Abstract
Previous evidences have indicated that granulosa cells play a critical role in follicular growth. Hydrogen peroxide (H2O2)-induced oxidative stress has been associated with ovarian granulosa cell apoptosis and ovarian function. Recently, a study highlighted the protective role of morroniside against H2O2-induced damage. In this study, we aimed to investigate the effects of morroniside on H2O2-stimulated rat ovarian granulosa cells and its underlying molecular mechanisms. Our results showed that H2O2 treatment suppressed cell survival and increased apoptosis in rat granulosa cells, while treatment with morroniside markedly increased H2O2-induced granulosa cell survival in a dose-dependent manner (0, 10, 50 and 100 µM). Moreover, treatment with 50 µM morroniside impeded H2O2-induced cell apoptosis. An elevation in intracellular ROS, MDA, SOD, GSH-Px, and CAT level was observed in H2O2-induced granulosa cells; however, this effect was abrogated by morroniside treatment. Further studies suggested that administration of morroniside inhibited H2O2-induced granulosa cell apoptosis and caspase-3 activity. In addition, after morroniside treatment of H2O2-stimulated granulosa cells, autophagy-related protein (LC3-II/LC3-I ratio) and beclin-1 expression was decreased and p62 level was increased. Interestingly, we found that morroniside treatment activated the PI3K/AKT/mTOR pathway in H2O2-stimulated granulosa cells. Finally, we showed that treatment with PI3K and mTOR inhibitors reversed the protective effects of morroniside on H2O2-induced granulosa cells. Taken together, our data suggest that treatment with morroniside decreased apoptosis, autophagy, and oxidative stress in rat granulosa cells through the PI3K/AKT/mTOR pathway.
Introduction
As important and complex developmental processes, ovarian follicular growth and maturation play a pivotal role in the reproductive life of females. 1 Dysfunction of granulosa cells, which surround the developing oocytes, results in ovulation problems and ovarian dysfunction. 2 Granulosa cells in the mammalian ovary form follicles, providing a suitable microenvironment for follicle development and oocyte growth. 3 Additionally, previous evidences have suggested that granulosa cells play an important role in regulating oocyte biology, including cell differentiation, genomic transcriptional activity, cytoplasmic maturation, meiosis, and cell growth. 4 Moreover, studies have reported that granulosa cell apoptosis is associated with follicular atresia. 5 A large number of studies have shown that a series of drugs have therapeutic effects on follicular development. 6 However, the effects of morroniside on H2O2-stimulated rat ovarian granulosa cells and its underlying mechanisms have not yet been explored.
Cornus officinalis is a commonly used Chinese herbal medicine and a widely applied vegetable drug in China. 7 Evidence has indicated that this medicine plays a therapeutic role in many injuries, including oxidative damage. 8 Morroniside, extracted from Cornus officinalis, is one of the most abundant iridoid glycosides. Its chemical structure is shown in Figure 1A. 9 A number of studies have shown that morroniside exhibits various bioactivities, such as anti-oxidative, 10,11 facilitating tissue regeneration, 12,13 pro-proliferation, 12,13 anti-inflammatory, 14 anti-apoptotic, 10 and anti-microbial activities in vitro. 15 However, the effects of morroniside on various cellular processes, including cell survival, oxidative stress, apoptosis, and autophagy in H2O2-induced granulosa cells have not been clarified. Thus, in the present study, we aimed to study the effects of morroniside on H2O2-induced granulosa cells.
