Abstract
Heparin has been in clinical use as an anticoagulant for the last eight decades and used worldwide in more than 100 million medical procedures every year. This lifesaving drug is predominantly obtained from ~700 million pig intestines or bovine organs through millions of small and medium-sized slaughterhouses. However, the preparations from animal sources have raised many safety concerns, including the contamination of heparin with potential pathogens, proteins, and other impurities. In fact, contaminated heparin preparations caused 149 deaths in several countries, including the United States, Germany, and Japan in 2008, highlighting the need for implementing sensitive and simple analytical techniques to monitor and safeguard the heparin supply chain. The contaminant responsible for the adverse effects in 2008 was identified as oversulfated chondroitin sulfate (OSCS). We have developed a very sensitive, facile method of detecting OSCS in heparin lots using a nanosensor, a gold nanoparticle–heparin dye conjugate. The sensor is an excellent substrate for heparitinase enzyme, which cleaves the heparin polymer into smaller disaccharide fragments, and therefore facilitates recovery of fluorescence from the dye upon heparitinase treatment. However, the presence of OSCS results in diminished fluorescence recovery from the nanosensor upon heparitinase treatment, because OSCS inhibits the enzyme. The newly designed nanosensor can detect as low as 1 × 10–9% (w/w) OSCS, making it the most sensitive tool available to date for the detection of trace amounts of OSCS in pharmaceutical heparins. In this report, we describe a simple methodology for the preparation of nanosensor and its application in the detection of OSCS contaminants.
Introduction
Heparin is a highly complex sulfated polysaccharide that has been in use as a blood thinner since the 1940s and is listed as an essential medicine by the World Health Organization.1–4 This lifesaving essential medicine is isolated predominantly from porcine intestine, and as a result, heparin lots have always been found to contain various amounts of other naturally occurring sulfated polysaccharides, such as dermatan sulfate and chondroitin sulfate. 5 Currently, more than 600 million pigs are annually used in the large-scale production of pharmaceutical-grade heparin for human use worldwide. 6 In 2008, there was a severe shortage of animal-derived heparin owing to the then foot and mouth infection of pigs on a large scale, and subsequently, several lots of heparin were found to be tainted with varying amounts of oversulfated chondroitin sulfate (OSCS) that caused severe life-threatening anaphylactic shock and several deaths worldwide. 7 This adulteration crisis exposed the vulnerability of the complex global heparin supply chain to various potential contaminants. Recent African swine fever (ASF) outbreaks in many Asian and European countries underline an emerging threat to the production of pharmaceutical-grade heparin and its purity.8,9 According to several conservative predictions, China, which supplies ~80% of the global need for porcine heparin, may have unfortunately lost as many as 200 million pigs to culling and/or death from current ASF outbreaks.10,11
In response to the 2008 crisis, the Food and Drug Administration (FDA) has revised the United States Pharmacopeia (USP) monograph for assessing heparin purity and its specific anticoagulant activity/potency. 12 Two analytical methods, nuclear magnetic resonance (NMR) and high-performance liquid chromatography (HPLC), are currently used by heparin manufacturers, as required by the FDA, to assess the molecular identity and purity of pharmaceutical-grade heparin products.13,14 These two analytical methods can only detect OSCS as low as 0.25%, and therefore a number of sensitive colorimetric methods have been developed.15,16 Unfortunately, these alternative methods can detect OSCS in a nonlinear manner. Furthermore, trace levels of OSCS in pharmaceutical heparins, far below the detection limits of NMR and HPLC, may still pose a risk to human life. Therefore, there is an urgent need to develop a novel sensor/technique that can detect OSCS in heparin lots in a linear manner at ultralow concentrations. Our lab has recently developed one of the most sensitive nanosensors, a gold nanoparticle (Au NP)–heparin dye conjugate, to rapidly and easily measure low levels of OSCS contaminants in heparin lots. 17 Heparitinase preferentially binds and cleaves heparin substrates. On the other hand, chondroitin sulfate is neither the substrate nor the inhibitor. However, after persulfonation, there is a change in the conformation of glucuronic acid residues owing to the presence of a 3-O sulfate on uronic acid, thus allowing OSCS to dock very well in the enzyme active site. However, the C5 proton of 3-O-sulfonated uronic acid is still further away from the critical H151 amino acid residue of the enzyme, prohibiting the cleavage of OSCS, which leads to the enzyme inhibition. 18
In this technical report, we discuss the synthesis and application of the gold–heparin dye nanosensor in assessing OSCS levels in pharmaceutical heparin lots for safeguarding the complex global supply chain.
