Abstract
In 2008, oversulfated chondroitin sulfate (OSCS) was identified as the main contaminant in recalled heparin. Oversulfated chondroitin sulfate can be prepared from bovine (B), porcine (P), shark (Sh), or skate (S) origin and may produce changes in the antithrombotic, bleeding, and hemodynamic profile of heparins. This study examines the interactions of various OSCSs on heparin in animal models of thrombosis and bleeding, as well as on the anticoagulant and antiprotease effects in in vitro assays. Mixtures of 70% unfractionated heparin (UFH) with 30% OSCS from different sources were tested. In the in vitro activated partial thromboplastin time (aPTT) assay, all contaminant mixtures showed a decrease in clotting times. In addition, a significant increase in bleeding time compared to the control (UFH/saline) was observed. In the thrombosis model, no significant differences were observed. The OSCSs significantly increased anti-Xa activity in ex vivo blood samples. These results indicate that various sources of OSCS affect the hemostatic properties of heparin.
Introduction
Pharmaceutical grade heparin is an extremely complex end product produced by various extraction and purification processes. Different preparation methods may cause changes in the physical, chemical, and biological properties of heparins. 1 If the preparation of heparin is not carefully performed under the strictest conditions and effective quality control measures are not practiced, contamination may result. Subsequently, issues of heparin contamination have been previously recognized.
In 1955, McGeown and colleagues 2 found traces of phosphate in commercial heparins. Additionally, other contaminants such as histamine 3 and EDTA 4 have also been identified in commercial heparins. In 1987, Perlin et al 5 found that dermatan sulfate was a common impurity of pharmaceutical heparin preparations, comprising 2% to 15% of the polymer mixtures in some cases. With the use of nuclear magnetic resonance (NMR) spectroscopy, the presence of other glycosaminoglycans, as well as sodium acetate and ethanol, has also been identified in pharmaceutical heparins.6,7
More recently (2008), some batches of heparin were found to contain a dermatan sulfate impurity and varying amounts (5%-30%) of a novel contaminant known as oversulfated chondroitin sulfate (OSCS). 8 Since OSCS is not a naturally occurring compound, many have formed the conclusion that the addition of OSCS to heparin was intentional. Additionally, Pan et al 9 have recently demonstrated the presence of other contaminants in heparin besides OSCS, which are presumably heparin by-products produced during the heparin purification process. Some patients receiving OSCS-contaminated heparin treatment experienced adverse allergic reactions such as hypotension and anaphylaxis and in some of these cases, death occurred.
Oversulfated chondroitin sulfate can be prepared from chondroitin sulfate (CS) derived from bovine (B), porcine (P), shark (Sh), or skate (S) tissue sources. The different sources of OSCS may vary in their molecular profile and biological actions and, therefore, may produce different interactions with unfractionated heparin (UFH). It is hypothesized that different OSCS preparations may alter the antithrombotic, bleeding, and hemodynamic profile of heparin. This investigation characterizes the effects of OSCS from different sources on the relative safety and efficacy of heparin in animal models of bleeding and thrombosis.
Materials and Methods
Materials
Different OSCS preparations produced from bovine, shark, skate, and porcine CS were used in this study. These agents were a generous gift from Dr C. Viskov (Sanofi-Aventis, Paris, France). An additional OSCS of porcine origin (P2-OSCS) produced by a different process was a generous gift from Dr R. Lindhardt (Renssalaer Polytechnic Institute, Troy, New York). Unfractionated heparin was obtained from Gentium (Corlo, Italy). The materials were obtained in the form of a white powder and stored at room temperature in a desiccator.
For the in vitro assays, the different OSCSs and UFH were reconstituted in saline to obtain a concentration of 100 µg/mL and then mixed in a ratio of 70% UFH to 30% OSCS. The mixtures were added to normal human plasma ([NHP] Loyola University Medical Center Blood Bank, Maywood, IL) and serially diluted to obtain concentrations ranging from 10 to 0.625 µg/mL. For the in vivo studies, heparin was mixed with the various OSCS samples at a ratio of 70:30 to make 2 mg/mL solutions in saline. Whole blood collected after the rats were sacrificed was spun down at 3000 rpm for 20 minutes to produce platelet-poor plasma (PPP).
