Abstract
Immunohistochemistry (IHC) provides clinically useful information on protein expression in cancer cells. However, quantification of colocalizing signals using conventional IHC and visual scores is challenging. Here we describe the application of quantitative immunofluorescence in angioimmunoblastic T-cell lymphoma (AITL), a peripheral T-cell lymphoma characterized by cellular heterogeneity that impedes IHC interpretation and quantification. A multiplexed immunofluorescence (IF) panel comprising T- and B-lymphocyte markers along with T-follicular helper (TFH) markers was validated for appropriate cellular localization in sections of benign tonsillar tissue and tested in two samples of AITL, using a Vectra microscope for spectral imaging and InForm software for analysis. We measured the percentage positivity of the TFH markers, BCL6 and PD1, in AITL CD4-positive cells to be approximately 26% and 45%, with 12% coexpressing both markers. The pattern is similar to CD4 cells within the germinal center of normal tonsils and clearly distinct from extragerminal CD4 cells. This study demonstrates the feasibility of automated and quantitative imaging of a multiplexed panel of cellular markers in formalin-fixed, paraffin-embedded tissue sections of a cellularly heterogenous lymphoma. Multiplexed IF allows the simultaneous scoring of markers in malignant and immune cell populations and could potentially increase accuracy for establishment of diagnostic thresholds.
Introduction
Mature T-cell neoplasms (also referred to as peripheral T-cell lymphomas) are a heterogeneous group of tumors arising from post-thymic T cells. 1 The World Health Organization (WHO) defines a number of entities under this category, of which peripheral T-cell lymphoma (PTCL) not otherwise specified (NOS) is the most common.2,3 PTCLs comprise 10% to 15% of non-Hodgkin lymphomas and have a higher incidence in Asia. 4 The diagnosis of PTCL requires integration of clinicopathologic findings and molecular diagnostics. 5 Immunohistochemistry (IHC) is crucial for the differentiation of PTCL subtypes; angioimmunoblastic T-cell lymphoma (AITL), for example, demonstrates CD10 and BCL6 expression, which are not found in most other PTCLs. 1 The prognostic role of IHC has been highlighted by the identification of a T-follicular helper subtype (TFH) of PTCL NOS, which has an adverse prognosis and clinical similarities to AITL. 6 The prognosis of PTCL with the exception of anaplastic lymphoma kinase (ALK)–positive anaplastic large cell lymphoma (ALCL) is poor, with a 5-year progression-free survival of less than 50%. 7 There is hence an urgent need to improve on the current standard of care, which is CHOEP (cyclophosphamide, doxorubicin, vincristine, etoposide, prednisolone) chemotherapy with a consideration of upfront autologous stem cell transplant for fit patients. 8 IHC is proving to be a valuable tool for directing targeted therapeutics. The use of brentuximab vedotin for CD30-expressing PTCL and PD1 (programmed cell death 1) antibodies for PDL1-expressing tumors is an instance when IHC is critical to guide therapy.9,10
Given the importance of IHC in PTCL, accurate and reproducible quantification of staining intensity is crucial. Intensity of staining may be affected by methodological variables such as fixation of tissue, specificity and dilution of antibodies, duration and type of antigen retrieval, and detection systems. 11 Although visual scoring of staining intensity is of clinical value in the hands of an experienced hematopathologist, the data generated remain subjective, with limited reproducibility. 12
Quantitative IHC (qIHC) relies on automated software to perform digital image analysis and provide an objective, accurate quantification of protein expression. 13 qIHC has been tested in epithelial cancers in several settings. For instance, it has been shown to correlate closely with visual scoring in ovarian cancer specimens stained for S100A1 12 and cytoplasmic levels of estrogen receptor β2 expression in prostate cancer. 14 qIHC for proteins in the MET/HGF pathway has been used to derive a predictive model for activation of the MET oncoprotein in a study using 18 different cancer types. 15 The Vectra Automated Quantitative (PerkinElmer, Waltham, MA) pathology imaging system was used to demonstrate concurrently the level of PD-L1 expression and cytotoxic T-cell infiltration in a melanoma specimen, before and after losing response to PD1 antibody therapy. 16 The same platform was used to identify protein biomarkers of lethality in prostate cancer. 17 These studies show that although qIHC techniques require further optimization, they are a promising tool to improve cancer diagnosis and therapy.
