Abstract
In vitro fertilization (IVF) has been one of the most exciting modern medical technologies. It has transformed the landscape of human infertility treatment. However, current IVF procedures still provide limited accessibility and affordability to most infertile couples because of the multiple cumbersome processes and heavy dependence on technically skilled personnel. Microfluidics technology offers unique opportunities to automate IVF procedures, reduce stress imposed upon gametes and embryos, and minimize the operator-to-operator variability. This article describes the rapidly evolving state of the application of microfluidics technology in the field of IVF, summarizes the diverse angles of how microfluidics has been complementing or transforming current IVF protocols, and discusses the challenges that motivate continued innovation in this field.
Introduction
In vitro fertilization (IVF) is a complex series of procedures used to treat infertility or genetic problems and assist with the conception of a child. 1 It is the most effective form of assisted reproductive technology (ART). During IVF, mature oocytes are retrieved from the patient’s ovaries and fertilized by a sperm in a lab. Then the fertilized oocyte is implanted into the patient’s uterus. Current IVF procedures include the selection of motile sperm, retrieval and processing of oocytes, insemination, in vitro embryo culture, and/or cryopreservation. Two main approaches of insemination in the lab are conventional IVF2,3 and intracytoplasmic sperm injection (ICSI). 4 During conventional IVF, each single oocyte is placed in an oil-covered microdrop in a culture dish in the presence of an optimal concentration of sperm, and fertilization occurs when one sperm penetrates the oocyte naturally. ICSI, on the other hand, is a technique in which a single sperm is injected directly into the oocyte’s cytoplasm through a thin needle for fertilization. ICSI has been the dominant treatment for almost all forms of male infertility, and it has also been increasingly adopted to overcome fertilization failure. 4 In 2012, for example, ICSI was used in 93.3% of fresh IVF procedures (i.e., using non–freeze-thawed embryos) to treat male factor infertility and was used in 76.2% of all fresh IVF procedures with and without male factor infertility. 5
According to the Centers for Disease Control and Prevention, a total of 182,111 IVF procedures with the intent to transfer at least one embryo were performed in the United States in 2015, contributing to 1.7% of all infants born in the United States during the same year. 6 Despite its immense potential of overcoming human fertilization failure, IVF treatment still remains inaccessible and unaffordable to the majority of infertile couples around the world, especially in resource-limited countries and areas. Current IVF procedures suffer from a variety of limitations, 7 such as being costly and time-consuming. 8 The IVF procedures also rely heavily on highly skilled embryologists, which often results in variation from operator to operator and a lack of standardization. 7
However, we can gain leverage on addressing the above challenges facing IVF applications by incorporating miniaturization and automation technologies that have emerged in recent decades, such as microfluidics. Microfluidics technology manipulates and controls fluids of microliters to picoliters within a network of channels with dimensions from tens to hundreds of micrometers. Microfluidics has many unique advantages, including reduced reagent volume, shorter reaction time, and the scalability for parallel operation. 9 Miniaturization and automation offered by microfluidics can further improve the precision of experiments, lower the limits of detection, and allow performance of techniques and experiments that are usually not possible on the macroscale. 10
Over the past two decades, microfluidics technology has demonstrated great potential for streamlining IVF procedures. 11 Microfluidics provides an alternative to almost every single step in the process of IVF, including but not limited to improved selection of motile sperm, automated processing of oocytes, in vitro follicle growth, embryo culture, and cryopreservation. Furthermore, the capability of reproducing a given single IVF procedure on an isolated microfluidic module offers the opportunity to investigate and understand the reproductive physiology in each step of assisted human reproduction.
In the literature, there have been several outstanding reviews discussing the applications of microfluidics in assisted human reproduction.7,8,11–13 However, the strong synergy between microfluidics and IVF has been constantly driving the evolution of concepts, designs, methodology, and applications in the field. Many new microfluidic approaches were proposed and investigated in the past 2 y, which has brought unprecedented outcomes or served the unmet needs for automating IVF procedures.14–25 This article reviews recent progress in employing microfluidics to process gametes, perform IVF, and/or culture embryos. It also discusses the state of the art of microfluidics-facilitated cryopreservation of gametes and embryos for IVF. In the end, several challenges and opportunities are outlined to motivate the continued innovation of microfluidics with implications for IVF.
Improved Selection of Motile Sperm on a Chip
The fundamental challenge of sperm selection for IVF is dictated by biology: a heterogeneous population of ∼108 sperm per milliliter with a short lifetime in vitro. Traditional sperm selection methods such as swim-up and density gradient centrifugation are not only time and labor intensive but also result in low yield of motile sperm or high DNA fragmentation. 26 To address these challenges, microfluidics has been integrated with procedures such as semen analysis for male infertility diagnosis27,28 and sperm selection based on motility14,29–33 and/or facilitated by chemotaxis,15,34,35 rheotaxis,36–38 thermotaxis,15,39 or inertial focusing. 40
Asghar et al.
