Abstract
The use of hydrazones presents an opportunity for enhancing drug delivery through site-specific drug release, including areas such as tumor tissue or thrombosis. Many researchers are experimenting on how to more efficiently form these hydrazones, specifically using heat and chemical catalysts. Hydrazones respond on the pH environment or are synthesized with particular functional groups of the hydrazone and are two of the many unique features that allow for their programmed drug release. Their flexibility allows them to be relevant in a diverse range of applications, from anti-inflammatory to anticancer to acting as a chelating agent. This review paper discusses efficient ways to optimize the properties of hydrazones and their utilization in various clinical applications, including anticancer, anti-inflammatory, the prevention of platelet aggregation, and roles as chelating agents.
Keywords
Introduction
Currently, many treatments methods for a variety of diseases require high doses of a drug, which can lead to toxic off-site effects; therefore, many researchers are now focusing on increasing the bioavailability of a drug at the diseased sites. 1 One promising new approach is to utilize hydrazone linkers to release drugs in response to specific disease physiology and environments. 2 Hydrazones are small, organic molecules with the common formula R1–NHN=CH–R2, where R1 and R2 represent different functional groups. 3 Since the imine (N=C) is cleavable via a hydrolysis reaction, drugs conjugated as the R group can be released in a controlled manner. 4 This change in the R group can allow for different applications and functionalities, which creates an opportunity for tunability and gives rise to anti-inflammatory, anticancer, and antiviral applications. The different molecules that may form from different functional groups are shown in Figure 1 .
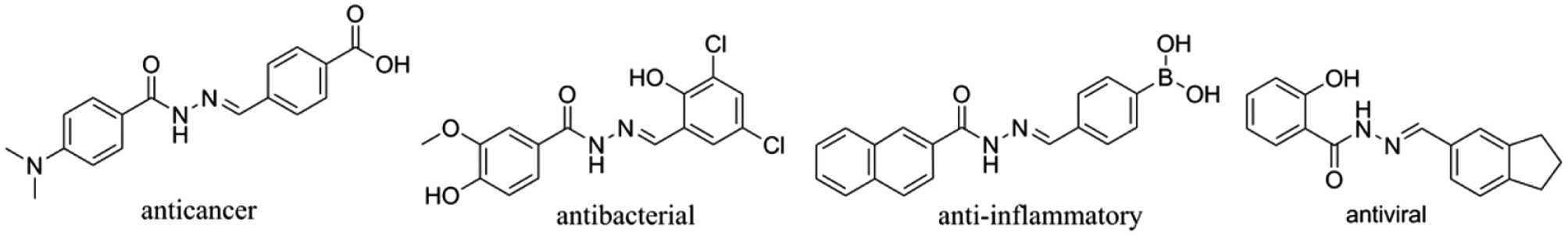
Compounds derived from a hydrazone for specific targeting applications. 7 Reproduced from Li, L.; Peng, J.; Zhou, W.; et al. Potent Hydrazone Derivatives Targeting Esophageal Cancer Cells. Eur. J. Med. Chem. 148, 359–371. Copyright 2018. Elsevier Masson SAS. All rights reserved.
To include different R substituent groups, various chemical reactions can modify hydrazones through the initially synthesized hydrazine reaction. 5 These molecules begin with a carbon carbonyl substituent group but react with aldehydes to form a carbon nitrogen double bond, to form hydrazones.5,6 This double bond possesses a pair of free electrons around the nitrogen bond, which can allow for reactions with other functional groups or substituent groups. 6 Additionally, the nitrogen atom has nucleophilic qualities while the carbon has both electrophilic and nucleophilic properties, which makes it very versatile. 7 Applications using this strategy include varying the tissue clearance rate or control drug release kinetics through the addition of various functional groups.7,8
By modifying the substituent groups for application purposes, researchers can embed drugs and program these molecules to be released in response to the appropriate environment. Most notably, pH environments can be utilized for the manipulation of drug release. In blood, which has neutral pH, hydrazones are stable. However, in regions of lower pH, such as near tumor sites or within the endosomes of cells, hydrazones can be cleaved to release active drugs. 9 These characteristics have allowed hydrazones to be linked to many drugs, such as doxorubicin (DOX) for cancer treatment as well as gold nanoparticles (Au-NPs) for prolonged retention times and controlled drug release. 2 DOX, for example, can be conjugated with a pH-sensitive hydrazone to avoid toxicity, which has resulted in a high survival rate.2,10,11 They can be extremely helpful in the formation of aromatic compounds, which are ideal for drug loading due to their stability and previous research proving their efficacy. 12 There are a variety of other modifications that come from changing the synthesis process of these molecules. In this review, we briefly describe some options for optimization via synthesis and go into more detail about the applications of hydrazones.
