Abstract
Life science research has been actively carried out in space for a long time using bioreactor equipment, in anticipation of manned space exploration and space tourism. Such studies have reported that the microgravity environment has a negative effect on the human body, including the musculoskeletal system, nervous system, and endocrine system. Bone loss and muscular atrophy are issues that need to be resolved before long-term exposure of the human body to a space environment. To address this problem, Y. K. Kim et al. designed a system in 2015 and performed an evaluation of an automated bioreactor development model (DM) for space experiments. In this study, we developed an automated bioreactor engineering model (EM) based on the previous literature, and conducted media exchange performance testing using the Bradford assay. We used a novel method that allowed quantitative assessment of the media exchange rate versus the conventional assessment method using visual observation with a camera. By measuring the media exchange rate of the automated bioreactor EM, we attempted to verify applicability for the system for space experiments. We expect that the experimental method proposed in this study is useful for logical determination of liquid exchange or circulation in different closed systems.
Introduction
The International Space Station (ISS) provides an experimental environment different from that of Earth, with microgravity (typically 10–6 g). A series of experiments are being performed in space representing a wide range of fields, including cell biology, plant science, fundamental physics, combustion, and material science. There have also been recent efforts to commercialize space travel for regular people in the United States, United Kingdom, and Russia, marking the beginning of the space tourism era in the near future. 1 As a result, interest in life science research in a microgravity environment continues to increase.
Life science research in microgravity environments has now been conducted for a significant amount of time, since the availability of Skylab, Mir, and the ISS. To carry out studies on the reaction mechanisms of cells and tissues under microgravity, the United States, Europe, and Japan have developed bioreactors for space experiments, including the BioServe Culture Apparatus (BCA), Multiple Orbital Bioreactor with Instrumentation and Automated Sampling (MOBIAS), Single Loop for Cell Culture (SLCC), Cell Culture Module (CCM), Cell Culturing (CellCult), and Biological Experiment Laboratory (BioLab). 2 Through these studies, various acute and chronic changes have been reported to occur in the human body in microgravity, involving the musculoskeletal system,3–5 endocrine system, 6 immune system, 7 and vestibular system. 8 Most recently, life science studies have been conducted on the development of medicine to restore cells exposed to radiation, and to regenerate aged cells, for the manned Mars exploration planned by NASA. 9
In particular, research on changes to the musculoskeletal system under microgravity has been actively conducted. In 2004, Hoffman and Nader 10 found that muscular strength and muscle power decreased by 35% and 55%, respectively, after space flight, and in 2010, Fitts et al. 11 concluded that the diameter and cross-section area of muscle cells decreased by 20% and 30%, respectively. This muscular atrophy increases the risk of cardiovascular disease; thus, the issue must be resolved before long-term human exposure in the upcoming space tourism era. Until now, exercise is the only known solution, but research has reported that bone loss and muscular atrophy cannot be prevented with 2.5 h of exercise daily (6 days/week). 12
Korean and Japanese scientists have proposed the joint research campaign onboard the ISS to develop medication to prevent muscular atrophy and accelerate muscle growth, and furthermore develop industrial sports drinks. To this end, the Korea Aerospace Research Institute (KARI) began the development of an automated bioreactor for space experiments. In 2015, Kim et al. 13 determined the system requirements for an automated bioreactor for space experiments and manufactured a bioreactor development model (DM) to carry out environmental testing with constant temperature and humidity, and observation-based performance testing of the media exchange performance using a camera.
This study deals with the design and implementation of an automated bioreactor engineering model (EM), following the previous study, 13 and verifies the performance of the media exchange module, which is one of the core components of the automated bioreactor. Details of the media exchange performance testing using the Bradford assay are provided. To establish a quantitative and logical assessment method for measuring the media exchange rate, the experiments performed in this study used the Bradford assay over the conventional method of visual observation using a camera that was used in a previous DM design.
Materials and Methods
Design and Implementation of Bioreactor Engineering Model
To design the bioreactor EM for space experiments, the overall configuration of the bioreactor was reorganized for the EM based on a previously developed bioreactor DM. 13 For example, the Double-Sized Cargo Transfer Bag (2CTB) standard transport bag is supposed to be used to deliver the bioreactor to the ISS. Therefore, the dimensions of the bag (approximately 482 × 413 × 470 mm) and the maximum allowable weight (52.4 kg) 14 were considered in the EM design. Also, the bioreactor was designed in the form of a drawer with rails so that cell culture operations, such as replacements of the disposable culture chamber (DCC) and media pack, could be carried out from the front side of the bioreactor, considering the workload of the astronauts. Figure 1 displays the overall composition of the automated cell culture system referred as to the bioreactor EM in this study. The most critical component for the automation of the bioreactor EM was four fluid flow control modules. Each module is composed of a 1-to-8 fluid splitter, a de-bubble filter, a vacuum pump, a micropump, and a pump driver, respectively. The 1-to-8 fluid splitter divides and supplies the media to each of eights DCCs, while the de-bubble filter and vacuum pump remove the air produced during the supply process to ensure adequate fluid flow. The micropump controls the supply rate of the media and is responsible for the provision of nutrients to the cells in the DCC located within the cell culture module.
