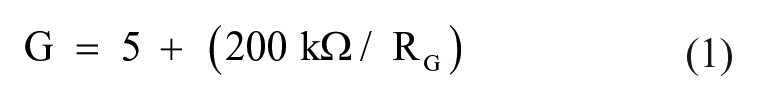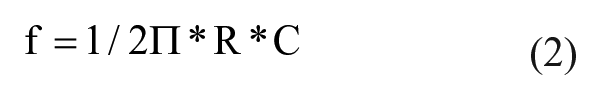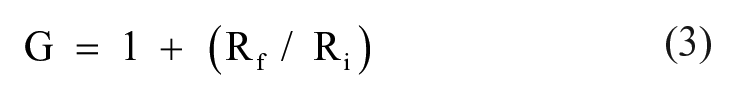Abstract
Measurement of the late potentials and His-bundle activity is crucial for many clinical studies using the noncontact and noninvasive magnetocardiography (MCG) technique; these weak signals are extracted by averaging many cardiac cycles aligned using the R-peak of the cardiac cycle identified using an electrocardiography (ECG) lead. ECG is measured simultaneously with MCG using a conventional dual-supply ECG amplifier, which requires either two separate batteries or a single battery with a switching voltage inverter circuit for its proper operation. The ECG circuitry based on two separate batteries requires a relatively large voltage supply (–18 to +18 V). The single-supply (low voltage: 0–9 V) ECG circuitry may be implemented using a switching voltage inverter; however, this mode of operation introduces switching noise in the system. The objective of the present work is to overcome these problems by carefully designing a low-voltage, single-supply ECG system, which can be used simultaneously with the MCG setup without introducing a significant level of additional noise in the MCG measurement system.
Introduction
Magnetocardiography (MCG)1–8 is a noninvasive, noncontact technique, for measuring the magnetic field associated with the electrical activity of the heart using superconducting quantum interference devices (SQUIDs), which have unparalleled sensitivity for the detection of extremely weak magnetic fields. The MCG offers many advantages over conventionally used electrocardiography (ECG), as the measurement carried out using MCG is noncontact and is less sensitive to the variations of the electrical conductivity of the intervening tissues. Recent studies show that the diagnosis of right atrial hypertrophy and right ventricular hypertrophy can be performed more accurately using MCG compared with ECG; 9 moreover, MCG can precisely detect the regional dominant frequencies of atrial fibrillation. 10 The noninvasive measurement of weak magnetic field associated with the activation of the His-bundle and the late ventricular potential associated with delayed conduction channels across scarred myocardial tissue are regarded as important and clinically useful information;11,12 however, a reliable extraction of such extremely weak signals depends crucially on significantly improving the signal-to-noise ratio (SNR) of the measured MCG by averaging a large number of nominally identical cardiac cycles; the averaged MCG data are commonly known as high-resolution MCG.13,14 To improve the SNR of the measured MCG signal, trigger-based averaging is commonly used. 15
Trigger-locked averaging improves the SNR by a factor of √m, where m is the number of samples taken for averaging. In the case of the cardiac signal, since the R-peak is the highest in amplitude among all the features in a cardiac cycle, trigger-based averaging was performed by taking the R-peak instants as the trigger in the case of the MCG data. The selection of the R-peak instant is generally automated by finding the maxima or minima above a particular amplitude level, commonly known as amplitude thresholding. However, it is difficult to correctly apply the amplitude threshold method on the raw MCG data when it is contaminated by the motion artifacts, as shown in Figure 1 .
