the bleaching mode of the halogen light curing unit (Optilux 501, Kerr, Orange, CA, USA). The bleaching procedure was performed with activations of 30 s each (4 × 30 s, interval 3 min between activations) for a total of 15 min according to the manufacturer’s recommendations. The bleaching gel was removed from the enamel surface using a saliva ejector with high-power suction.
Group 3—whitening gel (W): The tooth surface was covered with Whiteness HP Maxx (FGM) containing 35% H2O2 for 15 min according to the manufacturer’s recommendations. The bleaching gel was then removed, with care taken to avoid overflowing.
Group 4—control: The teeth were extracted without any whitening procedures.
Teeth extraction and sample preparation
The patients were directed to the Oral and Maxillofacial Surgery Clinic 24 h after the bleaching procedures. The teeth were extracted by same surgical dentist with the local anesthesia. After extraction, the teeth were separated from the enamel-cement junction, and the pulp tissue was removed from the pulp chamber. For storage, pulp and dentin tissues were placed in transport nutritive medium (Dulbecco’s modified Eagle medium (DMEM); Life Technologies, Rockville, MD) containing penicillin–streptomycin (Sigma, St Louis, Missouri, USA). The specimens were then frozen at 80°C and were dispatched immediately to the laboratory.
Proteolytic activities in dentin and pulp
The hard tissues of the teeth of the experimental and control groups were frozen at 80°C. Then, dentin tissues were powdered, and pulp tissues were thawed and homogenized. Cathepsin B activation in the dentin powder and pulpal tissues, as well as collagenolytic/gelatinolytic MMP activities, was evaluated spectrofluorometrically as in the Scaffa et al.’s study. 31
Detection of ROS in dentin and pulp
The hard tissues of the teeth of the experimental and control groups were frozen at 80°C. Then, dentin tissues were powdered, and pulp tissues were thawed and homogenized. Total ROS in the dentin and pulp tissues of all groups were determined by spectrofluorometry. The tissues were incubated with 4 mM 2′,7′-dichlorodihydrofluorescein diacetate (DCFDA;Molecular Probes, Eugene, Oregon, USA) fluorogenic probe for 30 min. After incubation, ROS values were quantitated from the supernatant by spectrofluorometry. 29
Statistical analysis
Three-way analysis of variance (ANOVA) was used for comparison between groups. A Bonferroni correction was made for multiple comparisons. p-Value < 0.05 was considered statistically significant. Analyses were performed using SPSS 19 (IBM SPSS Statistics 19, SPSS Inc., an IBM Co., Somers, New York, USA).
Results
The three-way ANOVA results revealed significant differences between the different test methods that were evaluated (cathepsin B, MMPs, and ROS activities (DCFDA); p = 0.000) and the bleaching methods (p = 0.003). There were also significant differences in the response of the dentin–pulp complex to bleaching methods (p = 0.000; Table 1). The test results investigated for the bleaching methods and teeth tissues are shown in Figures 1 to 3 and Tables 2 and 3.
Three-way ANOVA.

ANOVA: analysis of variance.

The bar chart represents the amount of cathepsin B in dentin powders and pulp tissues according to bleaching methods and control group-N (number of samples): 10 teeth, (p < 0.05).