31
developed a sperm-sorting device in which only the most motile and functional sperm could pass through a polycarbonate membrane filter against gravity and swim into a retrieval reservoir on top of the filter, leaving dead and less motile sperm behind (
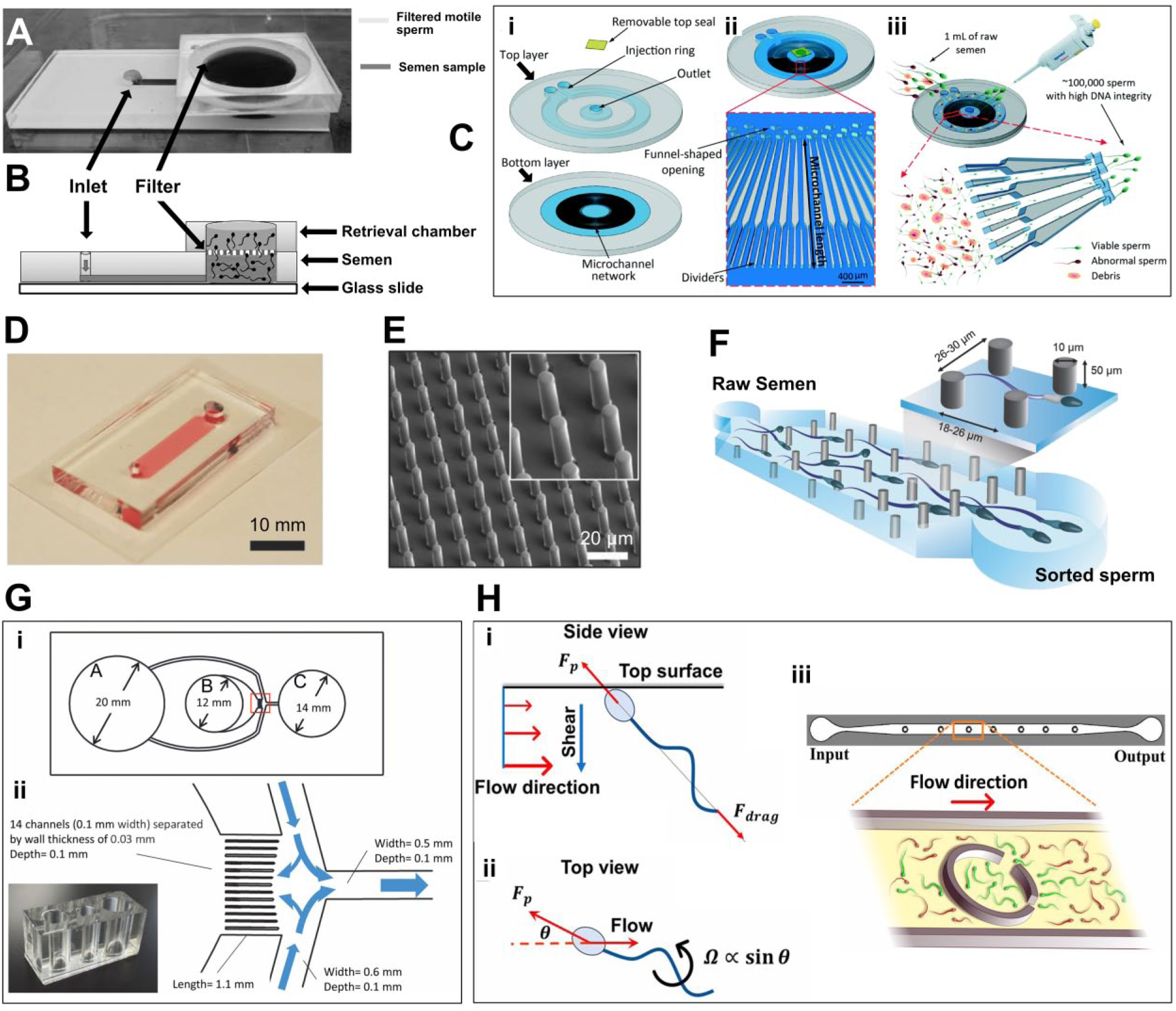
Recent microfluidic systems for sorting motile sperm. (
Chinnasamy et al.
14
developed a microfluidic device, named the Simple Periodic ARray for Trapping And isolatioN (SPARTAN), in which they designed an array of pillars to efficiently isolate motile and morphologically normal sperm from raw semen with lower epigenetic global methylation (
Furthermore, Nagata et al.