Optimization of Hydrazones through Varying Synthesis Methods
While many of the properties and applications of hydrazones, such as pH environments, are being tested for how they can manipulate hydrazone efficacy, few studies go into detail regarding how these hydrazones are actually made. This initial synthesis is lengthy, which is unfortunate because most research experiments need many hydrazones to have statistical significance. Chemical catalysts such as proton donors can be used to speed up a hydrazone reaction while providing a high yield. 12 In order to find the best-performing chemical catalysts, pKa values of catalysts can be studied for usage as a proton donor in expediting the reaction process. Having a pKa closer to basic would yield faster reaction rates in the formation of hydrazones due to their flexibility with protons. As expected, if a hydrazone can quickly accept the proton used in the carbon and nitrogen double bond, the formation can take place quicker. 12
Another way to optimize quick and stable hydrazone formation is using heat as a catalyst to increase reaction times. Heat can be applied in the synthesis of different hydrazone compounds. Studies have shown that the reaction time for forming a hydrazone can be significantly increased while holding a high yield using higher temperatures. 5 Not only does the temperature decrease reaction times, but by prolonging the initial heating of the mixture and monitoring the critical temperature closely—to ensure that no changes occur in the middle of the reaction—a more stable yield is produced within the same time frame. Understanding the relationship between the efficiency and stability of hydrazone formation is vital for formulating drugs for delivery in the body; hence, these studies are critical in ensuring that hydrazones can be more efficiently formed so that they can be more readily tested. For the rest of this mini-review, we discuss how hydrazones are applied in various biomedical applications.
Applications of Hydrazones
Anticancer Applications
The environment around a tumor is more acidic than blood, and hence designing a pH-sensitive drug delivery system using hydrazone linkers is advantageous to the success of targeted drug release. 11 Ye et al. developed a DOX-loaded micelle by conjugating the hydrophobic DOX to the hydrophilic polyethylene glycol (PEG) polymer with a hydrazone bond to efficiently target bone metastatic cancer. By introducing the hydrazone bond in between DOX and PEG, researchers observed enhanced circulation times in the bloodstream.11,13 In order to study the pH effects on this micelle, dynamic light scattering was used to analyze the micelle size change. Approximately 20 mg of DOX-hydrazone-PEG polymer was loaded into the micelle (114 nm) and measured over 4 h to observe stability and any morphology changes. With measuring the micelles, increased size comes from a broken hydrazone bond because it separates from the DOX and PEG. This then allows all the elements to separate and expand, thus increasing the overall size. This increasing size can be used to measure the pH at which the bond will be broken. At pH 7.4, the size of the micelle was not largely changed, whereas at pH 5.0 the size significantly increased. After proving this concept, an in vivo study on male nude mice with bone metastasis was conducted to compare free DOX with 5 mg/kg of the DOX embedded in the micelle. 11 Tumor volume after 30 days was significantly lower in the mice treated with the micelle (approximately 500 mm3 compared with close to 1000 mm3 for free DOX). Survival was also enhanced for the micelle-treated group, with 40% survival after 50 days compared with free DOX with 0% survival after only 40 days. 11 Additionally, the drug’s toxicity was tested with the MTT assay and the DOX decreased toxicity in the tumor-bearing mice. Overall, DOX with hydrazone proves to be a more reasonable and effective delivery method.