Methods
In vitro studies
The above-described mixtures in NHP were assayed using the aPTT, anti-factor IIa (anti-FIIa), and anti-FXa assays. The aPTT was performed on the ACL 300 Plus fast kinetic coagulation analyzer (Beckman Coulter; Fullerton, California) using the Platelin reagent (Trinity Biotech; Bray, Colorado). The amidolytic anti-FIIa and anti-FXa assays were run on the ACL 300 Plus using bovine FXa and human thrombin from Enzyme Research Laboratories (South Bend, Indiana). Spectrozyme Xa and Spectrozyme TH were obtained from American Diagnostica (Stanford, Connecticut).10,11
In vivo studies
This study was approved by Loyola University Medical Center Institutional Animal Care and Use Committee and was performed in accordance with the guidelines set forth by the National Institutes of Health.
Antithrombotic/bleeding effects
Male Sprague-Dawley rats (250-300 g; Harlan Co, Indianapolis, Indiana) were housed with excess rodent chow and water in the animal care facility, where temperature, humidity, and light cycle (7:00-19:00) were strictly controlled.
Rat tail transection bleeding model
The rat tail bleeding (RTB) model 12 was used to evaluate the hemorrhagic potential of the agent mixtures. Following the induction of anesthesia by an intramuscular injection of ketamine (90 mg/kg) and xylazine (10 mg/kg), a baseline blood pressure reading (Stoelting Manual Non-Invasive Blood Pressure System; IITC Life Science Inc; Woodland Hills, California) was obtained. Before drug administration, the abdominal area of the animal was shaved and the area of injection was sterilized with alcohol swabs. Intravenous (IV) injections of the UFH/contaminant mixture were administered (2 mg/kg) through the tail vein or femoral vein. A second blood pressure reading was taken immediately after drug administration and 5 minutes prior to tail transection. The percentage change in blood pressure was calculated using the equation: (original blood pressure reading − second blood pressure reading)/(original blood pressure reading). Ten minutes after IV injection, bleeding was induced by transecting the distal 2 mm of the rat tail. The tail tip was gently blotted with filter paper every 30 seconds until the bleeding stopped. Care was taken that no pressure was exerted on the tail tip, which could affect hemostasis. For each treatment group, the mean bleeding time ± standard error of mean was determined.
Rat jugular vein clamping thrombosis model
The rat jugular vein clamping thrombosis (JVT) model 13 was used to evaluate the antithrombotic effects of the agent mixtures. After the completion of the rat tail transection bleeding model, the JVT model began 50 minutes after the initial IV injection. The skin on the neck was shaved and an incision was made centrally above the trachea. The right jugular vein was isolated and covered with ultrasound transmission gel. An initial reading of blood vessel flow by a bidirectional Doppler probe was taken. Next, a manual clamping of the jugular vein for 1 minute was performed using mosquito forceps. Clamping of the blood vessel caused endothelial damage and initiated clot formation. Blood flow was measured 5 minutes after clamping. The procedure was continued until the vessel had thrombosed and blood flow ceased, as measured by the probe 5 minutes after each clamping. The effectiveness of the antithrombotic agent was determined by the number of clampings required to cause complete vascular occlusion.
Immediately after completion of the thrombosis model, a cardiac puncture was performed for blood collection (90 mins after drug administration). The rat was euthanized with an intracardiac injection of Beuthanasia D (0.3 mL). Ex vivo assays, aPTT (whole blood and plasma), anti-FXa (plasma), and anti-FIIa (plasma) were performed to determine the circulating levels of the agent mixtures.
Statistical Analysis
For the in vitro studies, the data represent mean ± standard error of 3 individual experiments. The JVT, RTB, and the ex vivo assays were analyzed using the one-way analysis of variance (ANOVA) model; followed by a post hoc Bonferroni analysis. In all cases, a P value < .05 was considered statistically significant.