25
developed a diffuser-type microfluidic sperm sorter to select functional and progressively motile bovine sperm with high DNA integrity (
Using human and bovine semen, Zaferani et al.
24
demonstrated a different high-throughput microfluidic device that could passively isolate motile sperm within corrals inside a fluid channel, separating them from the rest of the diluted sample (
It is challenging to isolate the most motile and presumably healthiest sperm from semen samples that have low sperm counts and/or low sperm motility. Microfluidic systems discussed previously have shown potential to sort motile sperm with flow systems. However, the small field of view (FOV) of conventional microscopes commonly used to image sperm motion limits the number of sperm cells that can be tracked simultaneously. 29 To address this challenge, Zhang et al. 29 integrated a lensless charge-coupled device with a microfluidic device to enable wide FOV and automatic recording as the sperm moved inside a microfluidic channel. The integrated system enabled the sorting and tracking of a population of sperm within a microfluidic channel. This channel was able to be monitored in both a horizontal and vertical configuration similar to the swim-up column method. Sperm motility was quantified by tracing the shadow paths for individual sperm. Moreover, motile sperm that reached the outlet were conveniently retrieved from the channel at the end of the process. This technological approach was suggested to be highly useful for isolating motile sperm from a pool of a small number of sperm confounded by low sperm motility. 29
Microfluidics is also a powerful tool to understand how sperm compete for insemination of the oocyte in vivo. Very recently, Zaferani et al. 17 revealed that sperm with the highest motility were most likely to make it through the narrow junctions inside the female reproductive tract, by using an “hourglass” shape microfluidic device to simulate the fluid mechanical properties of these narrow junctions in vitro. These pinch points were found to prevent sperm with a motility below a certain threshold from advancing through the pinch point to the fertilization site, as the slower sperm accumulated in front of the pinch point and were caught in oncoming currents.
Microfluidics-Facilitated Processing of Oocytes
In the ovary, individual oocytes are natively surrounded by cumulus cells (specialized granulosa cells), forming an organized structure known as the cumulus-oocyte complex (COC). The cumulus cells that are in close contact with the oocyte, also known as corona cells, develop cytoplasmic projections that cross the zona pellucida and form gap junctions with the oolemma. 41 In vivo, the cumulus cell mass is naturally removed from the oocyte upon fertilization. Following conventional IVF, the embryo has to be denuded from the cumulus cell mass within the first 24 h because the by-products from the interaction between hyaluronidase and cumulus cells can damage the zona pellucida and decrease embryo viability. 42 Furthermore, oocytes must be denuded before ICSI to allow the evaluation of oocyte morphology including the nuclear maturation stage 41 and facilitate the injection of a sperm. 43 In IVF clinics, oocytes are usually denuded from the cumulus cell mass via a combination of enzymatic action of hyaluronidase and mechanical pipetting, which are inefficient and suffer from operator-to-operator variation. However, microfluidics technology has helped automate the denudation procedures, which has the potential to reduce time and cost as well as improve standardization.