Materials and Methods
Materials
Recombinant heparitinases (heparitinase I, II, and III) were expressed and purified as described elsewhere. 19 Heparin was from Ming Han Company (Oakland, CA). OSCS was prepared as described in the published reports. 20 Au NPs were synthesized using established protocols and are briefly described in the following section. HAuCl4·3 H2O, sodium citrate tribasic dihydrate, chondroitin sulfates (CS-A, CS-B, and CS-C), N-(3-dimethylaminopropl)-N′-ethylcarbodiimide hydrochloride, Py:SO3, tributylamine, and Costar 96-well plates (UV transparent flat bottom) were purchased from Sigma-Aldrich (St. Louis, MO). Amberlite IR120-H+ form was purchased from Fluka. Bond-breaker TCEP solution and 3-[2-pyridyldithio]propionylhydrazide (PDPH) were obtained from Thermo Scientific (Waltham, MA). HiLyte Fluor 594 hydrazide–TFA salt was from Anaspec (Fremont, CA). Amicon Ultra 3000 molecular weight cutoff (MWCO) centrifugal filters were from EMD Millipore Corporation (Billerica, MA). All other chemicals were purchased from either VWR Scientific (Radnor, PA) or Sigma-Aldrich.
Preparation of Nanosensor
HAuCl4·3 H2O (0.02 g) was dissolved in 100 mL of deionized H2O in a 500 mL round-bottom flask. The solution was heated at 100 °C with a stirring speed of 1100 rpm. After 30 min, 10 mL of 150 mM sodium citrate dihydrate solution was added directly to the gold solution while it was kept under stirring and heating. When the color of the gold solution changed to red, it was removed immediately from the oil bath and allowed to reach room temperature. The Au NP concentration can be obtained using Beer–Lambert’s law. Au NPs are stable for ~14 days at 4 °C. Heparin (1 mg/mL) in 1 mL of 50 mM phosphate-buffered saline (PBS) buffer, pH 7.4 (1 mL), was treated with 230 μg of PDPH and 34 μg of EDC for 12 h at room temperature with shaking. The mixture solution was purified and concentrated through centrifugation using a 3000 MWCO filter at 10,000g for 10 min; this process was repeated three times. By adding Milli-Q water, the heparin–thiol concentration was adjusted to 10 μg/μL. TCEP (0.5 mL of 5 mM solution) was added to 1 mL/mg of heparin–thiol, and this mixture was incubated at 37 °C for 30 min and centrifuged using a 3000 MWCO filter at 10,000g for 10 min to remove the excess TCEP. A coupling reaction between 1 mg of heparin–thiol and 0.3 mg of HiLyte 594 hydrazide was carried out in the presence of 2 mg of EDC in 1 mL of 50 mM PBS buffer, pH 7.4, for 16 h at room temperature. The excess dye and EDC were removed by centrifugation using a 3000 MWCO filter at 1000g for 10 min; this process was repeated several times until the complete removal of excess unconjugated dye molecules. Thiolated heparin dye conjugate (1 mg) was incubated with 0.5 mL of 5 mM TCEP solution for 30 min, and subsequently the excess TCEP was removed by centrifugation. Finally, the thiolated heparin dye conjugate (1 mg) was mixed with 10 mL of 1.4 nM Au NPs and shaken gently at 37 °C for 2 days. The gold–heparin dye conjugate was concentrated by a 3000 MWCO filter at 4000g for 30 min.
