Abstract
A blood-typing assay is a critical test to ensure the serological compatibility of a donor and an intended recipient prior to a blood transfusion. This article presents a lab-on-disc blood-typing system to conduct a total of eight assays for a patient, including forward-typing tests, reverse-typing tests, and irregular-antibody tests. These assays are carried out in a microfluidic disc simultaneously. A blood-typing apparatus was designed to automatically manipulate the disc. The blood type can be determined by integrating the results of red blood cell (RBC) agglutination in the microchannels. The experimental results of our current 40 blood samples show that the results agree with those examined in the hospital. The accuracy reaches 97.5%.
Introduction
Blood is a body fluid in humans for delivering nutrients and oxygen to the cells and, simultaneously, transporting metabolic waste products away. In case of surgery or heavy bleeding, blood transfusion is a critical medical treatment to replace the lost blood components. Human blood, however, can be classified into several blood group (also called blood type) systems based on the presence or absence of inherited antigenic substances on the surface of red blood cells (RBCs). Prior to a blood transfusion, a blood-typing test must be conducted to ensure the serological compatibility of a donor and an intended recipient. Mistyping of blood group in transfusion is not allowed and might result in intravascular hemolysis, renal failure, and shock. Moreover, serious incompatible RBCs might even cause the recipient’s death. 1 A blood-typing test is based on the antigen–antibody interaction. If antigens on the surface of RBCs correspond to antibodies in the serum, agglutination of RBCs occurs. Therefore, the test result of blood typing can be determined by observing the agglutination of RBCs. In the case of agglutination, transfusion using the donor’s blood to that particular recipient is not allowed.
There are many stages to examining blood products for the sake of successful transfusion. In blood banks, considerable quantities of routine blood-typing tests are executed to examine the blood of all donors, and also to ensure that all individual recipients in scheduled transfusions are given blood that is compatible. Thus, fully automated blood-typing systems are required for blood banks (or medical research centers) to conduct routine blood-typing tests.2,3 These systems are expensive with a large footprint; however, they are not the choice of the local hospitals and clinics. Blood-typing tests usually rely on highly trained personnel in the hospitals or laboratories. Currently, the microplate method, which is carried out in a 96-well microtiter plate, is used by manual operation followed by a visual determination of the extent of RBC agglutination. This assay is tedious and laborious. Moreover, it is not amenable to giving an automated optical readout of agglutination.
To overcome the disadvantage of tediously manual operation, many researchers have reported microfluidic chips for blood typing. Microfluidic systems can manipulate minute amounts of microfluids to provide transportation in the microchannels for the purposes of dilution, particle separation, mixing, and incubation. Thus, microfluidic systems can provide an elegant solution for assays to be conducted in a smaller consumption volume of reagents, a shorter reaction time, and a parallel operation in a single chip, and they have been successfully applied to numerous biochemical analysis procedures. Kim et al. 4 presented a disposable blood-typing biochip by integrating flow-splitting microchannels, chaotic micromixers, reaction microchambers, and detection microfilters, allowing the observation of agglutination with the naked eye. Kline et al. 5 used the microfluidic droplet technique to encapsulate RBCs, antibody, and buffer in droplets to perform multiple agglutination assays. Makulska et al. 6 presented a method to determine the agglutination by measuring the speed of flow of microdroplets, containing RBCs and antibodies, in the microchannel. Charrière et al. 7 designed a fluidic biochip that can trap RBCs onto the surface if antigens and antibodies are complementary. The concentration of the captured RBCs was then detected by an optical absorbance detection module. Paper-based microfluidics is alternatively another branch developed for blood typing.8–13 Moreover, agglutination detection is also an important issue for blood typing.14–16
A blood-typing assay involves forward-typing, reverse-typing, and irregular-antibody tests. Forward typing (also called red cell typing) is performed by mixing a patient’s RBC blood sample with anti-A, anti-B, and anti-D, respectively. In a reverse-typing test (also called serum typing), a patient’s serum is used to react with A cells and B cells, respectively. The purpose of conducting these two major typing procedures is to ensure the accuracy of the agglutination results of each other. In addition, an antibody screening test is to detect the presence of “unexpected” antibodies other than anti-A or anti-B, which are the only RBC antibodies in normal human serum or plasma naturally. In this study, we developed a lab-on-disc blood-typing system, which is a centrifugal microfluidic platform including a multichannel microfluidic disc and a mechanical apparatus, to carry out the assay operation so that the forward-typing, reverse-typing, and irregular-antibody tests can be conducted simultaneously. This multichannel microfluidic disc allowing parallel operations can save time, which is important especially for emergency situations. Furthermore, this apparatus accepts insertion cases to handle emergency assays.