Results
Anticoagulant and antiprotease assays were run on 70% and 30% mixtures of UFH and OSCS from various sources, as shown in Figures 1 and 2. For the aPTT assay, a concentration-dependent response was observed. The aPTT was prolonged up to 300 seconds for all the UFH/OSCS mixtures as well as for the UFH/saline control at the 10 µg/mL concentration (Figure 1). However, at lower concentrations, the UFH/OSCS mixtures demonstrated less anticoagulation compared to the UFH/saline control. There was no significant difference observed in terms of inhibition of factor IIa (thrombin) by the UFH/OSCS mixtures compared to the UFH/saline control, as shown in Figure 2; however, similar trends as seen in the aPTT were observed.
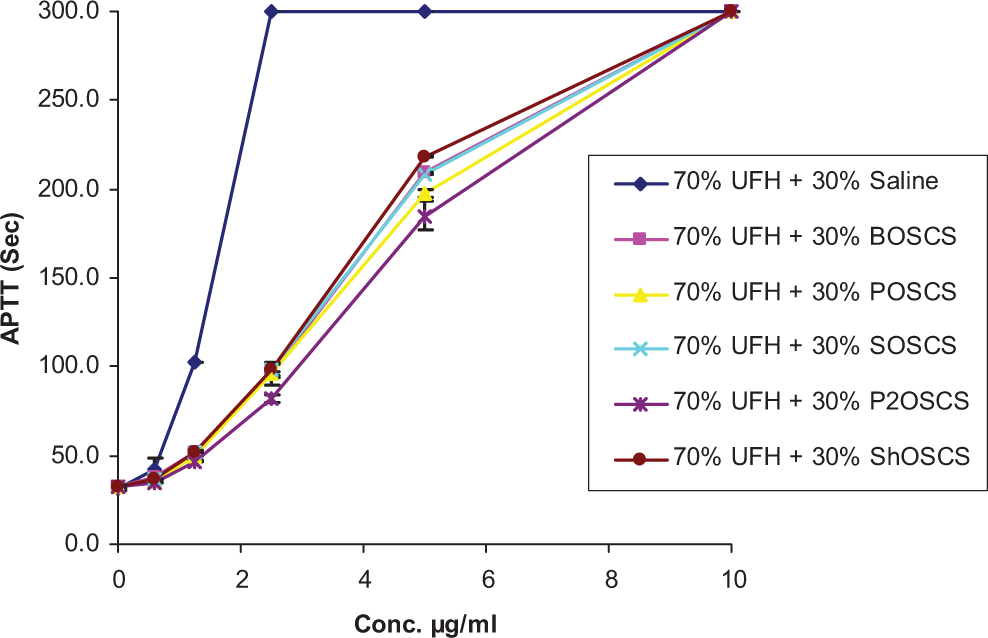
Comparative in vitro anticoagulant profile of UFH/contaminant mixtures and a UFH/saline control in the aPTT assay. The aPTT was prolonged up to 300 seconds for all the UFH/contaminant mixtures as well as for the UFH/saline control at the 10 µg/mL concentration. At the 5 to 0.625 µg/mL concentrations, the UFH/contaminant mixtures showed a significant decrease in aPTT compared to the UFH/saline control. The data represent the mean ± SEM of 3 individual experiments. aPTT indicates activated partial thromboplastin time; UFH, unfractionated heparin; SEM, standard error of the mean.