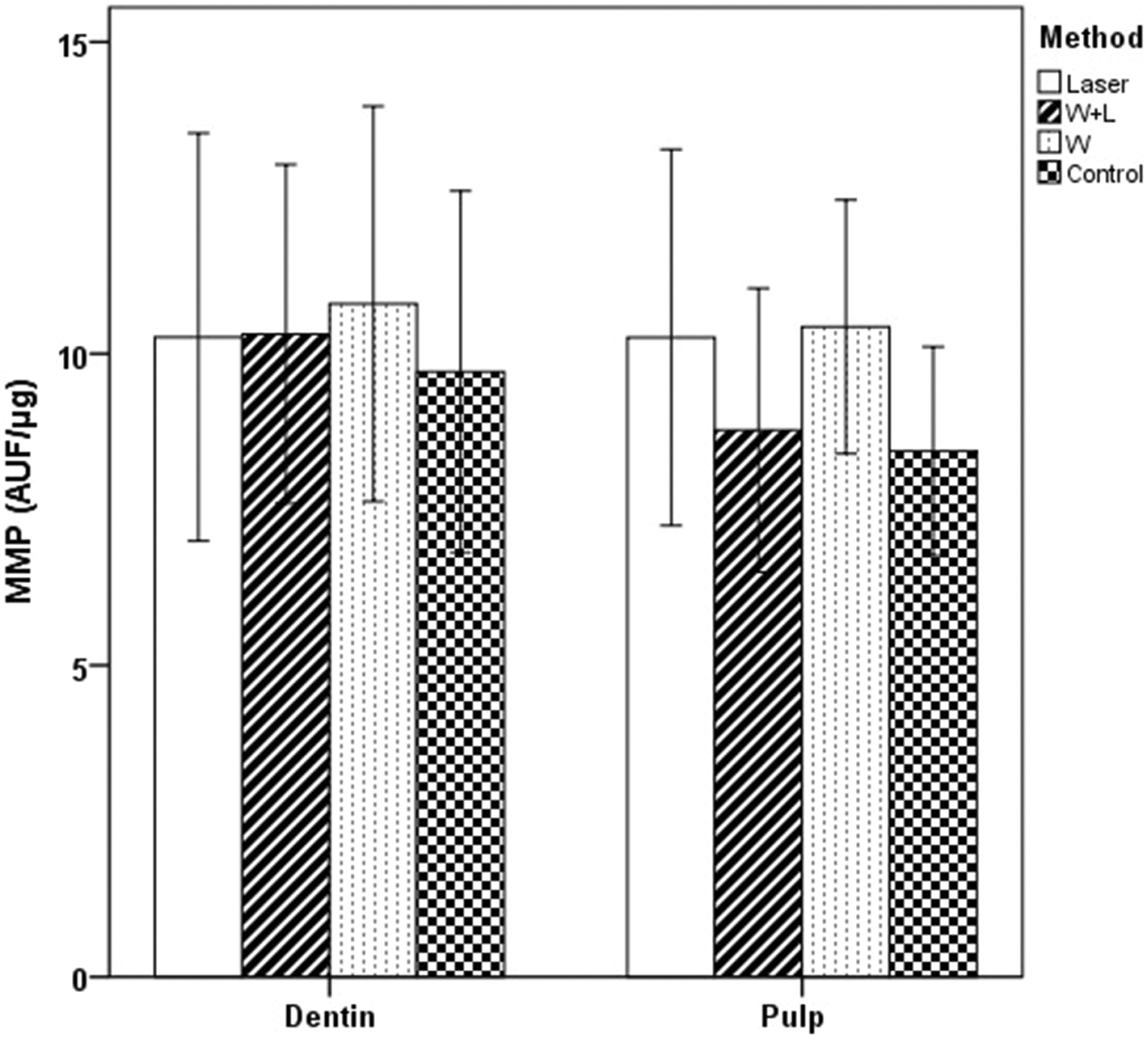
The bar chart represents the MMP activates (degradation unit per microgram of sample; mean with SD) dentin powders and pulp tissues according to bleaching methods-N (number of samples): 10 teeth (p < 0.05). MMP: matrix metalloproteinase.

The bar chart represents the amount of ROS (DCFDA) in dentin powders and pulp tissues according to bleaching methods-N (number of samples): 10 teeth, (p < 0.05). DCFDA: 2′,7′-dichlorodihydrofluorescein diacetate; ROS: reactive oxygen species.
Comparison of bleaching methods in dentin and pulp tissues according to test methods.

ROS: reactive oxygen species; MMP: matrix metalloproteinase; DCFDA: 2′,7′-dichlorodihydrofluorescein diacetate; W + HL: whitening gel and halogen light.
a,bSame letters indicate an insignificant difference among groups. In dentin tissue, for total ROS, there were significant differences between Laser versus W; Laser versus Control; W + HL versus W; W + HL versus Control group (p < 0.05).
Comparison of dentin and pulp tissues according to bleaching methods.