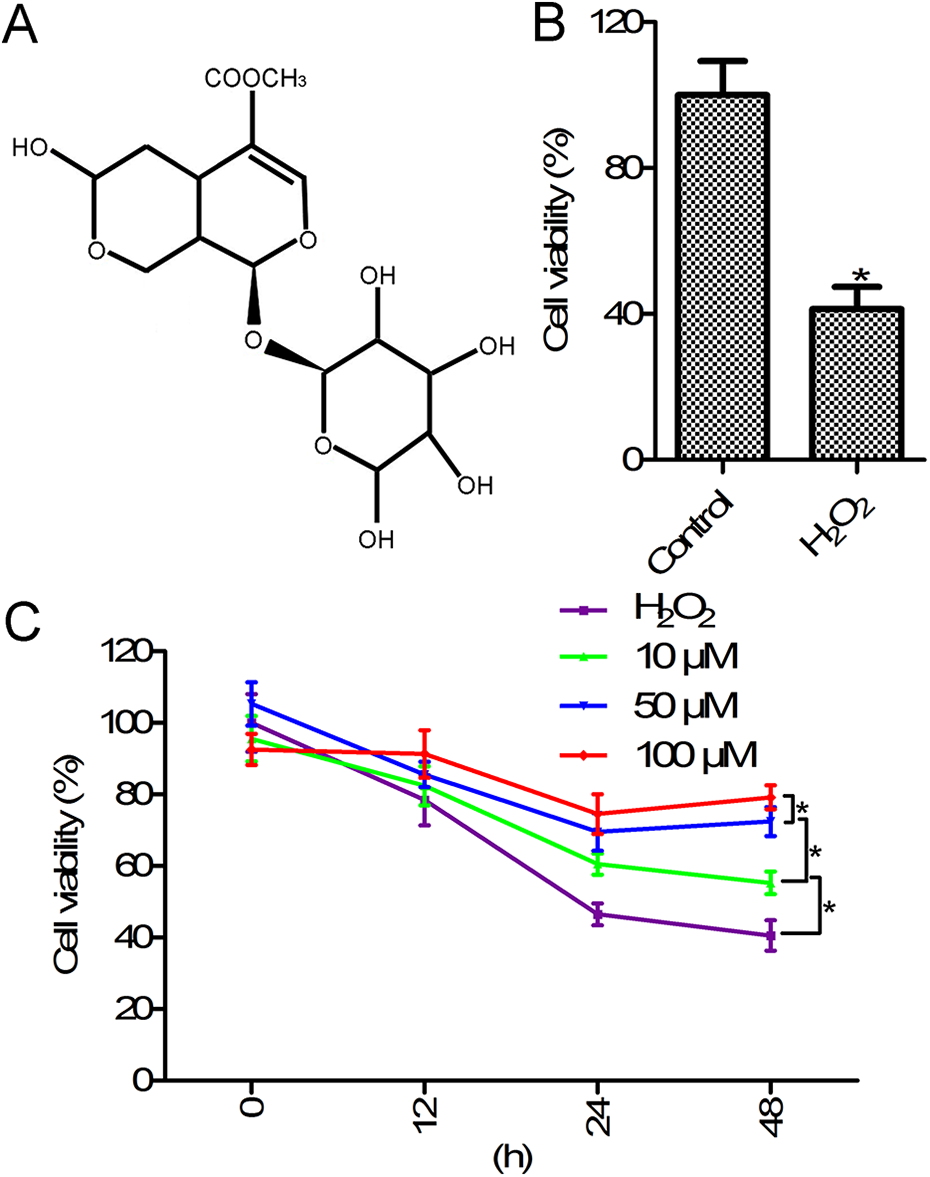
Morroniside increased cell viability in H2O2-stimulated rat ovarian granulosa cells. (A) The chemical structure of morroniside. (B) Cell viability of H2O2-stimulated rat ovarian granulosa cells. (C) Morroniside treatment enhanced the viability of H2O2-stimulated rat ovarian granulosa cells. CCK-8 assay was used to determine cell viability. Rat ovarian granulosa cells were exposed to H2O2 (300 µM) for 24 h, or pretreated with different concentrations of morroniside (0, 10, 50, and 100 µM) for 12, 24, and 48 h, and then exposed to H2O2 (300 µM) for 24 h to induce granulosa cell injury. *p < 0.05 compared with the control group or H2O2 group.
The phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT) pathway has been shown to be involved in cell apoptosis, metabolism, and growth. 16 Moreover, AKT activation was shown to affect oxidative stress-induced cell injury. 17 It is well known that PI3K/AKT are upstream regulators of the mechanistic target of rapamycin (mTOR) signaling pathway. 18,19 Previous evidences have suggested that the evolutionarily conserved serine-threonine kinase, mTOR, plays important roles in the inhibition of autophagy, and is associated with cell growth and proliferation. 16,20 In the present study, we aimed to investigate the role of PI3K/AKT/mTOR signaling in rat ovarian granulosa cell survival and apoptosis. We also investigated the function and underlying mechanism of the natural compound morroniside. Our results demonstrated that morroniside suppressed cell apoptosis and promoted cell survival in H2O2-stimulated rat ovarian granulosa cells.