Materials and Methods
This proposed lab-on-disc blood-typing system involves a disc with eight microchannels and an apparatus to manipulate the rotation of the disc for conducting bioassays. The disc was designed to execute three different kinds of blood-typing tests for a patient. It is disposable to avoid cross-contamination of blood samples from different patients.
Protocol of Blood Typing
The manual polybrene (MP) technique for blood typing is widely used in Asia. The conventional test protocol involves a series of mixing, incubation, and centrifugation steps.17,18 The blood-typing assay is usually carried out in a 96-well microtiter plate. First, RBCs, test serum (or plasma), and low ionic medium (LIM) are added to the microplate. LIM can promote the reaction between RBC antigens and antibodies if they are associated. After one minute of incubation at room temperature, the plate is centrifuged. Then, the supernatant is flicked out of the plate, and one drop of 0.05% polybrene solution is added. The function of polybrene is to cause nonspecific RBC aggregation. After mixing and centrifugation, the supernatant is discarded, and one drop of resuspending solution, which is for reversing the nonspecific polybrene-induced aggregation, is added. Agitation is thus performed. Finally, agglutination is observed. The procedure is shown schematically in Figure 1 .
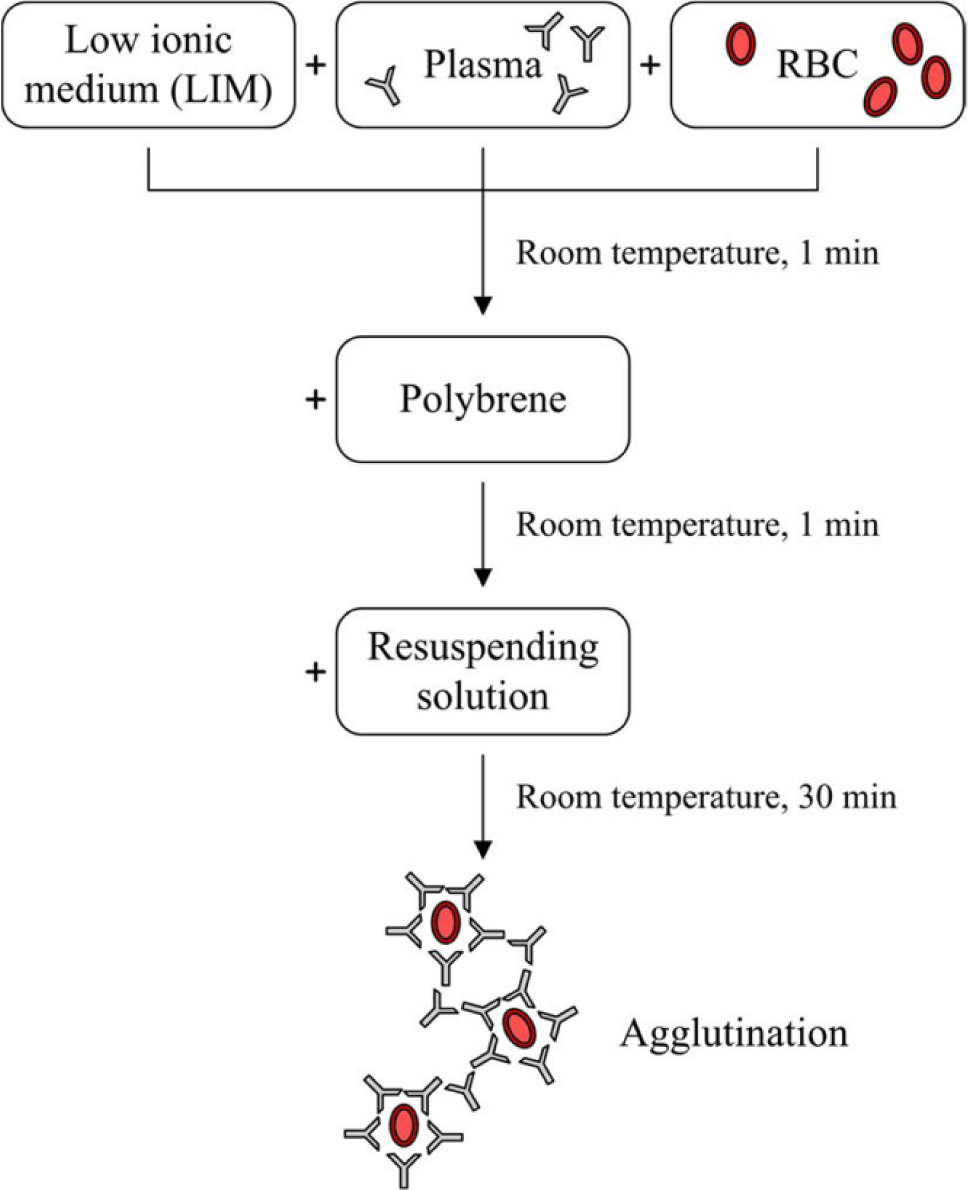
Conventional test protocol of the manual polybrene (MP) technique for blood typing.
The essential biomaterials for carrying out blood-typing tests, including A cells, B cells, anti-A, anti-B, anti-D, and antibody screening cells I, II, and III, were purchased from Formosa Biomedical Technology Corp. (Taiwan). In this study, a microfluidic technique was adopted so that only minute amounts of blood sample and reagents were required. Forward-typing tests, reverse-typing tests, and irregular-antibody tests can be carried out simultaneously. Because the two most important blood group systems are ABO and the RhD antigen, a total of eight tests must be conducted. Therefore, eight microchannels were designed on the disc. Three microchannels were used to perform the forward-typing tests using a blood sample of 3% diluted RBCs to react with anti-A, anti-B, and anti-D, respectively. Two microchannels were used to conduct the reverse-typing tests using serum to react with A cells and B cells, respectively. The other three microchannels were for irregular-antibody tests using serum to react with antibody screening cells I, II, and III, respectively.