Another example in which DOX has been conjugated to a nontoxic, nanoconjugate platform polymalic acid (PMLA) using a hydrazone linkage focuses on the PEG coating. 13 PEG was used to prevent uptake by the reticuloendothelial system (RES) and contained a fluorescent dye for tracking and imaging. The results of this study successfully showed stability of the DOX–nanoconjugate under the body’s conditions and was able to prevent in vitro cell growth of invasive breast carcinoma cell lines and primary glioma cell lines. 13 DOX is bound to the PMLA-based delivery system via a pH-sensitive linker that can be cleaved under endosomal acidic conditions before delivery to the targeted cell. The drug is frequently used in tandem with hydrazones and is proving to be an extremely effective complex in drug delivery to cancer patients. 13 This type of hydrazone linkage application has the potential to have a widespread effect on a variety of diseases with its ability to attach different drugs and target different diseases.
To further elaborate on the previous study and better understand what types of environments these hydrazones could be used in, Jin et al. investigated the drug release behavior of the peptide conjugate. Using four different buffer solutions and monitoring via high-performance liquid chromatography (HPLC), the AP2H-hydrazone-DOX degradation was graphed according to the release of DOX. 14 At pH 5.0, which is comparable to the low pH of the endolysosomal cell environment, the release of DOX reaches a level of 80% within 25 h. Similarly, the chromatographic peak for AP2H-hydrazone-DOX decreased as incubation time increased at pH 5.0. This signifies that the optimal conditions for cleavage of the pH-sensitive hydrazone linker are around 5.0. Comparing the release at different pH values, Figure 2 shows that the release of DOX was very efficient and approximately 80% after 40 h. On the other hand, also in Figure 2 , at a physiological pH of 7.4, only approximately 10% of DOX was released after 40 h. The same trends in release profile of DOX were observed at pH 5.0 and pH 7.4. 14 From these studies, the DOX nanoconjugate is able to maintain its stability under neutral physiological conditions (pH 7.4).2,13,14
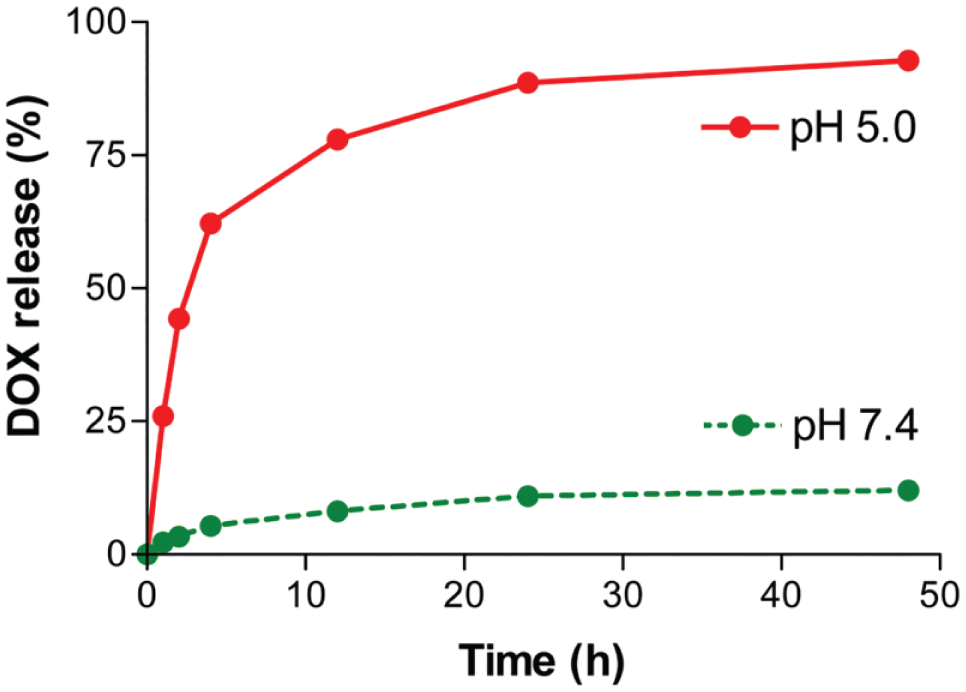
Release kinetics of DOX from nanoconjugate at pH 5.0 (red) and pH 7.4 (green) at 37 °C. 13 Copyright 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
In addition, hydrazone linkers have been used in combination with nanoparticles for targeted drug release toward cancer applications. 2 One study aimed to combat the issue of chemotherapy having limited clinical benefits due to its inability to penetrate tight junctions and cross the blood–brain barrier (BBB) in glioma applications. By using Au-NPs decorated with targeting antibodies and peptides, nanoparticles can act as drug delivery vehicles and specifically target the BBB. 2 Specifically, the cell-penetrating peptide, transactivator of transcription (TAT), and DOX were functionalized onto Au-NPs. An in vitro study was performed using DOX conjugated to TAT-Au-NP and free DOX to act as a control. 