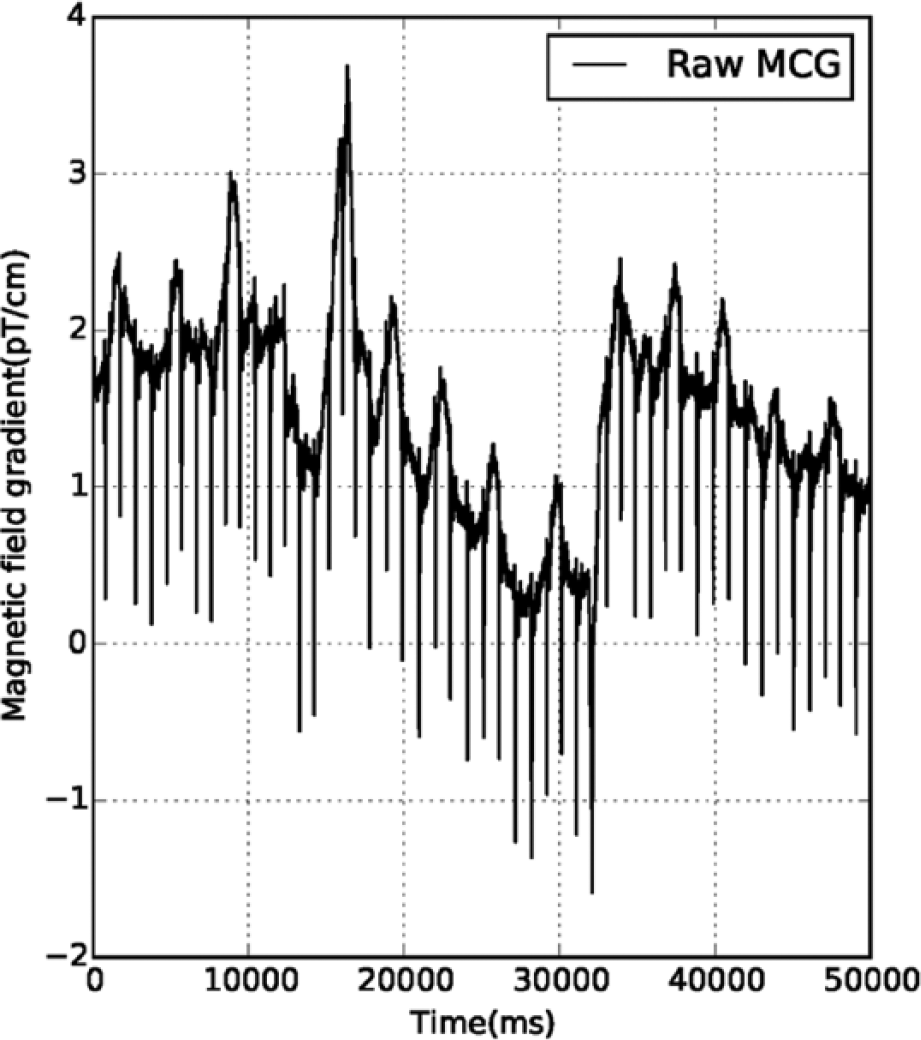
Section of recording from the raw MCG data contaminated by movement artifact.
Several techniques, such as principal component analysis (PCA), independent component analysis (ICA),16–18 and wavelet-based approaches, have been suggested to eliminate the common biological artifacts from the signal of interest. The main problem with these statistical approaches is that they require multichannel data (the number of recording sensors should be greater than or equal to the number of independent sources contributing to the signal), and hence they are unsuitable for the measurement performed using only a few sensors (the present MCG study was conducted using a configuration of four SQUID sensors). On the other hand, the wavelet-based approach is a simple technique for the elimination of artifacts due to its optimal resolution in both the time and frequency domains. 19 The wavelet-based technique relies on a choice of predefined basis functions (wavelets), as well as the selection of a suitable threshold by an expert with the intention of separating the noise and signal components without distorting the signal of interest.
To overcome the issue of identifying the R-peak instant from the contaminated MCG data for averaging the cardiac cycles, we have designed a single-supply ECG system compatible with the MCG system in the sense that it does not introduce a significant level of additional external noise. The designed single-supply ECG system is used simultaneously with the MCG system to acquire the cardiac electrical activity of the subject, and thereafter, the R-peak instant is extracted from the acquired single-channel ECG data. In the designed ECG system based on single supply, linear drifts are removed and ECG signals are smoothed using the low-pass and high-pass filter settings, enabling the easy identification of R-peak instants in the ECG data. To the best our knowledge, simultaneous MCG and ECG recordings were reported in the literature using the ECG system based on dual power supplies, which may not be considered an optimum setup in the context of its use while simultaneously recording the MCG data for the following reasons:
A biopotential amplifier working with bipolar signals requires dual power supplies for proper operation. While this is not an issue in a line-powered system, it is a critical issue in battery-operated applications. 20
It may be possible to operate a dual-power-supply amplifier with a single battery using a switching voltage inverter; however, these switching circuits could add switching noise to the recorded signal. 20
Recently, researchers21,22 have used single-supply-based biopotential amplifiers for designing the multichannel ECG system as an independent setup to measure the variation of electric potential associated with the electrophysiology of the human heart. However, the single-supply-based ECG setup has not been used simultaneously with the MCG measurement in previous studies. In the proposed system, the single-supply-based biopotential ECG amplifier is used with the MCG system, powered by a battery, and packed in a shielded aluminum box, in order to reduce the possibility of contamination of the signal of interest by external sources of noise, such as power line interference and interference between the two different measurement modalities (ECG and MCG).