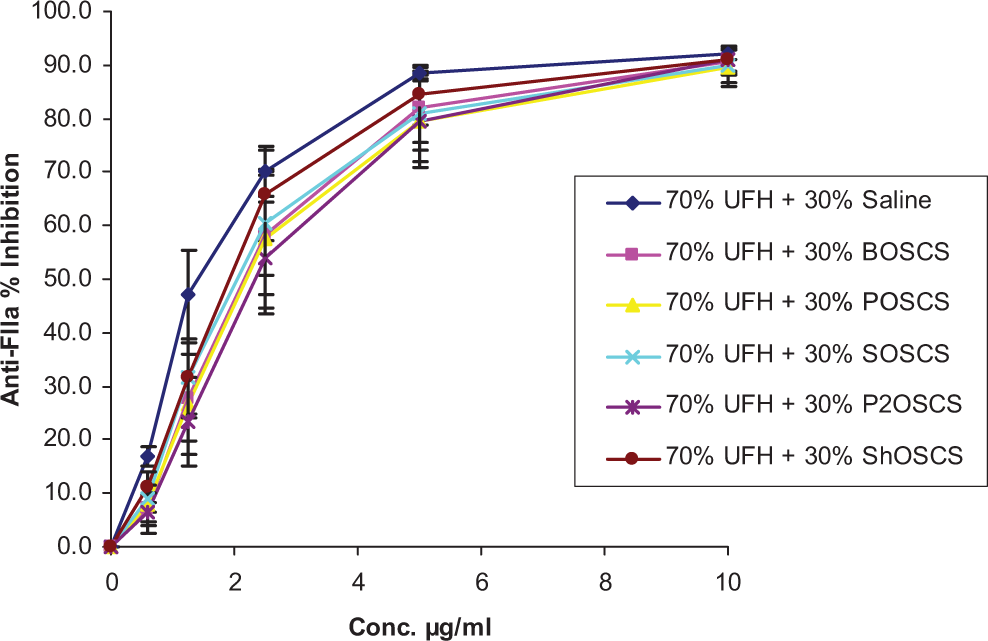
Comparative in vitro antiprotease profile of UFH/contaminant mixtures and a UFH/saline control in an anti-FIIa assay. No significant differences were observed in terms of inhibition of FIIa by the UFH/contaminant mixtures compared to the UFH/saline control. The data represent the mean ± SEM of 3 individual experiments. UFH indicates unfractionated heparin; FIIa, factor IIa; SEM, standard error of the mean.
In the RTB, 70% and 30% mixtures of UFH and OSCS from various sources were tested at 2 mg/kg IV. Significant increases in bleeding times between the rats treated with UFH/saline (21.8 ± 1.1 minutes) and the rats treated with UFH/POSCS (28.4 ± 4.6 minutes), UFH/ShOSCS (28.9 ± 2.7 minutes), and UFH/SOSCS (27.8 ± 2.6 minutes) were observed (P < .05 vs UFH/saline; Figure 3 ). Conversely, UFH/BOSCS (20.0 ± 3.6 minutes) did not show any increase in bleeding when compared to UFH/saline.
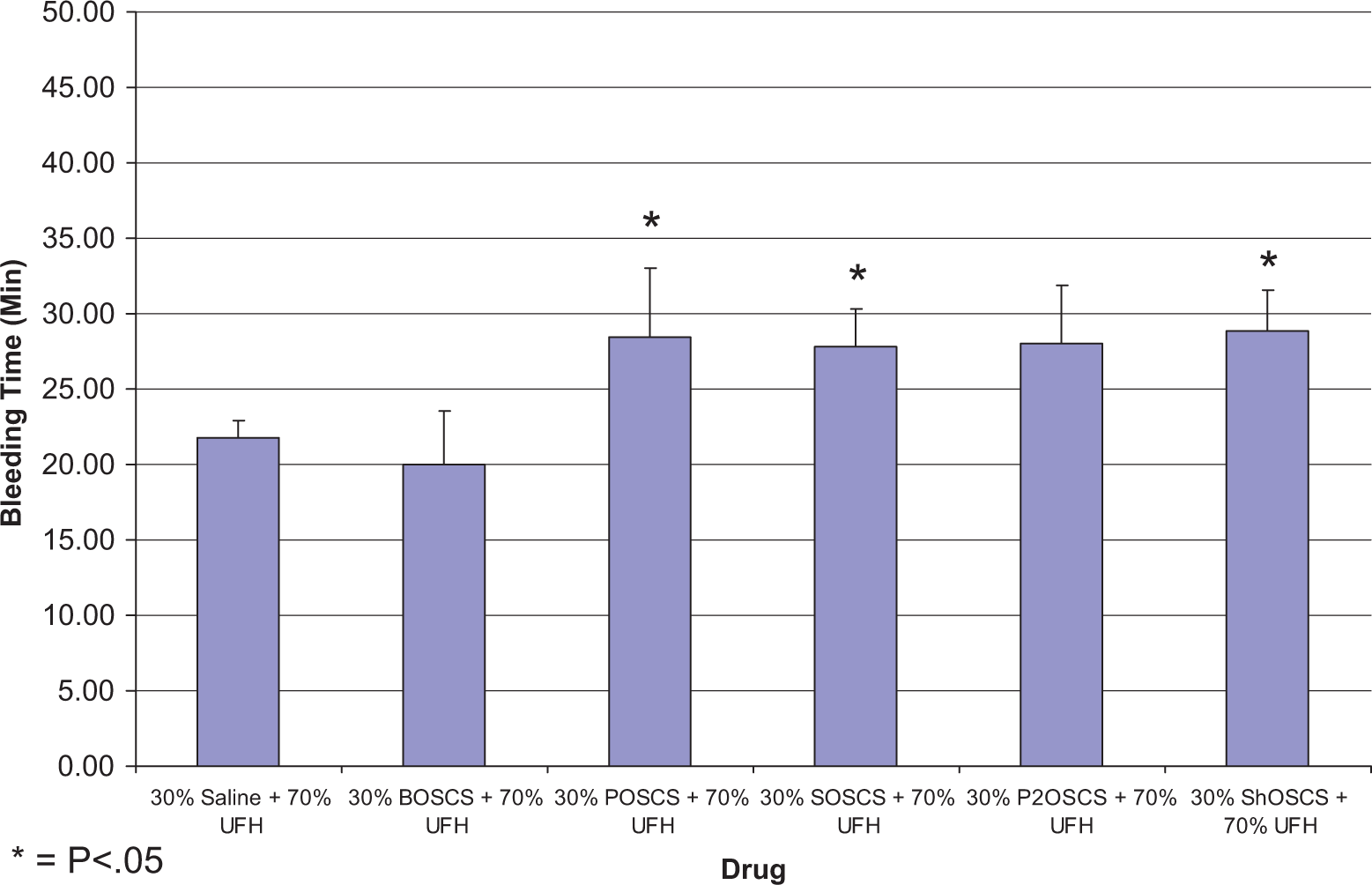
Hemorrhagic effects of UFH/contaminant mixtures following intravenous administration (2 mg/kg) in a rat tail bleeding model. Significant increases in bleeding times between the rats treated with UFH/saline (21.8 ± 1.1) and the rats treated with UFH/P (28.4 ± 4.6), UFH/Sh (28.9 ± 2.7), and UFH/S (27.8 ± 2.6) were observed. UFH/B (20.0 ± 3.6) did not show any increase in bleeding when compared to control. The data represent the mean ± SEM of 5 rats per treatment group. Statistical differences were determined using one-way ANOVA followed by the Bonferroni multiple comparison test. *P < .05 versus control. UFH indicates unfractionated heparin; SEM, standard error of the mean; B, bovine; P, porcine; Sh, shark; S, skate; ANOVA, analysis of variance.
The jugular vein clamping model was used to test the antithrombotic effects of the UFH/OSCS mixtures. Figure 4 shows the results of the jugular vein clamping model. None of the UFH/OSCS mixtures showed any significant change compared to the UFH/saline control in the JVT model or in terms of percentage change in blood pressure (Figure 5 ). Although no significant differences were observed in terms of percentage change in blood pressure, some of the UFH/OSCS mixtures exhibited a decreasing trend. The ex vivo whole blood aPTT was prolonged up to 300 seconds for all UFH/OSCS mixtures, demonstrating an excessive level of anticoagulation. This was significantly higher compared to the UFH/saline control (Figure 6 ).