2 Using a mouse model with glioma cells, TAT-Au-NP-DOX was intravenously injected. After 24 h, transmission electron microscopy (TEM) images were taken of the tumor to see where the Au-NPs were able to travel. Mice treated with TAT-Au-NP-Dox were able to survive approximately 44 days longer than mice treated with DOX only. The TEM images show that the Au-NPs were able to enter the glioma cell lysosomes and distribute themselves around the membrane. Importantly, no cytotoxicity was found regarding TAT-Au-NP-DOX particles, confirming their safety and efficacy for cancer applications. 2
The peptide AP2H (IHGHHIISVG) has also been used for cancer applications, specifically to target and identify the tumor marker lysosomal protein transmembrane 4 beta (LAPTM4B). 14 The hydrazone linker was placed between AP2H and DOX, which aided in controlled release of the drug to the tumor cells and subsequently decreased cell viability. Cell cytotoxicity was examined using a standard MTT assay on hepatoblastoma cells (HepG2) and human lung adenocarcinoma epithelial cells (A549), as well as normal human embryonic kidney 293 (HEK293). 14 Specifically, cell viability decreased significantly in both HepG2 and A549 cells (26.6% and 29.2%, respectively). On the other hand, the HEK293 cells still had a high cell viability (98.1%), which indicates that AP2H-hydrazone-DOX does not cause toxicity to the normal cell lines but is able to specifically target tumor cell lines. By identifying the target cells, delivery of the drug to unnecessary parts of the body decreased. 14 They were also able to identify a more efficient way to inhibit the cancer cells through a more privileged structure involving the linker and controlled release.
In cancer tissue, autophagy, which removes misfolded proteins and damaged organelles, suppresses the initiation of tumor growth in the early stages of cancer or allows longer survival times in later stages. 15 Because most tumors depend on the autophagy pathway to replenish nutrient homeostasis from the degraded proteins, lipids, and carbohydrates, autophagic inhibitors have been recently investigated as a new form of cancer therapy. 16 Hydroxychloroquine (HCQ) is an autophagy inhibitor and a derivative of the antimalaria chloroquine (CQ), but with the addition of a hydrazone. 15 Wolpin et al. conducted clinical trials using HCQ in 20 patients with metastatic pancreatic cancer that did not respond to chemotherapy. 17 Patients received 400 or 600 mg of HCQ twice daily. After 2 months of treatment, the tolerability and toxicity were similar at both levels of dosage, the median survival was 69 days, and two patients were without progressive disease. 17 This was one of the first studies to show beneficial effects of using a higher dosage of HCQ. By using HCQ to inhibit autophagy, tumors lose their nutrient source, which will slowly lead to the breakdown of the tissue, as they cannot grow. This paper highlights the ability of a hydrazone structure to be a transporter of high-dosage drugs while minimizing toxic effects.