Microfluidic Disc
The microfluidic disc, as shown in Figure 2c , was fabricated using injection molding. Polystyrene (PS) PG33 was adopted as the raw material so that the disc is transparent, allowing us to observe the agglutination of RBCs with the naked eye or to capture images of agglutinations. Eight microchannels, located symmetrically on the disc, have the same channel pattern. Figure 2a shows the design of a microchannel. There are five chambers for loading the necessary reagents. The reagent volumes needed for the assay are indicated in Figure 2a . All chambers link to a main channel that is used to transport the fluid into the reaction chamber. In this study, the width of the main channel is 200 µm. The injected reagent is kept in the chamber by the capillary valve. The capillary valve is a kind of passive valve without any moving parts. Its shape and dimensions are shown in Figure 2b . It has a sharp increased width in the cross section of the microchannel, producing the resistance of the capillary pressure for the microfluid in a capillary valve to stop the flow.19,20 Therefore, during rotation, the valve can prevent the reagent from flowing out of the chamber because of the balance of centrifugal force and capillary force, resulting from the surface tension in the junction where the microchannel meets the valve. When the centrifugal force is made greater than the capillary force by increasing the spinning speed, the reagent bursts through the valve and begins to flow to the reaction chamber. Thus, this spinning frequency is called the burst frequency. Because the capillary valve at the outer edge of the disc has the smaller burst frequency, the reagent in the chamber close to the reaction chamber will be transported first. The capillary valve at the inner edge of the disc having a larger burst frequency leads to a delayed reagent transportation. Thanks to the design of capillary valves, these different reagents flowing from their respective chambers can be driven one by one into the reaction chamber by increasing the rotation speed of the servo motor. That is, these reagents can be added in a specific sequence according to the test protocol of blood typing.
