Abstract
Misclassification of an acute disease condition as chronic and vice versa by electrochemical sweat biomarker sensors can cause significant psychological, emotional, and financial stress among patients. To achieve higher accuracy in distinguishing between a chronic condition and an acute condition, there is a need to establish a reference biomarker to index the actual chronic disease biomarker of interest by combinatorial sensing. This work provides the first technological proof of leveraging the chloride ion content in sweat for a combinatorial sweat biomarker benchmarking scheme. In this scheme, the sweat chloride ion has been demonstrated as the reference/indexing biomarker, while sweat cortisol has been studied as the disease biomarker of interest. Label-free affinity biosensing is achieved by using a two-electrode electrochemical system on a flexible substrate suitable for wearable applications. The electrochemical stability of the fabricated electrodes for biosensing applications was studied by open-circuit potential measurements. Attenuated total reflectance–Fourier transform infrared spectroscopy spectra validate the crosslinker–antibody binding chemistry. Concentration-dependent analyte–capture probe binding induces a modulation in the electrical properties (charge transfer resistance and double-layer capacitance) at the electrode–sweat buffer interface, which are transduced by nonfaradaic electrochemical impedance spectroscopy (EIS). Calibration dose responses for the sensor for cortisol (5–200 ng/mL) and chloride (10–100 mM) detection were evaluated in synthetic (pH 6) and pooled human sweat (
Keywords
Introduction
In the past 6 years, there has been an explosion of innovation in healthcare. The key focus has been to provide rapid access to disease diagnosis anytime, anywhere. The experience of healthcare is fast changing, because the focus has shifted from the doctors and has become patient-centric. 1 Owing to faster lives and on-demand lifestyles, people are at comparatively greater risks of diseases and injury than before. Earlier, patients had to face long wait times to get an appointment with doctors, and the per patient visit time was equally short. Growing awareness among people about health risks has made them become more vigilant and responsible about their own health. Growing traction toward individualized and personalized medicine has paved the way for a boom for at-home monitoring healthcare devices, so that patients can keep track of their unique physiology as copious amounts of analyzable and transferrable data at any given point in time. 2 This has not only reduced visits to doctor’s offices but also solved the financial and psychological stress associated with not knowing what’s going on in one’s body. The shift from a reactive healthcare to P4 healthcare (predictive, preventive, personalized, and participatory) ecosystem, 3 the rise in the Internet of Medical Things (IoMT), Moore’s law and lowering smartphone prices, and greater internet connectivity have made this possible. It is estimated that by 2025, the average number of connected devices per person will be nine—most of which will be healthcare sensors. 4 Most users of these connected medical devices are not trained healthcare professionals or caregivers, and thus unlimited access to healthcare data can be counterproductive to patients if they are unable to collect and interpret these data with precision. The decision of whether to visit the doctor is now more at the discretion of the patients than their doctors. This necessitates high precision, reliability, and robustness in these at-home healthcare monitoring devices.
Among wearable devices, disease biomarker sensors capable of detecting the presence of a disease, and tracking its progression in body fluids like blood, urine, sweat, saliva, and tears, have gathered much traction throughout the years. 5 Noninvasive sampling and easy handling of sweat make it a suitable candidate biofluid for these applications.6–10 Furthermore, as the wearable sweat sensor technology advances in its development, strategies such as the use of smart materials for self-powering will further enhance their functionality in the near future.11–13 Among sweat-based wearable devices, electrochemical biosensors are considered most suitable for disease diagnosis and biological sample analysis, because they directly transduce the biochemical signals to faithfully quantifiable electrical outputs, are low in cost, and are easily integrable to portable wearable electronics due to miniaturization and higher sensitivity and signal-to-noise ratios.14–16 In the currently available sweat monitors, however, the focus is narrowed down to a single biomarker level. The influence of fluctuations of a sweat constituent, such as an electrolyte, on a given biomarker and the corresponding sensor readout is not accounted for. To achieve higher precision in preventive medicine, it is important that the sensing