Heparitinase Treatment and Fluorescence Recovery Assay
The efficacy of the heparitinases on the Au–heparin dye probe was tested by incubating the enzyme cocktail (10 µL) with the probe (100 µL, 0.20 µg/µL) in the presence of 10× heparitinase buffer (10 µL) at 37 °C. The 96-well plate was excited at 488 nm, and the emission maxima readings were taken at 600 nm after exciting the samples at 488 nm using a fluorescence plate reader. The fluorescence recovery spectra were recorded at different incubation times of 15 min, 30 min, 1 h, 2 h, 3 h, and 4 h. For OSCS quantification in heparin, the following steps were followed. First, a logarithmic serial dilution of OSCS from 1 µg to 0.1 fg was performed. The OSCS samples were then added to the heparin stock (10 µL, 1 µg/µL), making a total of 11 OSCS samples containing heparin standards. A positive control of 100% heparin (10 µL, 1 µg/µL) and a negative control of 100% OSCS (10 µL, 1 µg/µL) were also used. Second, a heparitinase enzyme cocktail (5 µL) was added to all of the above vials and allowed to incubate at room temperature for 1 h. Third, a Au–heparine dye nanosenor (100 µL, 0.20 µg/µL) was transferred (for a duplicate study) to a 96-well plate. Fluorescence intensities (F0) of these wells were read with the plate reader. Next, the samples from 13 vials were added to the nanosensor, and fluorescence intensities (F) were recorded at incubation times of 1 min, 30 min, 1 h, 2 h, 3 h, and 4 h.
Results and Discussion
Au NPs are known to quench fluorophores up to ~ 200 Å distances.21,22 Therefore, we predicted that a Au NP–heparin dye conjugate would exhibit strong fluorescence quenching when the dye is conjugated to heparin polymers (~8000 Da), which in turn linked to Au NPs. When a Au NP–heparin dye conjugate is treated with a heparin cleaving enzyme, heparitinase, the fluorescence should be recovered as heparin dye fragments are released from the Au NPs. On the other hand, OSCS is known to strongly inhibit heparitinase enzyme, and thus enzymatic treatment of the nanosensor should not lead to fluorescence recovery in the presence of inhibitory OSCS, as heparin dye fragments cannot be released from the Au NPs. 17
In the following sections, we describe the preparation and application of the nanosensor.
Preparation of Nanosensor
The nanosensor containing gold–heparin dye was synthesized as shown in Figure 1. 17 In brief, the synthesis involves the following four steps: (1) thiolation of a naturally occurring commercial heparin polymer by reacting it with PDPH in PBS buffer (pH = 7.4), followed by TCEP treatment to cleave the disulfide bond, resulting in the preparation of thiolated heparin [heparin–SH]; (2) thiolated heparin [heparin–SH] is then subjected to conjugation with HiLyte 594 hydrazide dye, giving the required heparin dye–SH conjugate; (3) synthesis of Au NPs using HAuCl4·3 H2O and sodium citrate; and (4) conjugation of heparin dye–SH with Au NPs of different sizes generates the final nanosensor, a Au NP–heparin dye conjugate.