Zeringue et al.44,45 developed a microfluidic device to mechanically remove the cumulus cell mass from single bovine zygotes (
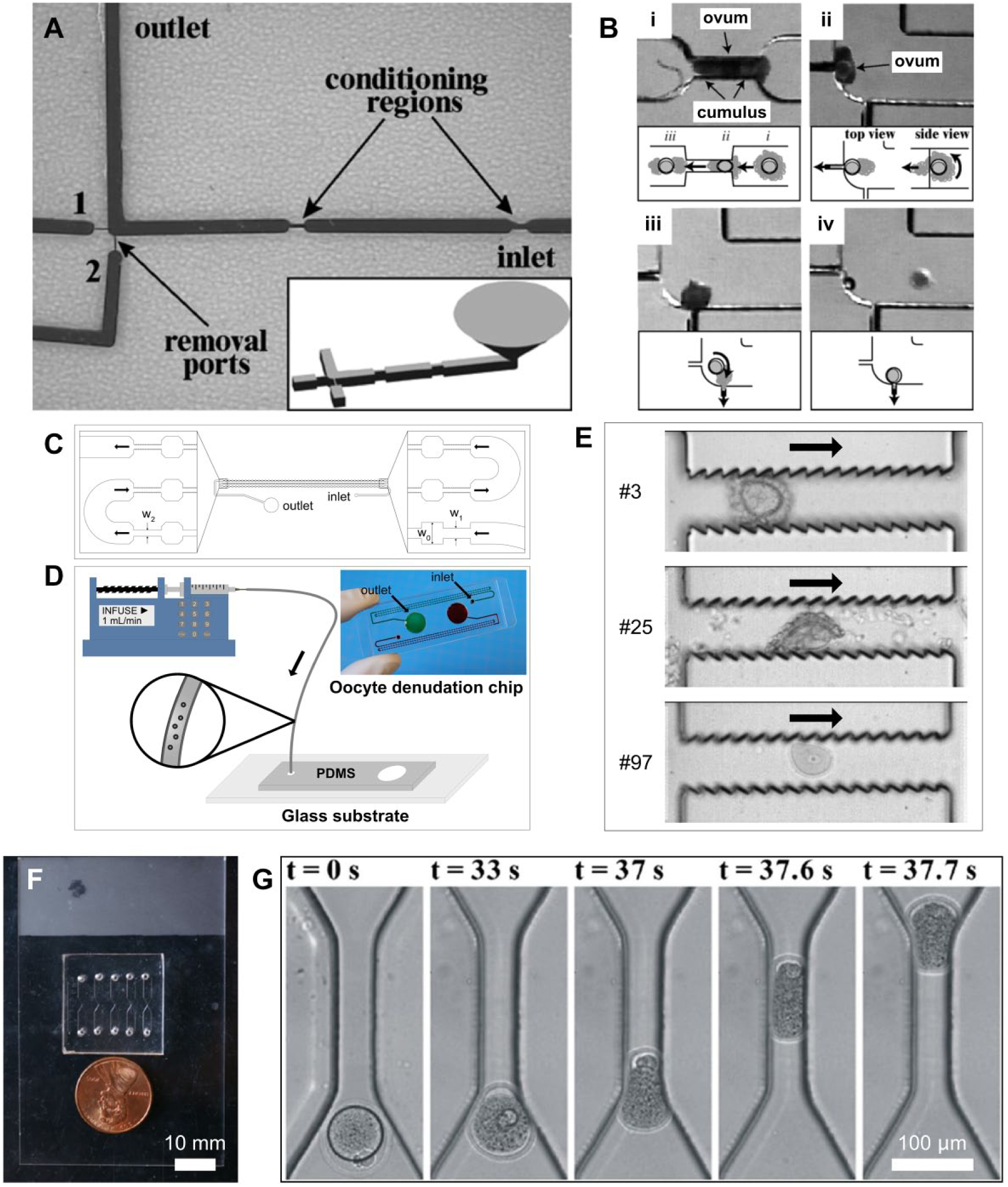
Current microfluidic systems for processing of female gametes and zygotes. (
Very recently, Weng et al.
16
developed a microfluidic device to denude mouse oocytes from the surrounding cumulus cell mass, facilitating the evaluation of oocyte quality and the injection of sperm for ICSI (
While the oocytes travel through microchannels, mechanical stress arises from the physical contact between the cells and channel walls, which may damage the fertilization capacity of processed oocytes. Luo et al.
46
studied the deformation of mouse oocytes under shear flow and its subsequent impact on their spindle structure as the oocytes traveled through constricted microfluidic channels of different sizes at various flow rates (
Taking advantage of the correlation between oocyte quality and its sedimentation rate, Iwasaki et al. 22 developed a microfluidic device to separate high-quality bovine oocytes to improve the conception rate of IVF. Their device featured a long separation channel of 10 mm long, 700 µm wide, and 1 mm high. After denuded oocytes were injected into the separation channel, they traveled toward the outlet at an optimal flow rate controlled by a syringe pump and settled gradually. The outlets of the separation channel were divided into upper and lower reservoirs. High-quality oocytes settled faster than poor-quality ones in a phosphate-buffered saline buffer with 0.05 wt% polyvinyl alcohol and 0.3 M sucrose. Thus, high-quality oocytes were collected from the lower outlet reservoir. IVF results showed that the developmental rate of blastocysts of oocytes collected from the lower outlet (36.0%) was significantly higher than those collected from the upper outlet (14.1%).
In Vitro Growth of Ovarian Follicles on a Chip
In vitro culturing of ovarian follicles provides a promising strategy to restore fertility for female patients facing premature infertility due to cancer therapies. Woodruff and coworkers47–49 developed a three-dimensional (3D) culture system to support in vitro development of follicles, producing mature oocytes of fertilization capacity. Immature follicles were encapsulated within sterile alginate beads, which maintained the oocyte’s 3D architecture and cell-cell interactions.47–49 On the other hand, microfluidics approaches of encapsulation of ovarian follicles for 3D culture have also been reviewed previously. 50 A flow-focusing microfluidics technique was used to generate core-shell droplets encapsulating Peromyscus early preantral follicles. 50 The collagen core and alginate shell of these hydrogel droplets mimicked the cortex and medulla, respectively, reproducing the native milieu in a mammalian ovary, as mechanical heterogeneity was found to be critical in regulating follicle growth. 50 The results showed that 5 MII oocytes out of 11 COCs (45.5%) were obtained by culturing early preantral follicles in the optimal biomimetic ovarian microtissue with a 0.5% collagen core and 2% alginate hydrogel shell. Moreover, these MII oocytes and in vitro maturation of the COCs could be further activated to obtain two-cell stage embryos.