Vogl et al. combined bortezomib with HCQ to target multiple myeloma in a phase I clinical trial. 18 Bortezomib is a standard therapy drug for myeloma, 18 but only about 6% of patients achieve a total response to the treatment and only 38% a partial response (i.e., not a complete disappearance of myeloma cells as with the total response). 18 In 25 subjects, HCQ was administered for 2 weeks before the combined HCQ and bortezomib were administered for an additional 3 weeks. Six different dosage levels were tested: 100, 200, 400, 800, and 1200 mg of HCQ, all with 1.3 mg of bortezomib, and 100 mg of HCQ with 1 mg of bortezomib. 17 Three of the patients were not able to be evaluated due to severe toxicity or infection from HCQ. Of the 22 patients assessed, 3 (14%) had very good partial responses, 3 (14%) had minor responses, 10 (45%) had stable disease, and 6 (27%) had immediate progression without achieving stable disease. In a follow-up study, researchers gave three patients the highest dose (1200 mg of HCQ and 1.3 mg of bortezomib). Two patients yielded a very good partial response and one patient ended the trial with total, stable disease. 18 The combination of HCQ with bortezomib proved more successful in treatment, which may be due to the hydrazone’s ability to target the plasma cells in myeloma more efficiently. Like Wolpin et al., this study shows the promise of inhibiting autophagy to improve cancer treatment efficacy using hydrazone-coupled cancer therapies. Anticancer applications are the most commonly studied for hydrazone usage. All the studies reviewed here have shown success in the different hydrazones modified, which continues to prove hydrazone’s increased efficacy and decreased toxicity in cancer patients. Its ability to target specific tumor sites, decreasing the drug delivery to unnecessary areas and locating tumors faster, will greatly increase the success of cancer treatments in the future.
Anti-Inflammatory
One major application of hydrazones is to link anti-inflammatory drugs for controlled release. To achieve its anti-inflammatory properties, the hydrazine core will react with aldehydes or ketones to form a new substituent group to provide additional chemical, thermal, and hydrolytic stability. 19 The drug piroxicam is commonly used as an anti-inflammatory free drug. Piroxicam has a long half-life and can be administered orally to manage chronic inflammation such as rheumatoid arthritis and osteoarthritis. 20 One study investigated various hydrazone derivatives from the parent benzothiazine N-alcyl-hydrazone (NAH) structure using piroxicam to find the most effective linker to inhibit inflammation. 19 The three main compounds tested were 2-thiazol, 2-biphenyl, and 3,5-diterbutyl-4-hydroxy-phenol. 21 Each of the three compounds were administered orally in an in vivo study with 100 µmol/kg on carrageenan-induced peritonitis and zymosan-induced peritonitis, an inflammation of abdominal tissue lining, in mice. In the zymosan-induced peritonitis test, all NAH derivatives inhibited inflammation; however, not all inhibited inflammation at a higher percentage than piroxicam by itself (57.4%). 21 The three compounds listed above had the highest percent inhibition: 2-thiazol (78.7%), 2-biphenyl (81.3%), and 3,5-diterbutyl-4-hydroxy-phenol (82.6%). As for the carrageenan-induced peritonitis test, piroxicam had a 25.8% inhibition of inflammation, while 2-thiazol had 73.2% inhibition, 2-biphenyl had 77.5% inhibition, and 3,5-diterbutyl-4-hydroxyl-phenol had the highest, with 81.8% inhibition. 21 Their research highlights the advantage of drug carriers compared with simply administering standard drugs into the body, particularly with a hydrazone structure included in the compound.
Ketoprofen is another common anti-inflammatory free drug used to inhibit the growth of the inflammation. Previously, ketoprofen was modified with a hydrazone incorporating a benzothiazole moiety. 22 Benzothiazole derivatives have recently been found to bind to tissues enabling disease and inflammation in the brain and hydrazones as useful bioactive agents. With this study, we wanted to combine the binding characteristics of the benzothiazole derivatives and a hydrazone, with its bioactive agents, to test whether its anti-inflammatory effects would exceed that of the current drugs on the market. Two of the most notable compounds the study compared were (1Z,2E)-2-(2-(4-methylbenzoyl)hydrazono)-N′-p-tolylpropanehydrazonoyl chloride and (1Z,2E)- 2-(2-(4-nitrobenzoyl)hydrazono)-N′-p-phenylpropanehydrazonyl chloride because of their anti- inflammatory responses and NAH properties. The therapeutic activity was also studied through rat paw edema via oral administration. The small-molecule derivatives were given at 100 mg/kg, and after 1 h 0.1 mL of 2% formaldehyde was administered intravenously through the left paw of each rat to induce swelling. Percent inhibition of the inflammation was measured using the thickness of each paw before and after 3 h of administration. Ketoprofen yielded a 91.23% inhibition, determined by the change in paw size. The methylbenzyl compound also yielded 91.23%, while the nitrobenzoyl compound ended with 78.95% inhibition after 3 h. 22 The compounds were also tested for their ulcerogenic liability due to the fact that anti-inflammatory drugs have a common side effect of developing stomach ulcers. Ketoprofen and the compounds were delivered orally in doses of 10 mg/kg body weight once a day for 3 days, and were found to have similar numbers and severity of ulcers, which shows that these hydrazone-modified drugs perform at the same level as ketoprofen in terms of toxicity and their percent inhibition. 22 Anti-inflammatory is another commonly studied application for hydrazones as they are able to control the release of drugs. These studies have shown different physical and chemical modifications affecting the ability of hydrazones to inhibit inflammation. The outcomes of these studies can be further researched for the specific composition of each hydrazone in anti-inflammation applications for increased success rates.