Single-Supply, One-Channel ECG Amplifier
The proposed approach uses an ECG amplifier, which employs an instrumentation amplifier (IA; INA 122), followed by a high-pass filter, a low-pass filter, and a noninverting amplifier, as shown in Figure 2a . INA 122 (Texas Instruments, Dallas, TX) 23 was used as an IA since it has low noise, a low offset voltage, and low power consumption; indeed, these characteristics make it well suited for several medical applications, such as ECG and electromyography (EMG). The gain is computed using the equation
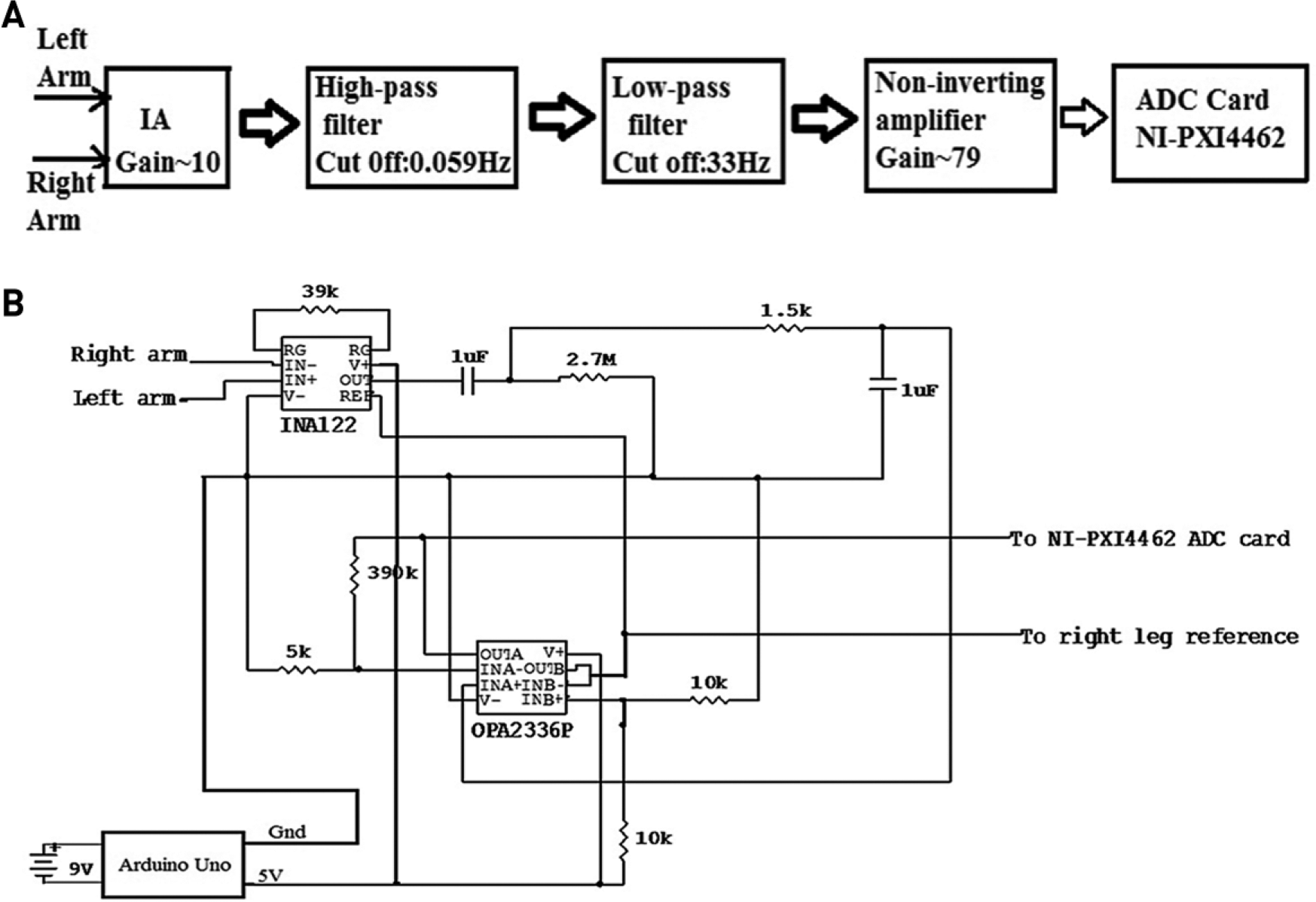
(
We have chosen RG to be 39 kΩ so that IA achieves a gain of 10. The gain is set to values as low as 10 to avoid saturation.24–26 The input signal to be amplified is connected to the input pins of INA 122. After the IA, a high-pass filter stage was used to eliminate the DC offsets and very low-frequency drifts, while a low-pass filter was used to limit the noise bandwidth. In the proposed approach, since the aim was limited to find only the R-peak of the cardiac cycle using the ECG amplifier, we selected frequency cutoffs of 33 and 0.059 Hz for the low-pass and high-pass filters, respectively. The high-pass filter was designed using passive elements, that is, resistor (2.7 MΩ) and capacitor (1 µF). After this, a low-pass filter stage was used, which allowed low-frequency signals to pass through, but attenuated frequencies higher than the chosen cutoff frequency value. The low-pass filter was also designed using only passive components, resistor (5 kΩ) and capacitor (1 µF), which limits the bandwidth to 33 Hz. The values of the passive components for implementing the low-pass and high-pass filters were calculated using the equation
where R is the resistor and C is the capacitor. An operational amplifier OPA2336P (Texas Instruments) was used in the last stage of amplification. This noninverting operational amplifier amplifies the signal with high precision with a gain of 79. For the noninverting operational amplifier, the gain was computed using the equation
where Rf is the feedback resistance of about 390 kΩ and Ri is the input resistance of 5 kΩ. Figure 2b shows the schematic circuit of the single-supply ECG amplifier used in this study to correctly identify the R-wave peaks along with the MCG system. Figure 3 shows a photograph of the single-channel ECG system designed and built during the course of this work. The system was packed properly into an aluminum box with all the wires terminating at the LEMO connector to avoid any interference with the MCG system. The output cables were interfaced to NI-PXI 4462 (National Instruments, Austin, TX) cards using a standard LEMO connector for digitization of the data, while two other wires coming out of the aluminum box were used as the power and system ground, respectively. Generally, a 9 V battery would be enough