Abstract
Keloids are disfiguring pathological scars that could cause pain and pruritus. The conventional treatments, such as bolus injection of drugs or surgery, are invasive and require a personal visit to clinic. Microneedle (MN) technology has great potential to offer a self-administered and minimally invasive treatment of keloids. However, drugs delivered using MNs suffer from limited penetration in keloid tissue. This study demonstrates enhanced drug penetration in human keloid scar tissue by combining MN and sonophoresis.
Introduction
Keloids are tumor-like pathological scars that can overgrow the borders of the original wound site. 1 As a disfiguring disease with a high prevalence rate in Asian and African populations, 2 keloids accompanied by concomitant pain and pruritus can cause great life inconvenience and a psychological burden. Unfortunately, since their mechanism has not been fully understood, existing treatments remain limited and often the keloids recur. Steroids such as triamcinolone are a common medication. However, the regular injection of steroids often causes great pain, while tapes and creams are too weak to efficiently deliver drugs across the stratum corneum. Therefore, improvements in chemical composition and modality are of equal importance.
Microneedles (MNs) are miniaturized needles with a microscale length. They can pierce through the stratum corneum and create transient pores in the skin for trans- and intradermal delivery. 3 This platform has been intensively explored for drug delivery in the treatment of both local and systemic diseases. The drugs delivered using MNs include small molecules, peptides and proteins, and oligonucleotides.4–6 Our team first explored utilization of the MN platform for keloid treatment.7,8 Both preclinical and clinical results showed promising outcomes for controlling the growth of keloids. 9 However, we also noticed that MNs alone could not get the drugs into the deep layers of the scar tissue.
One way to enhance the skin penetration of MN-delivered drugs is to integrate this method with other strategies, such as sonophoresis. For example, Han and Das improved the penetration of bovine serum albumin in porcine ear skin to 1 μm/s with the combination of an MN patch and ultrasound, which resulted in about 10 times higher penetration than the permeability obtained in passive diffusion. 10 Nayak et al. showed that pretreatment with both stainless steel MN and 20 kHz sonophoresis for 5–10 min would result in an initial faster permeation of lidocaine in porcine skin by 23-fold, with an average 4.8-fold increase over 30 min of application when compared with separate device treatments and passive diffusion. 11 Thus, we hypothesized that the integration of sonophoresis into an MN-based strategy would further enhance the drug pepentration in keloid scar tissue.
The purpose of this study was to examine this hypothesis using a polylactic acid (PLA) MN array, medical sonophoresis device, and living human scar tissue. Methylene blue with or without triamcinolone acts as the model drug. Briefly, scar tissue was pretreated with MNs before the drug was topically applied. Later, the ultrasound was applied to enhance the drug penetration. A total of 94 views of keloid scars and 50 views of normal scars were collected from frozen sections to calculate the drug penetration depth and contact sides.
Materials and Methods
Methylene blue dye (2 mL/20 mg) was obtained from Jumpcan Pharmaceutical Group Co., Ltd. (Taixin, China). Triamcinolone acetonide injection (Transton, 1 mL/40 mg) was obtained from Kunming Jida Pharmaceutical Co., Ltd. (Kunming, China). PLA MN arrays were purchased from Smicna Pte Ltd. (Singapore). Each MN tip has a pyramid shape and length of 600 µm. There are a total of 339 MN tips on one patch ( Fig. 1A ).