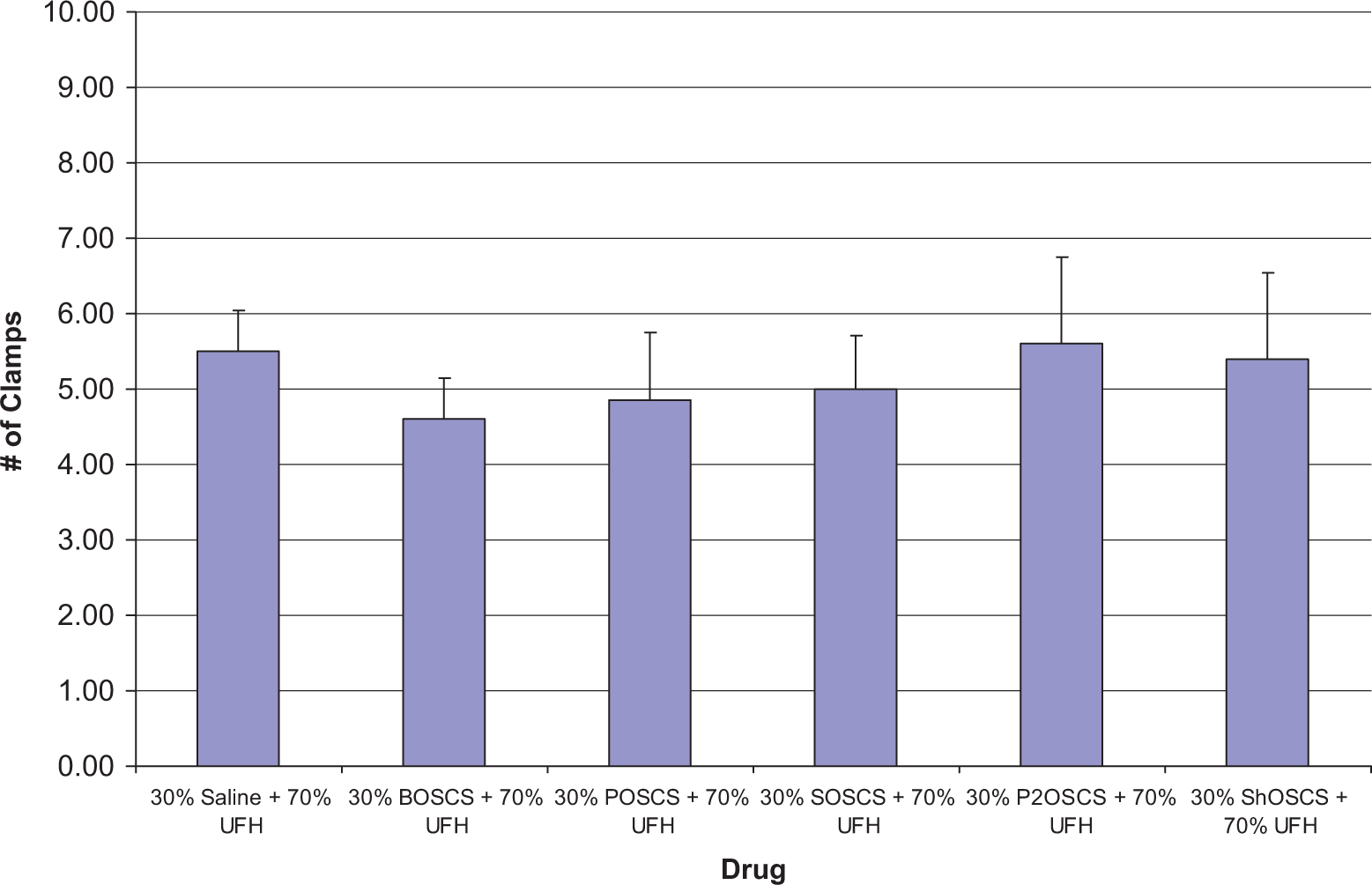
Antithrombotic effects of UFH/contaminant mixtures following intravenous administration (2 mg/kg) in a rat jugular vein clamping model. None of the UFH/contaminant mixtures showed any significant change compared to the UFH/saline control. The data represent the mean ± SEM of 5 rats per treatment group. Statistical differences were determined using one-way ANOVA followed by the Bonferroni multiple comparison test. UFH indicates unfractionated heparin; SEM, standard error of the mean; ANOVA, analysis of variance.