Although qIHC has been evaluated extensively in epithelial cancers, few studies have explored this technology in lymphoma. Chabot-Richards et al. 18 investigated Ki-67 expression in diffuse large B-cell lymphoma (DLBCL) and demonstrated good correlation between visual scoring and qIHC. Multiplexed qIHC has been used in a murine model of B-cell lymphoma to study the homing of chimeric antigen receptor (CAR) T cells to malignant B lymphocytes. 19 The authors subsequently used qIHC to investigate PD-L1 expression on lymphoma cells, which they proposed may guide patient selection for immunotherapy trials. 19 Quantitative immunofluorescence (qIF) has also been used to demonstrate increased B-cell receptor signaling in DLBCL with concurrent MYC and BCL-2 expression, suggesting the therapeutic option of Bruton tyrosine kinase inhibition in these patients. 20 These data demonstrate that qIHC and qIF can be applied to lymphoma specimens, although much work remains to be done before this can be translated to clinical use. However, quantitative techniques in histology remain relatively unexplored in T-cell lymphomas.
Methods
Case Selection
The samples were retrieved from the archives of the Department of Pathology, National University Hospital of patients diagnosed with AITL and chronic tonsillitis between 2016 and 2017, under an institutionally approved study (Singapore NHG Domain Specific Review Board B 2014/00693). We used benign tonsil tissues as a comparison to AITL because lymphomas evolve from and often retain epitopes present in normal lymphoid cell types. The repertoire of normal lymphoid cell types is well represented in benign tonsil samples.21,22 Two AITL and three tonsil samples were selected for this study. All patient identifiers were removed, and the samples were assigned a study number to comply with the guidelines of our institutional review board.
Immunofluorescence Staining
The specimens were fixed in formalin and embedded in paraffin at the time of biopsy/surgical excision. For the immunostaining, 3-µm-thin sections of the AITL and tonsil samples were cut. Multiplex immunofluorescence (IF) staining was performed for CD4/CD8/CD20/PD1/BCL6 on 3-µm formalin-fixed, paraffin-embedded (FFPE) tissue sections using the Opal 7-color Fluorophore TSA plus Fluorescence Kit (NEL 797001KT; PerkinElmer). We performed multiplexed IF instead of IHC because combining four or more antibodies was technically challenging using IHC.
Slides were deparaffinized in xylene and rehydrated in ethanol. Heat-induced epitope retrieval (HIER) was performed using target retrieval solution pH 9.0 (S2367; Dako, Santa Clara, CA) with a microwave oven (Dako milestone RHS-1 microwave vacuum histoprocessor) for 25 min. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide for 10 min, followed by 10% albumin for 10 min. First step: slides were incubated with anti-PD1 (ab52587, 1:150; Abcam, Cambridge, UK) for 30 min, incubated with Anti-Mouse HRP (K4001; Dako) for 10 min, and amplified with TSA FITC for 10 min. Second step: slides were subjected to HIER for 10 min and blocked with 10% albumin for 10 min (these two steps were also conducted at the beginning of steps 3 to 5). Slides were incubated with anti-BCL6 (NCL-564, 1:50; Leica, Newcastle, UK) for 30 min, followed by Anti-Mouse HRP incubation for 10 min and amplification with TSA Cy5 for 10 min. Third step: slides were incubated with anti-CD20 (L26, 1:4000; Dako) for 30 min, followed by Anti-Mouse HRP incubation for 10 min and amplified with TSA Cy3.5 for 10 min. Fourth step: slides were incubated with anti-CD4 (NCL-368, 1:100; Leica) for 30 min, followed by Anti-Mouse HRP incubation for 10 min and amplification with TSA Cy3 for 10 min. Fifth step: slides were incubated with anti-CD 8 (C8/144B, 1:100; Dako) for 30 min, followed by Anti-Mouse HRP incubation for 10 min and amplification with TSA Opal 690 for 10 min. Slides were subjected to HIER for another 10 min and counterstained with DAPI.
