Abstract
Transdermal drug delivery (TDD) provides a direct drug administration route bypassing gastrointestinal and liver metabolism. Until now, topical nanocarriers responsible for efficient TDD are predominantly polymeric or lipid based. The size-dependent skin penetration ability of framework nucleic acids (FNAs) has recently been reported, along with their efficacy in delivering doxorubicin for skin melanoma therapy. This commentary is to highlight the paradigm shift of nucleic acid delivery from being a cargo moiety to serving as a drug carrier instead. Further development directions to maximize the potential of FNAs for TDD are also discussed.
Transdermal drug delivery (TDD) provides a direct drug administration route bypassing gastrointestinal and liver metabolism. However, most drug molecules are incapable of penetrating the skin barrier, thus requiring the assistance of carriers to encapsulate and improve delivery efficiency. Following skin penetration, drugs are liberated either swiftly or in a sustained manner. Until now, several TDD carriers have been identified and demonstrated but are predominantly polymeric or lipid-based nanoparticles (e.g., liposomes, solid lipid nanoparticles). Over the past few years, framework nucleic acids (FNAs; generated through programmed nucleic acids’ “origami” folding) have seen adaptation into cancer therapy and immunomodulation. However, further translation has been limited by the poor lifespan of FNAs in the harsh microenvironment of the blood circulation. 1 Considering this, we explored whether FNAs can be delivered through topical application.
The results were recently published and highlighted in Nature Communications, 2 where we discovered the size-dependent skin penetration of FNAs. Comparing several FNA configurations between 20 and 200 nm in mice and pig skin, we noted a penetration depth up to 350 µm for 20 nm tetrahedral FNAs. This was accompanied by the retention of integrity, as revealed using Förster resonance energy transfer (FRET)-incorporated FNAs. Finally, topically applied FNAs enabled efficient doxorubicin delivery to melanoma tumor, with a twofold greater inhibition performance than other topical carriers (i.e., polymeric or liposomal nanoparticles).
One compelling fact associated with this study is the paradigm shift regarding nucleic acid delivery. Conventionally, nucleic acids are primarily cargo moieties, requiring the assistance of a carrier platform for cell and tissue delivery. As shown with FNAs, however, it is now feasible to employ nucleic acids as the carrier platform. In fact, a therapeutic agent made entirely from nucleic acids (e.g., small interfering RNAs [siRNAs] or antisense oligonucleotides [ASOs]) is conceivable. 3 This ensures biocompatibility and complete biodegradation and provides improved drug loading efficiency (i.e., drug-to-carrier ratio) relative to carriers fabricated through the stochastic encapsulation process (e.g., nanoprecipitation, emulsification).
Future studies are expected to streamline translation toward the topical delivery of FNAs. Like spherical nucleic acids, we have observed major localization of FNAs at skin appendages. 4 Comprehensive study is thus required to elucidate the extent that each penetration pathway influences FNA penetration and its stability. Exploration of more FNA structures (having distinct shapes and tighter size differences) would allow the generation of an FNA “toolbox” to match the intended topical application, based on its propensity to localize to different regions of skin anatomy. For instance, 20–30 nm FNAs capable of reaching the dermis layer can be adopted for scar management. Meanwhile, slightly larger FNAs (50–100 nm) can be utilized to manage the epidermis layer (e.g., dermatitis skin tearing) or skin appendages (e.g., excessive sebum production). Concurrently, different drug types can be loaded on FNAs.5,6 Aside from nucleic acid-intercalating drugs, chemical moieties can be covalently conjugated through inclusion of functional linkers to the FNA backbone. Nucleic acid moieties (e.g., siRNAs and ASOs) can be hybridized by providing extensions to specific sites on FNAs. In this manner, precise and simultaneous delivery of drug moieties can be guaranteed for optimal therapeutic efficacy.