Methods
Animals
Immature female Sprague-Dawley rats (22-day old) were obtained from Chengdu University of Traditional Chinese Medicine (Shanghai, China). Animals were housed and maintained in a clean room under illumination (12 h light, 12 h dark). All animal experiments performed in this study were approved by the Hospital of Chengdu University of Traditional Chinese Medicine.
Cell isolation and culture
Rat ovarian granulosa cells were isolated as previously described, with some modifications. 21,22 Briefly, follicular development was induced in Sprague-Dawley rats by stimulation with 20 IU pregnant mare’s serum gonadotropin (PMSG; Sigma-Aldrich, St. Louis, MO, USA). After 48 h, rats were sacrificed and the ovaries were removed and placed in ice-cold Dulbecco’s modified Eagle’s medium (DMEM). Primary rat granulosa cells were isolated using a needle puncture under a microscope (40×). Single cell suspensions were filtered and resuspended in DMEM/F12 medium, and cell viability was evaluated by the Cell Counting Kit-8 (CCK-8) assay.
Cell treatment
Rat ovarian granulosa cells were pretreated with different concentrations of morroniside (0, 10, 50, and 100 µM; Phytomarker Ltd., Tianjin, China) for 24 h, followed by treatment with H2O2 (300 µM) for 24 h to induce granulosa cell injury. For molecular mechanism studies, cells were first treated with the PI3K inhibitor LY294002 or the mTOR inhibitor rapamycin, and then administered morroniside for 24 h, followed by stimulation with 300 µM H2O2.
CCK-8 assay
Rat ovarian granulosa cell viability was determined by CCK-8 assay according to the manufacturer’s instructions. Briefly, cells were treated with morroniside for 24 h, followed by treatment with 20 µL CCK-8 solution (5 mg/ml) for another 2 h. The OD value was recorded at 450 nm.
Measurement of ROS, MDA, SOD, GSH-Px, and CAT level
Rat ovarian granulosa cells were cultured in cell culture flasks (25 cm2) and the ROS, MDA, and SOD levels were measured. ROS level was evaluated by the ROS assay kit (Beyotime Biotechnology, Shanghai, China), according to the manufacturer’s instructions. In brief, cells were stained with the fluorescent dye 2′,7′-dichlorofluorescein diacetate (DCFH-DA; 10 μM) for 20 min. The MDA content was assessed using the lipid peroxidation MDA assay kit (Beyotime Biotechnology, Shanghai, China). Superoxide dismutase (SOD), GSH-peroxidase (GSH-Px), and catalase (CAT) activities were assayed by the corresponding assay kits.
Enzyme-linked immunosorbent assay (ELISA)
Cell apoptosis was detected using a cell death detection ELISA kit, following the manufacturer’s instructions.
Caspase-3 activity
Caspase-3 activity in rat ovarian granulosa cells was measured using a commercial caspase-3 activity assay kit, according to the manufacturer’s instructions. Briefly, cells were lysed using lysis buffer and the supernatant was collected. Next, 10 µl Ac-DEVD-pNA (2 mM) was added to the supernatant and incubated at 37°C for 2 h in the dark. The absorbance values were recorded at 405 nm.
Western blotting
Primary rat ovarian granulosa cells were lysed using radioimmunoprecipitation assay buffer. The BCA protein assay kit (Beyotime, China) was used to measure protein concentration. Proteins (30 µg/lane) were separated on 10% sodium dodecyl sulfate-polyacrylamide gels and detected using the following antibodies: rabbit polyclonal phospho (p)-AKT antibody (ab38449; 1:500), rabbit polyclonal AKT antibody (ab8805; 1:500), rabbit polyclonal p-mTOR antibody (ab137133; 1:1000), and rabbit polyclonal mTOR antibody (ab2732; 1:2000).
Statistical analysis
Results are presented as the mean ± standard deviation (SD) of three independent experiments, each performed in triplicate. Statistical analysis was performed by one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test. p < 0.05 was considered statistically significant.
