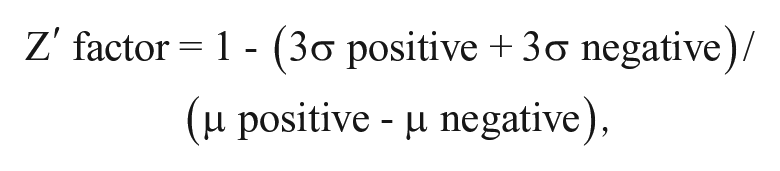Abstract
miRNAs play a key role in the regulation of gene networks in mammalian cells. However, little is known about their roles and functions in the apoptosis pathway. Here, we conducted a whole-genome miRNA screening for apoptosis and identified more than 100 miRNAs as apoptosis inducers. To further explain the roles of these mRNAs in apoptosis, a second round of screening was conducted between p53 +/+ and –/– cells. Among the hits, miR-596 was identified as a regulator of p53. The overexpression of miR-596 significantly increased p53 at the protein level, thereby inducing apoptosis. We also demonstrated that Smurf1 was the direct target of miR-596. Previously, Smurf1 was reported to attenuate the level of p53 through binding and stabilizing MDM2, a p53 inhibitor. Consequently, by targeting Smurf1, miR-596 indirectly increased the p53 level in mammalian cells. Moreover, our study demonstrated that miR-596 had other antitumor characteristics, such as inhibiting migration and proliferation. The data from the GEO dataset revealed that the high expression of miR-596 contributed to survival benefits among cancer patients. These results make miR-596 a potential antitumor factor for future biomedical applications.
Introduction
Apoptosis has been a hot topic for its potential application in antitumor studies.1,2 Identifying novel apoptosis-inducing factors in cancer cells would undoubtedly promote medical progress in cancer therapy.3–5
miRNAs are 19–24 nt small noncoding RNAs that have been reported to play important roles in metabolism, angiogenesis, and other pathways. 6 Notably, as many as 40% of the human genes and 70% of mature proteins are regulated by miRNAs. Certain miRNAs have the potential to resist carcinogenesis. They bear certain antitumor characteristics, such as inhibiting proliferation, inducing apoptosis, and attenuating metastasis. For example, the miR-29 family induces apoptosis through upregulating the p53 level by targeting p85 alpha and CD42. 7 Some miRNAs function as antiapoptosis factors, and their expression levels are elevated in cancer tissues. For example, miR-125b attenuates p53 by functioning on BAK1. 9 A single miRNA targets hundreds of genes, which in turn regulate different pathways at the same time. This gives miRNAs a potential advantage in tumor therapy. 6
Although previous studies have demonstrated some miRNAs as apoptosis-inducing factors, a whole-genome-scale miRNA apoptosis screening has not been conducted. Here, based on a cell microarray developed in our lab,10,11 we conducted apoptosis screening for a library containing 895 miRNAs covering the majority of human miRNAs. We identified 104 miRNAs and validated them as proapoptotic factors. Most miRNAs were reported as apoptosis inducers for the first time. Furthermore, all these candidates were selected for a second round of screening between p53 +/+ and –/– cells to investigate if their proapoptosis effects were p53 dependent. To this end, we identified miR-596 as a potent p53-dependent apoptosis-inducing factor.
In this study, we found that miR-596 was able to increase p53 via targeting Smurf1. miR-596 was a potential biomarker for cancer, and its expression level in patients was significantly lower in tumor tissues and was correlated with their survival life spans.