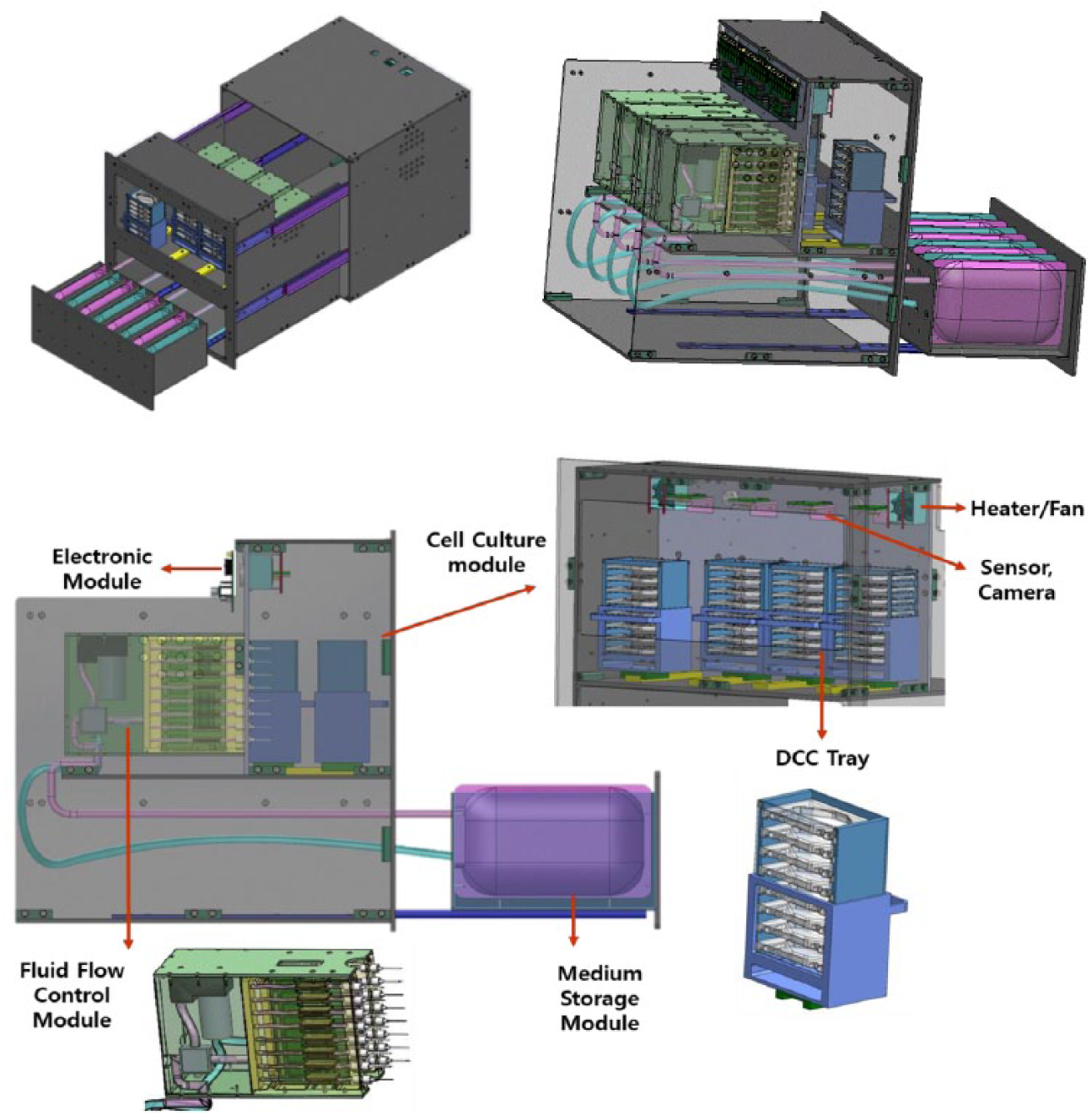
The contents the bioreactor for the space experiment.
The first model (DM) of the research bioreactor developed for space experiments was created in 2015, and the automation design and functionality of the total system were verified. The model included experimental equipment developed to perform a comparative analysis, by conducting the same experimental process for cell culture experiments on the ground as would be done in the ISS.
The bioreactor EM detailed in this study was designed using the manufacturing requirements listed in Table 1 and operational requirements summarized in Table 2 . The focus of the bioreactor EM was to assess the bioreactor automation functionality, implementation, and performance for maintaining temperature and humidity and stable media circulation. Generally, since cells are a form of life, they require appropriate temperature and humidity along with the provision of nutrients necessary to sustain life, and these conditions are the most important automation performance factors in the development of the bioreactor.
Part List of Bioreactor Components.