to power up the entire hardware, but there was apprehension that running the system solely on a 9 V battery based on an unregulated power supply would damage the integrated circuits (ICs). The Arduino Uno microcontroller board was used in this setup, which served two purposes: it allowed one to derive the input power supply since it has an in-built linear voltage regulator, 27 and it also allowed one to measure additional basic functions of the human body, such as temperature near the nostrils for the purpose of correcting baseline wander in the recorded MCG by eliminating the breathing artifact.28,29

Photograph of the single-supply ECG packed into an aluminum box.
Methods
Experimental Protocol
In our experiments, we used a four-channel system for MCG measurements, consisting of four SQUID sensors housed inside a liquid helium cryostat, and the distance between the adjacent SQUID sensors was 4.2 cm. The signal associated with the physiological activity of the heart was coupled to the SQUID sensor using the first-order axial gradiometer to attenuate the distant sources of magnetic noise. The first-order axial gradiometer consisted of two loops of superconducting wire wound in opposition having a diameter of 15 mm and separated by a distance (baseline) of 5 cm. The output of the SQUID was processed by flux-locked-loop (FLL) electronics to provide a linearized SQUID readout signal, which was routed via shielded cables for digitization using analog-to-digital converter (ADC) cards (NI-PXI 4462). The MCG was recorded inside a magnetically shielded room (MSR) to attenuate the external magnetic noise. The MSR consists of two layers of mu-metal (2 and 3 mm thickness) for low-frequency shielding and two layers of aluminum (4 and 8 mm thickness) for high-frequency shielding. 30 The shielding factor of MSR was measured to be 70 dB at 1 Hz and 110 dB beyond 100 Hz. The sensitivity of the MCG setup is typically 20f Trms/cm/√Hz and remains nearly the same after adding the proposed ECG setup. The MCG was measured sequentially on the subject’s chest at 36 locations covering an area of 21 × 21 cm on a 6 × 6 square grid, as shown in Figure 4 . Of three input electrodes of the ECG amplifier used during the present work, two were placed on the left arm and right arm of the subject, whereas the third electrode was placed on the right leg of the subject, which acted as a reference electrode. All three electrodes, used for measuring the ECG, were standard disposable electrodes composed of Ag-AgCl. As our ECG amplifier was powered by using a single supply, it was important that the reference be kept at 2.5 V, allowing the ECG pattern to swing in a positive direction (2.5–5 V) as well as a negative direction (0–2.5 V). Vref (2.5 V) was derived using a buffered resistive divisor, as shown in the schematic circuit diagram (OUTB of OPA2336P in Fig. 2b ).