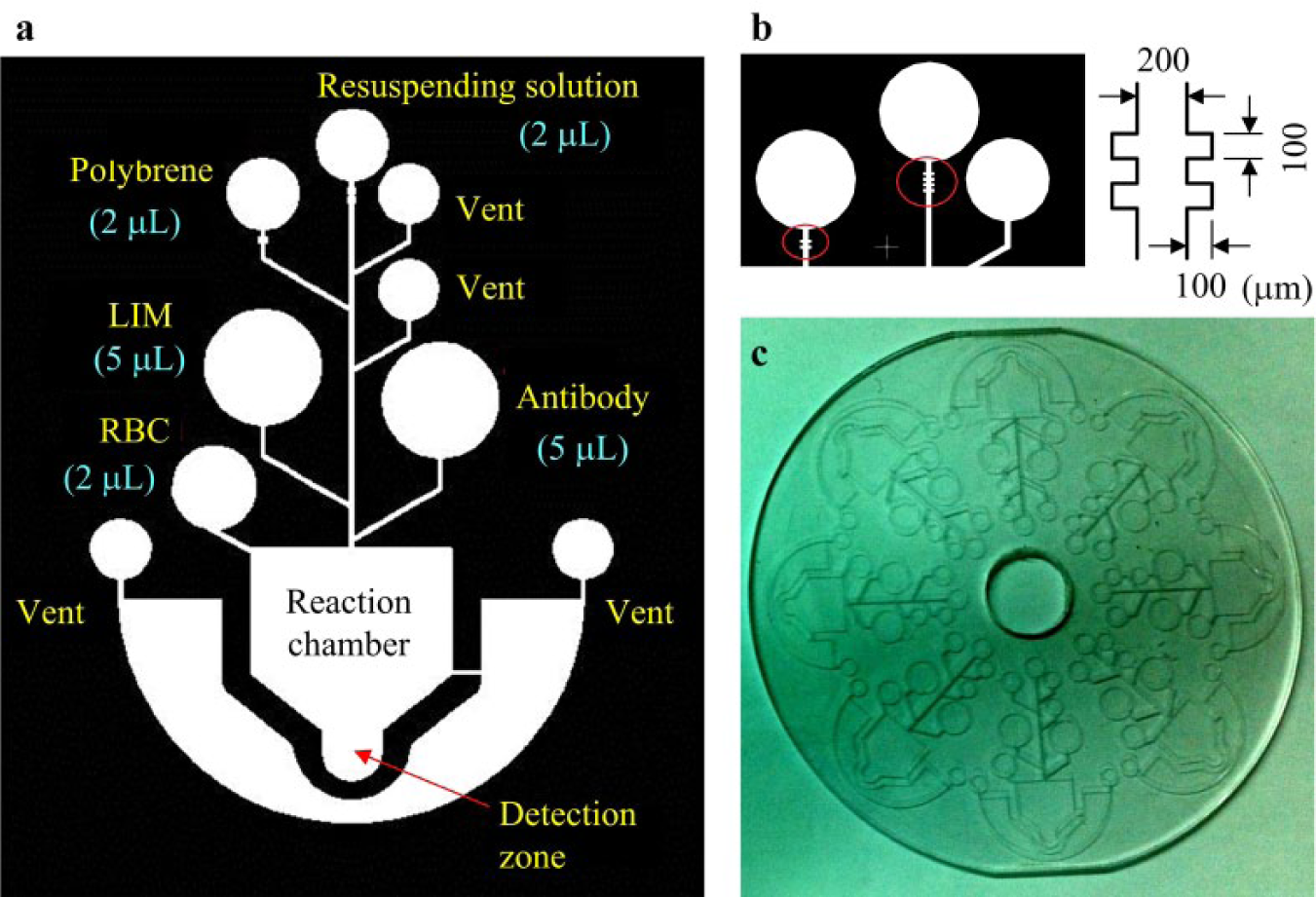
Microfluidic disc: (a) Design of microchannel. (b) Design of capillary valve. (c) Disc fabricated using injection molding.