device is able to tell the difference between fluctuation in biomarker levels due to a serious chronic underlying pathophysiological condition and that due to a short-lived acute biomarker-level shift (false positives) and vice versa (false negatives). This is key for at-home sweat monitors because reduced false positives are essential, making early disease detection possible to retard or stop disease progression, while reduced false negatives save patients and their families from unnecessary emotional, financial, and psychological burdens. One way to reduce the possibility of such misclassification is by normalizing the disease biomarker of interest to some reference biomarker—when a person suffers from a transitory acute condition like dehydration, respiratory infection, or acidosis, variation in the sweat salt content may cause a global departure from homeostatic limits for all biomarkers in the patient’s sweat. A ratio of the disease biomarker of interest to the reference for such an acute condition will be almost equal to that observed for the benchmark healthy state. In contrast, if a person suffers from or is susceptible to a chronic underlying disease condition, then shifts in the levels of the biomarker for that disease will be significantly higher than that for the reference biomarker and the disease biomarker: The reference biomarker ratio will be significantly higher or lower, depending on the disease. To leverage the benefits of sweat biomarker normalization, combinatorial detection of the reference biomarker along with the disease biomarker of interest for a given patient sweat sample can save the day. This method of biomarker benchmarking and normalization has been used for detecting urinary diseases like chronic kidney disease and acute kidney injury, in which relevant urinary biomarkers are normalized to urinary creatinine (UCr) to account for fluctuation in biomarker concentrations due to variation in time of urine collection, urine concentration, urine flow rate, and so on. 17 Sweat harbors a wealth of physiological information in the form of biomarkers like proteins, electrolytes, and neuropeptides, but to our knowledge, no biomarker benchmarking scheme exists for electrochemical biosensing based sweat diagnostics.
This article serves as the first-time demonstration of combinatorial sensing of a reference biomarker in addition to the disease biomarker of interest for sweat-based electrochemical biosensing. For the proof of concept, sweat chloride—the most abundant anionic electrolyte in the extracellular fluid matrix—was chosen as the reference or benchmarking biomarker because it fluctuates with acute conditions like dehydration, acute respiratory acidosis, and respiratory tract infection.18–21 Sweat cortisol, a biomarker associated with a host of diseases associated with urbanized diets, environment, and lifestyle like chronic stress, obesity, type 2 diabetes, heart disease, gut and immune issues, inflammation, and insomnia, was selected as the “disease biomarker of interest” for this study.22–27 The electrochemical sensor, in this work, is a simple coplanar two-electrode system deposited on a flexible nanoporous substrate to leverage the benefits of nanoconfinement, resulting in surface area–to-volume enhancement and higher sensitivity. Detection of chloride (1–100 mM) and cortisol levels (5–200 ng/mL) was achieved in ultralow volumes (~1 µL) of pooled human sweat (three donors) and synthetic sweat buffer of pH 6. Nonfaradaic electrochemical impedance spectroscopy (EIS), an AC-based electroanalytical technique, was used to transduce the changes in concentration in the sweat sample to changes in impedance at the frequency of interest.28–30 Nonfaradaic EIS-based sensor calibration allows for reliably tracking the modulation in the capacitive behavior at the electrode–buffer interface due to analyte–capture probe binding events, driven by the biomarker concentration, with high resolution.31,32 Optimization of sweat volume required for reliable output was done by wicking tests. Open-circuit potential (OCP) measurements were done to probe the electrochemical stability of the electrode system.28,30 Attenuated total reflectance–Fourier transform infrared spectroscopy (ATR–FTIR) analyses were done to optimize the incubation times and concentration of the crosslinkers and capture probes for the cortisol-sensing assay. To further underline the necessity of detection of the reference biomarker along with the disease biomarker of interest, combinatorial detection of cortisol levels for sweat samples containing low (healthy) and high chloride content (dehydrated), to capture both bookends, was demonstrated. The effect of an acute condition such as dehydration, simulated as sweat chloride fluctuation, on the reported cortisol concentration was studied to emphasize that both absolute and normalized values of a disease biomarker should be reported to get the truest idea of disease onset or progression.