Results
Morroniside improved cell viability in H2O2-stimulated rat ovarian granulosa cells
We observed that rat ovarian granulosa cell viability was significantly inhibited in the H2O2 group (300 µM) compared with that in the control group (Figure 1B, p < 0.05). Next, we investigated the protective effects of morroniside on H2O2-stimulated rat ovarian granulosa cells. As shown in Figure 1C, the viability of H2O2-stimulated rat ovarian granulosa cells treated with different concentrations (10, 50, and 100 µM) of morroniside for different time periods (12, 24, and 48 h) was increased compared to that of cells treated with H2O2 alone. These results indicated that morroniside improved H2O2-stimulated cell viability in rat ovarian granulosa cells in a concentration- and time-dependent manner.
Morroniside inhibited oxidative stress in H2O2-stimulated rat ovarian granulosa cells
To further study the effects of morroniside on oxidative stress in H2O2-stimulated granulosa cells, the levels of ROS, MDA, SOD, GSH-Px, and CAT were measured. As shown in Figure 2, the levels of ROS and MDA levels were increased, while those of SOD, GSH-Px, and CAT were reduced in the H2O2 group compared with those in the control group. Further studies suggested that the expression of ROS and MDA was significantly suppressed, while that of SOD, GSH-Px, and CAT was increased in the H2O2 + morroniside (50 µM) group compared with that in the H2O2 group. These results indicated that morroniside inhibited oxidative stress in H2O2-stimulated granulosa cells.
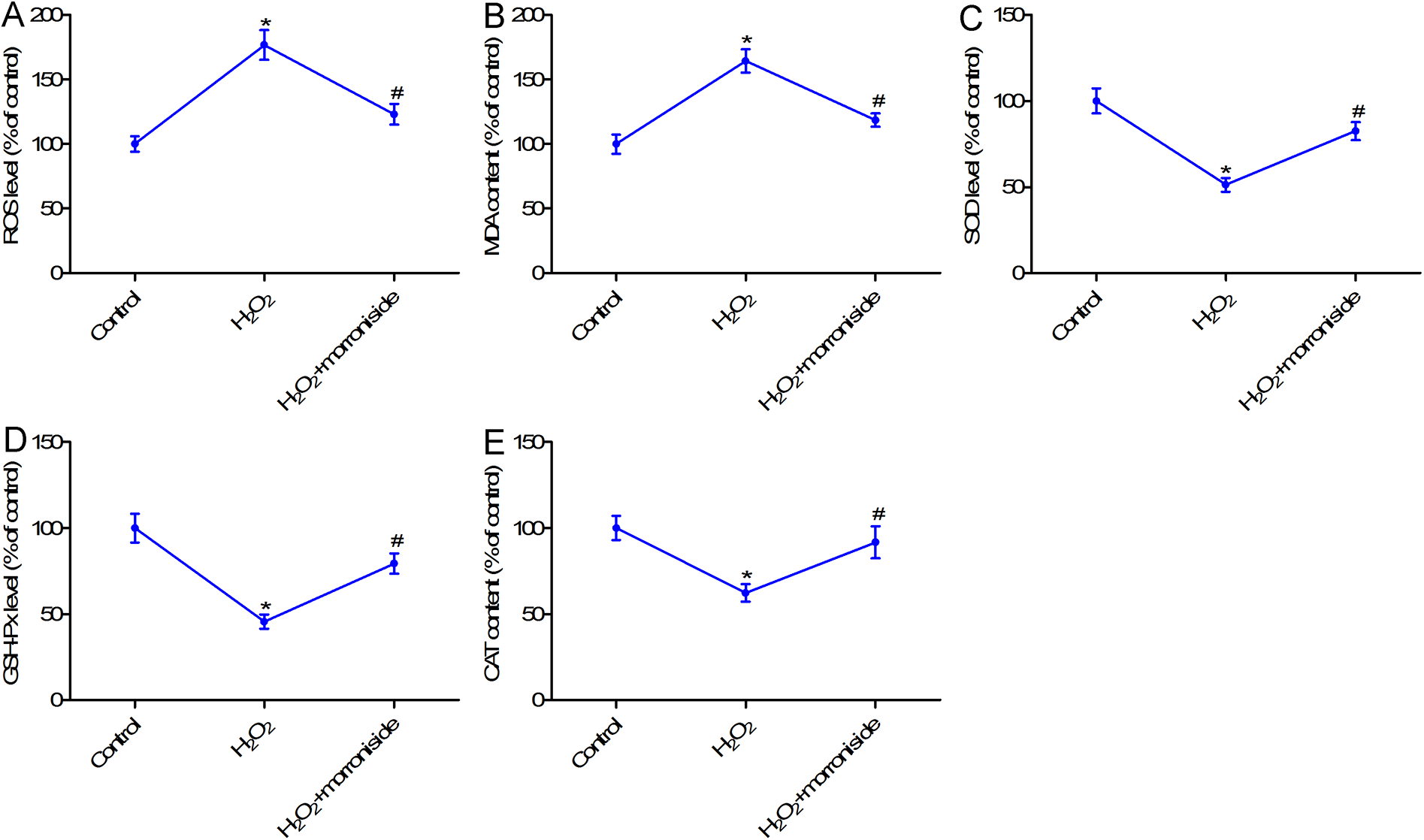