Materials and Methods
Cell Microarray Preparation
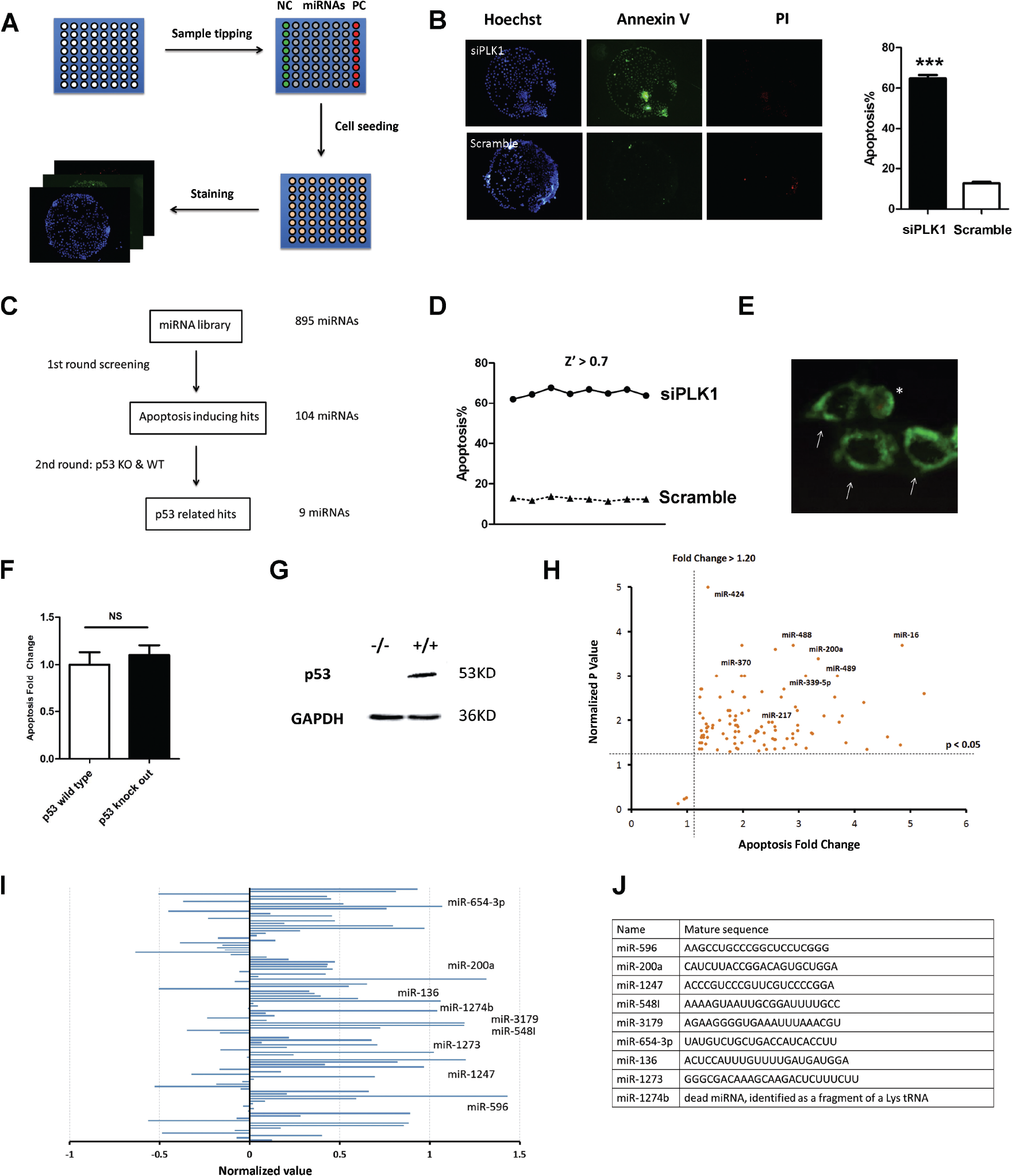
Cell microarray technology has been developed and applied in high-content screening by Zhang et al. 10 Briefly, the slides were covered with poly N-isopropylacrylamide and etched via a shadow mask by oxygen plasma. Each chip had 64 (8 × 8) tiny wells in which cells could grow ( Fig. 1A ). After ultraviolet (UV) sterilization, a reverse transfection reagent containing miRNA mimics was printed on each tiny well using a nanodispenser (Phoenix, Art Robbins Instruments, Sunnyvale, CA). Next, the slides were fixed in a six-well plate with melted wax. Approximately 3 mL of 37 °C culture medium containing 1 × 105 cells was transferred to each well. Cells were able to grow inside each tiny well and form cell islands ( Fig. 1B ). The reverse transfection mixture was gradually absorbed by cells within several hours; the transfection efficiency was tested in previous work. 10 Approximately 24 h later, the dishes were moved to room temperature for 5 min, and the chips were carefully washed with phosphate-buffered saline (PBS) to remove the polymer. Apoptosis staining assay was performed on the cell islands.