Materials and Methods
Materials and Reagents
The crosslinker DSP [dithiobis (succinimidyl propionate)] and the solvent it is dissolved in, DMSO, were obtained from Thermo Fisher Scientific (Waltham, MA). Cortisol (hydrocortisone) and the monoclonal α-cortisol antibody were purchased from Abcam (Cambridge, MA). Chloride ionophore IV Selectophore (4,5-Bis-[N′-(butyl) thioureido]-2,7-di-tert-butyl-9,9-dimethylxanthene), which was used as the capture probe for chloride ions in sweat, was purchased from Sigma-Aldrich (St. Louis, MO). Ionophore was dissolved in DMSO and vortexed for 30 min at 1000 rpm to obtain a clear solution. Pooled human sweat from three donors was obtained from Lee Biosolutions (Maryland Heights, MO) and stored at −20 °C without the addition of any preservatives. Potassium chloride (KCl) was used to prepared desired chloride doses and was ordered from Thermo Fisher Scientific. Synthetic sweat (pH 6) was prepared as per the recipe described by Mathew et al. for the calibration of the cortisol sensor. 33 For the chloride sensor calibration using synthetic sweat, the same recipe was followed minus the sodium chloride (NaCl) and KCl dissolution. Lactic acid (85%) and ammonia (NH3), used to tune the pH of the synthetic sweat buffer, were obtained from Sigma-Aldrich. Urea was obtained from Thermo Fisher Scientific . The coplanar microelectrode sensor system was a silver screen-printed standard two-electrode system deposited on nanoporous flexible hydrophilic substrate.
Fluid-Wicking Study
To ensure uniform spreading of the dispensed sweat sample on the sensor, a hydrophilic substrate was the suitable choice.
34
On evaluation of the sensing area, size and the dynamic concentration range of the biomarker, wicking profiles for volumes throughout the range of 0.5–5 µL were evaluated, as shown in
Open-Circuit Potential Measurements
For simplicity, a two-electrode system was chosen for this study. The electrochemical stability of the two-electrode system was probed by measuring the OCP of the system in a 1× phosphate-buffered saline (PBS) buffer medium for a duration of 1 h to ensure that a steady state is reached. For OCP measurements, a high-impedance voltmeter is connected in parallel to the working electrode (WE) and the reference electrode (RE) to measure the inherent potential gradient when no current flows (Gamry Instruments, Warminster, PA). 30
ATR–FTIR Analyses