Morroniside inhibited oxidative stress in H2O2-stimulated rat ovarian granulosa cells. (A) ROS content was measured with the ROS assay kit. (B) The MDA level was determined using an MDA assay kit. (C) The level of SOD was measured with the SOD assay kit. (D) The GSH-Px level was measured using an GSH-Px assay kit. (E) The CAT level was assessed using a CAT assay kit. Rat ovarian granulosa cells were exposed to H2O2 (300 µM) for 24 h, or pretreated with 50 µM morroniside for 24 h and then exposed to H2O2 (300 µM) for 24 h to induce granulosa cell injury. *p < 0.05 compared with the control group; #p < 0.05 compared with the H2O2 group.
Morroniside inhibited cell apoptosis in H2O2-stimulated rat ovarian granulosa cells
To further investigate whether morroniside affects cell apoptosis in H2O2-stimulated granulosa cells, cell death detection ELISA assay was performed. As shown in Figure 3A, treatment with 50 µM morroniside markedly impeded H2O2-induced apoptosis in granulosa cells (p < 0.05). To confirm cell apoptosis, caspase-3 activity was evaluated. Results showed that treatment with H2O2 enhanced caspase-3 activity in rat ovarian granulosa cells; however, treatment with morroniside reversed H2O2-induced caspase-3 activity.
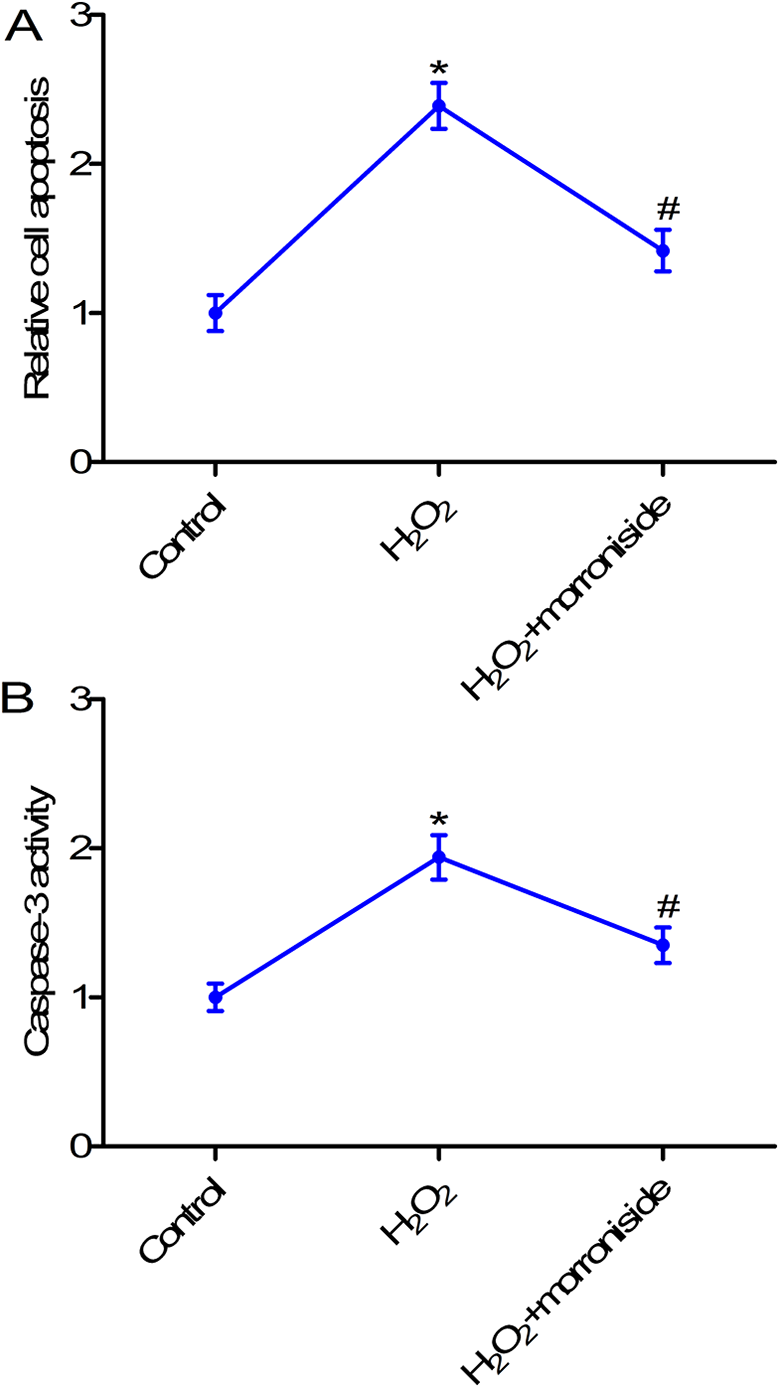
Morroniside inhibited cell apoptosis in H2O2-stimulated rat ovarian granulosa cells. (A) The cell death detection ELISA kit was used to measure cell apoptosis. (B) Caspase-3 activity was detected using caspase-3 assay. Rat ovarian granulosa cells were exposed to H2O2 (300 µM) for 24 h, or pretreated with 50 µM morroniside for 24 h and then exposed to H2O2 (300 µM) for 24 h to induce granulosa cell injury. *p < 0.05 compared with the control group; #p < 0.05 compared with the H2O2 group.
Morroniside inhibited autophagy in H2O2-stimulated rat ovarian granulosa cells