Role as a Chelating Agent
Hydrazones can also be used as chelating agents in reducing toxic effects caused by metals such as iron and cobalt.23–26 Chelating agents refer to compounds that react with metals to form a stable and hydrophilic complex. Hydrazones, in particular, are good chelating agents due to their ability to form stable complexes with many transition metals. 23 The binding of these metal compounds, however, promotes the excretion of the metals. 23 Excess accumulation of cobalt (Co) in the body can result in metal poisoning, and Co (II) complexes with 3-methoxysalicylaldehyde-4-hydroxybenzoyl hydrazone and 3-methoxysalicylaldehyde isonicotinoyl hydrazone were synthesized. After characterization, researchers determined that their stability and insolubility in water are important, but the affinity to cobalt ions that aroyl hydrazones possess make them a very practical option for use as a chelator in cobalt poisoning treatment. 23
In addition, iron (Fe) overload disorders such as beta thalassemia increase the risk of heart and liver diseases, and the only Fe chelator currently in widespread clinical use is desferrioxamine (DFO).24,27,28 The use of DFO is not only expensive but also inefficient in intestinal absorption. According to an early study, 25 pyridoxal isonicotinoyl hydrazone (PIH) is able to act as an Fe-chelating agent and was shown to mobilize iron from rabbit reticulocytes constraining high levels of nonheme Fe. In particular, the ability of PIH to mobilize iron, through release, was greater than that of DFO, with a 72.2% release of iron with PIH and a 5.1% release with DFO. Intraperitoneal administration of PIH to mice over a period of 6 days decreased the levels of iron-59 in the liver, spleen, and kidney. 26 These studies support PIH and its analogs, displaying high iron chelation efficacy, and should be used for the treatment of iron overload diseases. Hydrazones as chelating agents could be utilized with great success due to their ability to form stable complexes with metals. This study serves as a basic starting point for accelerating more tests for hydrazone agents for limiting iron release.
Prevention of Platelet Aggregation
Various methods for antiplatelet therapy have been explored in recent years due to the rise in cardiovascular disease. 29 Antiplatelet drugs can benefit individuals who have suffered from myocardial infarction or strokes. One study synthesized nonsteroidal anti-inflammatory drugs (NSAIDs) containing an NAH due to the ability of NAH to inhibit platelet activity by acting as a calcium chelator to treat cardiovascular diseases.30,31 NSAIDS alone can have detrimental effects to the cardiovascular system because while they inhibit platelet aggregation, they also reduce the expression of prostaglandin, which in the long term can increase a patient’s risk of stroke. 31 Hence, by introducing an NAH unit, the analgesic activity can be increased to reduce the overall risk of further diseases after treatment in the cardiovascular system. This compound was studied in a rat carrageenan-induced paw edema model with five different substituent groups, as seen in Figure 3 , to find the highest inhibition of platelet aggregation. 31 The control, acetylsalicylic acid, commonly known as aspirin—one of the only NSAIDS left in pharmacies—showed an 80% inhibition rate. Compound 2, as seen in Figure 3 , was the most comparable, showing 80.8% inhibition, while compound 5 showed the least at 65.9%. Although some of the NAH derivatives did not perform as well as aspirin, their survival rates in rats exceeded that of the aspirin. 31 Aspirin had a 30% survival rate, whereas compound 1 had a 40% rate and compound 5 a 33% rate. Overall, they found that compounds 1 and 5 were the most active in inhibiting platelet aggregation due to their inhibition of the aggregation and minimal toxic side effects. 31 In finding a substitute for aspirin to treat platelet aggregation, the toxic effects to the body can be significantly reduced and, as this study shows, the survival rate increased.