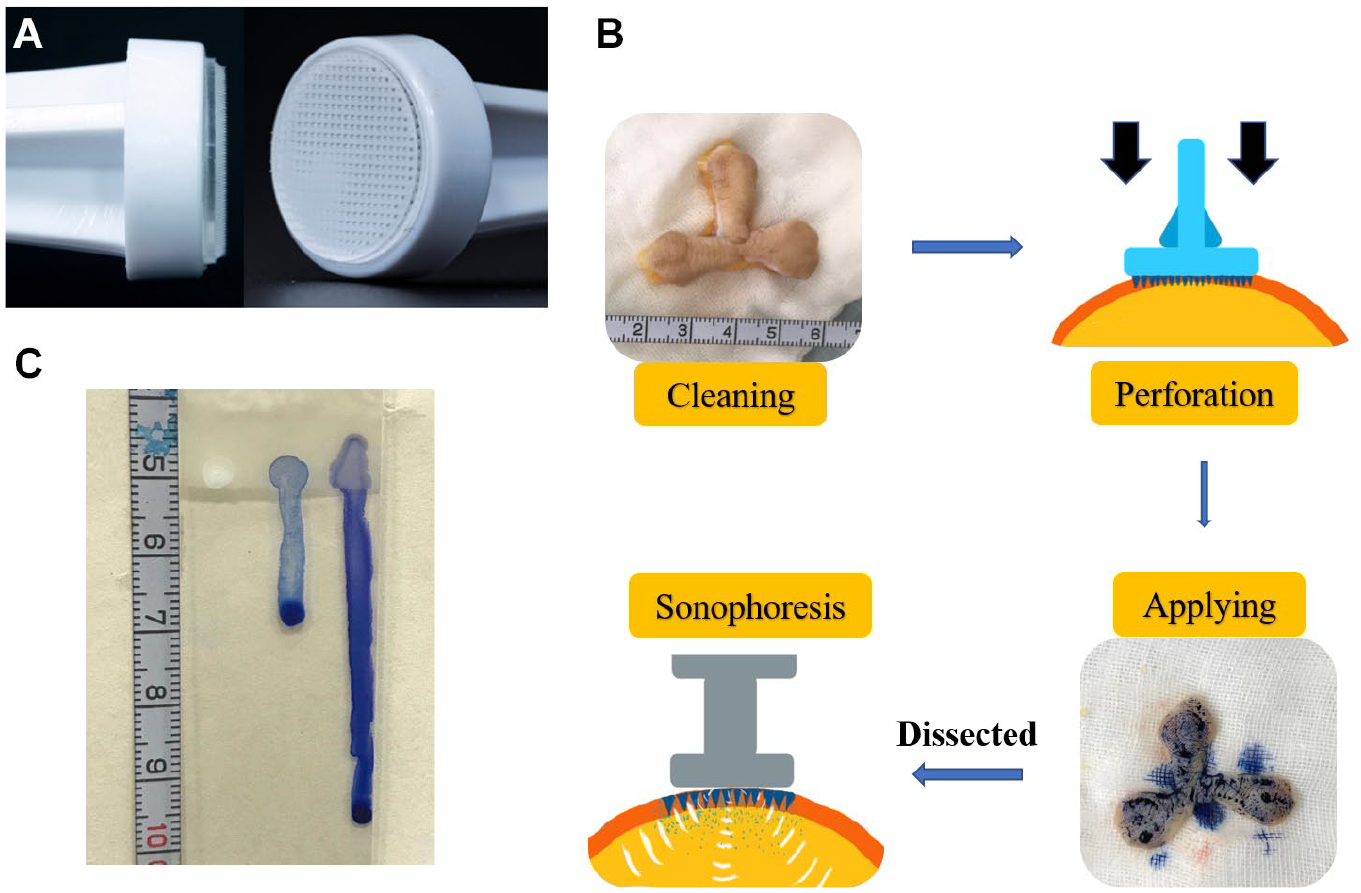
Setup of the experiment. (
Sonophoresis Device
The medical sonophoresis device was purchased from Accent Pro (Alma Lasers Ltd., Israel). Thirty hertz is recommended by the manufacturer as a routine parameter in the treatment protocol for keloids. The frequency at which the device emits ultrasound is 30/50 Hz, while the working frequency of the ultrasound is 40.68 MHz.