Visualization of the binding chemistry between the capture probe and the crosslinker and the optimization of incubation times and concentration were achieved via ATR–FTIR. The crosslinker was anchored to the silver electrode by strong thiol bonds. The spectra were obtained by using Nicolet 6700 FTIR (Thermo Fisher Scientific). To validate the cortisol assay binding chemistry, a liquid sample containing a DSP (10 mM)–cortisol antibody (10 µg/mL) complex, incubated for 1 h, was studied.
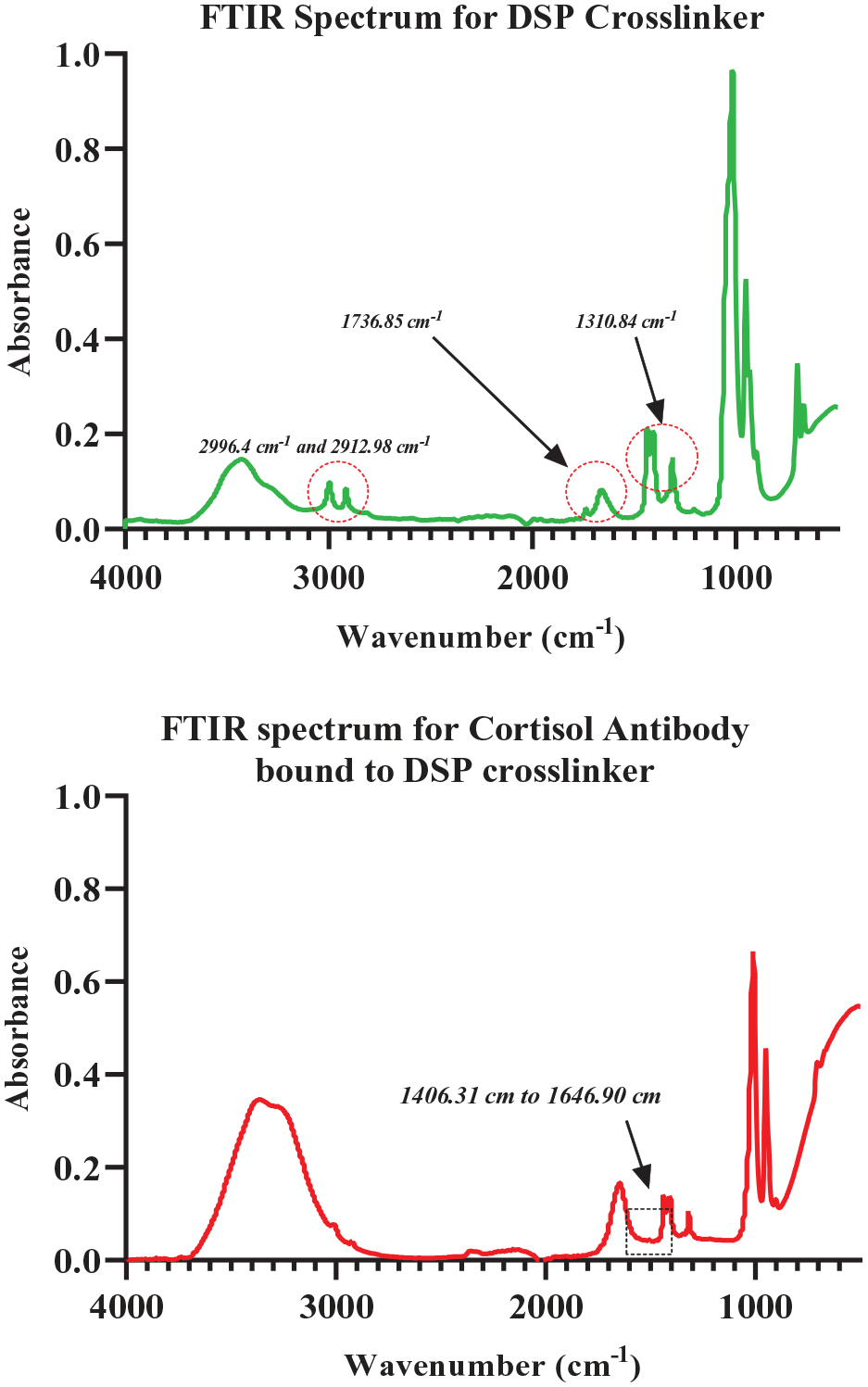
Infrared (IR) spectra for a crosslinker and a disease biomarker capture probe.
Sensor Calibration for the Detection of Chloride in Synthetic and Pooled Human Sweat
The sensor stack was optimized for chloride detection in pooled human sweat and synthetic sweat (pH 6) buffers. A self-assembled monolayer of the 10 mM DSP crosslinker was formed on the conducting electrode/sensing area by functionalizing it for an optimized incubation duration and volume of 2 h and 15 µL, respectively, in darkness. This was followed by the functionalization of 15 µL of 30 mM chloride ionophore for ~1 h. Strong multitopic hydrogen binding causes chloride ions in sweat to preferentially bind to the chloride ionophore capture probe.35,36 One microliter of synthetic sweat at pH 6 (not containing chloride ions) was placed onto the sensing region. This was considered as the baseline or zero-dose step for measuring changes in impedance values. Potassium chloride was dissolved in zero-dose synthetic sweat buffer to obtain sweat chloride solutions in the dynamic range of 1–100 mM. One-microliter chloride doses were dispensed in increasing concentration from 1 mM to 100 mM. Approximately one minute after every dose-dispensing step, a single-frequency EIS measurement was taken to determine the modulus of impedance corresponding to the level of the chloride in the detection buffer. A similar protocol was followed for the pooled human sweat solution, in which the blank sweat solution was taken as the zero dose and was spiked with potassium chloride to obtain 1–100 mM human sweat chloride solutions. Calibration dose–response curves for chloride detection in synthetic sweat (
Sensor Calibration for the Detection of Cortisol in Synthetic and Pooled Human Sweat
Calibration dose responses were obtained for cortisol detection in synthetic sweat (n = 3) and pooled human sweat (n = 4) buffers for a dynamic cortisol range of 5–200 ng/mL. The cortisol-sensing immunoassay has been adapted from a protocol described in a previous work done by our group. 37 Fifteen microliters of 10 mM of DSP crosslinker were functionalized on the electrode sensing area by 2 h of incubation in darkness. This was followed by the immobilization of 10 µg/mL of cortisol antibody for 30 min, for a volume of 15 µL.