Effects on percentage change in blood pressure following intravenous administration (2 mg/kg) of UFH/contaminant mixtures. No significant differences were observed in terms of percentage change in blood pressure for any of the UFH/contaminant mixtures compared to the UFH/saline control. The data represent the mean ± SEM of 5 rats per treatment group. Statistical differences were determined using one-way ANOVA followed by the Bonferroni multiple comparison test. UFH indicates unfractionated heparin; SEM, standard error of the mean; ANOVA, analysis of variance.
Comparative ex vivo anticoagulant profile of UFH/contaminant mixtures and a UFH/saline control in the aPTT assay. Whole blood aPTT was prolonged up to 300 seconds for all UFH/contaminant mixtures, which was statistically significant compared to the UFH/saline control. The data represent the mean ± SEM from 5 whole blood samples taken from rats. Statistical differences were determined using one-way ANOVA followed by the Bonferroni multiple comparison test. *P < .001 versus control. aPTT indicates activated partial thromboplastin time; UFH, unfractionated heparin; SEM, standard error of the mean; ANOVA, analysis of variance.
The ex vivo analysis of the rat PPP demonstrated that UFH/POSCS, UFH/SOSCS, and UFH/ShOSCS exhibited a moderate inhibition (55%-70%) of factor Xa compared to the UFH/saline control (P < .01; Figure 7 ). Additionally, UFH/BOSCS inhibited factor Xa but to a lesser extent (P < .05). UFH/P2OSCS showed no effect compared to the UFH/saline control. UFH/P2OSCS, UFH/BOSCS, UFH/ShOSCS, and UFH/POSCS significantly inhibited (P < .001) factor IIa (thrombin) when compared to the UFH/saline control (Figure 8 ). Additionally, UFH/SOSCS inhibited factor IIa but to a lesser extent (P < .01). No significant difference compared to control was observed for APTT in rats treated with UFH in combination with each of the contaminants (data not shown).
Comparative ex vivo antiprotease profile of UFH/contaminant mixtures and a UFH/saline control in a chromogenic anti-FXa assay. Ex vivo analysis of rat platelet-poor plasma demonstrated that UFH/P, UFH/S, and UFH/Sh exhibited a moderate inhibition (55%-70%) of FXa compared to the UFH/saline control. UFH/B also inhibited (50%) FXa but to a lesser extent. UFH/OSCS showed no effect compared to the UFH/saline control. The data represent the mean ± SEM from 5 platelet-poor plasma samples taken from rats. Statistical differences were determined using one-way ANOVA followed by the Bonferroni multiple comparison test. *P < .05 versus control. **P < .01 versus control. UFH indicates unfractionated heparin; FXa, factor Xa; OSCS, oversulfated chondroitin sulfate; ANOVA indicates analysis of variance; B, bovine; P, porcine; Sh, shark; S, skate; SEM, standard error of the mean.
Comparative ex vivo antiprotease profile of UFH/contaminant mixtures and a UFH/saline control in a chromogenic anti-FIIa assay UFH/OSCS, UFH/B, UFH/Sh, and UFH/P significantly inhibited FIIa when compared to the UFH/saline control. UFH/S inhibited factor IIa but to a lesser extent. The data represent the mean ± SEM from 5 platelet-poor plasma samples taken from rats. Statistical differences were determined using one-way ANOVA followed by the Bonferroni multiple comparison test. **P < .01 versus control. ***P < .001 versus control. aPTT indicates activated partial thromboplastin time; UFH, unfractionated heparin; FIIa, factor IIa; OSCS, oversulfated chondroitin sulfate; B, bovine; P, porcine; Sh, shark; S, skate; SEM, standard error of the mean.