Two-round screening of apoptosis. (
Apoptosis Detection Assay
Annexin V-FITC Kit (Biosea Biotechnology, Beijing, China, cat. CX1001) was used to detect apoptosis. After Dulbecco’s modified Eagle medium (DMEM) was replaced with PBS, the staining buffer was carefully added to the cell microarray. Hoechst33342 stained both live and dead cells and was used to calculate the total cell number. Hoechst33342 was selected because its use in staining does not ruin membrane integrity or induce cell death. It was used to determine the total cell numbers. Propidium iodide (PI) stained only necrotic cells and was used to determine the number of necrotic cells. Only annexin v–positive and PI-negative cells were identified as apoptotic ( Fig. 1E ). The staining process must be conducted rapidly and carefully so as not to induce cell death. No paraformaldehyde (PFA) stabilization was allowed during the entire process. Apoptotic cells were recognized and counted automatically using a high-content imaging system (Molecular Device Company Ltd., Shanghai, China, cat. ImageXpress Micro2). The apoptosis ratio was calculated based on total cell number.
Proliferation Detection Assay
HCT116 cells were transfected with miRNA and seeded at the same density in a 48-well plate. At 24, 48, and 72 h, the cells were labeled with Hoechst33342, and the cell number in each well was calculated by a high-content imaging system (Molecular Device). All experiments were conducted in duplicate to avoid potential bias.
Cell Culture and Transfection
Cells were cultured in DMEM containing 10% fetal bovine serum (FBS), and 100 U mL−1 penicillin and 0.1 mg mL−1 streptomycin (PS) under humidified conditions in 95% air and 5% CO2 at 37 °C.
The HCT116 p53 knockout cell line is a widely characterized p53 double-knockout cell that originally derives from the Volgelstien lab. We obtained this cell line from Dr. Huang Xingxu of Nanjing University. Its p53 level was tested via Western blotting ( Fig. 1G ). The background apoptosis ratio in p53 wild-type and knockout cell lines was similar after transfection with scrambled RNAs ( Fig. 1F ).
Plasmids were transfected into cells by lipo2000 (Invitrogen, Waltham, MA). PS was removed 1 day before plasmid or siRNA transfection. DMEM was replaced with fresh media 6 h after transfection.
Western Blot
Forty-eight hours after transfection, the cells were collected and lysed. Proteins were extracted and resolved by sodium dodecyl sulfate (SDS)–polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride membrane (Millipore Corporation, Billerica, MA). Membranes were probed with antibodies against Smurf1 (Cell Signaling Technology [CST] no. 2174, Danvers, MA), MDM2 (CST no. 3521), p53 (CST no. 9282), and GAPDH (CST no. 2118). The images were obtained and quantified by Quantity One software (Bio-Rad, Hercules, CA). The final dilutions of primary and secondary antibodies were 1:1000 and 1:3000, respectively.
Plasmid Construction and Luciferase Experiment
Smurf1 CDS was constructed into a pcDNA3.1 vector for overexpression. The 3′UTR or 5′UTR of candidate genes, respectively located at the 3′ and 5′ ends of the firefly luciferase gene ORF, were cloned into the pGL3 vector. Mutations were introduced into the 3′UTR through bridge PCR. Cells (104) were cotransfected with 200 ng of the indicated pGL3 firefly luciferase construct and 20 ng of a pGL3 Renilla luciferase normalization control. The indicated miRNA expression plasmid or mimics were transfected. After 48 h, the cells were lysed, and luciferase activities were measured using the Dual Luciferase Reporter Assay System (Promega, Madison, WI).
siRNA and miRNA Silencing
A whole-genome miRNA library and all siRNA duplexes were ordered from GenePharma (Shanghai, China). The siRNAs’ efficacy was tested by quantitative real-time (qRT)-PCR. The sequence of Smurf1 siRNA was as follows:
Sense: CCUGCCCAGAGAUACGAAAtt
Antisense: UUUCGUAUCUCUGGGCAGGaa
The sequence of PLK1 was as follows, and its efficacy was tested by qRT-PCR.
Sense: CGAGCUGCUUAAUGACGAGtt
Antisense: CUCGUCAUUAAGCAGCUCGtt
Scrambled RNAs were synthesized with random sequences and acted as a negative control.
Statistical Analysis
The Z′ of screening was calculated using the following equation: 14
where σ positive is the standard deviation of the positive control, σ negative is the standard deviation of the negative control, µ positive is the mean of the positive control, and µ negative is the mean of the negative control.