Highly specific antibody–antigen immunochemistry is used to capture cortisol in the sensing stack. Sensor calibration and data reporting were done in a manner similar to the process described for the chloride sensor.
Combinatorial Detection of Cortisol and Chloride: The Need for Sweat Biomarker Benchmarking
A 1:1 concoction of pooled human sweat, spiked with the desired chloride levels (both high and low) and cortisol concentrations, was prepared. Here, the effect of the fluctuation of chloride on the cortisol concentration, reported as the impedance change from baseline by the cortisol sensor, was evaluated. Pooled human sweat solutions containing chloride solutions at concentrations of 10 mM (healthy) and 70 mM (dehydrated-acute condition) devoid of cortisol were considered as zero doses or baselines for the low-chloride and high-chloride cases, respectively. The effect of chloride was observed for low- (5 ng/mL), medium- (100 ng/mM), and high-cortisol (150 ng/mL) doses spiked in both low-chloride and high-chloride baseline doses. The table depicted in
Results and Discussion
Characterization of the Combinatorial Sensor and Affinity Capture Assay
The wicking profiles of the sweat sample were studied for sample volumes ranging from 0.5 to 5 µL. It is evident from
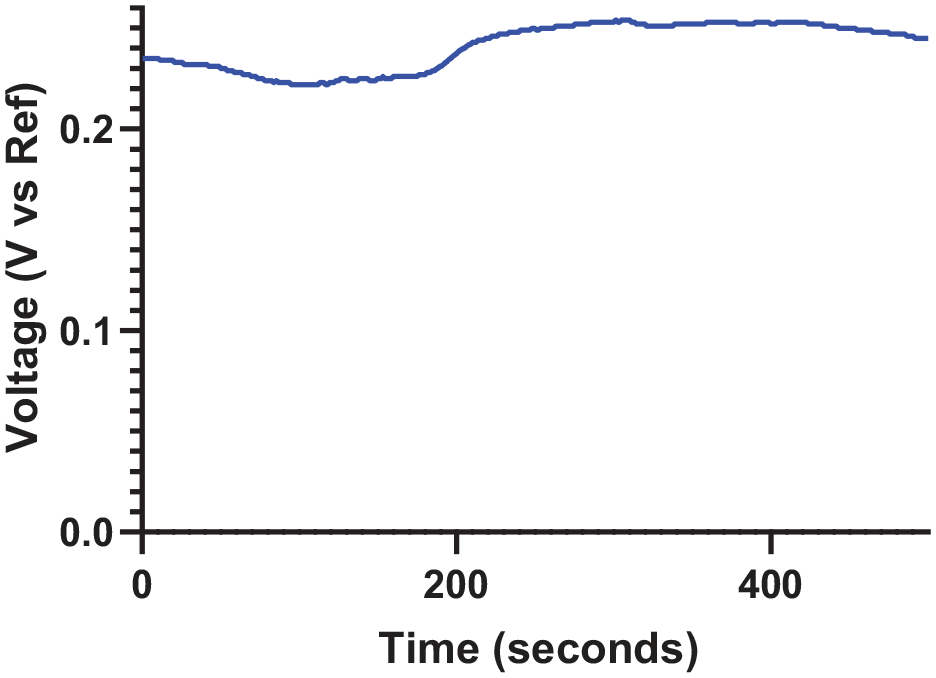
Open-circuit potential for the two-electrode system.