Discussion
Heparin is one of the oldest and most widely used drugs. In 2008, a contaminant that mimicked heparin activity but produced numerous adverse effects was identified in the active pharmaceutical ingredient used to manufacture heparin. In the same year, 249 deaths were linked to the administration of heparin of which 149 of those deaths were associated with one or more allergic symptoms which were attributed to the contaminated heparin. 14 The contaminant was eventually identified as OSCS 8 and confirmed to be in finished product vials of heparin manufactured by Baxter Healthcare. 15 Additionally, the OSCS contaminant was shown to activate the kinin–kallikrein pathway in vitro and in vivo, 16 thereby establishing the possible link between the contaminant and the adverse reactions seen in affected patients.
Commercially manufactured heparin may be derived from various animal tissues; but in the United States, only porcine-derived heparin is approved. The OSCS contaminant is suspected to be synthetically prepared from naturally occurring CS. Chondroitin sulfate can be extracted from a variety of animal cartilages, including bovine, porcine, shark, and skate. 17 The exact animal source of OSCS found in batches of contaminated heparin is still not known; therefore we wanted to determine whether OSCS derived from different animal sources can affect heparin function in different ways.
The highest known concentration of OSCS found in the batches of contaminated heparin 8 was 30%. Therefore, we selected this concentration of contaminate to mix with heparin in order to observe the maximal effect the OSCS contaminant may have on the various properties of heparin in vitro and in vivo.
We wanted to test whether the addition of OSCS to heparin would cause changes in its anticoagulant and antiprotease activity; therefore, we supplemented mixtures of 70% UFH and 30% OSCS from various sources in NHP. Our in vitro data demonstrated that the addition of OSCS to heparin decreased its anticoagulant activity. There were no significant differences observed between the various sources of OSCS. Additionally, OSCS had no effect on the antiprotease activity as measured by an anti-FIIa assay.
One of the major side effects of heparin therapy is the risk of severe bleeding. Therefore, we wanted to test whether the addition of OSCS to heparin increased its potential hemorrhagic effects. To address this issue, we administered rats with 2 mg/kg IV mixtures of 70% UFH and 30% OSCS from various sources and a 70% UFH and 30% saline control. Oversulfated chondroitin sulfate from porcine, shark, and skate sources but not from bovine sources significantly increased bleeding in rats compared to control, indicating that the various sources of OSCS can differentially affect heparin’s hemorrhagic activity in vivo. Consequently, these data suggest that patients receiving contaminated heparin could have a greater potential risk of bleeding complications depending on the source of the OSCS.
Normally, CSs lack clinically relevant levels of anticoagulant activity due to their low levels of sulfation 18 ; however, oversulfated CSs with 2 to 3 sulfate groups per disaccharide unit have been shown to exhibit enhanced antithrombotic activity. 19 To test whether the addition of OSCS to UFH would increase antithrombotic activity, we used a rat jugular vein clamping model. As discussed previously, none of the UFH/contaminant mixtures increased the antithrombotic effect of heparin when compared to control.
Blossom et al 15 did a comprehensive analysis of the contaminated heparin clinical case reports and determined that one of the most common adverse events associated with the use of the contaminated heparin was hypotension. Therefore, we measured changes in blood pressure before and after administration of the UFH/contaminant mixtures in rats. Our data showed there was no significance difference compared to control. This finding was similar to what was presented by Kishimoto et al 16 which showed that not all rats treated with an UFH/contaminant mixture developed hypotension. Both findings are consistent with the fact that not all patients who were administered the contaminated heparin experienced adverse effects.
After treating the rats with the UFH/contaminant mixtures, whole blood and plasma were collected and analyzed. All the UFH/contaminant mixtures increased the whole blood aPTT up to 300 seconds, which was significantly higher compared to control. There were no changes observed in the aPTT performed on PPP. The different sources of OSCS, when added to UFH, were able to inhibit factor Xa and factor IIa but to varying degrees.
These studies demonstrate how the addition of OSCS to UFH can produce various changes in its anticoagulant and antithrombotic properties. In addition, various sources of OSCS have been shown to have an assay-dependent affect on UFH.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