Autophagy has been implicated in cell death. 23 Thus, we next assessed autophagy in rat ovarian granulosa cells by western blotting. As shown in Figure 4, the expression of the autophagy-associated proteins, LC3-II, LC3-I, beclin-1, and p62, was analyzed. Results showed that the ratio of LC3-II/LC3-I and the protein expression of beclin-1 were increased, while p62 protein level was reduced in the H2O2 group compared with the control group. However, treatment with morroniside inhibited H2O2-induced autophagy through decreasing LC3-II/LC3-I ratio and beclin-1 expression and increasing p62 expression (Figure 4, p < 0.05).

Morroniside inhibited autophagy in H2O2-stimulated rat ovarian granulosa cells. LC3-II, LC3-I, beclin-1, and p62 expression was measured by western blotting. Rat ovarian granulosa cells were exposed to H2O2 (300 µM) for 24 h, or pretreated with 50 µM morroniside for 24 h and then exposed to H2O2 (300 µM) for 24 h to induce granulosa cell injury. *p < 0.05 compared with the control group; #p < 0.05 compared with the H2O2 group.
Morroniside induced the activation of the PI3K/AKT/mTOR pathway in H2O2-stimulated rat ovarian granulosa cells
PI3K/AKT/mTOR signaling has been shown to exert proliferative and anti-apoptotic effects in goose granulosa cells. 24 Hence, we next determined whether or not morroniside treatment affected the PI3K/AKT/mTOR signaling pathway in H2O2-stimulated rat ovarian granulosa cells. Western blot analysis showed that treatment with H2O2 suppressed p-AKT and p-mTOR expression in rat ovarian granulosa cells; however, morroniside treatment reversed this effect and activated the PI3K/AKT/mTOR pathway through increasing p-AKT and p-mTOR expression in H2O2-stimulated rat ovarian granulosa cells (Figure 5). There results indicate that morroniside activates the PI3K/AKT/mTOR pathway in H2O2-stimulated rat ovarian granulosa cells.