ATR–FTIR Analysis
ATR–FTIR spectra obtained for DSP and cortisol antibody bound to the DSP crosslinker validate their successful functionalization and binding (
Sensor Performance Evaluation for Cortisol Detection in Synthetic and Pooled Human Sweat
The physiologically relevant cortisol level in human sweat ranges from 8 to 140 ng/mL. Calibrated sensor dose responses for cortisol detection in synthetic sweat and pooled human sweat are depicted in
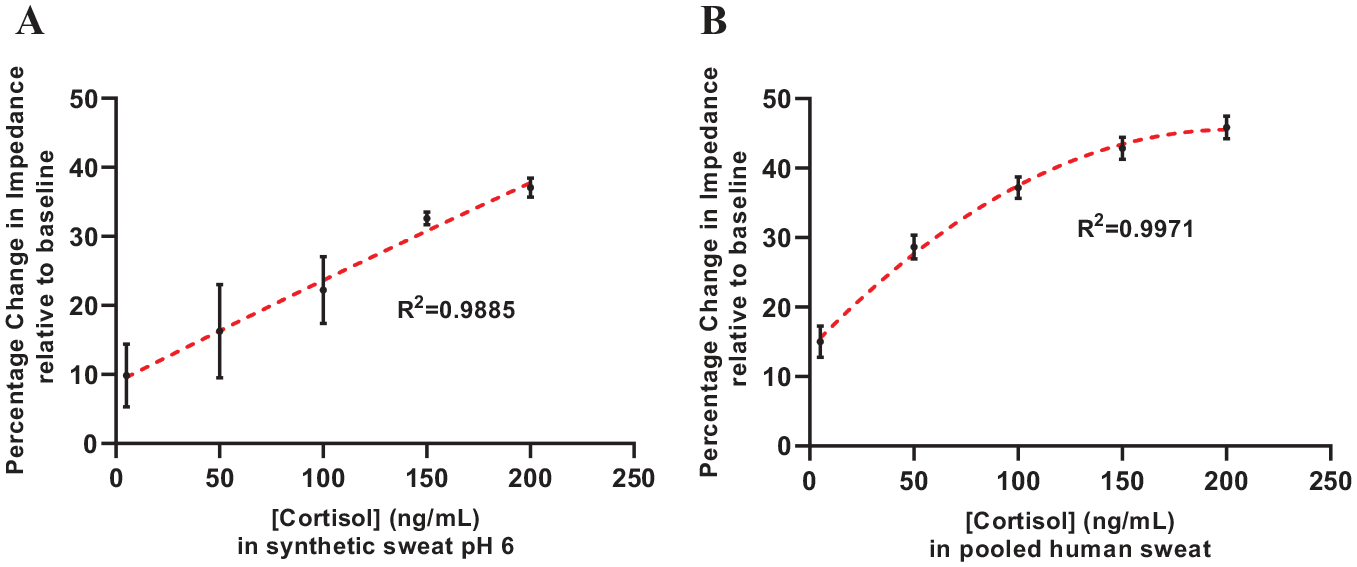
Calibration dose–response curve for cortisol spiked in (
Sensor Performance Evaluation for Chloride Detection in Synthetic and Pooled Human Sweat
Sensor dose responses for chloride detection in synthetic sweat and pooled human sweat were studied for a dynamic range of 10–100 mM sweat chloride. Linear calibration dose–response curves were obtained with
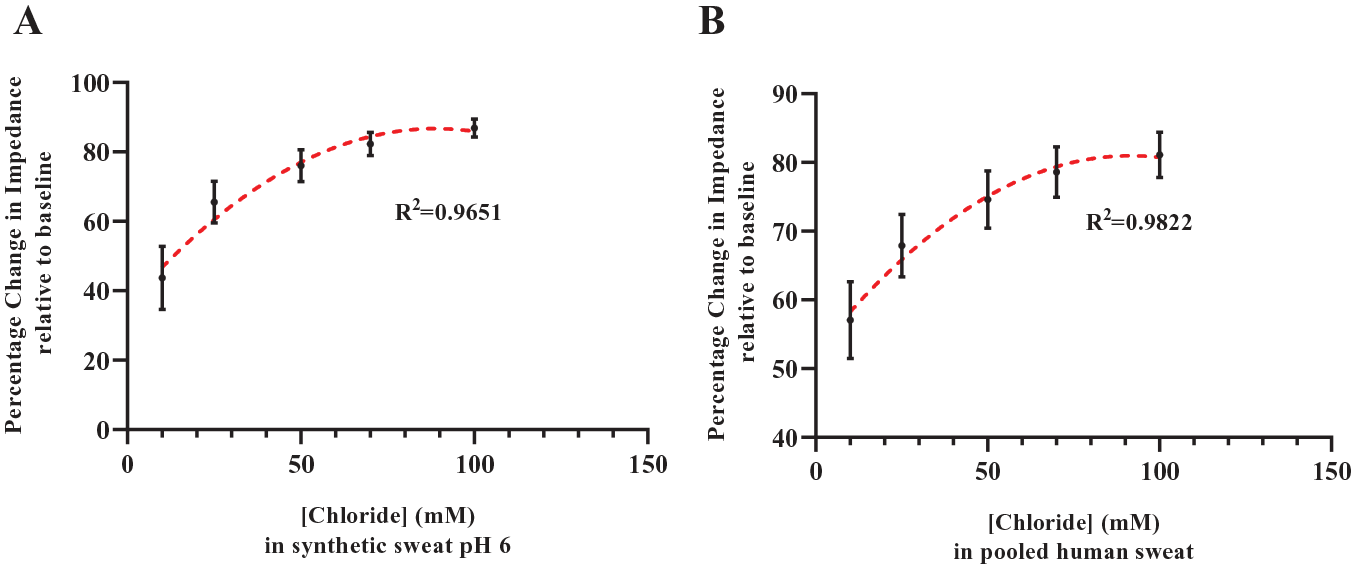
Calibration dose–response curve for chloride spiked in (
Sensor Performance Evaluation for Combinatorial Detection of Disease and Reference Biomarkers in Pooled Human Sweat
As shown in
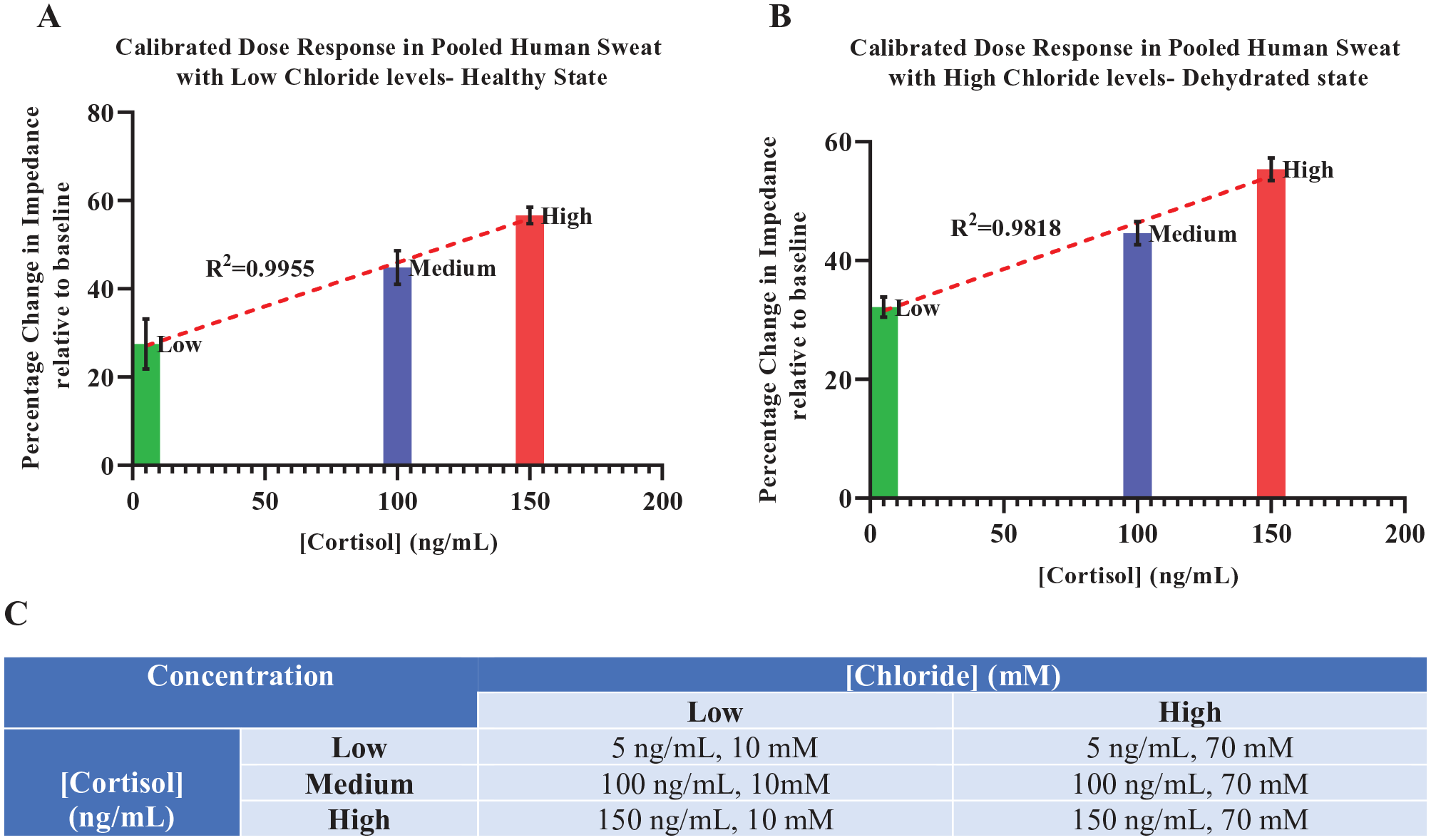
Calibration dose–response curve for cortisol for a relative impedance modulus value of 180 Hz for (
Conclusion
With the rapid rise of IoMT devices, easier internet connectivity, and an increase in diseases due to urbanized lifestyles, an increase in vigilance among patients toward their own health is being witnessed. We are inching toward a world in which patients generate, store, and securely share volumes of medical data by continuously obtaining their physiological snapshots in real time using these at-home monitoring devices. Since the patients are enabled by these devices to interpret their medical data and plan hospital visits