Multispectral Imaging and Analysis
Acquisition and image analysis on the tonsil and AITL slides were performed with Vectra 2 multispectral automated microscope (PerkinElmer) and InForm 2.2 image analysis software (PerkinElmer), an interactive image segmentation system. For each markers of interest (CD4/BCL6/PD1), the fluorescent intensity count cutoff value for positivity was decided by the pathologist according to the staining pattern and intensity on each selected image. For normal tonsil tissue, each image included germinal center area and extra–germinal center area. These two areas were differentiated by manual tissue segmentation function of the software and analyzed separately.
Results
Validation of a Multiplexed Panel of CD4/CD8/CD20/PD1 and BCL6 Using Normal Tonsillar Tissue
We used a tyramide-based IF protocol to simultaneously stain for a collection of markers in lymphoid tissue. CD4 is a marker of T-helper cells, while CD8 marks cytotoxic T cells and CD20 marks B lymphocytes. A small subset of T-helper cells is normally present within the germinal center. They express the markers PD1 and BCL6 and are termed T-follicular helper cells (TFH). Angioimmunoblastic T lymphomas are presumed to arise from this subset of T cells, and therefore a panel comprising CD4, PD1, and BCL6 was chosen for further analysis. CD8 and CD20 were included as relevant cellular controls and to highlight other immune cells of interest.