Morroniside activated the PI3K/AKT/mTOR pathway in H2O2-stimulated rat ovarian granulosa cells. p-AKT, AKT, p-mTOR, and mTOR protein expression was assessed by western blotting. Rat ovarian granulosa cells were exposed to H2O2 (300 µM) for 24 h, or pretreated with 50 µM morroniside for 24 h and then exposed to H2O2 (300 µM) for 24 h to induce granulosa cell injury. *p < 0.05 compared with the control group; #p < 0.05 compared with the H2O2 group.
PI3K/AKT or mTOR inhibitor reversed the protective effects of morroniside on H2O2-stimulated rat ovarian granulosa cells
To further confirm the role of PI3K/AKT/mTOR pathway in the action of morroniside in granulosa cell survival, oxidative stress, apoptosis, and autophagy, cells were treated with the PI3K inhibitor LY294002 or the mTOR inhibitor rapamycin. Results showed that both LY294002 and rapamycin suppressed cell survival compared with H2O2 + morroniside group (Figure 6A, p < 0.05). Further studies indicated that LY294002 or rapamycin treatment reversed the effects of morroniside on ROS and SOD levels, cell apoptosis, caspase-3 activity, and LC3-II/LC3-I ratio in H2O2-induced rat ovarian granulosa cells (Figure 6B–F).

Morroniside exerted its action in H2O2-stimulated rat ovarian granulosa cells via the PI3K/AKT/mTOR pathway. (A) CCK-8 assay was used to determine cell viability. (B) ROS content was measured with the ROS assay kit. (C) The level of SOD was measured with the SOD assay kit. (D) Cell death detection ELISA kit was used to measure cell apoptosis. (E) Caspase-3 activity was detected by caspase-3 assay. (F) LC3-II and LC3-I protein expression was assessed by western blotting. Rat ovarian granulosa cells were subjected to either of the following treatments: (1) pretreated with 50 µM morroniside for 24 h and then exposed to H2O2 (300 µM) for 24 h to induce granulosa cell injury, or (2) treated with the PI3K inhibitor LY294002 or the mTOR inhibitor rapamycin, then administered morroniside for 24 h, and then stimulated with 300 µM H2O2. *p < 0.05 compared with H2O2 group; #p < 0.05 compared with the H2O2 + morroniside group.
Discussion
Granulosa cell from follicle is the component of somatic cell in the oocyte, and the cells were closely associated with follicular development. 25 Accumulated evidences have implicated that oxidative stress is associated with granulosa cell injury, which affects oocyte quality and fertility. 26,27 In this study, we used H2O2 to induce cell injury in rat ovarian granulosa cells, and then investigated the effects and specific molecular mechanism of morroniside in cell survival, apoptosis, oxidative stress, and autophagy in H2O2-stimulated granulosa cells (Figure 7). Our results confirmed that stimulation with H2O2 inhibited granulosa cell survival. Further studies showed that morroniside treatment markedly improved cell survival, and impeded apoptosis, oxidative stress, and autophagy in H2O2-stimulated rat ovarian granulosa cells, and that the effects of morroniside were mediated by the PI3K/AKT/mTOR pathway, which is a key regulator of cell survival.