accordingly, it is of utmost importance that these devices report with the highest possible fidelity and accuracy. A large chunk of these devices are electrochemical sweat biosensors, which suffer from issues of false alarms and false negatives due to inaccurate measurements of biomarker levels. This work attempts to address this issue by proposing a combinatorial electrochemical sweat-biosensing scheme, which points out the need to report not only the absolute disease biomarker levels but their levels relative to a suitable reference biomarker as well. As a technological proof, combinatorial detection of cortisol hormone along with chloride ion, selected as the indexing marker, was studied in sweat samples. Linear calibration dose responses for chloride and cortisol detection and their combinatorial detection were obtained using a standard metal coplanar two-electrode electrochemical cell. Validation of binding chemistry between the cortisol capture probe and the thiol crosslinker was studied using ATR–FTIR analysis. Variation in the reported cortisol impedance due to sweat chloride ions has been demonstrated, which proves that more precise and robust electrochemical biosensors can be designed by the proposed combinatorial benchmarking sensing scheme. It was observed that the fluctuation in chloride ions affected the reported impedance change corresponding to the cortisol concentration, which may result in misleading diagnoses. To combat this issue, a benchmarking solution is proposed in which a ratio of the disease biomarker to a suitable reference biomarker is considered. It was found that on normalization, a clear distinction between acute and chronic disease conditions could be made, because the chronic disease states