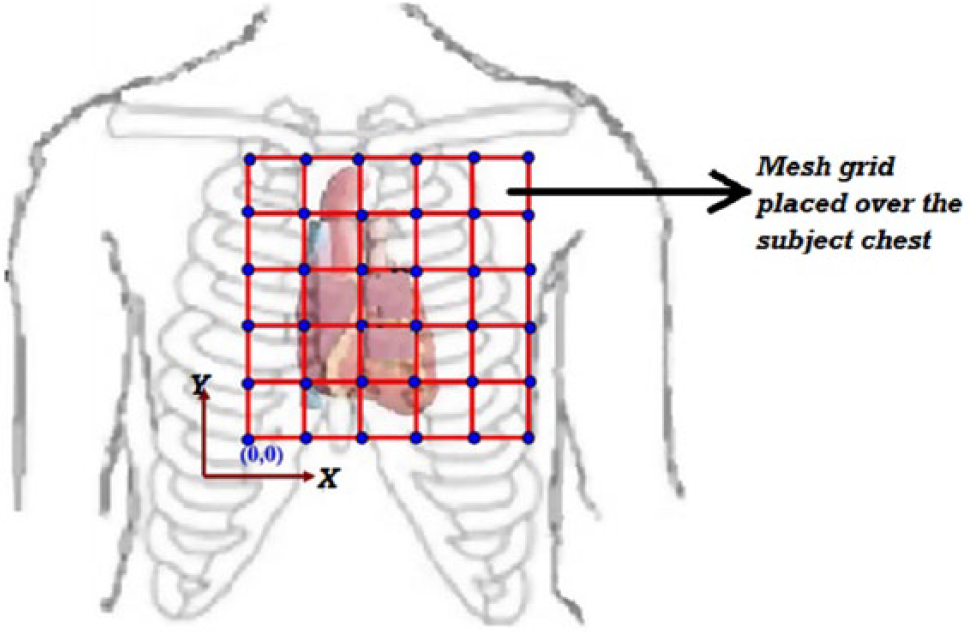
Square grid pattern (the jugular notch over the sternum provided the reference) over the subject’s chest for the MCG measurement.
A four-channel MCG system and the single-supply, one-channel ECG system were used to simultaneously and synchronously acquire the ECG and MCG signals associated with the cardiac activity of a human subject in the supine position at a sampling rate of 1 kHz using NI-PXI 4462 data acquisition cards as indicated in Figure 5 . A total of five male subjects took part in this experiment. The subjects belonged to the age group of 33–40 years; written informed consent for participation was obtained from all the subjects.
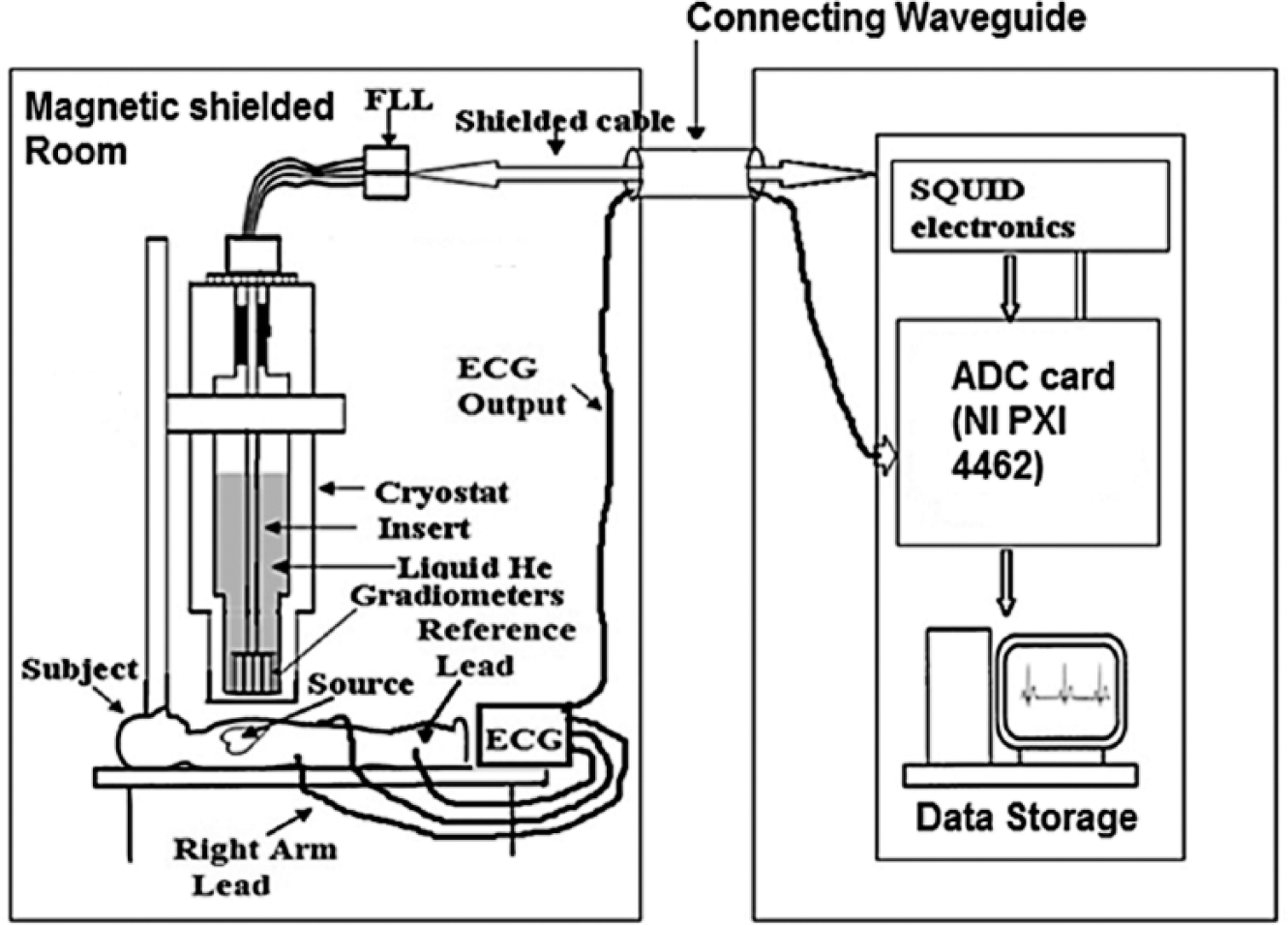
Block diagram of the setup used to simultaneously measure single-channel ECG with MCG.
Data Analysis
Both MCG and ECG data, as shown in Figure 6 , were acquired simultaneously; in both sets of data, the R-peak instants were expected to be identical, as both modalities were measuring the same underlying physiological activity. To improve the SNR of the raw MCG data contaminated by the movement artifacts, R-peak instants were taken as a trigger for averaging a large number (approximately 60) of cardiac cycles. Since the R-peak amplitude of the MCG data was contaminated by artifacts, there was a problem in the correct detection of the R-peak time instants from the contaminated raw MCG data using simple techniques like amplitude thresholding. Figure 6 shows that the ECG data recorded using the single-channel ECG