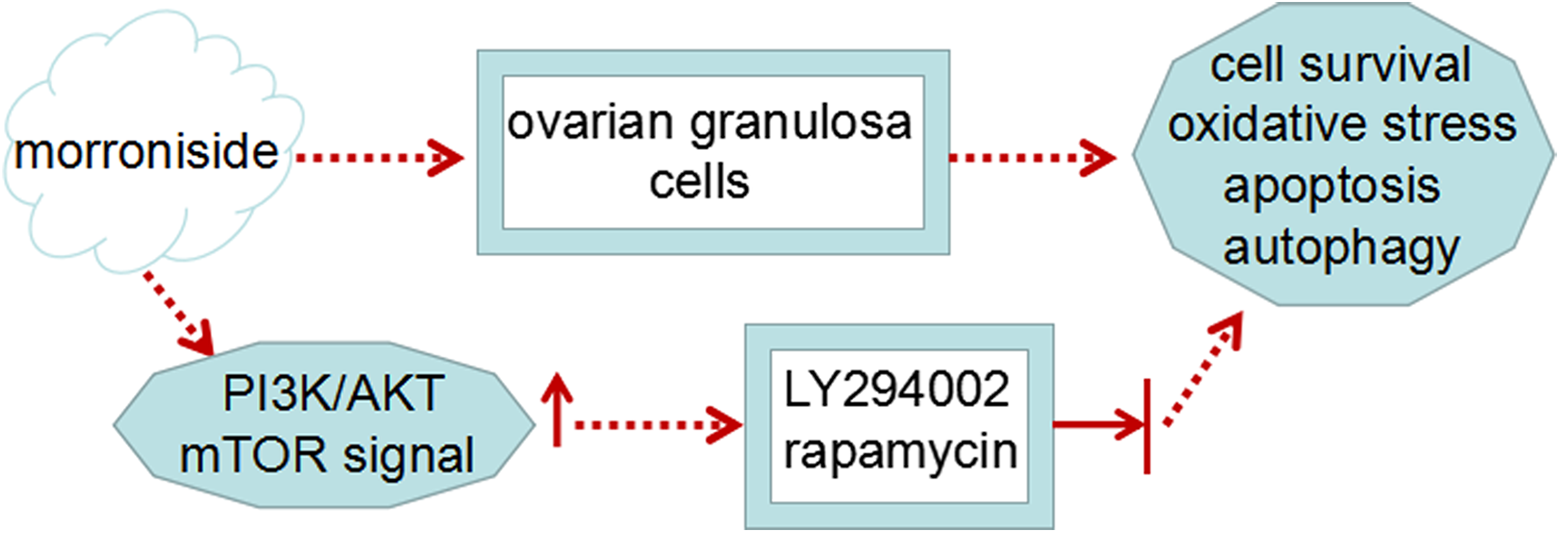
A schematic diagram of the role and mechanism of action of morroniside in rat ovarian granulosa cells.
Almost all follicles undergo atretic degeneration prior to ovulation in mammalian ovaries. 28,29 Granulosa cell apoptosis has been associated with follicular atresia. 30,31 Certain cell survival- and death-related molecules, including intraovarian regulators (intracellular proteins, gonadal steroids, and cytokines) and hormones (gonadotropins) have been shown to contribute to follicular development and atresia. 30 Besides, multiple evidences have suggested that oxidative stress can induce apoptosis in rat ovarian granulosa cells, which may lead to follicular atresia. 32 In this study, we showed that morroniside treatment inhibited apoptosis in H2O2-stimulated rat ovarian granulosa cells. Recent studies have suggested that the non-apoptotic forms of programmed cell death, such as autophagy may be associated with granulosa cell biology and atretic follicles. 33,34 To assess the molecular mechanism of morroniside in ovarian granulosa cell apoptosis, oxidative stress and autophagy were evaluated. We found that morroniside treatment suppressed oxidative stress and autophagy in rat ovarian granulosa cells.
Abramovich D and his colleagues confirmed that the PI3K/AKT signaling pathway is involved in follicular cell proliferation and apoptosis. 35 Mary E. Hunzicker-Dunn et al. showed that PI3K/AKT signaling is involved in ovarian granulosa cell proliferation, apoptosis, and differentiation. 36 Moreover, the protective effect of resveratrol on ovarian function in a rat model of premature ovarian insufficiency has been shown to be mediated by the PI3K/AKT/mTOR signaling pathway. 37 Consistent with these results, our findings indicated that morroniside treatment markedly activated the PI3K/AKT/mTOR pathway, and treatment with PI3K/AKT or mTOR inhibitor reversed this effect.
In conclusion, our study revealed that morroniside inhibits cell survival, apoptosis, oxidative stress, and autophagy in H2O2-stimulated rat ovarian granulosa cells, and that the effects of morroniside are mediated via the PI3K/AKT/mTOR pathway. Hence, our findings provide evidence that morroniside may play a key role in follicular development and atresia. However, further studies are needed to determine the importance of morroniside in human tissue, cells and animal models.
Footnotes
Authors’ note
Disi Deng and Jin Yan contributed equally to this work.
Author contributions
Keming Wu conceived and designed research. Jin Yan and Yeke Wu conducted experiments. Wanjing Li contributed new reagents or analytical tools.Jin Yan and Li wanjing analyzed data. Disi Deng wrote the manuscript. All authors read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (Grant numbers: 81873334 and 81973684).