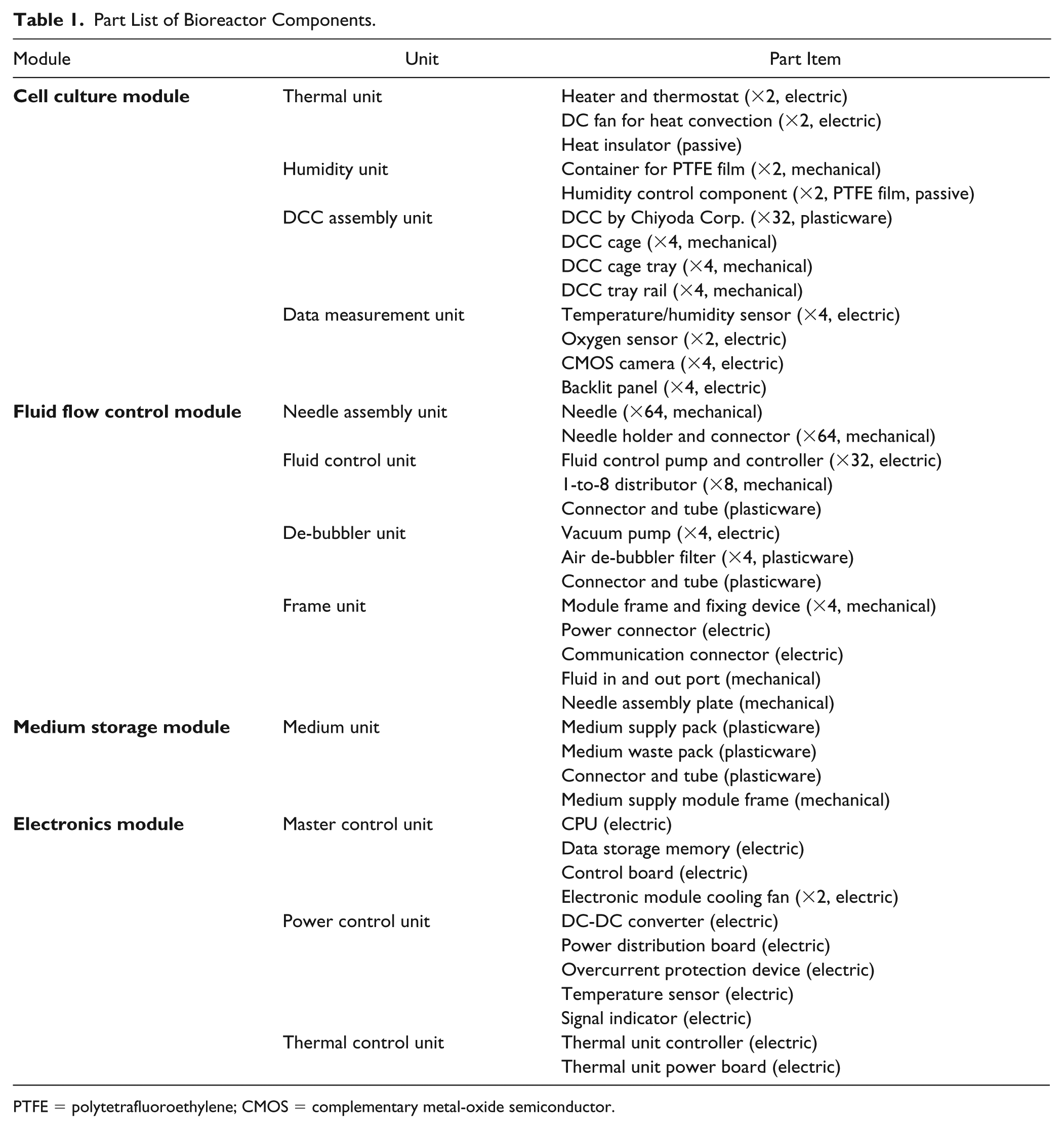
PTFE = polytetrafluoroethylene; CMOS = complementary metal-oxide semiconductor.
Operational Steps of the Bioreactor.
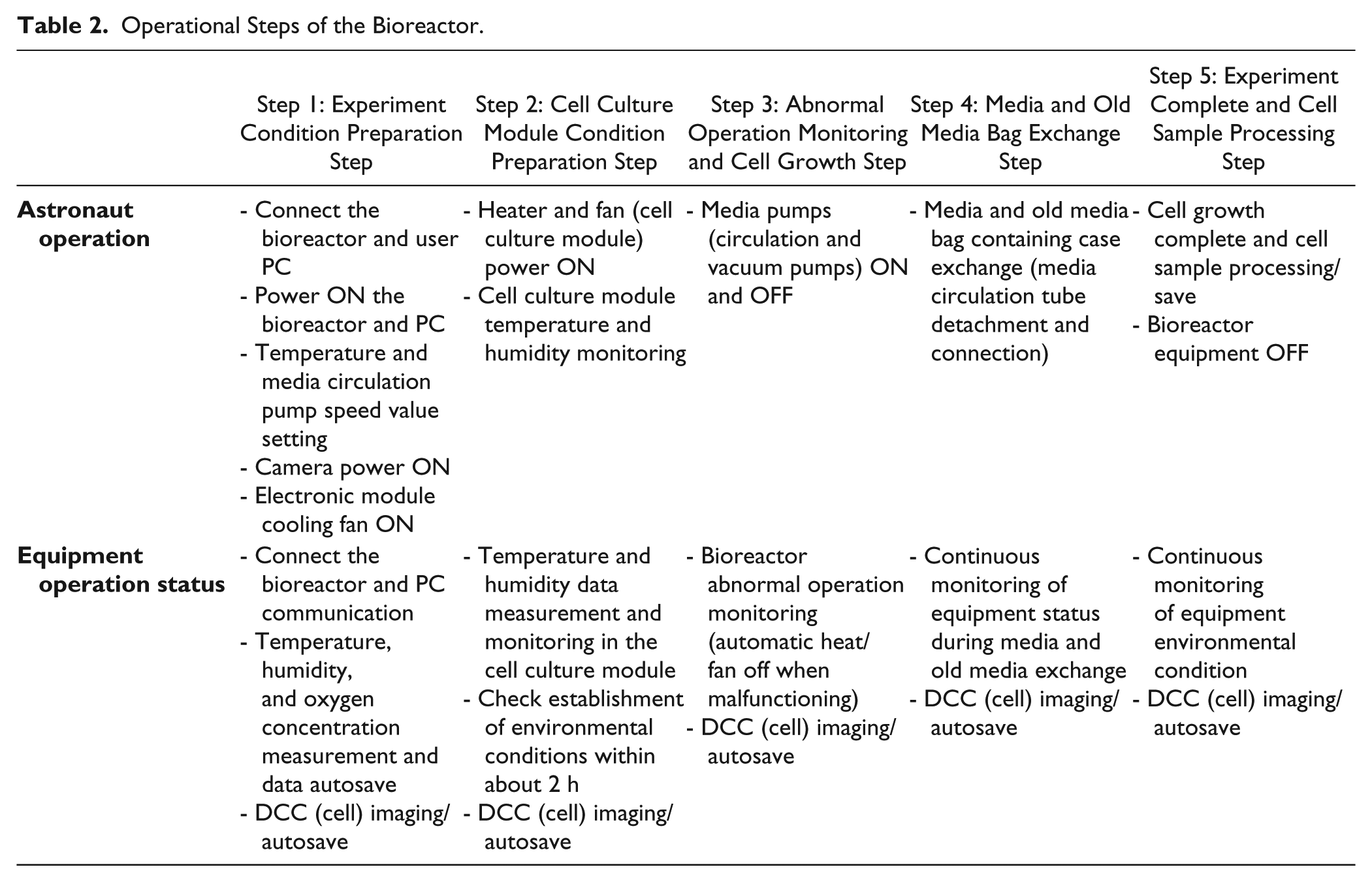
Automated systems to maintain constant bioreactor temperature and humidity are already commercialized in many bioreactors that are used on the ground, and have been applied to various previously developed bioreactors for space experiments. To automate media circulation, several bioreactor automation studies have been conducted for use on the ground. However, applications for space experiments are generally limited. Therefore, it is necessary to design an appropriate mechanism to achieve this media exchange performance, since there has been no case where the media is automatically supplied to and discharged from 32 cell culture dishes, as in the bioreactor developed in this study.