showed a significant change from the benchmark ratio while the acute conditions showed negligible change. Thus, we can move a step closer to more precise sweat diagnosis, particularly for at-home monitoring, by using the proposed indexing scheme based on sweat biomarker normalization.
Supplemental Material
Supplemental_Material_for_A_Combinatorial_Electrochemical_Biosensor_for_Sweat_Biomarker_Benchmarking_by_Ganguly,_et_al – Supplemental material for A Combinatorial Electrochemical Biosensor for Sweat Biomarker Benchmarking
Supplemental material, Supplemental_Material_for_A_Combinatorial_Electrochemical_Biosensor_for_Sweat_Biomarker_Benchmarking_by_Ganguly,_et_al for A Combinatorial Electrochemical Biosensor for Sweat Biomarker Benchmarking by Antra Ganguly, Paul Rice, Kai-Chun Lin, Sriram Muthukumar and Shalini Prasad in SLAS Technology
Footnotes
Supplemental material is available online with this article.
Author Contributions
A.G. and S.P. conceived the project framework. A.G. performed the modeling and experiments. A.G., P.R., K.C.L., S.M., and S.P. optimized the fabrication protocol and assisted in the fabrication process. A.G. and S.P. wrote the article.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Shalini Prasad and Dr. Sriram Muthukumar have a significant interest in EnLiSense LLC, a company that may have a commercial interest in the results of this research and technology. The potential individual conflict of interest has been reviewed and managed by the University of Texas at Dallas, and it played no role in the study design; the collection, analysis, and interpretation of data; the writing of the article; or the decision to submit the article for publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