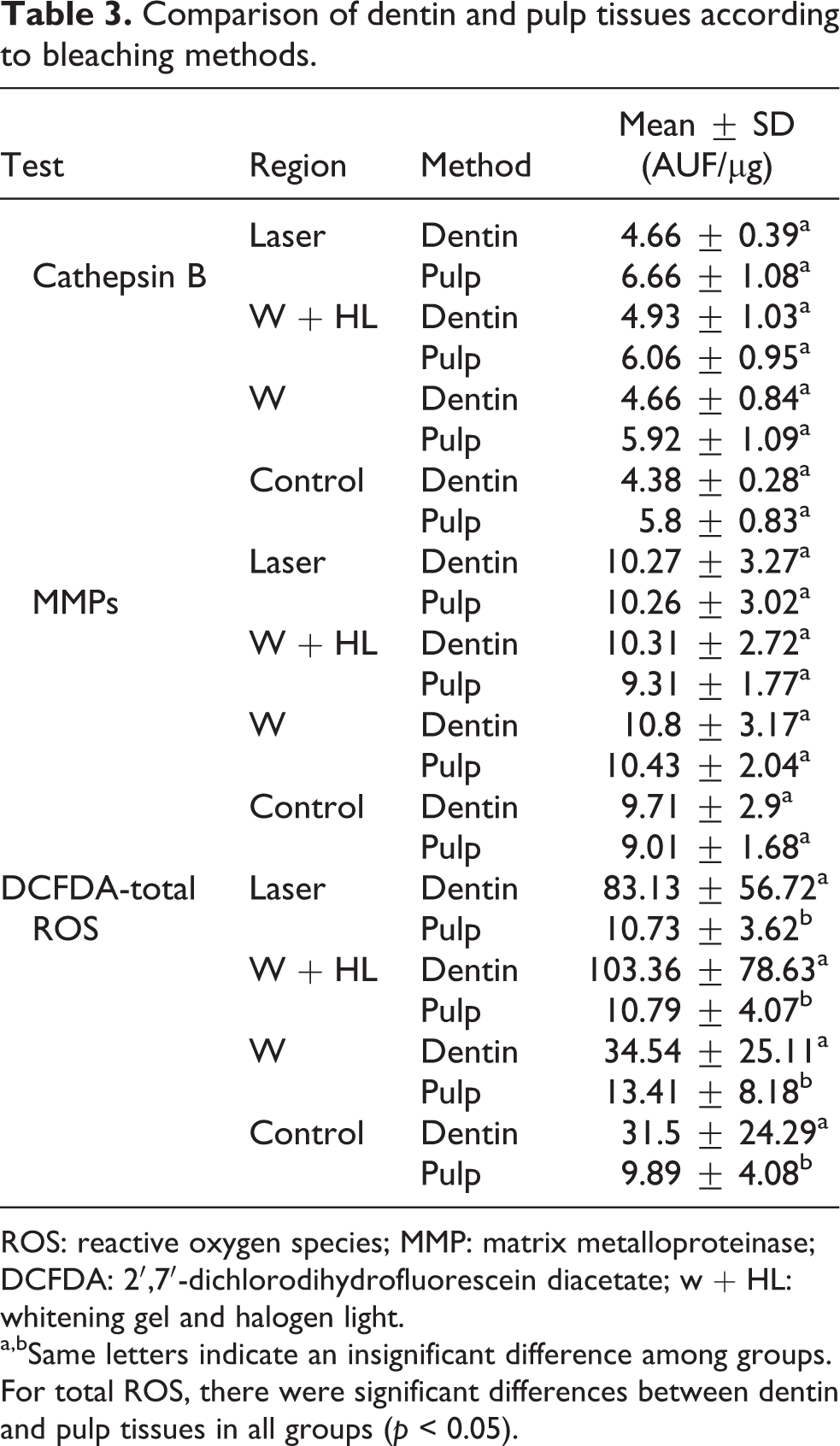
ROS: reactive oxygen species; MMP: matrix metalloproteinase; DCFDA: 2′,7′-dichlorodihydrofluorescein diacetate; w + HL: whitening gel and halogen light.
a,bSame letters indicate an insignificant difference among groups. For total ROS, there were significant differences between dentin and pulp tissues in all groups (p < 0.05).
Cathepsin B test results
According to the test results, there were no significant differences among bleaching groups in the values of cathepsin B released from dentin tissues (p > 0.05). Again similarly, there were no significant differences among bleaching groups in the values of cathepsin B released from pulp tissues (p > 0.05; Table 2 and Figure 1).
In all study groups, including the control group, there were no significant differences between the values of cathepsin B released from dentin and pulp tissues (p > 0.05; Table 3 and Figure 1).