Figure 2a displays the bioreactor EM fabricated in this study. The bioreactor system consists of a cell culture module, fluid flow control module, medium storage module, and electronic module. The bioreactor is designed to be automated and controlled by the electronic module. Figure 2b shows the electric diagram of the automated bioreactor system. The multipurpose small payload rack (MSPR) on the ISS (where the bioreactor is supposed to be installed) provides 28 V of DC power to the electronic module, which is composed of a power control unit, master control unit, and thermal control unit. The power control unit converts the 28 V voltage to 24 and 5 V to provide power to electrical components in each module, including fans, heaters, sensors, and pumps. The master control unit delivers the temperature sensor data of the cell culture module through a feedback loop to the thermal control unit and controls the fan and heater. The media flow rate is controlled by issuing commands to the subcontroller of the fluid flow control module.
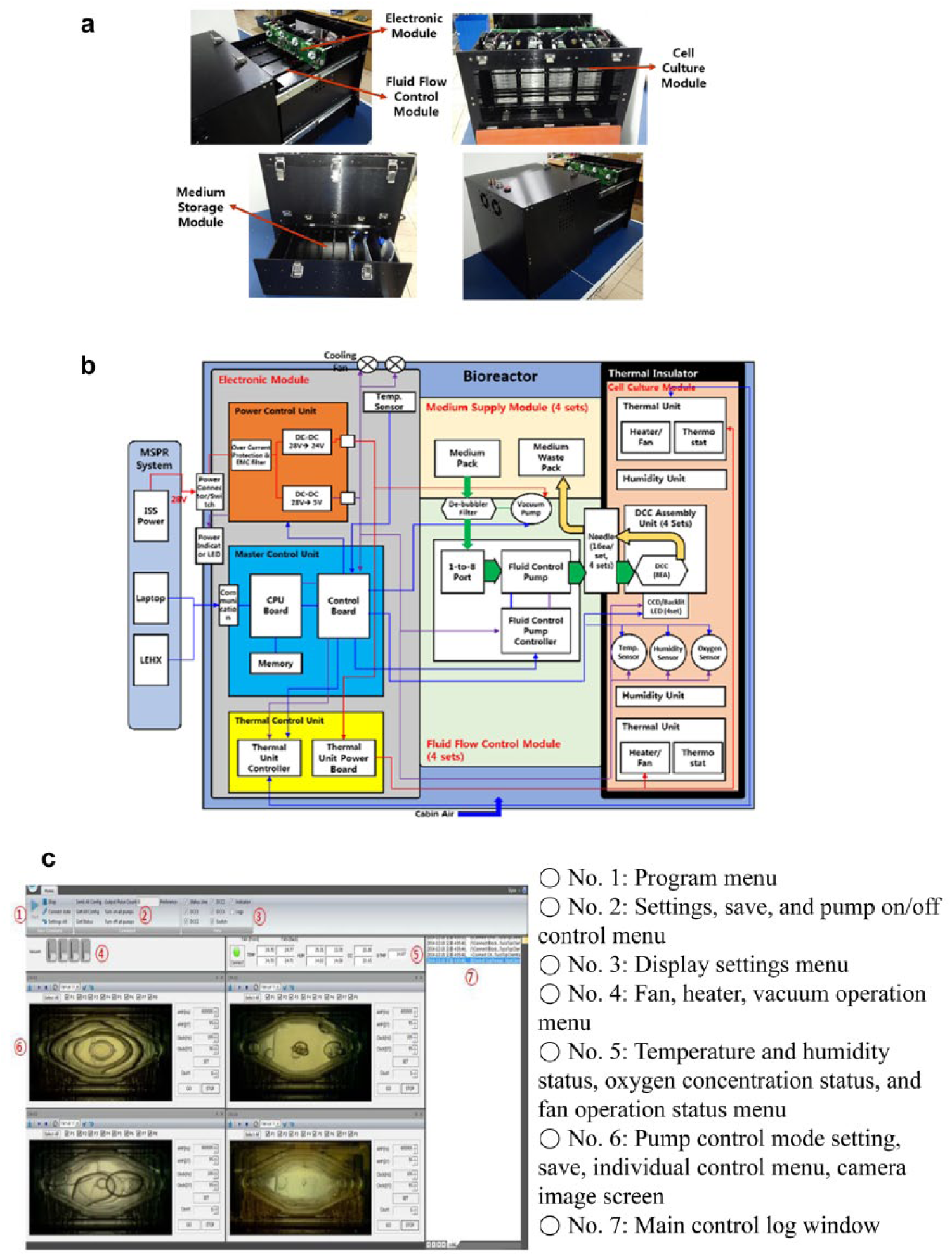
(
Figure 2c shows the graphical user interface (GUI) provided on the user PC that controls and monitors the bioreactor operation through various settings and operation commands. The set values of the clock, amplitude, frequency, and duty ratio for the media circulation were determined experimentally. As shown in the figure, the presence of air bubbles can be observed in the DCC layered within the bioreactor. Since the presence of air bubbles in the DCC negatively impacts the media circulation and cell growth, the de-bubble filter and vacuum pump were used to remove air bubbles. As the fluid flow control works, the air bubbles are filtered and removed (see the lower right image in Fig. 2c ).