Parallel screening result analysis was conducted using the following equation:
where FC is fold change, which is normalized by the negative control.
When comparing two groups, the two-tailed Student’s unpaired t test was used. For all the tests, a p value of <0.05 was considered significant. In the bar chart, * indicates p < 0.05, ** indicates p < 0.01, and *** indicates p < 0.001. Error bars represent the standard deviations of at least three independent experiments.
Imaging Capture and Analysis
A high-content screening machine was purchased from Molecular Device (ImageXpress Micro2). With the kind help of Mr. Zhou Xuan, we revised the original ImageXpress Micro System and allowed it to recognize and photograph each cell island on the chips automatically. Pictures were transferred to a Micro XLS System data pool for apoptosis analysis. Only the annexin V–positive and PI-negative cells were identified as apoptotic ( Fig. 1E ).
Patient Data Analysis
The miRNA/mRNA expression data and patient overall survival (OS) data were downloaded from GEO datasets. The GEO numbers were GSE49355, GSE49246, GSE73582, and GSE17536. The p value of the expression data was based on Student’s t test. The p value of the OS data was based on the Mantel–Cox log-rank test.
Results
High-Content Screening Was Conducted for miRNA Library on Cell Microarrays
Cell microarray has been developed and applied for high-content screening by Zhang et al. 10 Briefly, we first printed a reverse transfection mixture containing miRNA mimics or scrambled siRNAs onto each chip, and positive and negative controls were located at the two ends of chips for quality control screening.
In this study, HeLa cells were seeded at the proper density onto the chips, and apoptosis detection was conducted 24 h later ( Fig. 1A ). PLK1 siRNA and scramble RNA were used as positive and negative controls, respectively ( Fig. 1B ). Only annexin V–positive and PI-negative cells were identified as apoptotic. For a typical apoptotic cell, annexin V was stained with a green circle around the nucleus ( Fig. 1E ). Since the background apoptosis ratio was rather low (approximately 9%, Fig. 1B ), this assay could only be used to screen the apoptosis-inducing factors.
Two-Round Screening Identified miR-596 as a p53-Related Apoptosis Inducer
We designed the study workflow depicted in Figure 1C . The whole miRNA library was screened with a HeLa cell line for the first-round screening. A second-round screen of the hits was conducted in HCT116 p53 wild-type and knockout cell lines. The Z′ of the screening was more than 0.7, indicating that the screening data were of high quality ( Fig. 1D ). 12 We validated 107 hits from the first-round screening, and the false-positive rate was lower than 3% (three false-positive hits) ( Fig. 1H , Suppl. Table S1 ). Twenty-six miRNAs (25% of the hits) had been previously reported as apoptosis inducers. This was the first time that the roles for the remaining miRNAs in apoptosis were identified ( Suppl. Table S1 ).
To identify p53-related apoptosis-inducing miRNA, a second-round screening was conducted between p53 wild-type and knockout cell lines ( Fig. 1I , Suppl. Table S2 ). If a miRNA induced significantly higher apoptosis in p53 wild-type cells than p53 knockout cells, this indicated that the miRNA-induced apoptosis was in a p53-dependent manner. We tested the apoptosis ratio in both cell lines for each miRNA and found that nine miRNAs induced significantly higher apoptosis in p53 wild-type cell lines ( Suppl. Table S3 ). However, their mature sequences did not share any similarities ( Fig. 1J ). miR-200a was reported to induce apoptosis in a p53-dependent manner. 8 Based on the second-round screening, miR-596-mediated apoptosis was highly dependent on p53. Moreover, its correlation with p53 was even stronger than that of miR-200a. Since little was known about miR-596, we conducted further experiments to unveil its role in apoptosis.