Scar Tissue Collection
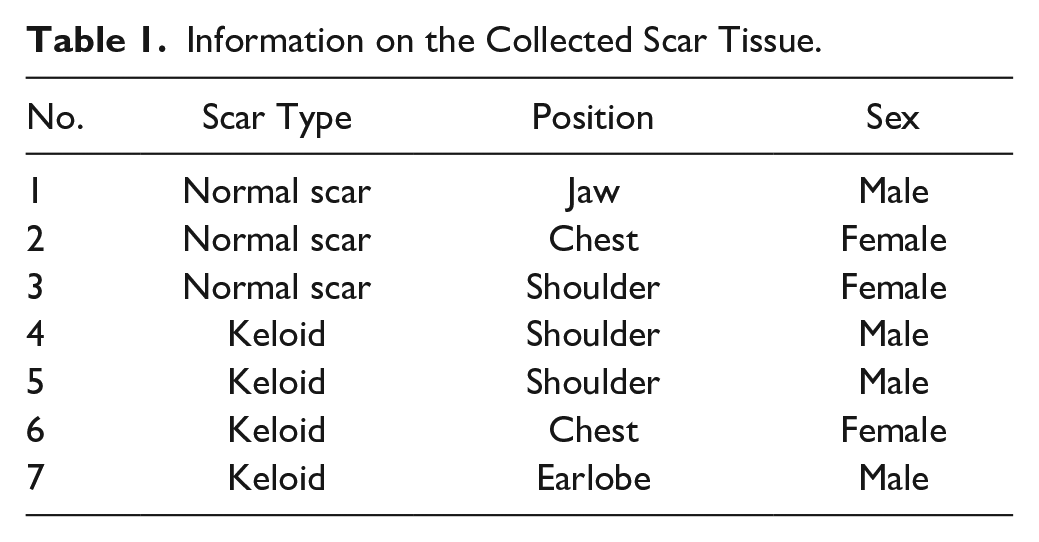
All scar tissues (three normal scar tissues and four keloid tissues) ( Table 1 ) were medical waste from scar repair operations. Typical pictures of normal scar (top) and keloid (bottom) are presented in Figure 2B . Patients were diagnosed by two expert plastic surgeons of Shanghai 9th People’s Hospital and were fully informed and signed written consent. This study was approved by the ethics committee of Shanghai 9th People’s Hospital (no. 120 of year 2016).
Information on the Collected Scar Tissue.
(
Application Protocol
Scar tissues collected from the operating room were stored at 4 °C and used within 3 days. As shown in Figure 1B , the sample surface was first cleaned with an alcohol pad. Then a sterilized MN patch was thumb pressed into the scar tissue for 30 s. After the MN patch was removed, methylene blue dye or the mixture dye (methylene blue/triamcinolone acetonide injection, 1:1, v/v) was applied evenly. Finally, the scar tissue was treated with an ultrasonic probe (30 or 50 Hz) for 1 min.
Dye Binding Studies
The isolated scar tissues were fixed in O.C.T. compound medium. Transverse cryosections of 8 μm thickness were cut from the frozen samples and placed on glass slides for examination with Leica DM 2500. Pictures were automatically analyzed by ImageJ version 1.53c.
Liquid Mobility Evaluation
Twenty microliters of methylene blue, injectable triamcinolone acetonide, and their mixture (1:1, v/v) was dropped respectively on the same line of a horizontally positioned glass slide. Then the slide was positioned upright. Their flowing tracks were evaluated to roughly represent their mobility ( Fig. 1C ).
Statistical Analysis
All results for binding studies were presented as an average of three replicates to determine statistical significance. A p value of 0.05 was used to determine statistical significance. The statistical analysis (paired-t test) and statistical chart were performed using GraphPad version 6.02.