There is a vent on each side of the reaction chamber. The vent is a small opening to permit the escape of air so that the microfluid can flow inside the closed space of the microchannel. When all of the reagents and sample are driven eventually into the reaction chamber, the reactant solution will be swung to the rim of the reaction chamber in the radial direction due to the centrifugal force. Hence, a detection zone is designed allowing the reactant solution to enter. This zone is an extension area protruding from the reaction chamber to collect the RBC agglutinations together for image observation. That is, the detection zone allows us to capture images of agglutination without scattering over the reaction chamber. After mixing and incubation, the redundant solution is drained to the waste reservoir according to the MP protocol to flick away the supernatant. Only 20% of reactant solution needs to be kept for further detection. Different formations of the agglutinations are produced in the eight reaction chambers. Thus, all of these eight results are integrated to determine the blood type.
Blood-Typing Apparatus
The blood-typing apparatus consists of many subsystems, including a dosing module, a spinning module, a control module, an image capture module, and so on. Figure 3a shows the apparatus.

Blood-typing apparatus: (a) Photo of the apparatus. (b) Dosing module. (c) Operation of dosing module to add the reagent into the chambers on the disc.
The dosing module, as shown in Figure 3b , has similar functionality to the commercial pipettes. It is a specially designed piping mechanism comprising several long thin pipes. Every pipe in this module is controlled by a motor, so that it can draw a constant quantity of liquid such as a specimen (3% diluted RBCs or serum) or a reagent from the test tube and distribute the liquid to the associated chamber on the disc. Figure 3c shows that the reagent is introduced into the chambers on the disc, respectively. After adding specimens and different testing reagents onto the disc, the spinning module rotates the disc to produce the centrifugal force to manipulate the flow of reagents. Then, these reagents are added to the reaction chamber in a specific sequence. The test protocol of blood typing, however, involves a series of mixing steps. To mix the specimen and reagents thoroughly and rapidly, the disc was controlled to rotate a small angle back and forth. After the procedure of a series of mixing and incubation steps, the images of bioassay results were captured using a charge-coupled device (CCD) camera that was mounted atop the spinning module. These images are recorded in a database under the file of a patient. The blood type can be determined based on these eight images.
The entire procedure, from dosing to spinning to recording, is precisely controlled by the control module using an industrial personal computer. This procedure follows the protocol of the MP method, but it is specifically modified for this lab-on-disc system.
After the specimens and reagents are introduced into their respective chambers, the electrical motor begins to spin at 1400 rpm for 10 s to drive LIM into the reaction chamber. Then, the electrical motor rotates back and forth six times to mix the specimen and LIM. Next, the spinning speed of the electrical motor is raised to 1700 rpm for 10 s to add polybrene. Mixing is performed once again to mix the solution thoroughly. At the spinning speed of 1800 rpm for 10 s, the supernatant is drained out to the waste reservoir. Then, the spinning speed of the electrical motor is raised to 2150 rpm for 10 s to add the resuspending solution, and mixing is performed for 20 times. The burst frequency for each aforementioned step was tested using the mercurochrome solution and, finally, confirmed by experiments because the viscosity of the mercurochrome solution is different from those of the reagents.
The external dimensions of this blood-typing apparatus are W890 × H890 × D810 mm. Hence, it can be easily installed in medical facilities and laboratories, which have the need to test few or important specimens. Up to 20 discs can be loaded into this apparatus, so that it can perform continuous blood-typing tests. In addition, this apparatus can be interrupted for an emergency test. It provides flexible operations for either the scheduled tests or the exigent tests.
Results and Discussion
Freshly drawn venous blood samples from 40 informed and consenting patients were used for the experiments. Their blood types were examined in the hospital by medical technologists; however, this piece of information about the results was masked for blind tests to eliminate bias of experiments using this proposed system.