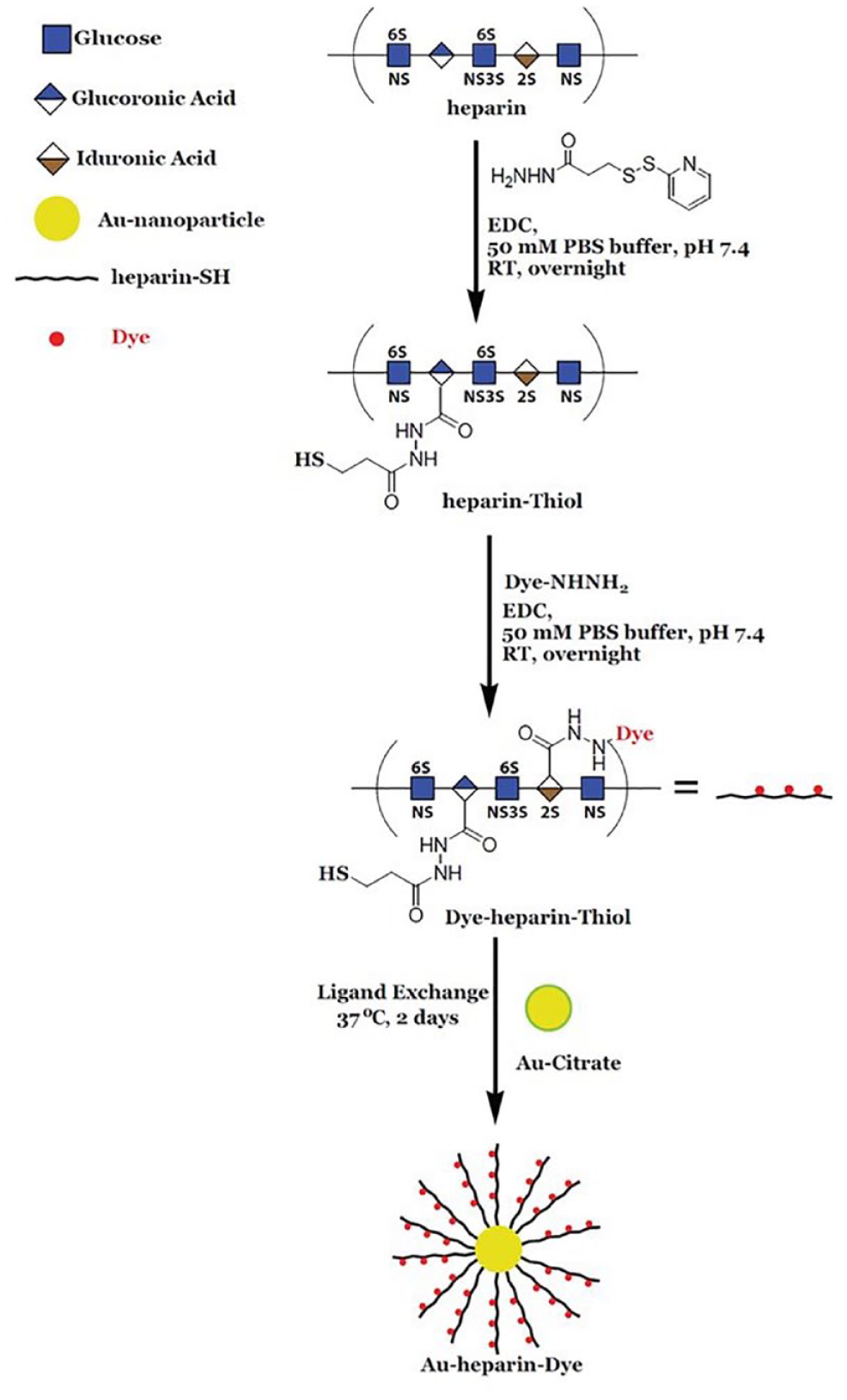
Synthesis of Au–heparin dye nanosensor.