Results
MNs Penetrated Keloid and Normal Scars with Equal Depths, but Barely Reached Keloid Dermal Layer
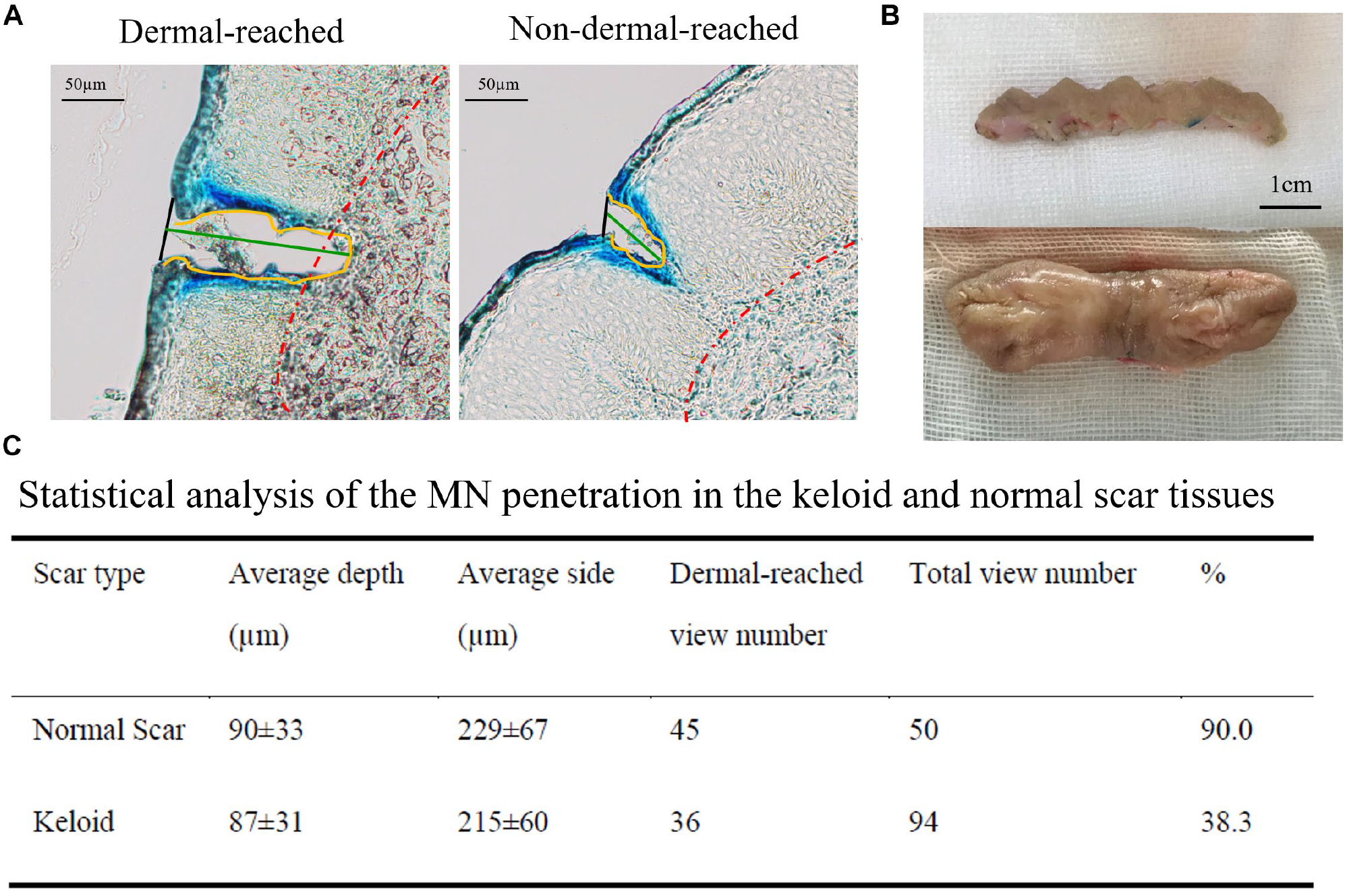
The penetration depth of MNs was defined as the distance from the skin surface to the bottom of the notch ( Fig. 2A ). The length of the contour was defined as the contact side, while the tip parts beyond the epidermis baseline (red) were defined as dermal-reaching areas. Figure 2C summarizes the penetration performance of MNs in normal scar tissue and keloid tissue.