Results of Forward- and Reverse-Typing Tests
Determination of blood type relies on the results of forward-typing tests and reverse-typing tests. That is, the blood type can be concluded based on the recorded images of the first five microchannels. In the detection zone of a reaction chamber, the RBC agglutination results in a visible alteration of the appearance of the blood sample to a crimson spot, whereas non-agglutination remains limpid. Hence, by observing the result of agglutination or non-agglutination in the reaction chamber of each microchannel, the final decision of blood type can be made. If, for instance, a blood sample of 3% diluted RBCs agglutinates with anti-A but non-agglutinates with anti-B, and, moreover, if serum agglutinates with B cells but non-agglutinates with A cells, the blood type is A. The rules for determining the blood types are listed in Table 1 . Figure 4 shows the images captured in all eight microchannels for blood types A, B, O, and AB, respectively. Because anti-A is blue-dyed, the reactant solution in microchannel 1 displays in blue color, resulting in a fuscous image. The discrimination between agglutination and non-agglutination was obvious, however, and the agglutinative determination was not influenced by this color appearance.
Determination rules of blood type.
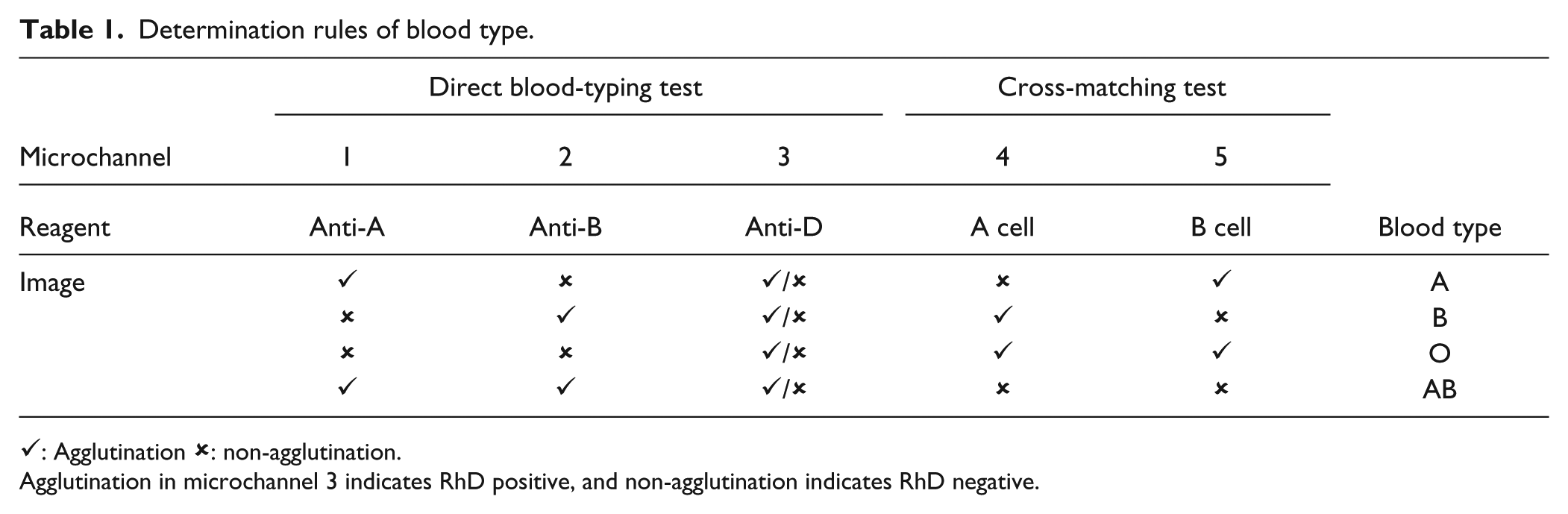
✓: Agglutination x: non-agglutination.
Agglutination in microchannel 3 indicates RhD positive, and non-agglutination indicates RhD negative.