Last but not the least, therapeutic FNAs can be incorporated into a hydrogel cream formulation (
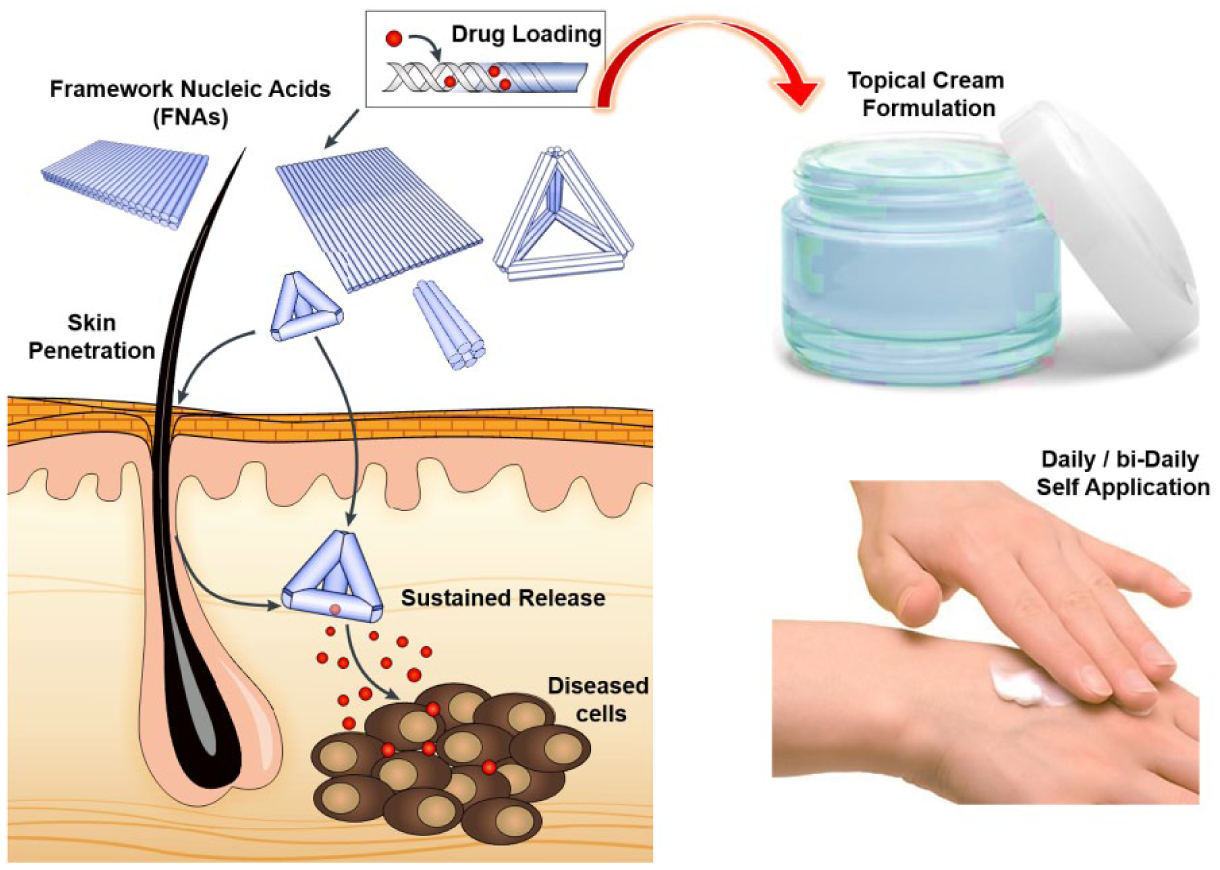
FNAs as the prospective carriers for TDD. Showing size-dependent penetration capability, FNAs can be loaded with various drug moieties and incorporated into a hydrogel cream formulation for facile self-application.
Footnotes
Acknowledgements
The authors would like to acknowledge Ying Zhu, Daniel Lio, Mo Xie, Weina Fang, Qian Li, Mengjia Zheng, Maurice Van Steensel, Lihua Wang, and Chunhai Fan for close collaboration on the original study, and hence their contribution to these ideas.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