FFPE normal tonsillar tissue sections were used for the validation and optimization of the multiplexed stain. The spectral detection system on the Vectra allows for the generation of unique spectra of each individual fluorophore and of autofluorescence from the sample, which can then be used to unmix the multiplexed sample for its individual constituents. The unmixed images and the composite image are depicted in Figure 1 . A pixel threshold for “positivity” of each of these markers was determined by manual inspection of the images by an expert hematopathologist, using a simulated brightfield image and measuring pixel intensity in cell types that were expected to be positive and negative visually ( Fig. 1B–F ). A pixel intensity cutoff that excluded negative cells and included positive cells in the tonsil was chosen for further analysis.
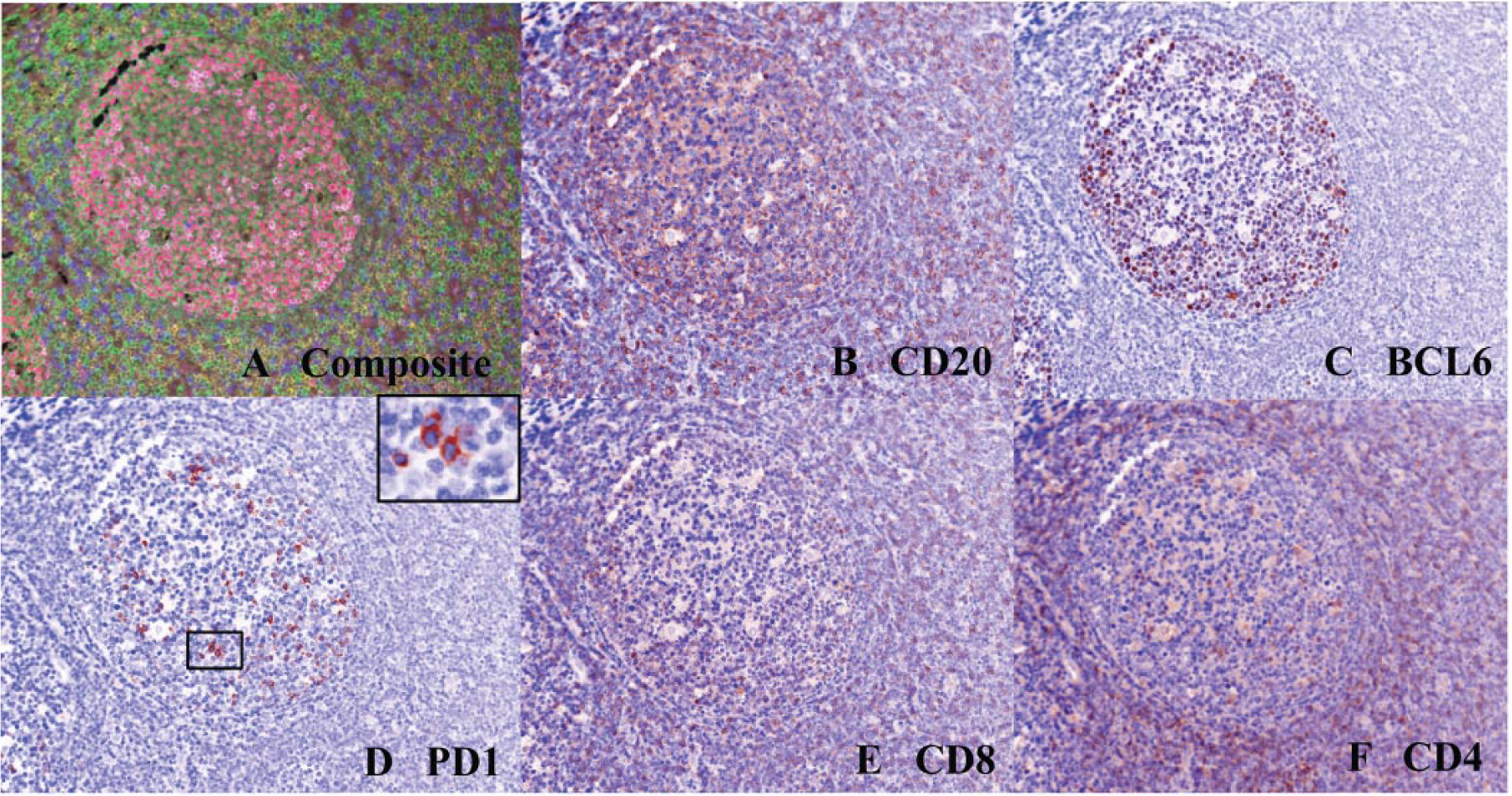
(
Using these derived thresholds for all the above markers, the percentage positivity of cells was then estimated in the normal tonsil. As expected, approximately 10% of cells within the germinal center were positive for CD4. Most of these cells (~75%) also showed expression of PD1, consistent with a TFH phenotype ( Fig. 2 ). The CD8-positive cells in the sample were not positive for PD1. The staining of BCL6 was also noted throughout the germinal center, consistent with its published localization as a germinal center marker. The TFH cells were also positive for BCL6 (~34%).
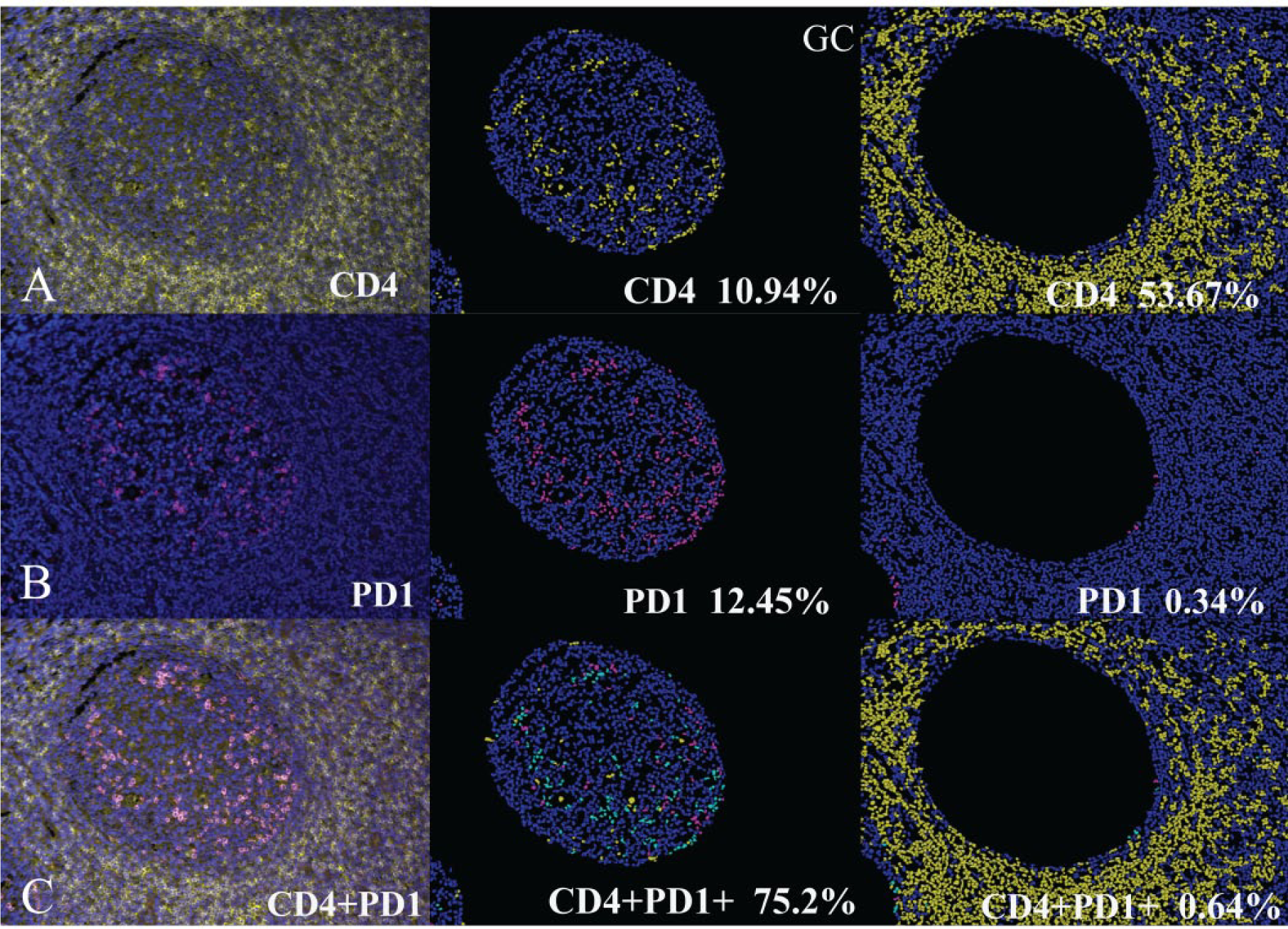