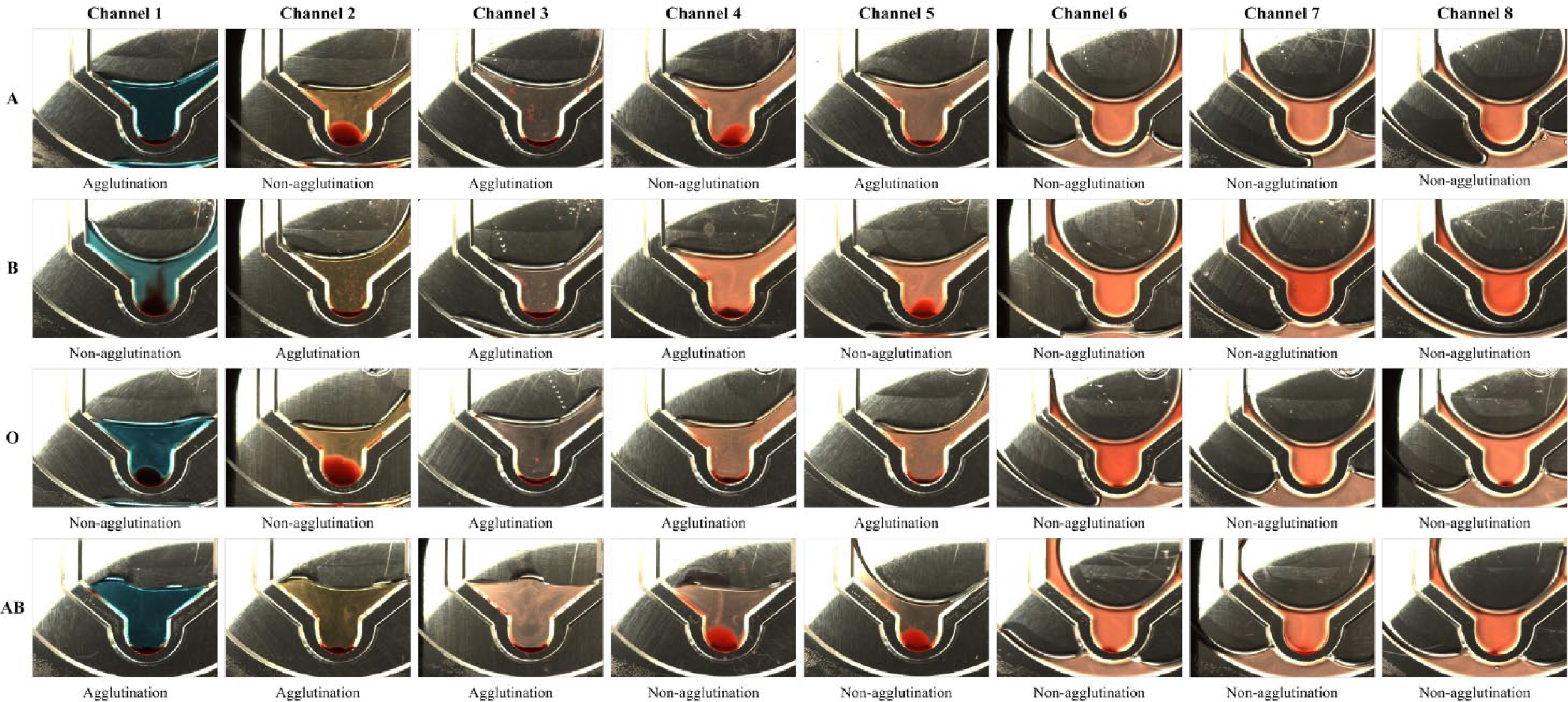
Images of eight microchannels for blood types A, B, O, and AB, respectively.
After the image capture module records all of the images, it reports the result of blood type according to the rules listed in Table 1 . If the images for a certain blood type agree with the corresponding rule, conclusion reaches without any debate. Any weak agglutination, however, resulting in an undiscriminative appearance from the non-agglutination phenomenon leads to a controversial conclusion. Then, the test must be reconducted by highly trained medical technologists because mistyping of blood group in transfusion is not allowed.