Xiao et al. 51 developed a microfluidic system to support mouse ovarian follicles to produce the human 28-d menstrual cycle hormone profile, which controlled human female reproductive tract and peripheral tissue dynamics in single, dual, or multiple unit microfluidic platforms. Mouse ovarian tissue was cultured on a microfluidic platform based on pneumatic actuation, simulating the in vivo female reproductive tract and the endocrine loops between organ modules including ovary, fallopian tube, uterus, cervix, and liver. 51 This microfluidic platform achieved organ-organ integration of hormonal signaling and pregnancy-like endocrine loops and also presented a powerful tool for drug discovery and toxicology research.
IVF and Embryo Culture on a Chip
An integrated microfluidic system has the prospect of achieving IVF and embryo incubation with reduced human intervention, high efficiency, and minimal operator-to-operator variation. Suh et al. 52 demonstrated in vitro insemination, co-incubation, and fertilization of murine oocytes in a microfluidic chamber. This microfluidic system featured a barrier gate that allowed only media and sperm to flow through, whereas the oocyte was blocked without deformation. Such selective passage resulted in the increase in the local sperm concentration around the trapped oocyte. Therefore, the sperm-oocyte interaction was increased compared with the microdrop-in-a-dish technique. This study demonstrated that a smaller total number (i.e., 1000–4000) and a lower concentration of sperm (i.e., 2 × 104 to 8 × 104 sperm/mL) were required to achieve a similar fertilization rate to the corresponding control using the microdrop-in-a-dish technique, which required an optimal concentration of 1 × 106 sperm/mL. Similarly, using a geometrical constraint to block the oocyte within a microfluidic chamber, Clark et al. 53 demonstrated that the incidence of polyspermy IVF of porcine oocytes was statistically significantly lower (p < 0.05) than the oocytes fertilized in the traditional microdrop system, whereas the sperm penetration and male pronucleus formation rates were comparable. The flow velocity and profile of sperm traveling through this microfluidic chamber were investigated in a follow-up study by Lopez-Garcia et al. 54 It was suggested that the sperm-oocyte interaction could be controlled by regulating the local flow rate and adjusting the channel geometry, thereby further reducing polyspermy by maintaining a low sperm concentration around the trapped oocyte.
Han et al.
55
developed a microfluidic chamber featuring an array of microwells that could facilitate single oocyte trapping, fertilization, and embryo culture (
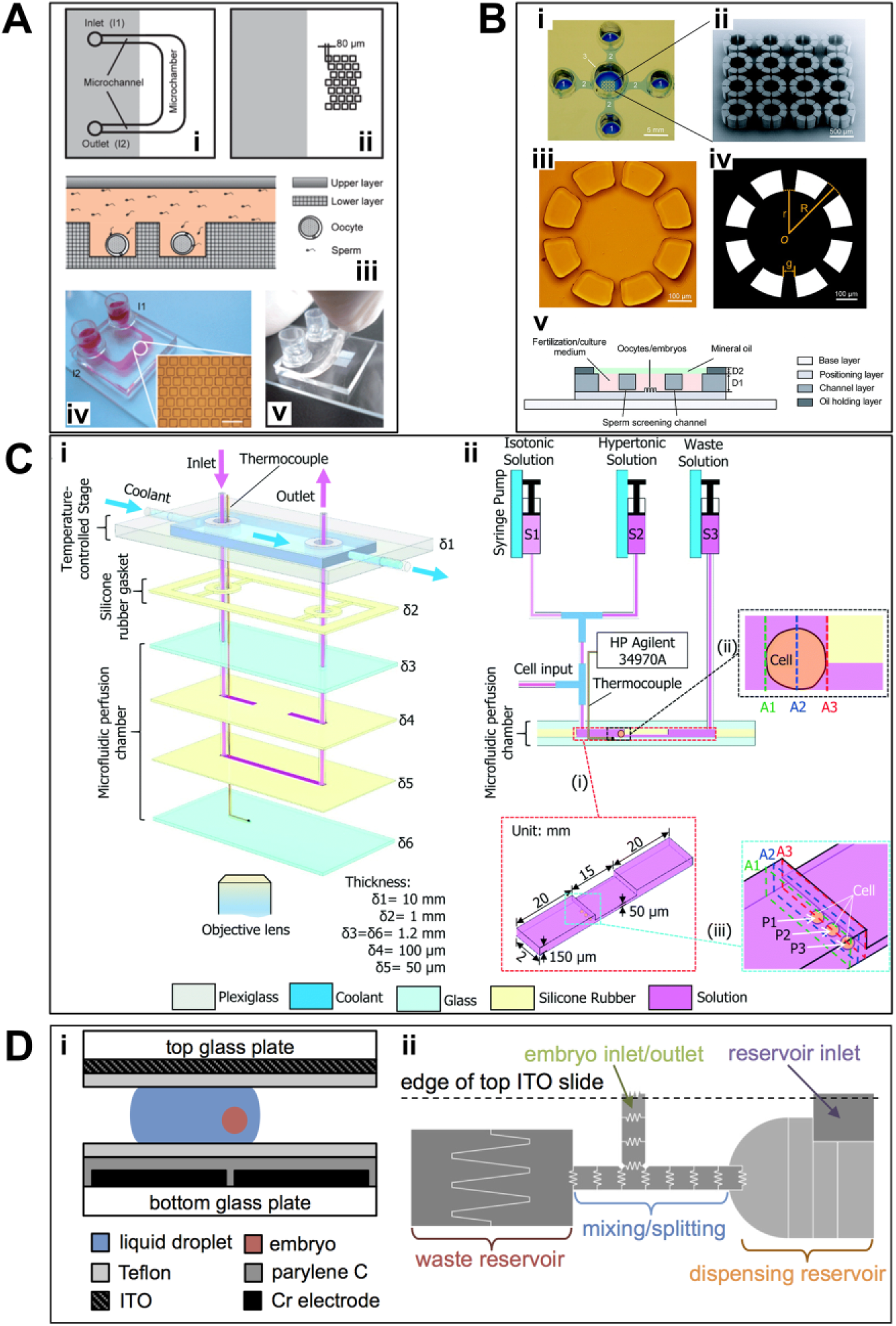
Recent microfluidic systems for manipulation or cryopreservation of oocytes/embryos. (
Furthermore, Ma et al.
57
developed a different microfluidic device that integrated each step of IVF, including oocyte positioning, sperm screening, fertilization, medium replacement, and embryo culture. Individual oocytes were positioned in a 4 × 4 array of octa-column units (