Additionally, the user PC regularly receives and displays the control data for all heaters, fans, and vacuum pumps and the temperature, humidity, and oxygen concentration data from sensors. The temperature control of the bioreactor can be set to be automatic or manual. When set to automatic mode, the temperature within the cell culture module is configured and the heaters are automatically turned on and off, while the manual control mode allows the user to control the temperature manually.
Media Circulation to the DCC
The DCC used in this study is a type of the cell culture plate developed by JAXA specifically for in vitro studies in space.15,16 The cell culture dishes used on the ground range in diverse sizes, with their top being open. However, the DCC is a closed chamber (dimensions 86 × 49 × 8 mm, culture area 15.5 m2) with a total volume of 4.65 mL. An air-permeable film is attached to the upper surface so that the carbon dioxide and oxygen necessary for cell growth can be supplied. Also, there are two ports, sealed with silicon, in the DCC for space experiments, and the ports are used to supply the media, which is the nutrient source for the cells. This DCC was used as one of the core components of the bioreactor developed in this study for space experiments.
The DCC was adapted to investigate the automation of the bioreactor media circulation for cell growth. Here, media circulation refers to the process of supplying and discharging the media to and from the bioreactor interior, and this replacement of the media is automated using fluid flow control modules, since the consumable media needs to be replaced at regular time intervals for cell growth. The DCC developed for space experiments is in the form of a hexagon or rhombus in the horizontal direction to facilitate the supply and discharge of the media through the ports.
The fluid flow control module controls the entire media circulation process, supplying the media from the medium pack to the DCC. After the media passes through the 1-to-8 fluid splitter, the media supply rate is controlled by eight piezo micropumps. When the media is delivered, the existing media in the DCC has to be discharged. The newly supplied media pushed by the eight micropumps is sufficient to discharge the existing media. Figure 3a shows the media circulation loop. In order to predict the circulation process of the culture fluid, we performed a computational fluid dynamics (CFD) analysis on the fluid flow control module. Figure 3b displays the calculated pressure distribution inside the fluid flow control module when the media is supplied. For the CFD analysis, the inner fluid was set to distilled water. The fluid velocity at the supply port was 0.001 kg/s, which minimizes the effects of shear stress on cell culture induced by fluid motions. The outlet was set to atmospheric pressure condition as the boundary condition. In order for the culture medium to be exchanged well, a difference in pressure between the supply and outlet ports should exist. Results obtained from the CFD analysis clearly demonstrate variations in the pressure distributions that allow media to flow through the DCC. Further analysis on the media flow through the DCC can be found in our published work. 17