On the normal scar tissue, MNs produced an average contact side of 229 ± 67 µm and an average penetration depth of 90 ± 33 µm. They also reached the dermal layer in 90% (45/50) of the tissue samples. For the keloid group, the average contact side was 215 ± 60 µm and the average penetration depth was 88 ± 31 µm for MNs. Different from the situation with normal scar tissue, MNs could only reach the dermis layer in 38.3% (36/94) of the keloid tissue samples.
Statistical analysis showed that MNs had a similar penetration performance between the group of normal scars and keloid scars (penetration depth, p > 0.05; contact side, p > 0.05). However, the percentage of dermal-reaching MNs in normal scars was significantly higher than that in keloids (p < 0.0001). This significant difference may be caused by the thickened and abnormal differentiated epidermal layer in keloids.
Sonophoresis Combined with MNs Promoted the Permeation of Drugs with Poor Mobility in Keloid Tissues
Methylene blue is a fluidic dye commonly used for staining tissues in plastic surgery. Triamcinolone acetonide is the most commonly used steroid injection in keloid treatment. It can inhibit fibroblast proliferation and suppress inflammation.12,13 Methylene blue and triamcinolone acetonide have shown different mobilities ( Fig. 1C ). Here we took methylene blue and its mixture of triamcinolone acetonide as two model drugs with different mobilities. Before applying the model drugs, keloid tissues were treated with three strategies: MNs alone, MNs and sonophoresis of 30 Hz, and MNs and sonophoresis of 50 Hz. The treated tissues were then cryosectioned for analysis.