Once the embryo is formed, it takes nearly a week to travel down the fallopian tube to enter the uterus. While it travels in the oviduct, the fertilized egg is subjected to dynamic physical stimulation such as muscular contraction. 61 Kim et al. 61 developed a microfluidic in vitro culture system for bovine embryos, in which peristaltic muscle contraction was mimicked by using a partially constricted channel. The system also generated a gravity-driven dynamic flow using a micro-modulated tilting machine. The results showed that the proportion of eight-cell development among total embryos in the constricted channel (56.7%) more than doubled that in the straight channel (23.9%), indicating the vital role of constriction in the early development of bovine embryos after IVF.
Role of Microfluidics in Cryopreservation of Gametes and Embryos
Cryopreservation of gametes and embryos is highly desirable for many reasons, primarily to store excess genetic material and to control the timing and precision of embryo transfer. 62 As IVF was introduced in the clinics to treat human infertility, cryopreservation of embryos using slow freezing and rapid thawing with dimethyl sulfoxide (Me2SO) was soon applied to human embryos in the early cleavage stage and led to the first live birth from cryopreserved human embryos. 63 Cryopreservation of human embryos has now become a routine procedure in ART clinics. 64 Also, when single men or women require urgent cancer treatment such as chemotherapy, they are at risk of losing their fertility. In such cases, cryopreservation of gametes may be the only option for future restoration of reproductive potential. 63
Slow freezing and vitrification are two main approaches of cryopreservation. Slow freezing is a classic preservation method for living cells in which the sample is frozen at an optimal rate. The freezing rate is selected such that it is slow enough to avoid detrimental intracellular ice formation but is fast enough to limit solute toxicity due to excessive dehydration in the presence of extracellular ice. 65 Alternatively, vitrification is an ice-free process in which the liquid is cooled rapidly to outrun ice crystallization and transits into a noncrystalline solid state (a glass) below the glass transition temperature, 66 which typically requires a combination of an ultra-rapid cooling rate and a very high concentration of glass-forming solutes (which can themselves be toxic), although vitrification with low concentrations of cryoprotective agent (CPA) were also reported in the literature.67,68 During thawing, the rewarming rate must be sufficiently high to prevent devitrification, especially in large-volume samples. 69
To help the preserved gametes or embryos overcome the detrimental stresses induced by cryopreservation, the addition of CPA, such as Me2SO and glycerol, has become routine. CPAs can depress ice nucleation, promote glass transition, help preserve the integrity of plasma membranes, or stabilize the native structure of proteins. 70 Permeable CPAs, including alcohols, sulfoxides, and amides, can readily penetrate the cell membrane by diffusion, acting in both extra- and intracellular spaces.