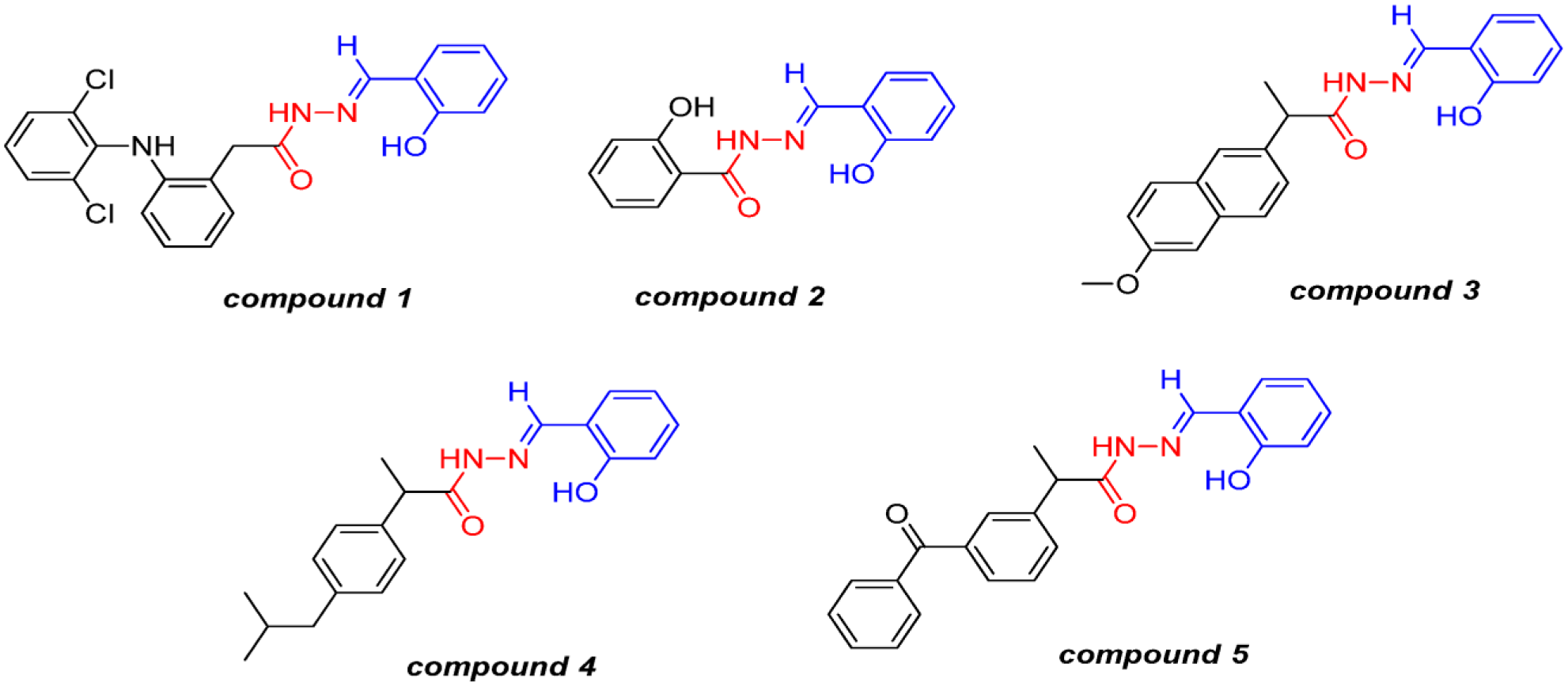
Compounds 1–5 obtained by molecular hybridization of NSAIDs, acetylsalicylic acid (ASA), and an NAH prototype. 31 Copyright 2014 by the authors. Licensee Molecular Diversity Preservation International, Basel, Switzerland. This is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
In another study looking at cardiovascular diseases and platelet aggregation, a new series of thienylacyl-hydrazone compounds were synthesized analogous to LASSBio-294 ((2-thienylidene) 3,4-methylenedioxybenzoylhydrazine). LASSBio-294 has been extensively studied for its effect in cardiac antiplatelet therapies, yielding findings that support its ability to increase cyclic AMP to decrease aggregation. 29 The compounds synthesized were LASSBio-785, -786, -787, -788, and -789. These five compounds have the same parent structure of a thiophene ring, but differ in the substituent group attached. The substituent groups added were N-methyl (785), N-benzyl (786), 5-methyl-2-thienylidene (787), 2-thienylidene (788), and 5-bromo-2-thienylidene (789). These different groups were introduced due to the varying structures of the substituent groups. An extra benzene ring was introduced in 786 and an extra double bond in 788; all these possessed new structural features that could lead to potent antiplatelet applications. Platelet aggregation was evaluated in New Zealand white rabbits by obtaining blood from the central ear artery of platelet-rich and platelet-poor rabbits and then injected with 5 µm of ADP, 5 µg/mL of collagen, and 200 µm of arachidonic acid to induce the aggregation of platelets. This specific aggregation was induced by arachidonic acid, whereas in humans, most platelet aggregation is collagen induced. 29 A collagen-induced platelet aggregation study was performed as well, and it yielded the same outcome for compounds with the most inhibition. In the collagen aggregation, the control, LASSBio-294, showed an inhibition of 14.7%, while LASSBio-785 inhibited aggregation by 39.6%, while LASSBio-786 only exhibited 3.2% inhibition. LASSBio-785 was able to outperform the current standard drug delivered. This specific compound had the N-methyl addition. Although a direct correlation cannot be made from this specific study, it can be inferred that this methyl group may increase aggregation prevention rates and could lead to future studies. In the collagen and arachidonic acid-induced aggregation, the same compounds yielded the best results but with different inhibitions. LASSBio-785 showed a 77.7% inhibition compared with the control with a 50.9% inhibition. 29 LASSBio-294 has proven, in previous studies, to have many positive effects on cardiovascular properties, but many antiplatelet therapies exhibit toxicity issues and limited efficacy, and therefore the development of new therapies is relevant. 