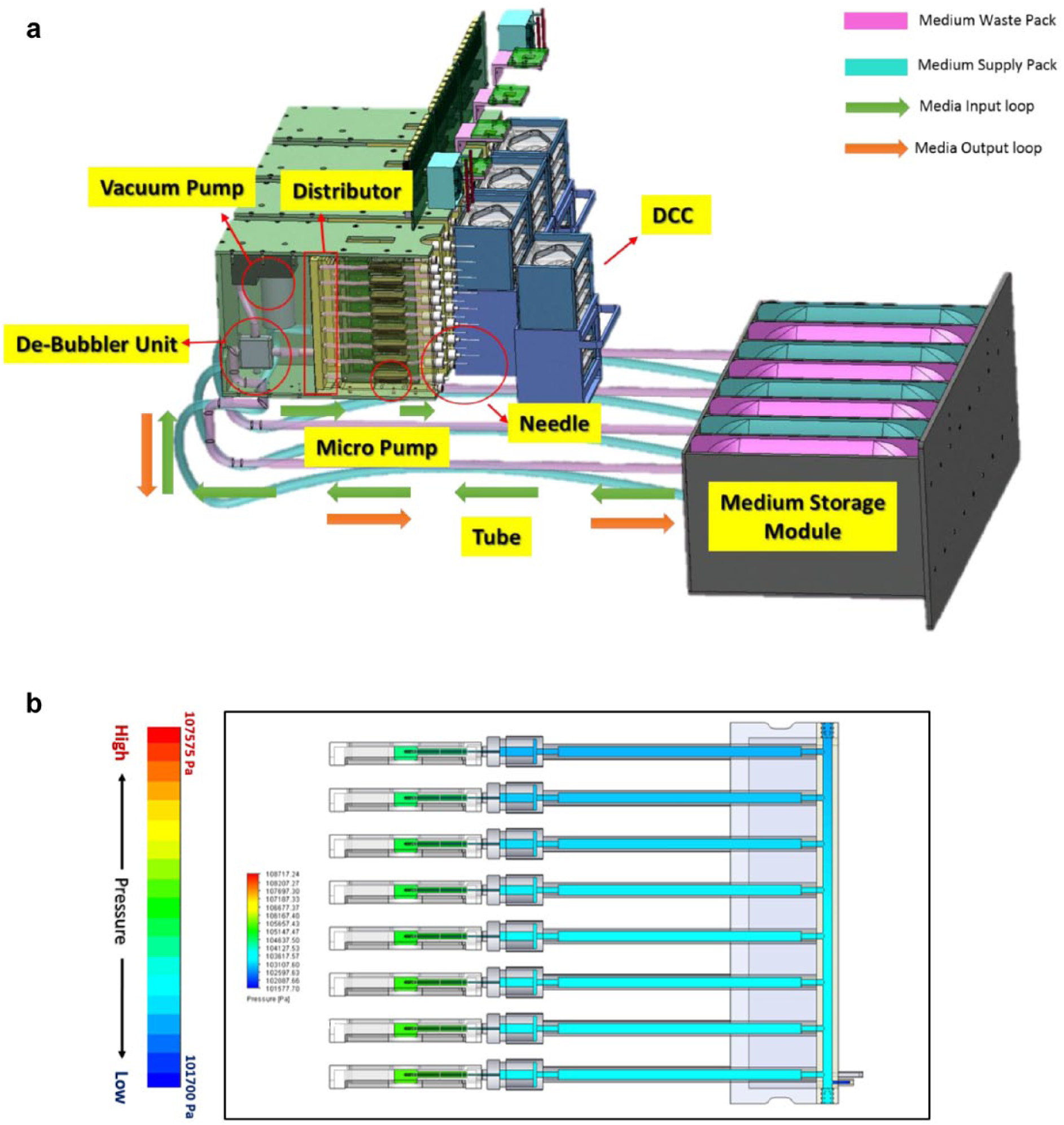
(
Nonetheless, it is still necessary to evaluate whether the media is being effectively replaced using this media circulation process. Although there have been studies where the overall performance of the DCC or developed cell growth system was investigated, by the analyses of cell growth number and density in the DCC, no studies have carried out quantitative performance assessments regarding media circulation using real components. Thus, to use the bioreactor for actual space experiments, such a quantitative assessment is necessary. This is especially important because the microgravity environment in space affects the flow of fluids, so a quantitative evaluation and analysis of fluid flow needs to be carried out on the ground to predict the media flow that will occur in the different environmental conditions of space. Consequently, this study adopted a Bradford assay for quantitative verification of the media circulation performance.
Results and Discussion
Media Exchange Performance Testing Using the Bradford Assay
The equipment developed for cell culture experiments in the microgravity environment was focused on the requirements of the cell culture environment, media, air bubble, system, and ISS, as shown in Table 3 . These requirements pertain to the safety of the user and system performance. Among those, the environmental factors regarding constant temperature and humidity, along with functionality factors concerning media circulation, are the most vital performance indices for the developed bioreactor. Therefore, this study aimed to verify whether the developed automated bioreactor was adequate for use in space experiments by determining, among all the performance requirements, whether the performance criterion of 90% media exchange rate was satisfied.
Top Requirements for Cell Culture Experiment in ISS.
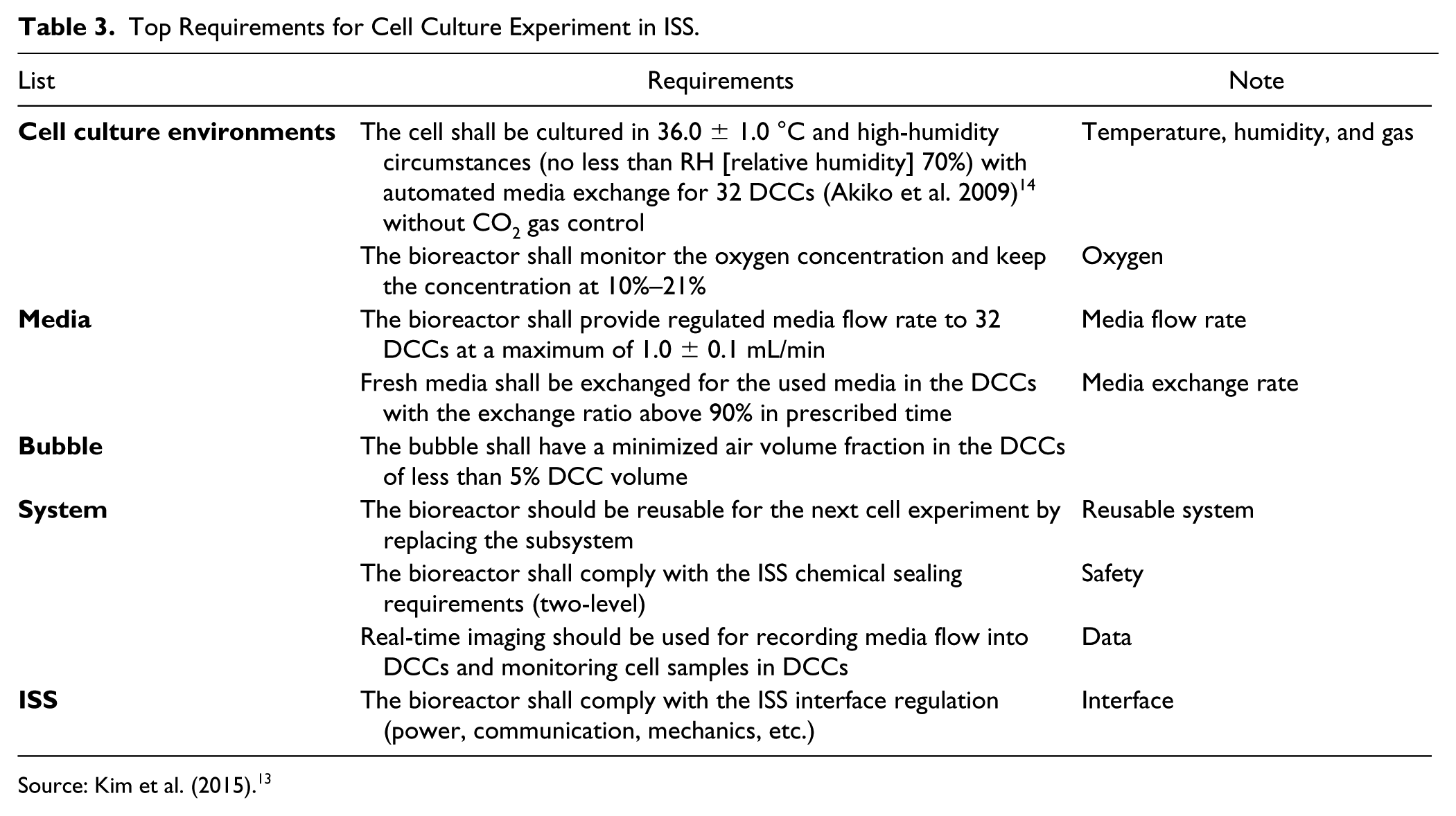
Source: Kim et al. (2015). 13
In order to conduct this assessment, conventional methods of using a camera 13 or verification through cell growth15,16 might not be adequate. Instead, the media exchange performance was assessed using the Bradford assay in a spectrophotometer. The Bradford assay is a general bioengineering experiment where the protein bovine serum albumin (BSA) of a known concentration is diluted to a certain concentration and then tested to determine whether the slope of the graph plot is close to the value of 1. Although this method requires expertise, it can provide much more quantitative data than the camera-based method, such as supplement time, the exchange rate of a medium. In this paper, the media exchange process was simulated by using a BSA protein solution of a certain concentration and distilled water to determine whether the solution was exchanged in the bioreactor. The reason for using distilled water is to simulate the actual space experiment. For space experiments, the DCC will be filled with distilled water on the ground and sent to the ISS to minimize the formation of air bubbles in the fluid flow module due to the presence of air in the DCC.