MMP test results
There were no significant differences among bleaching groups in the values of MMP proteolytic activities released from dentin tissues (p > 0.05). As in the results of cathepsin B, there were no significant differences among groups in the amount of MMP released from pulp tissues (p > 0.05; Table 2 and Figure 2).
When all groups were evaluated including the control group, there were no significant differences between the amount of MMP released from dentin and pulp tissues (p > 0.05; Table 3 and Figure 2).
DCFDA (total ROS) test results
There were no significant differences among bleaching groups in the amount of total ROS released from pulp tissues (p > 0.05). However, there were statistically significant differences among study groups in the amount of total ROS released from dentin tissues (p < 0.05; Table 2 and Figure 3). The highest ROS value was obtained in the whitening gel with halogen light (W + HL) group (p < 0.05). Although there was no difference between the control and the W group, the lowest DCFDA value was seen in the control group (Table 2).
The total ROS amount obtained from dentin powder was significantly higher than pulp tissue (p < 0.05; Table 3 and Figure 3).
Discussion
H2O2 is a ROS that is applied widely in dental bleaching, because it has the capacity to dissociate into other ROS with high levels of oxidative activity. 32 However, H2O2, together with its degradation products, can result in damage to the pulp cells, in particular the odontoblasts underlying the dentin. 24 It has been reported that increased ROS levels have deleterious effects on various cell components, including lipid peroxidation, oxidative changes of proteins, and DNA cell damage. 33 In this in vivo study, we compared the potential effects of three different bleaching methods on proteolytic activities (cathepsin B, MMPs) and ROS designation responses of the human premolar dentin–pulp complex. The results of this study did not support the null hypothesis that there would be no differences in the proteolytic activities induced by different bleaching methods, because significant differences were found between the effects of bleaching methods on proteolytic activities (cathepsin B, MMPs) and ROS designation responses of human teeth (p = 0.003). Also, the second null hypothesis that there would be no differences between the proteolytic activities of the dentin and pulp tissues was not supported: significant differences were observed in the response of the dentin–pulp complex to bleaching methods (p = 0.000).
The efficiency of the bleaching agent depends on the release of free radicals such as oxygen (1O2) and hydroxyl (OH−). 13 It is reported that the bleaching agent did not affect cysteine cathepsin or MMP activities in gingival crevicular fluid, suggesting that the procedure does not induce gingival damage. 29 H2O2 can diffuse through the enamel and the dentin, reaching the dental pulp. 34 Vital teeth present dentinal fluid flow produced by intrapulpal pressure and cytoplasmatic prolongations of odontoblasts, as well as other intratubular components, which may impede the diffusion of the bleaching gel components through the dentinal tubules. 19 In the presence of oxidative stress after bleaching, the defense system of the pulp cells is activated, releasing several endogenous antioxidant agents, such as peroxidases and catalases, which promote an enzymatic degradation of H2O2 to avoid excessive tissue damage. 35 Thus, in this in vivo study, we investigated the proteolytic activities (cathepsin B, MMPs) and ROS designation responses in the human premolar dentin–pulp complex after vital bleaching.
The available techniques for vital tooth bleaching differ in terms of the type and concentration of the bleaching agent, the time of application, product presentation, mode of application, and light activation. 19,36 Bleaching reaction is also affected by heat. 12 It has been reported that heat application enhances the penetration of hydrogen peroxide into the tooth pulp. 37 For in-office procedures, the use of curing lights (including halogen curing lights, plasma arches, LED, and lasers) has been recommended to accelerate the action of the bleaching gel. 38 It is assumed that irradiation from a diode laser light source heats the H2O2, thereby accelerating the decomposition of H2O2 and the formation of hydroxyl and oxygen free radicals 39 ; it thus reduces the treatment time and possibly enhances the results. Kashima-Tanaka et al. 40 have shown that the amounts of OH- generated from H2O2 are higher when the peroxide is activated by light sources (plasma arc lamp and halogen lamp) than when it is activated by laser irradiation (He–Ne laser and yellow He–Ne laser). Robertson and Melfi 41 reported that disastrous pulp response in bleached premolars was observed only when the bleaching agent was heated. No pulp damage was reported in the unheated bleaching group. In the current study, we aimed to evaluate the proteolytic activities and ROS designation responses in the human dentin and pulp tissues after the bleaching material was activated by a diode laser and a halogen light curing unit.