system described in this paper were less prone to the movement artifacts since the ECG electrodes were fixed on the skin surface, unlike the SQUID sensors in the MCG setup, which were inside the cryostat; hence, the problem of the detection of R-peak time instant in the measured ECG data could be addressed relatively easily using the amplitude thresholding technique.
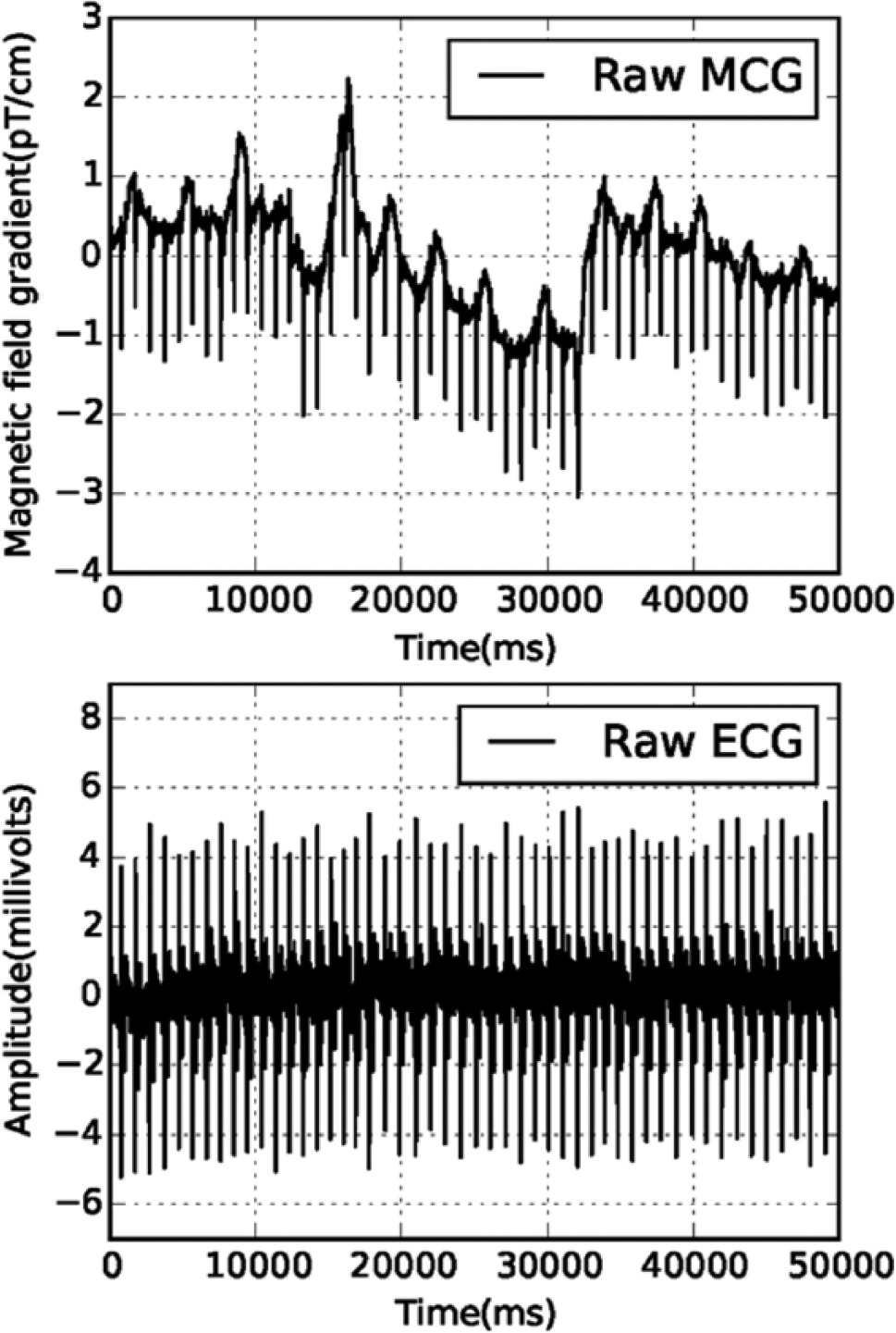
Results and Discussion
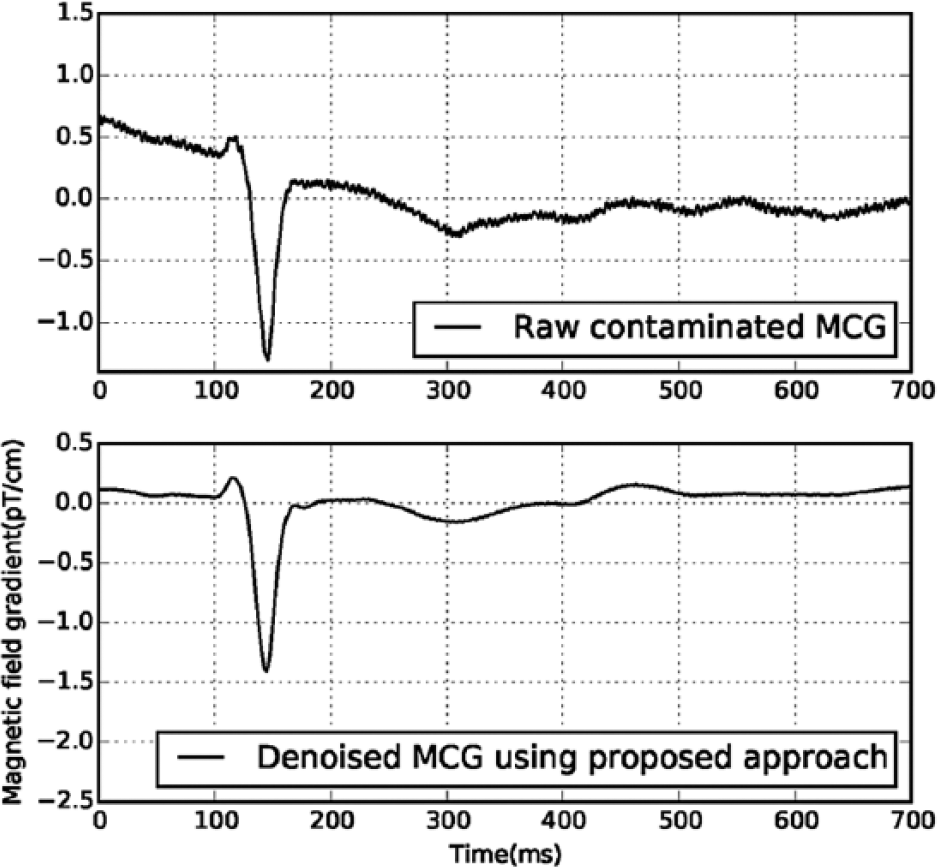
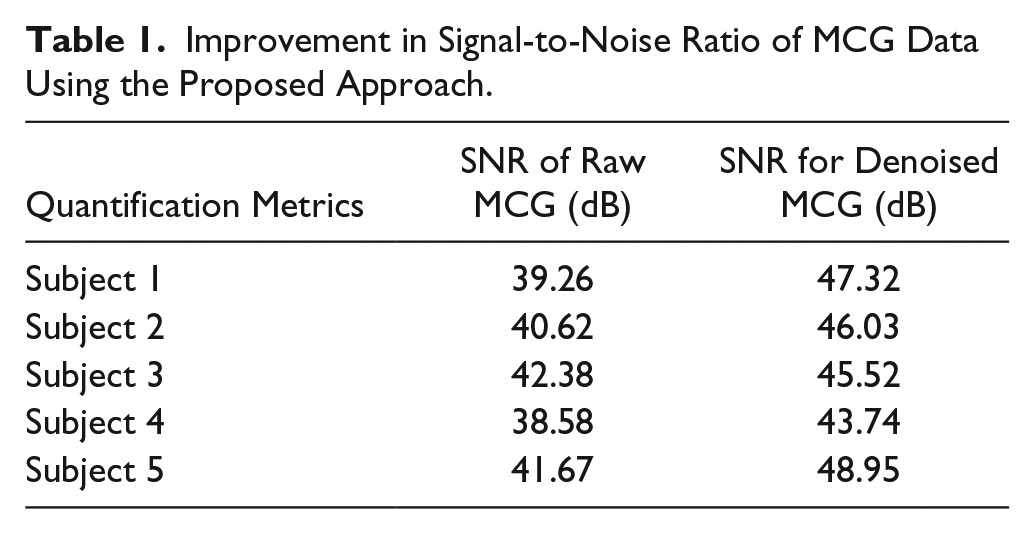
Figure 7 shows a contaminated section of MCG data and MCG data denoised using our approach. Table 1 shows a significant improvement in the SNR of the denoised MCG data using the proposed approach, which is achieved by detecting the R-peak instants from ECG, aligning MCG cardiac cycles on the R-peak instants derived from ECG, and averaging about 60 cycles of aligned MCG data to suppress uncorrelated noise; it may be noted that the system does not introduce a significant level of additional external noise to the MCG measurement system due to the addition of the proposed low-cost, single-supply ECG. Our approach is much simpler, as it involves less human intervention and no careful selection of threshold or noisy components, which is essential for the signal processing techniques based on wavelets and ICA. The proposed approach can be easily automated for processing a large volume (recorded for long duration) of MCG data, as it involves less human intervention or expertise, which is a major limiting factor in artifact suppression using software-based signal processing tools.