When exposed to a hypertonic solution of a permeable CPA, cells initially lose and then uptake water in response to the osmotic gradient, accompanied by a constant influx of CPA. As a result, cell morphology undergoes a shrink-and-swell behavior. 71 Oppositely, a swell-and-shrink behavior is observed when permeable CPAs are removed from freeze-thawed cells before fertilization or transplantation. Such abrupt volumetric changes can potentially collapse or explode the cells that are subjected to cryopreservation, especially for large cells such as oocytes and embryos. Therefore, it is critical to understand the transport of water and solute across the membranes of oocytes or embryos and then optimize the loading and unloading processes of CPAs, such as the number of addition and/or removal steps and the increment of concentration in each step.
In recent years, a number of microfluidic perfusion chambers have been developed specifically for the measurement of cell membrane permeability.58,71–73 Heo et al.
73
proposed a microfluidic device for the quantitative measurement of oocyte volume during various CPA loading protocols. Stepwise, linear, or complex CPA concentration profiles were created to load CPAs into a trapped oocyte, and the oocyte volumetric response to each profile was measured. With both linear and complex profiles, 1.5 M propanediol could be loaded into the oocyte in less than 15 min with a volumetric change of less than 10% compared with the isotonic condition. Very recently, Zhao et al.
58
designed a perfusion chamber with a “road block” to hold individual oocytes in place and coupled the chamber with a temperature-controlled stage to measure the osmotic behavior of human oocytes at 4, 15, and 25 °C (
Despite the characterization of membrane permeability of gametes and embryos to various CPAs, the attempt to eliminate osmotic stress is still limited by the practicality of performing numerous precise pipetting steps in a short period of time. Miniaturization and automation offered by microfluidics provide a promising solution to overcome this limitation. Lai et al.
75
developed an automated CPA addition protocol based on microfluidics to minimize the osmotic stress during the vitrification of bovine oocytes or murine zygotes, yielding better morphology, higher cell quality, and improved developmental competence than CPA exposure using the manual pipetting method that is currently used in clinics. In the proposed device, a number of individual oocytes or embryos were held within the exposure chamber by suction, being exposed to a continuous temporal gradient of CPA concentration that was generated by synchronizing two programmable syringe pumps that were connected to the microfluidic device.
75
In another study, a digital microfluidic device was developed to automate the preparation of embryos for vitrification (
We have seen a growing number of microfluidics devices developed in the field of cryopreservation during the past two decades.78–81 Although many of these devices were designed for other types of cells, some of them may be adapted relatively easily to facilitate the cryopreservation of gametes and embryos. Several representative examples are discussed in detail below.
Deutsch et al. 82 developed an Individual Cell-based Cryo-Chip (i3C) made of mold-injected polycarbonate and BK7 glass. The device featured an array of picowells, each of which was designed to maintain an individual cell in position during cryopreservation and accompanying treatments. More than 97% of the cells tested (human promonocytic U937) were found to retain their position in the picowells throughout the entire freeze-thaw cycle and medium exchange. This device enabled the comparison between prefreezing and postthawing data at single-cell resolution. In a subsequent study, Afrimzon et al. 83 demonstrated that the functional activity of U937 cells that were freeze-thawed on the i3C devices had no statistically significant difference from the control cells that were cryopreserved via conventional cryo-vials. Most importantly, the combination of the multiparametric analysis at a single-cell resolution and the optical and biological features of the device enabled the accurate determination of the functional status of individual cells and subsequent retrieval and utilization of the most valuable cells, which are highly desirable for on-chip cryopreservation of oocytes or embryos.