Among the 40 blood samples, the results of 39 samples agree with those examined in the hospital. The accuracy reaches 97.5%. The image capture module reported that the other blood sample needed to be reexamined. As shown in Figure 5 , the image of microchannel-5 was classified as non-agglutination due to the small quantity of RBC agglutination. This proposed system could not conclude a blood type and gave a proper warning. Our inference is that this blood sample was kept for a longer period before assay, leading to insufficient reaction.
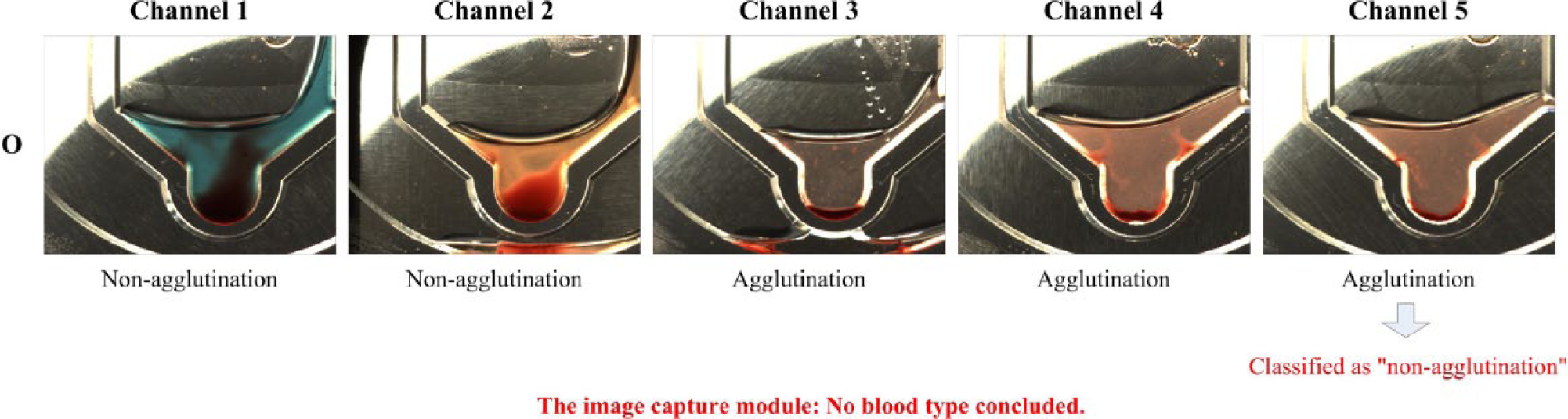
The blood type could not be concluded due to the small quantity of RBC agglutination in microchannel-5.
Results of Irregular-Antibody Tests
Irregular antibodies are all non-ABO antibodies. They appear because of infection, vaccination, or exposure to foreign antigens. In blood transfusions, irregular antibodies may react with the corresponding antigens and, therefore, cause the recipient’s incompatibility. On the disposable disc, microchannels 6, 7, and 8 are arranged to conduct these irregular-antibody tests. Similarly, these tests are based on the antigen–antibody interaction, and thus the result can be determined by observing the agglutination phenomenon. Once the agglutination occurs in any microchannel, further examination must be manually carried out to investigate the category of antibodies. As shown in Figure 4 , no irregular antibodies were discovered for these four patients.
Conclusion
The degree of agglutination in a manual blood-typing test usually depends on visual inspection of the trained personnel. For different medical laboratory scientists, their determinations of agglutination might not be identical, especially in the situation of weak agglutination. In addition, the result is merely qualitative. This proposed lab-on-disc blood-typing system was designed to conduct a total of eight assays for a patient, including forward-typing tests, reverse-typing tests, and irregular-antibody tests. Only small consumption volumes of reagents are required. An image capture module was developed to take pictures of RBC agglutination in every microchannel. The captured images are recorded in the database for every patient and analyzed by the system to give a report of blood type. Currently, the image capture module is successful in accurately determining the blood type. This apparatus, having a smaller footprint and allowing to be interrupted for an emergency test, provides flexible operations for either the scheduled tests or the exigent tests. It is especially suitable for local hospitals and clinics.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Technology (MOST) of Taiwan under grants MOST 103-2622-B-033-003 and MOST 104-2622-B-033-001.