Expression of CD4 and PD1 in a tonsillar section (left column) with corresponding segmented image masks—within the germinal center (center column) and in the extra–germinal center region (right column). (
Application of the CD4/CD8/CD20/PD1/BCL6 Panel to AITL
Having validated the appropriate staining conditions for our markers of interest, we next performed a multiplexed analysis of the abovementioned panel in FFPE samples from two samples of AITL. In comparison to benign lymphoid tissue such as in normal tonsil where B and T lymphocytes home to specific compartments, 2 the samples of AITL show heterogeneity in cellular composition and architecture that is expected of this subset of T-cell lymphoma, as highlighted in an overview image (tissue map) of the multiplexed sample ( Fig. 3A ).
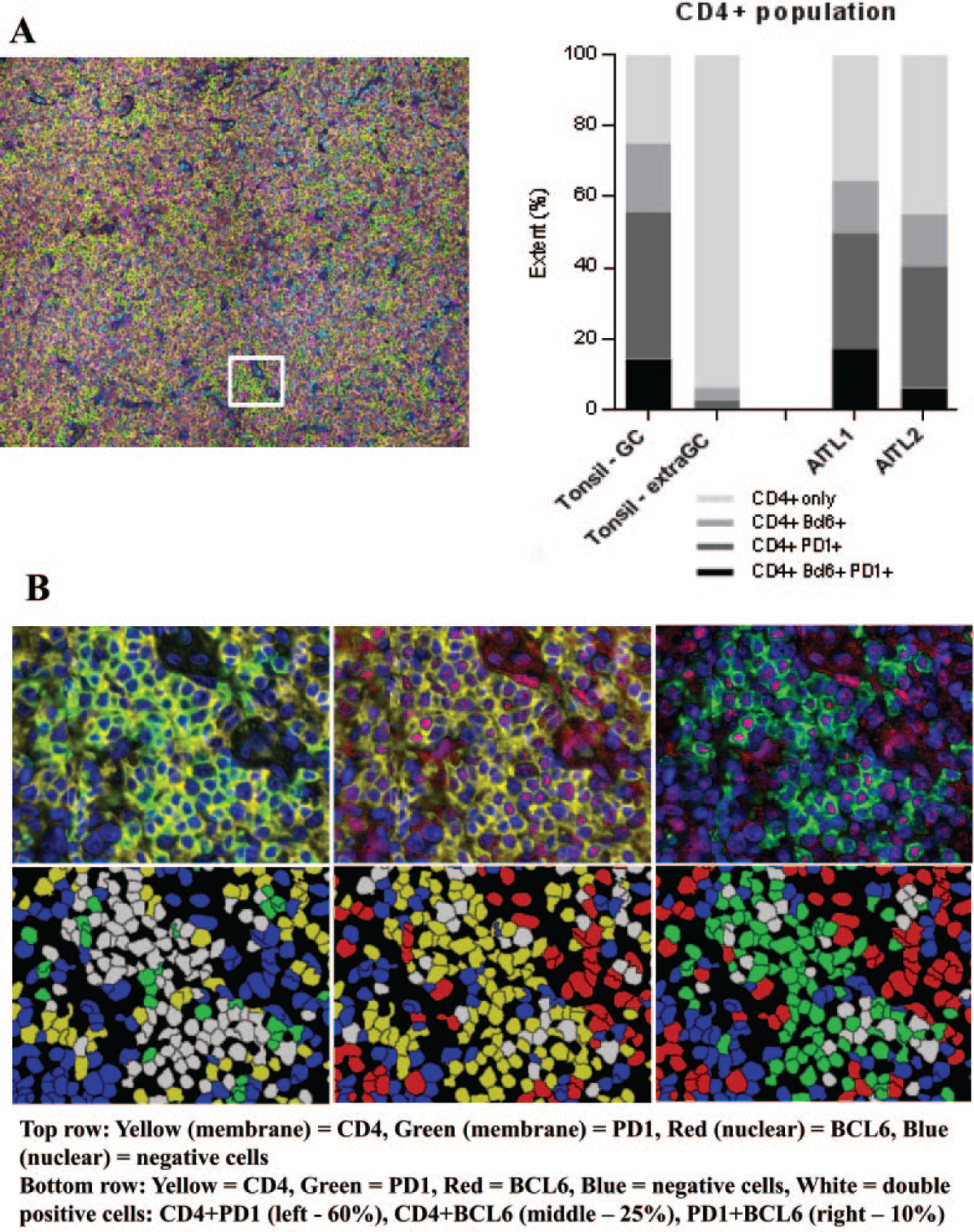
Low-magnification composite images of an angioimmunoblastic T-cell lymphoma (AITL) sample (tissue map) and the analysis of double positivity of markers in interest. (
One of the features that distinguishes AITL from other PTCL is the frequent expression of TFH markers (such as BCL6 and PD1) in the tumor population. 1 A specific challenge with traditional single-marker IHC has been quantitating the precise expression of a marker of interest such as BCL6 or PD1 within malignant cells, which are intimately admixed with abundant immune cells, including reactive T cells, B cells, histiocytes, and stromal cells. The quantitation algorithm within the InForm software allows the specific determination