HPLC-Based Assay
Disaccharide fragments of heparin, obtained from the digestion of heparin polymers with heparitinases, can be analyzed on an analytical HPLC to confirm the presence of OSCS through its inhibitory effect on enzyme activity. After digestion by heparitinase I, II, and III, heparin is cleaved into the following three major disaccharide units in addition to other minor disaccharide units: NS2S (N-sulfated and 2-O-sulfated), NS6S (N-sulfated and 6-O-sulfated), and NS2S6S (N-sulfated, 2-O-sulfated, and 6-O-sulfated). The dissacharides can be detected using an anion-exchange column, such as Dionex CarboPac PA1 IC columns under the appropriate mobile phase conditions. The individual peaks of disaccharide fragments can be detected through UV absorbance at 232 nm. However, an HPLC-based assay is somewhat less sensitive and can indicate only the presence of OSCS without providing much needed quantitative information, especially when it is present at ultralow levels.
Fluorescence Recovery Assay
A fluorescence spectrophotometer can be used to determine the quenching of heparin dye fluorescence after its conjugation with Au NP. The fluorescence of the heparin dye, which was quenched upon the conjugation with Au NP, can be recovered up to ~70% following incubation with an enzyme cocktail (heparitinase I, II, and III) that releases heparin dye fragments from the Au NP (Fig. 2). 17 Thus, a simple plate reader-based spectrophotometer assay for fluorescence detection can easily be exploited to quantify impurities such as OSCS in heparin lots. Since OSCS will inhibit enzyme activity, the fluorescence signal is maximal at low OSCS concentrations and minimal at high OSCS concentrations ( Fig. 3 ). Here, we show an example of heparin spiked with an OSCS sample to explain how the fluorescence release assay is performed (Fig. 2). A positive control of 100% heparin and a negative control of 100% OSCS are also used in the assay, and a serial dilution of OSCS in heparin standard is used for quantification. The above samples are incubated with heparitinase I, II, and III enzyme cocktail at room temperature for 1 h. Then the Au–heparin dye nanosenor is added to a 96-well plate, and the background fluorescence intensities (F0) at excitation/emission = 488 nm/600 nm are measured using a fluorescence plate reader. Next, the above enzyme-treated samples, heparin spiked with varying amounts of OSCS, are transferred to the 96-well plate that contains the nanosensor in each well, and fluorescence intensities (F) are recorded at different time points up to 4 h. In the positive control, a fluorescence recovery of 70% can be observed over a period of 4 h, while the heparin sample spiked with OSCS results in a diminished fluorescence recovery. To be noted, this method is very sensitive (with a detection limit at 1 × 10−9% [w/w] OSCS) and specific (no fluorescence interference with other naturally occurring glycosaminoglycan impurities such as CS-A, CS-C, or dermatan sulfate; see Fig. 4 ). In addition, the fluorescence recovery, following the enzymatic treatment of the nanosensor mixed with heparin samples spiked with varying OSCS amounts, can also be measured through the capture of emitting photons using a state-of-the-art in vivo imaging system (IVIS) equipped with a CCD camera and a DsRed filter. Based on the radiant efficiency, a heat map can be constructed to achieve a better sensitivity for the detection of as little as 1 × 10−9% (w/w) OSCS contaminant within a relatively short period of time. Thus, the use of an IVIS can substantially improve the detection limits of OSCS in commercial heparin lots. If a quality control lab or federal regulatory lab wishes to compare a large number of various heparin lots in a single, simple assay to assess the presence of OSCS, the nanosensor allows rapid detection, as one can use a multiwell fluorescence detector and obtain the data in less than 30 min to 1 h. On the other hand, although NMR is an essential, undisputable method for validating the presence of OSCS, analyzing a large number of heparin lots might be very time-consuming, as current conventional NMR instruments are not well suited for characterizing thousands of samples in a multiwell format in a single run. However, one must note that a nanosensor cannot provide the structural details of OSCS definitively. Thus, once a potential lot with poor fluorescence recovery with the nanosensor is identified, then the sample needs to undergo further structural analysis using sophisticated NMR techniques to unequivocally confirm the presence of an OSCS contaminant and its structural attributes.