29 This study enhanced the pharmacological properties of LASSBio-294, but studying different structural elements. The methyl group addition seemed to be the most effective in its inhibition rates, but human testing must be conducted to validate therapeutic efficacy. Both studies covered for platelet aggregation show promise for utilizing hydrazones to better inhibit the aggregation. Although not many studies have been carried out relating to these specific therapeutics, this field shows possibilities for increased efficacy and decreased toxicity.
Hydrazones show much potential for modifying drugs and have been widely applied for cancer treatments. Their tunability and chemical diversity in terms of drug conjugates allow for broad applications, including in breast cancer, glioblastoma, kidney cancer, esophageal cancer, and antiplatelet aggregation. At the root of hydrazones’ importance is the double bond between carbon and nitrogen that allows for the targeted release of drugs. This targeted release is essential for proper drug function and limited off-target side effects. Hydrazones are currently being used to assist in targeted drug delivery for applications such as anticancer, anti-inflammatory, the prevention of platelet aggregation, and a role as a chelating agent. The studies reviewed in this article have consistently shown an increase in efficacy and decrease in toxicity. The importance of these research findings is instrumental in each respective application. The use of hydrazones will be able to better treat diseases and decrease excess toxicity from harsh therapeutics. Hydrazones have immense possibilities for their modification and treatment and thus need to be thoroughly tested and researched for their application. However, out of thousands of potential derivatives, there may be only a handful that are ideal for clinical applications.
As seen in this review, though, many structures can be tested and only one may succeed. Different modifications can be made to the chemical composition or physical structure that may better the hydrazones’ performance. Due to the immense possibilities, these derivatives can be tested against commonly used hydrazones, as many of these studies have done. One possible direction could be testing a multitude of different structures against a specific disease, identifying the one with the most benefits and expanding that study to test the specific modification with different current drugs, diseases, or couplings. In order to best improve the usage of hydrazones, future improvements should focus on the high-throughput screening of drug candidates and their structures. This will decrease the amount of structures formulated and tested and allow for more potent possibilities to be researched for their therapeutic benefit.
Footnotes
Acknowledgements
We would like to acknowledge Dr. Eun Ji Chung for her continued support and guidance through the publishing process.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