of the percentage of CD4-positive malignant cells within the sample that are also positive for PD1 and/or BCL6 ( Fig. 3 ). This approach allows discrimination from other immune cell types, including B and cytotoxic T cells, which may also show positivity for these markers but are negative for CD4.
In these examples of AITL, 50% of CD4-positive cells in sample 1 and 41% of CD4-positive cells in sample 2 are positive for PD1. Similarly, this quantitative platform allows the estimation of positivity of BCL6 within the CD4-positive cells (32% for sample 1 and 21% for sample 2). Importantly, the ability to multiplex allows the determination of dual positivity (for both PD1 and BCL6) in CD4-positive tumor cells, which we estimated to be 17% and 6% for the two samples studied ( Fig. 3C and Suppl. Table S1 ). The determination of these values in larger data sets of AITL and other T-cell lymphomas may have a potential role in determining diagnostic, prognostic, and/or predictive thresholds for clinical and translational use.
Discussion
This is a feasibility study to demonstrate the applicability of a multiplexed and quantitative IF protocol in a challenging subtype of T lymphoma. Here we discuss some of the challenges and opportunities offered by this protocol.
Challenges to the Approach
1. Unmixing of fluorescent signals:
In a multiplexed IF experiment, the accurate unmixing of fluorescent signals is essential to ensure accurate quantitation of colocalization in these experiments.