As mentioned above, vitrification is an ice-free cryopreservation approach in which the aqueous phase is cooled rapidly to enter the glassy state in the absence of ice both inside and outside the cells. A major challenge to cell vitrification is intracellular ice formation induced by devitrification (i.e., formation of visible ice in a vitrified sample) during rewarming of the vitrified cells at a moderate rate while they are still at cryogenic temperature. 67 Therefore, high concentrations (up to 8 M) of permeable CPAs and small sample volume (∼2.5 µL) have to be used to facilitate vitrification during cooling and minimize devitrification during rewarming, yielding high toxicity and limiting scale-up capacity. Huang et al. 67 revealed that hydrogel microencapsulation could effectively inhibit devitrification during rewarming. Cells were encapsulated into alginate micro-droplets using a nonplanar microfluidic device. Their results showed that if ice formation was minimized during cooling, intracellular ice formation became negligible in hydrogel microencapsulated cells during the entire cooling and rewarming cycle. The proposed approach enabled vitrification of mouse embryonic stem cells and human adipose-derived stem cells with low concentration of permeable CPAs (1.5 M 1,2-propanediol) and nonpermeable CPAs (0.5 M trehalose) in a relatively large volume (up to 250 µL) using conventional plastic straws. 67 It is worth mentioning that these plastic straws are also the commonly used closed system for the vitrification of oocytes and embryos. 84 Very recently, alginate hydrogel microencapsulation and Fe3O4 nanoparticle-mediated rewarming were leveraged to suppress ice formation during the vitrification of mesenchymal stem cells in polytetrafluoroethylene capillaries, presenting another promising approach to achieve low-CPA (∼2.5 M) and large-volume (10 mL) vitrification. 85
Summary
Although it has significantly transformed the landscape of treating human infertility, IVF remains inaccessible and unaffordable to the majority of infertile couples, especially those living in remote, resource-limited regions. This is mainly due to its complicated, error-prone procedures and strong dependence on technically skilled personnel. During the past two decades, microfluidics technology has demonstrated its potential of automating IVF procedures, reducing stress on gametes and embryos, and improving standardization. Although the state of the application of microfluidics in IVF has been rapidly evolving, future challenges and opportunities still exist for microfluidics to advance our understanding of assisted human reproduction and optimize IVF protocols in clinical settings.
Most of the microfluidic systems were designed to specifically handle either male or female samples or to focus on the postfertilization stages such as embryo culturing. However, multimodular microfluidic systems hold the promise of fully automating and standardizing clinical IVF procedures, which would alleviate the dependence on human intervention and reduce the financial burden on patients. Despite recent advances, 57 further development of such IVF-on-a-chip systems may draw synergistic support from technologies such as organ-on-a-chip, 86 3D printing,87–89 and paper-based microfluidics.90,91 Also, microfluidic devices that mimic the in vivo structure of reproductive tissues and organs will offer more accuracy and flexibility when coupling different modules that represent different IVF procedures.
Most of the microfluidic devices fabricated for IVF so far are made of PDMS. However, this type of transparent and gas-permeable elastomer potentially has negative effects for cell studies at the microscale, such as deformation, evaporation of culture medium, molecular absorption, leaching of un-cross-linked oligomers, and hydrophobic recovery. 92 Other biocompatible materials may be superior when used for handling gametes and embryos. Thermoplastics such as poly(methyl methacrylate), polystyrene, and cyclic olefin copolymer have emerged as alternatives to manufactured microfluidic devices for biological applications. 93 For example, cyclic olefin copolymers are a relatively new group of polymers with desirable properties, 94 such as chemical resistance, excellent optical transparency, nonabsorption of small molecules, and biological compatibility.95,96 Moreover, fabrication techniques such as micro-milling, hot embossing, and injection molding are available for the production of microfluidic devices made of cyclic olefin copolymers to overcome the bonding issues. 92 Berenguel-Alonso et al. 97 have fabricated two microfluidic devices using hot embossing and micro-milling on cyclic olefin copolymers. One device was used to trap and fertilize bovine oocytes, and the other was used to store sperm and assess its quality over time.
There are relatively fewer microfluidic systems16,46,98 that can be used to facilitate the preparation and quality evaluation of oocytes for IVF in comparison with those used for sorting sperm or culturing embryos. In fact, the current clinical procedures used for female patients demand a great amount of intervention from well-trained embryologists, limiting the accessibility and affordability of IVF treatment. Therefore, an all-in-one microfluidic system is highly desirable to enable automation of the procedures, including oocyte retrieval from follicular fluid, selection of high-quality oocytes, and/or oocyte denudation for ICSI. To echo the first challenge discussed above, it would also be interesting to integrate oocyte-processing devices with those handling sperm or embryos to strengthen the prospect of IVF-on-a-chip.
The application of microfluidics in cryopreservation has been largely limited to the characterization of biophysical properties of gametes and/or embryos. Other microfluidic devices are suggested to meet the clinical interests more closely, such as freezing or vitrifying gametes or embryos on a chip. In particular, the miniature nature of microfluidics has the advantage of handling extremely small volume of samples, which may provide favorable characteristics to facilitate ice-free vitrification if materials of sufficiently high thermal conductivity are used in device fabrication. Further integration of microfluidics with cryopreservation has the potential to support automation and reduce user-specific variability, enhance discovery of new CPA exchange protocols, and improve outcomes in assisted human reproduction. 99
Footnotes
Acknowledgements
The author would like to gratefully thank Dr. Edward Kai-Hua Chow for the invitation to contribute this review article to the journal.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