As shown in
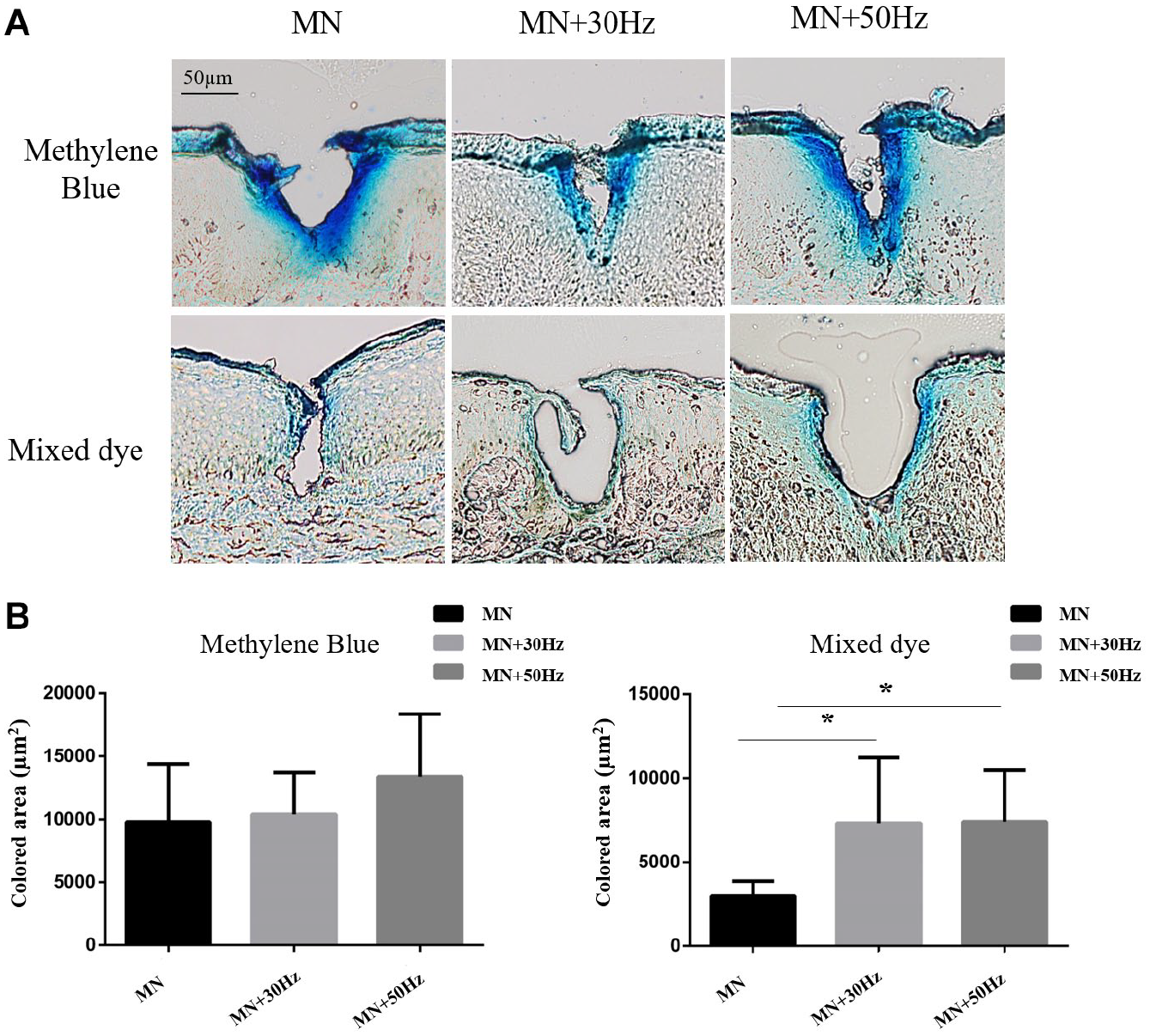
Sonophoresis improves the permeation of the model drugs with different mobilities into keloid tissues via MN-created channels. (
However, the situation is different for the mixture of methylene blue and triamcinolone acetonide, which has a relatively low mobility. The promoting effect of sonophoresis was statistically significant in both two sonophoresis-treated groups compared with the non-sonophoresis-treated group (MN, 3007 ± 801 µm2; MN + 30 Hz, 7328 ± 3787 µm2; MN + 50 Hz, 7418 ± 2979 µm2; p < 0.05). We noticed that there was no significant difference between two sonophoresis-treated groups with different frequencies (p > 0.05).
Sonophoresis Combined with MNs Promoted Drug Permeation in Normal Scar Tissues
As different parameters were tested in order to determine an optimal manner of drug permeation for future keloid therapy, a desired drug delivery method (MNs combined with sonophoresis of 50 Hz) was determined and applied to normal scar investigation. As shown in Figure 4A , the combination of sonophoresis and MNs could significantly promote drug permeation in scar tissues compared with the group without sonophoresis. The dye binding areas of two groups were 4895 ± 1740 µm2 (– sonophoresis) and 7023 ± 3056 µm2 (+ sonophoresis), respectively ( Fig. 4B ).