23
The testing of the multiplexed stain in a normal lymphoid control where staining patterns are well understood is therefore essential, which we performed here using the normal tonsil tissue section. As the multiplexed staining is sequential, it is essential to ensure that the primary and secondary antibodies are stripped completely from the sample after each tyramide reaction to ensure that there is no cross-reactivity between the different antibodies.
24
Residual primary antibody on the sample can lead to tyramide deposition of a different color during a subsequent step, leading to a false-positive colocalization.23,25 The choice of fluorophore for each antibody is also critical and has to be optimized for each specific tissue type, as the spectral unmixing protocol is influenced by the autofluorescence present in the tissue.
25
Importantly, the validity and reproducibility of the staining protocol can be ensured by testing it on appropriate control tissues and comparison to the corresponding single IHC markers.
2. Definition of positivity:
Once the antibodies are validated and unmixing protocol optimized, the next challenge is in the determination of a positive cutoff during image analysis. It is important to highlight that the percentages for each marker that we derived are dependent on the pixel intensity threshold used for defining the positive cutoff.
23
In our study, these thresholds are set up by manual inspection of a representative field by a pathologist (S.-B.N.), with identification of the threshold intensity in positive cells that are visible in the simulated brightfield image. Once the thresholds are set, they are maintained across samples for consistency. In larger data sets, the inclusion of positive and negative control samples in the staining batch will help account for batch artifacts.
26
Potential Uses
1. Quantitative scoring and multiplexing in lymphoma:
Quantitation of immunohistochemical expression in lymphoma has been hindered by wide variability in IHC protocols and definitions of positivity for cells of interest.
27
The use of automated and quantitative imaging is a step toward improving this, by removing interobserver and intraobserver variability.
12
In this study, we demonstrate that it is feasible to estimate the percentage positivity for the TFH markers, BCL6 and PD1, in AITL tumor cells. The ability to multiplex stains in a single section allows the estimation of these markers within malignant cells, which can be identified by their CD4 expression. Multiplexed IHC has been used in pathology and is typically restricted to two to three chromogens, but quantitation by manual visual scoring using brightfield microscopy lacks precision and is not feasible.
24
With the combination of spectral imaging and automated quantitation, it is now possible to generate a precise numerical score for each marker in different cells.
28
The added ability to multiplex and analyze the expression of multiple colocalized signals will enhance the applicability of this platform beyond routine diagnostic setting. In particular, this approach will be a valuable tool for predicting response to specific treatment by enabling quantification of markers of interest in specific cell types (e.g., immunotherapy in PD1-positive tumors).
2. Spatial resolution:
The use of multiplexed staining and spectral unmixing also allows the creation of a tissue map that clearly demonstrates the distribution of different cell populations and their spatial relationships within the tumor.
23
Subsequent studies using nearest neighbor analysis will allow the quantitation of these spatial relationships to determine their clinical relevance. Such studies will also be of significance for the evaluation of patients undergoing immunotherapy clinical trials, where the tumor cell–microenvironment–immune system relationship is understood to be relevant to outcomes.
29
In summary, in this study, we demonstrate that it is feasible to use an appropriate normal control tissue to establish a multiplexed panel of CD4/CD8/CD20/PD1/BCL6 stains for lymphoma and apply it to two samples of a challenging lymphoma subtype, AITL. The quantitative score for the percentage of PD1- and BCL6-positive CD4 cells in AITL is similar to germinal center CD4 cells and distinct from extra–germinal center CD4 cells. Specific advantages of multiplexed IF over conventional IHC include more objective assessment of antigen expression and the ability to quantify multiple colocalized signals and, hence, the capacity to distinguish tumor from surrounding immune cells. These advantages have potential clinical and therapeutic implications and may help to identify pertinent immunoregulatory targets for future clinical trials of PTCL but require further validation on larger retrospective cohorts.
Footnotes
Acknowledgements
The authors would like to acknowledge the NUH Lymphoma Group for helpful discussions and support.
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: S.-B.N. and A.D.J. are supported by the Singapore Ministry of Health’s National Medical Research Council Transition Awards (NMRC/TA/0020/2013 and NMRC/TA/0052/2016). We also acknowledge a Yong Siew Yoon Research Grant to A.D.J. from the National University Cancer Institute of Singapore toward the purchase of a Vectra Spectral Imager.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