First, a simulation experiment was conducted to determine whether systemwide media exchange took place in the bioreactor EM. The interior of the bioreactor was filled with distilled water and the medium supply pack of the media storage module was filled with 1 mg/mL BSA. Afterwards, the eight micropumps of the fluid flow control module were collectively operated at specific control values set in the GUI. Then, the BSA was supplied for approximately 30 min, followed by replacement of the medium supply pack with one containing distilled water, which was supplied for 25 min. During the experiment, the fluid discharged from the medium waste pack was collected at specific time intervals and its protein concentration was measured. Figure 4a shows the measured protein quantification value (outer diameter [OD]) as a function of time. As observed in the figure, the OD reached a value of 1 at around 8 min, signifying that the distilled water existing within the bioreactor was replaced with the BSA solution due to circulation by the pumps. For the next 22 min, it can be observed that the solution within the bioreactor maintained a value of 1 as BSA was supplied. Even after the BSA provision, the same state was maintained. For the distilled water supplied from 30 to 55 min, the OD value was 0 at around 40 min, which signified that the BSA solution within the bioreactor was completely replaced by distilled water. This simulation experiment allowed us to measure the OD values for the medium supply pack in the media storage module and for the medium waste pack. These outcomes thus indicate that the media was being exchanged throughout the system.
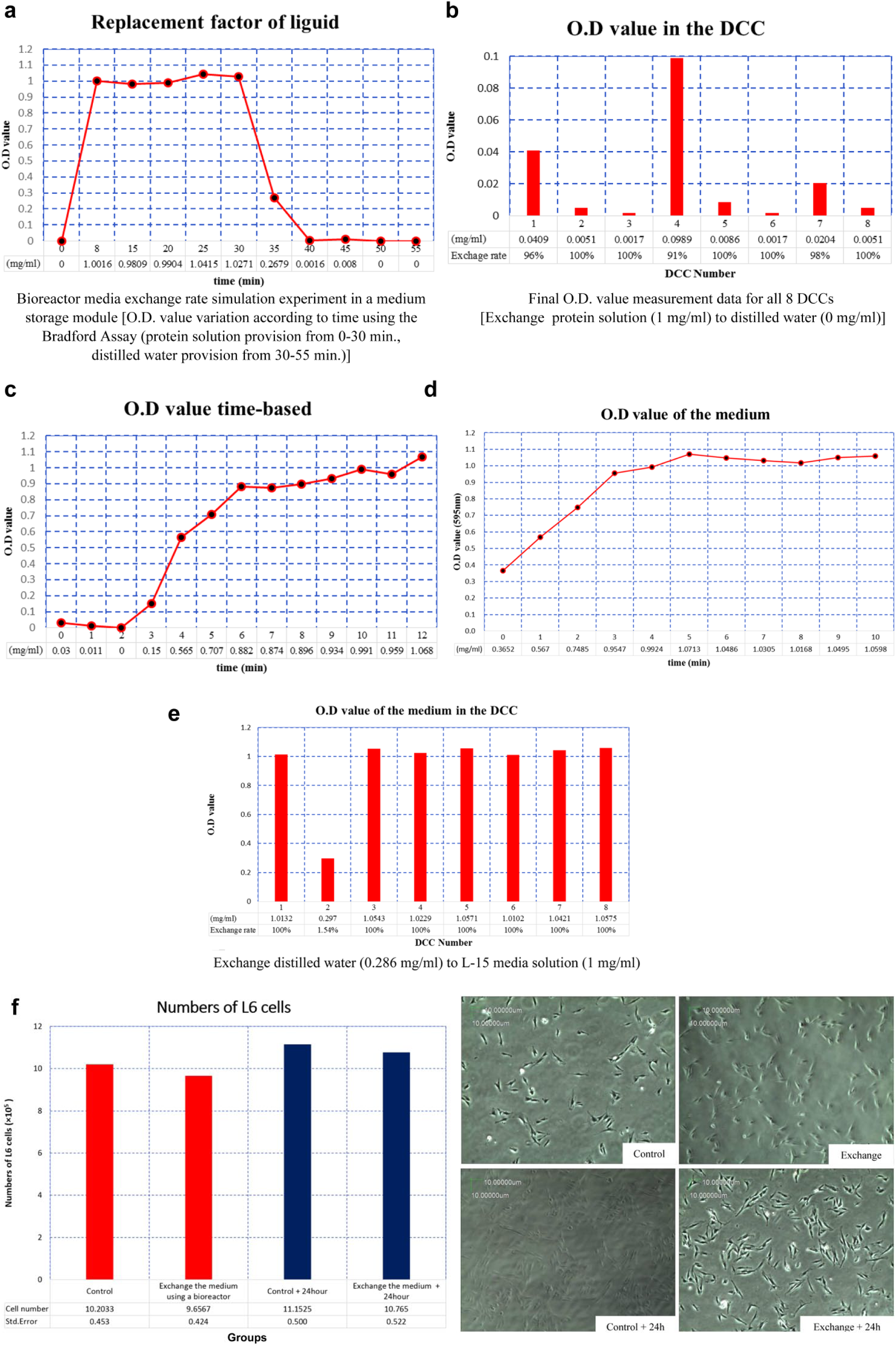
(
Next, it was necessary to determine whether the media exchange occurred within the DCC, where the cell growth actually took place. Syringes were used to extract the solutions from all eight DCCs in the cell culture module where the above experiment had been conducted, and the OD value for each extracted solution was measured. Figure 4b shows the obtained results. Since BSA was exchanged with distilled water in the experiment process, the OD value should be close to 0. As observed in the figure, all eight DCCs underwent at least 90% exchange to distilled water (no. 1 = 96%; no. 4 = 91%; no. 7 = 98%; nos. 2, 3, 5, 6, and 8 = 100%) This result satisfied the performance requirements.