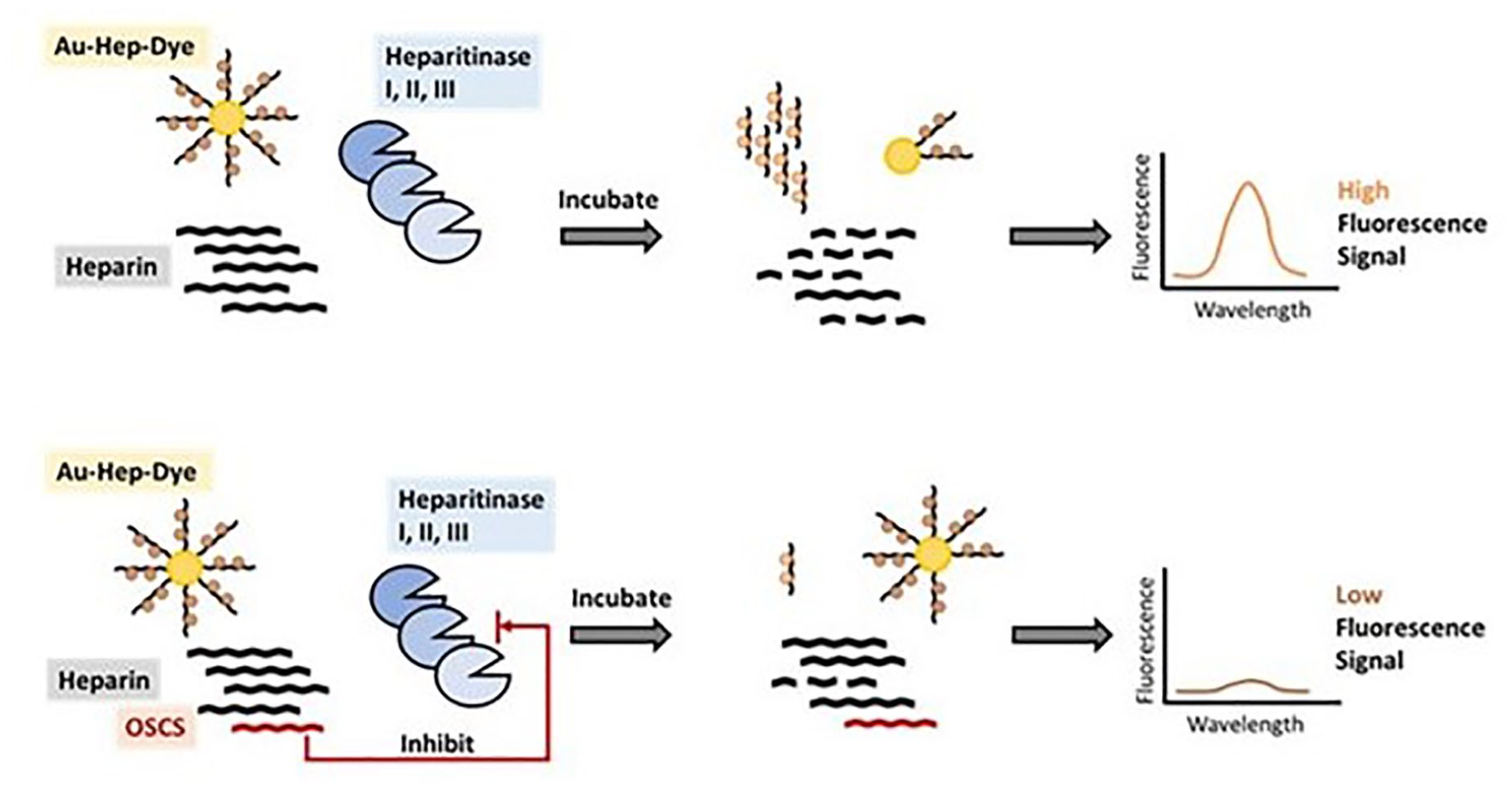
The workflow outlining the use of a spectrophotometer to measure the fluorescence recovery upon treatment of a nanosensor with the heparitinase cocktail in the presence of heparin lots containing varying levels of OSCS.