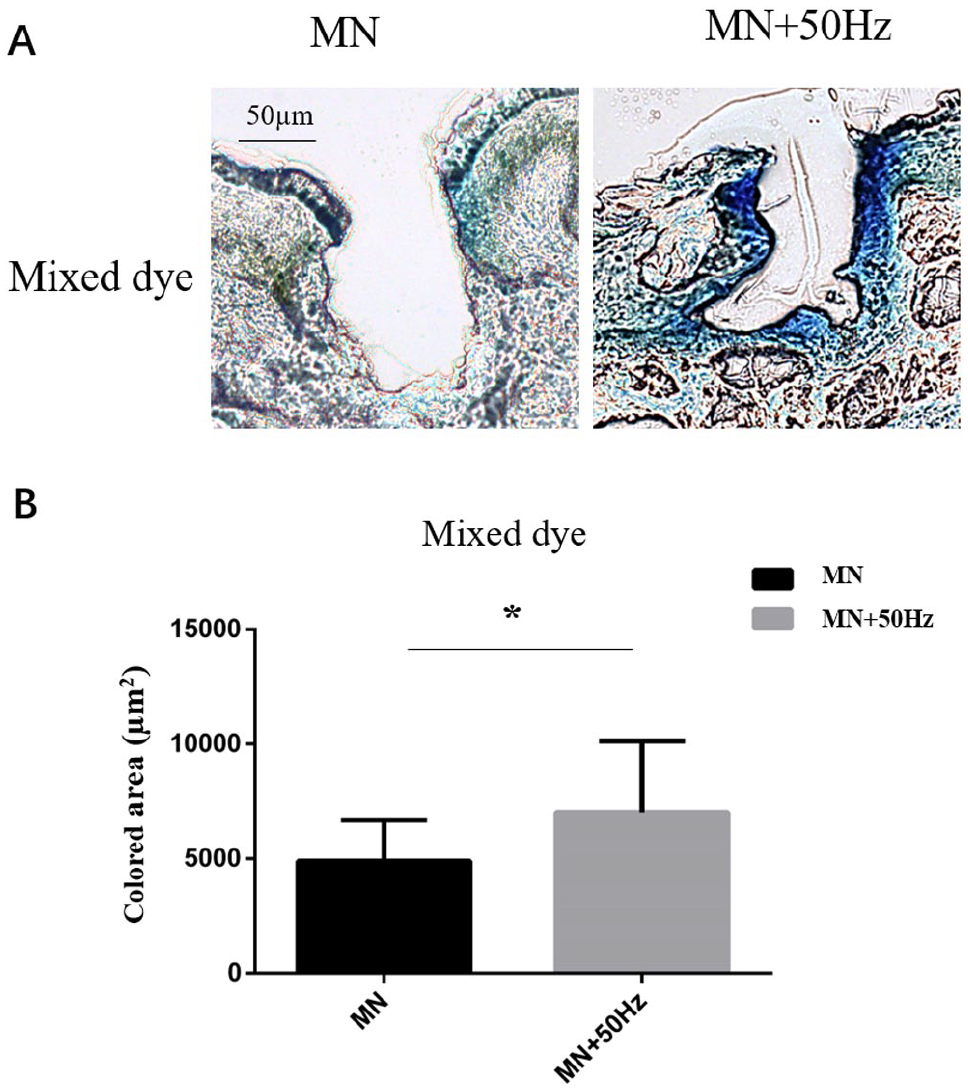
Sonophoresis improves the skin penetration of poor-mobility drug into normal scar tissues via MN-created channels. (
Discussion
Keloids are disfiguring fibroproliferative disorders with excessive collagen deposition. Elongated inflammation and enhanced skin tension are local factors that may lead to keloids at the wound site. 14 Steroid injection, cryotherapy, radiotherapy, and surgical removal are the most frequently used methods in clinical practice,2,14,15 but recurrence is still a major problem because of the limited understanding of keloid pathogenesis. Moreover, keloid patients suffer from both annoying symptoms (e.g., erythema, pain, and pruritus) from the keloid itself and the painful treatment process, such as surgery and injection. 15 In the past, pathological changes such as persistent inflammation in the dermal layer have been well demonstrated as the core of the keloid mechanism. 16 Recent studies have found that the altered epidermal structure and physiological functions in keloids, 14 which indicates the synergistic effects and interactions between the epidermis and dermis, are the keys to keloid pathogenesis. Therefore, we assume that the ideal drug delivery method should penetrate the full thickness of the epidermis and reach the superficial layer of the dermis in keloid tissues.