Figure 4c shows the OD values for the solution discharged from the medium waste pack according to time, when BSA was supplied to the bioreactor filled with distilled water. This experiment was conducted to measure the time needed to replace at least 90% of the fluid in the bioreactor and to establish the future space experiment protocol. The experimental result revealed that 90% exchange was attained at 8 min, and when 10 min passed, the distilled water was completely exchanged with BSA. Based on the results of the four performance experiments, it was confirmed that the bioreactor requirements summarized in Table 2 were satisfied.
Tests for Exchange Rate Competence of the System Using CO2-Free Media
For this experiment, L6 cells were cultured in DCCs, with CO2-free L-15 media that contained 10% FBS and 1% penicillin/streptomycin. The BSA was added to the medium until the final concentration reached 1 mg/mL. The experiment proceeded with distilled water, which was exchanged to the culture medium in DCCs. During the experiment, the fluid discharged from the medium waste pack was collected at 1 min intervals for 10 min and absorbance was measured with ELISA to check the quantity of protein from each DCC. The absorbance wavelength was 595 nm.
From the Bradford assay, the OD value of distilled water was 0.286 and that of the culture medium containing BSA was 1.046. As shown in Figure 4d , e , the medium was well exchanged by 6 min, which was 2 min earlier than seen in Figure 4c . From measurements of protein quantity in the solutions of eight DCCs, seven showed the exchange rates of 95% or higher, except one DCC that was exchanged little. This might be due to malfunction of a pump or backward flow of the medium.
Tests for Cell Growth with or without Medium Exchange
To test whether the medium exchange affected cell growth in the automated bioreactor, the L6 cells were divided into two groups, noted as control (no exchange) and exchange. The cells were cultured in eight DCCs per group for 3 days using a conventional incubator. For the control group, four out of eight DCCs were allocated to determine the number of cells in the conventional incubator. The remaining four DCCs were placed in the automated bioreactor, and the cells were cultured for 1 day at 36 ± 1 °C and 72% relative humidity with no CO2 supply and the number of cells was counted.
For the exchange group, the media of the eight DCCs were replaced with the fresh CO2-free L-15 culture medium in the automated bioreactor. The protocol for the replacement followed information of the culture exchange rate and the time span obtained previously. Here, the eight DCCs were divided into two: (1) Four were used to count the number of cells right after replacement, which was comparable to that of the control cells except that the latter were cultured in the conventional incubator with no medium exchange. (2) The remaining four were used to culture cells for 1 day under the same conditions as the control, and the number of cells were counted. The results are shown in Figure 4f . The number of cells were slightly greater in the control than in the exchange before the 1-day culture. However, the number of cells of the exchange group increased 1.12-fold after the 1-day culture, which overrode the increase of the control group, indicating that the bioreactor had sufficient competence to culture cells in the automated environment.
Many of the early models of cell culture equipment that were previously developed for space experiments on the ISS have been manually controlled by astronaut manipulation. At this time, the exchange of the culture medium is the most important task in the automatic cell culture experiment. In this paper, we also intensively examined the culture exchange performance. In fact, we cannot prove that this is suitable for space experiments without actual space experiments, but we have developed a cell incubator that meets the media exchange and cell culture requirements set out by Kim et al. 13
The bioreactor EM was developed through flow analysis and used to conduct a series of experiments that revealed that the automated bioreactor developed for space experiments satisfied the requirement of at least a 90% media exchange rate. In this study, a method of verifying media circulation was proposed using the Bradford assay. Also, we carried out a simple cell culture experiment. The proposed method showed that quantitative evaluation was possible and verified that the developed bioreactor EM had application potential for space experiments. This method is also proposed as a solution for logically determining fluid exchange or circulation in any closed system, such as a plant factory system.
KARI plans to continue research and development to improve the performance of the developed equipment, including the optimization and realization of detailed components. This life science research is expected to act as a catalyst for microgravity environment research in preparation for future manned space exploration. Furthermore, the muscular atrophy prevention material developed by experiments using the equipment developed in this study is expected to contribute to the Mars exploration and space tourism era in the near future.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