After exposure to bleaching agents, it is reported that the loss of collagen contributes to a reduction in the mechanical properties of the dentin. 22 Sato et al. 29 reported that a bleaching agent containing 35% H2O2 induced a significant in vivo alteration in the enamel and the dentin, which could potentially trigger biological and/or mechanical responses of dental structures. Sato et al. also investigated the potential effect of 35% H2O2 on the in vivo activity of dentin cysteine proteases and MMPs after vital bleaching. In their study, the effect of the H2O2 on human pulp tissue was evaluated by measurement of the amount of ROS and by the level of the lysosomal cathepsin B proteolytic enzyme in pulp induced by bleaching. They reported that statistically significant increases in both cathepsin B and ROS were observed in the pulp, and that a statistically significant increase in both cathepsin B and MMP proteolytic activities was observed in the dentin. 29 The acidity of the bleaching agents may trigger the autocatalytic activation of MMPs in the dentin–pulp complex and the stabilization of the cysteine proteases. 27 Vaz et al. 42 evaluated the macrophages migration, mast cell density, inflammation intensity, and pulp damage after the use of two bleaching techniques. Teeth were extracted 7 days after bleaching in their study. They reported that variability in the severity of inflammation factors may be due to the differences in time between bleaching procedures and tooth extraction. 42 Robertson and Melfi 41 reported that 4-day intervals between treatment and extraction are the optimal period to evaluate pulpal reaction. For this in vivo study, all teeth were extracted 24 h after bleaching, except those in the control group. According to our findings, although the values of cathepsin B and MMPs obtained from the pulp and dentin tissues of teeth subjected to bleaching methods were higher than those obtained from the control group, there were no statistically significant differences between the different bleaching methods. These results for cathepsin B and MMPs are valid for both the pulp and the dentin tissue. In this study, unlike our cathepsin B and MMP findings, we obtained total ROS (DCFDA) values in the dentin and pulp tissues. The ROS values obtained from the dentin tissue were statistically significantly higher than those obtained from the pulp tissue. Moreover, there were statistically significant differences among the bleaching groups. The highest ROS value was obtained in W + HL group. The lower ROS value in the pulp tissue may be ascribed to the dentin preventing diffusion of the bleaching agent components.
Although the effects of LLLT on the viability of cells exposed to bleaching agents have been evaluated in several studies, the outcomes are controversial. Dantas et al. 43 indicated that irradiation from a low power 780-nm laser at energy density of 10 J/cm2 was capable of compensating for the cytotoxic effects of 35% H2O2 on human pulp fibroblasts. It was reported that LLLT may be capable of attenuating the damage and inflammation induced by in-office bleaching products in pulp tissue and can thus possibly reduce the risk and intensity of tooth sensitivity arising from bleaching. 44 Furthermore, it was also reported that laser irradiation can modulate cell responses by acting on the mitochondria, increasing the levels of ATP and intracellular calcium. 45 The results of the current in vivo study suggest that the values of cathepsin B, MMPs, and ROS were not reduced as much in the diode laser group as in the other experimental groups. In the present study, the bleaching gel containing 46% H2O2 was activated by the diode laser according to the manufacturer’s recommendations (continuous mode, 200j, 7 W with 30 s of power). There were no significant differences among the experimental bleaching groups. The inadequacy of diode laser in reducing proteolytic activities may be due to the use of 46% H2O2 or to the variable parameters of the laser protocols tested (wavelength, potency, energy density). In Lima et al.’s 46 study, different parameters of low-level laser irradiation (4, 10, and 15 J/cm2) were applied to bio-stimulate ROS-damaged pulp cells. They reported that the laser protocols tested were not capable of influencing the metabolism of previously damaged odontoblast-like cells.
Conclusion
Within the limitations of this study, the following conclusions were obtained:
– When cathepsin B and MMPs were evaluated in the dentin and pulp separately, no differences were observed between the bleaching groups in either tissue.
– The Total ROS values released from the dentin tissue were higher than those obtained from the pulp tissue. Moreover, there were significant differences among the bleaching groups for the dentin tissue.
– Dentin tissue showed more biochemical changes than pulp tissue.
– There is a need for further studies to evaluate different extraction times of the teeth, as well as different laser parameters and H2O2 concentration.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Gaziosmanpasa University Scientific Research Projects Commission (project no: 2015/37).