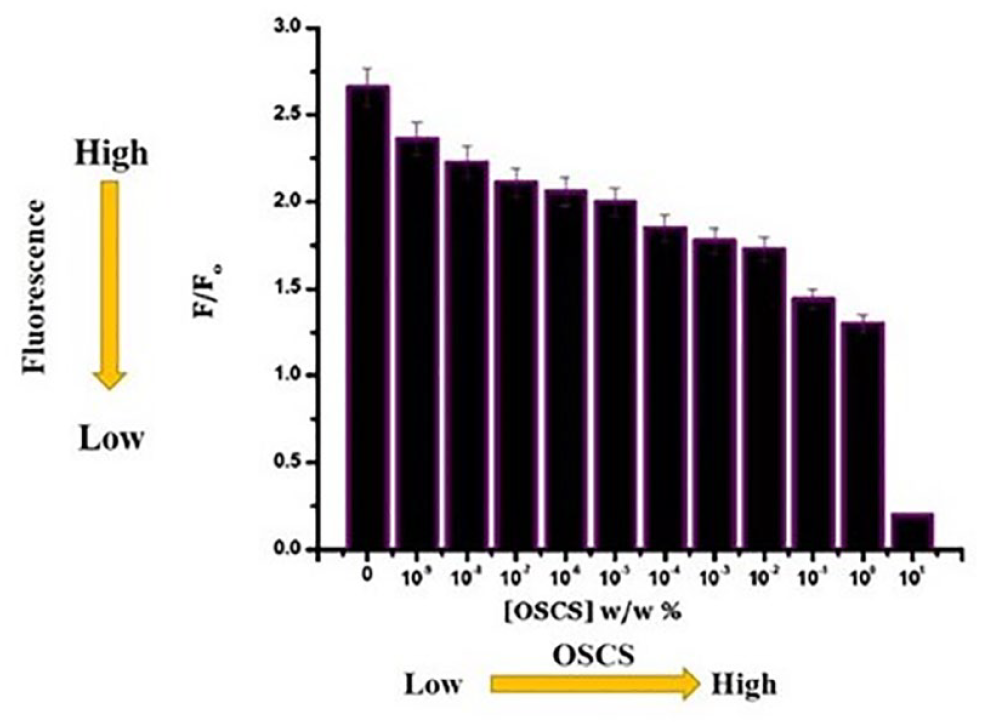
The relative fluorescence signal of the Au NP–heparin dye with serial dilution of OSCS contaminant shows the decreased fluorescence signal with increasing OSCS concentrations, and measurements were obtained using a fluorescence plate reader. F = fluorescence intensity of Au–heparin dye nanoprobe after incubation with heparitinase enzymes in the presence of OSCS at various concentrations; F0 = fluorescence intensity of the nanosensor before incubation with heparitinase enzymes. (Adapted from Kalita et al. 17 )
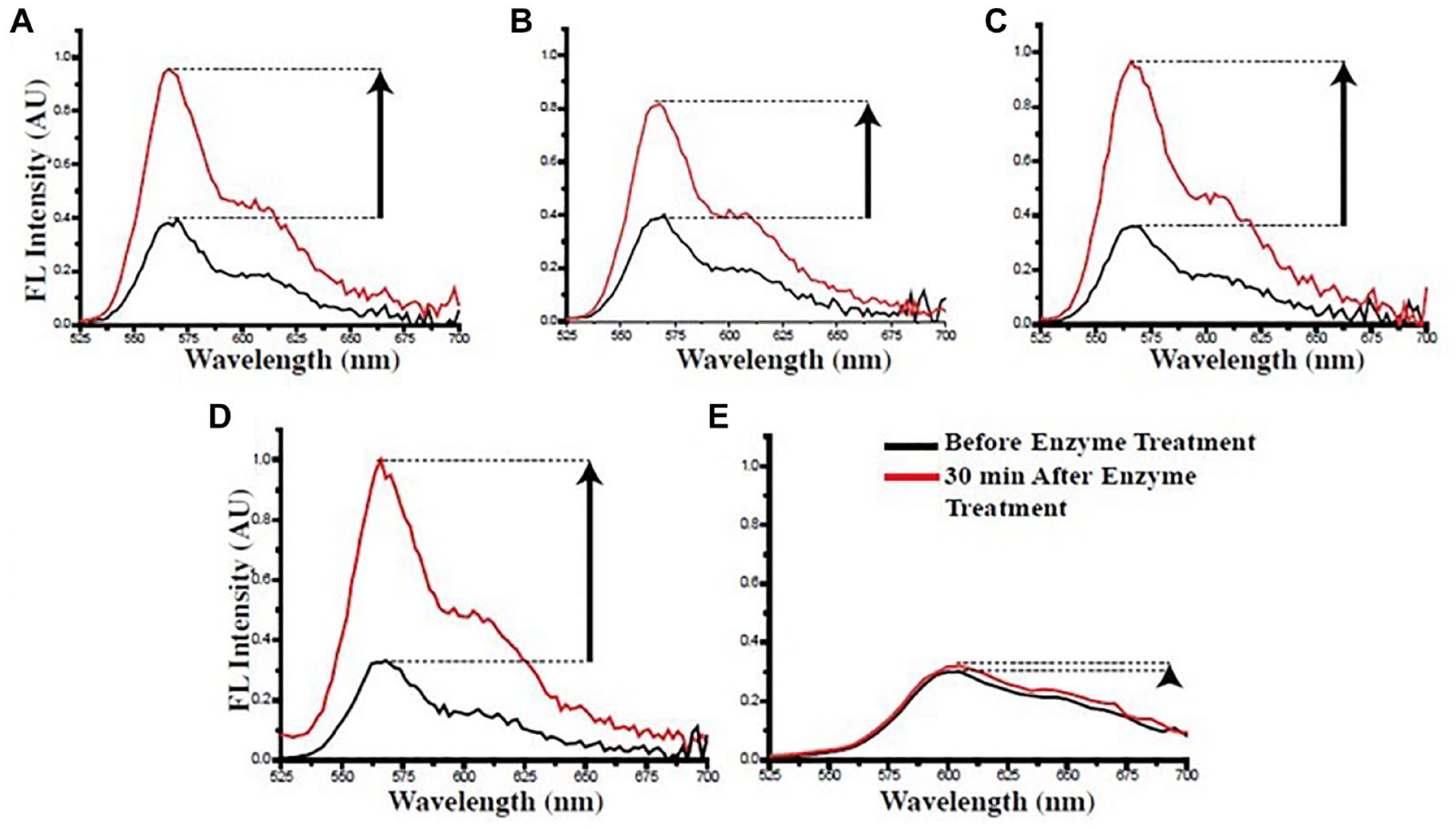
Fluorescence recovery from the heparin nanosensor was determined after treatment of the nanosensor (75 µL, 0.2 µg/µL) with heparitinases in the presence of following the samples containing various naturally occurring GAGs that are commonly found in heparin lots: (
In conclusion, a nanosensor, Au NP–heparin dye, was developed to efficiently and seamlessly screen a large number of heparin lots, which may be procured from thousands of different heparin manufacturing facilities in a relatively short time, with the final goal of assessing the presence of femtogram levels of life-threatening OSCS impurity and thus saving human lives through safeguarding the global heparin supply chain. Furthermore, other GAG–Au NP conjugates can be prepared using the protocol provided herein to detect the activity of GAG cleaving enzymes such as heparanase and hyaluronidase, which are associated with several human pathological conditions.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grant HL107152.