Improvement in Signal-to-Noise Ratio of MCG Data Using the Proposed Approach.
Significance of the Proposed Methodology
We have proposed a new methodology for improving the SNR of the MCG signal using the R-peak instants derived from the single-supply ECG system to align and average a large number of cardiac cycles in the recorded MCG. The proposed approach is capable of eliminating the motion artifacts from the contaminated MCG data recorded using even a few measurement channels, without the use of any sophisticated signal processing techniques. The significance of the present method lies in the fact that it is simple and easy to use and enables automated suppression of artifacts after a proper selection of detection threshold for finding the R-peak instant in the single-channel ECG data.
Limitation of the Proposed Methodology
The proposed methodology depends on the proper detection of R-peak instants in the contaminated MCG data segment by using the R-peak instants derived from the single-channel ECG data as auxiliary information for aligning and averaging a large number of nominally identical cardiac cycles in the MCG data. The use of a different ECG amplifier setting may possibly influence the process of R-peak detection. Hence, some fine-tuning of the threshold parameter used for the detection of R-peak may be required to generalize the proposed method. The present approach allows motion artifact suppression in MCG data using a low-cost, single-supply ECG amplifier design; it may also be noted that there is no universal single approach available for efficient suppression of all the different types of artifacts in the biomedical studies, as mentioned by other researchers. 31
Conclusions
We have implemented a new approach to improve the SNR of the MCG data by eliminating the artifacts associated with the subject’s motion from the contaminated MCG recordings without using any additional signal processing techniques. A single-supply, one-channel ECG amplifier was designed using low-cost passive elements and enables extraction of the R-peak instants from the ECG data without introducing a significant level of additional external noise in the MCG system. The proposed methodology is able to successfully capture the R-peak instants from the ECG data using the single-supply ECG amplifier described in this article, and use these R-peak instants as a trigger for aligning and averaging many cardiac cycles measured using the MCG system based on only a small number of channels (four). The experimental results demonstrate significant improvement in the SNR of the averaged MCG signal compared with the raw MCG data. It may also be possible to extend the use of the system in the future by investigating and comparing cardiac anomalies by acquiring data simultaneously using both the MCG and ECG modalities.
Footnotes
Acknowledgements
The authors would like to thank Dr. G. Amarendra, director of the MSG, for his valuable support throughout this research work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.