MNs with a variety of lengths are promising to treat keloids in the future. As a novel transdermal drug delivery method characterized with limited injury and irritation,17,18 MNs have been introduced into the treatment of pathological scars. The MN patch has been proven to significantly inhibit keloid fibroblast proliferation in vitro, 7 prevent tissue growth in a rabbit ear hypertrophic scar model, and reduce the volume and associated pain and itch of keloids. 9
According to our clinical experience, a comprehensive strategy may have better performance than a single treatment in keloids. For example, a CO2 fractional laser combined with triamcinolone is frequently used in clinical treatment with satisfactory outcomes. 19 Excitingly, the painless and self-administrated feature of the MN patch enables it to be a substitute for the CO2 fractional laser to deliver triamcinolone in keloid tissues since laser therapy is device-dependent and irritating to the skin. The published literature has also confirmed that combining sonophoresis with MNs can significantly promote the permeation of calcein (623 Da), 20 bovine serum albumin (6.6 kDa),10,20 and fluorescein isothiocyanate–dextran (4.4 kDa) 21 without causing noticeable damage to the skin. Ex vivo permeation studies using Franz diffusion cells measured lidocaine permeation through porcine skin after pretreatment with stainless steel MNs and sonophoresis and showed accelerated drug accumulation. 11
However, most previous studies were based on porcine skins10,11,20,21 or gelatin gels, 22 and only a few were conducted on human normal skin. Since human keloid tissues are characterized by extra collagen deposition and potential epidermis defects, 23 their physical features might be different from those of animal models or human normal skin. In addition, the micromechanical property of human and porcine skins was reported to be different. 24 Therefore, previous studies of the efficacy of the transdermal drug delivery of MNs may not truly reflect the effect on human keloid and human normal scar tissue.
As shown in this report, MNs with a length of 600 µm could penetrate into keloid scars and normal scars, but their tips reached different layers of the skin, possibly because of the altered epidermal thickness in keloids. 23 Ninety percent of MNs successfully penetrated through the full layer of epidermis and delivered drugs to the superficial layer of the dermis in normal scars, while only 38.3% did so in keloids. Therefore, the length of 600 µm might not be ideal for MNs used in keloid treatment. Since the stratum corneum of the skin has a depth of 5–20 µm and the epidermis has a variable depth of 20–100 µm (normal skin), some have argued that the penetrating length of MNs should be less than 100 µm. 20 However, one study declared that MN arrays with longer needles (>600 µm) were more effective in creating pathways across skin and enhancing drug flux. 25 MNs longer than 1000 µm were also frequently used,10,11 and improved penetration and permeation were observed when the needle length increased from 600 to 1500 µm. 26 Furthermore, another study declared that a needle length below 750 µm is painless and bloodless. 10 Therefore, the best length of MNs for different scar types should be further studied and balanced between comfort and penetration depth.
According to direct histological evidence from the dye binding images of frozen sections, we found that MNs combined with the existing medical sonophoresis device were sufficient to promote triamcinolone application in both normal scars and keloid scars. Compared with laser therapy, the advantages of MNs are that they are painless, cost-effective, safe, and self-administered, which may improve the treatment experience for keloid patients.
Furthermore, experiments have shown that the vibration of ultrasound can promote the melting of dissolving MNs and then improve drug delivery efficacy. 22 Thus, MNs assisted with advanced material technology should be further studied in the future. Our future work will focus on improving patients’ treatment experience by developing advanced MN-related strategies in the treatment of pathological scars.
Footnotes
Acknowledgements
I would like to express my gratitude to Mrs. Aijuan Zong for her invaluable assistance and patience throughout the preparation of frozen sections.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (NSFC) (no.81671921) (W.L.) and the City University of Hong Kong (no. 9610472), General Research Fund (GRF) of the University Grant Committee of Hong Kong (UGC) Research Grant Council (RGC) (no. 9042951), and NSFC/RGC Joint Research Scheme (N_CityU118/20) (C.X.).