miR-596 Induced Apoptosis but Not Cytotoxic Effect
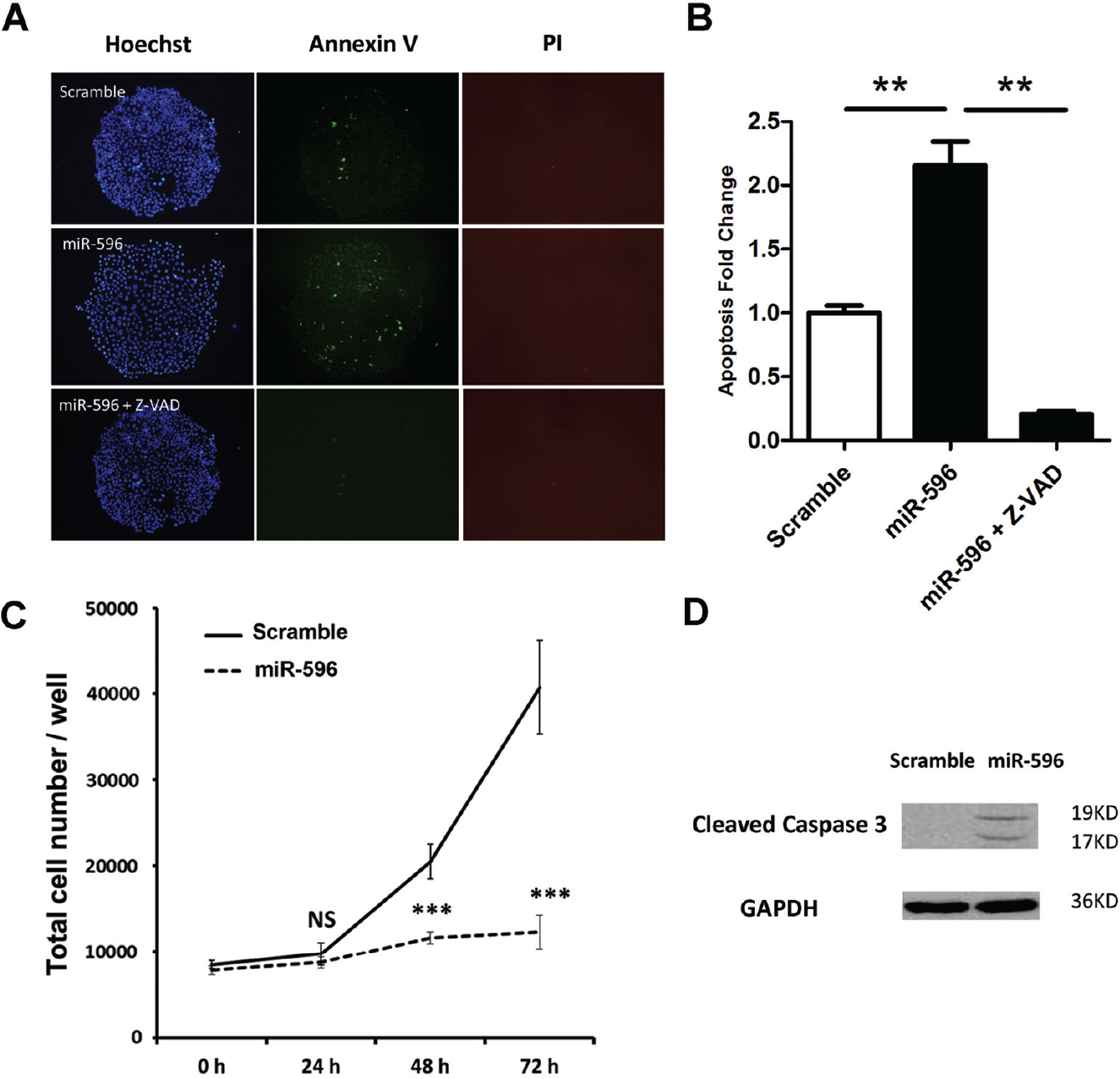
To eliminate the possibility that miR-596 induced a cytotoxic effect, we used the caspase inhibitor Z-VAD. Compared with scrambled RNA, miR-596 induced 2.13-fold apoptosis at the 24 h time point ( Fig. 2A ). This was abolished when Z-VAD was added, indicating that miR-596-induced apoptosis was dependent on caspase ( Fig. 2B ). Further, Western blot analysis demonstrated that the overexpression of miR-596 significantly induced cleavage in caspase 3, which was an apoptosis biomarker ( Fig. 2D ). To eliminate the possibility that miR-596 elevated the apoptosis ratio by inhibiting proliferation, we conducted a proliferation inhibition detection assay at 24, 48, and 72 h. As is shown, when apoptosis was tested at 24 h, there was no significant inhibition of proliferation ( Fig. 2C ).
miR-596 was validated as an apoptosis-inducing miRNA. (
miR-596 Downregulates Target Gene Smurf1
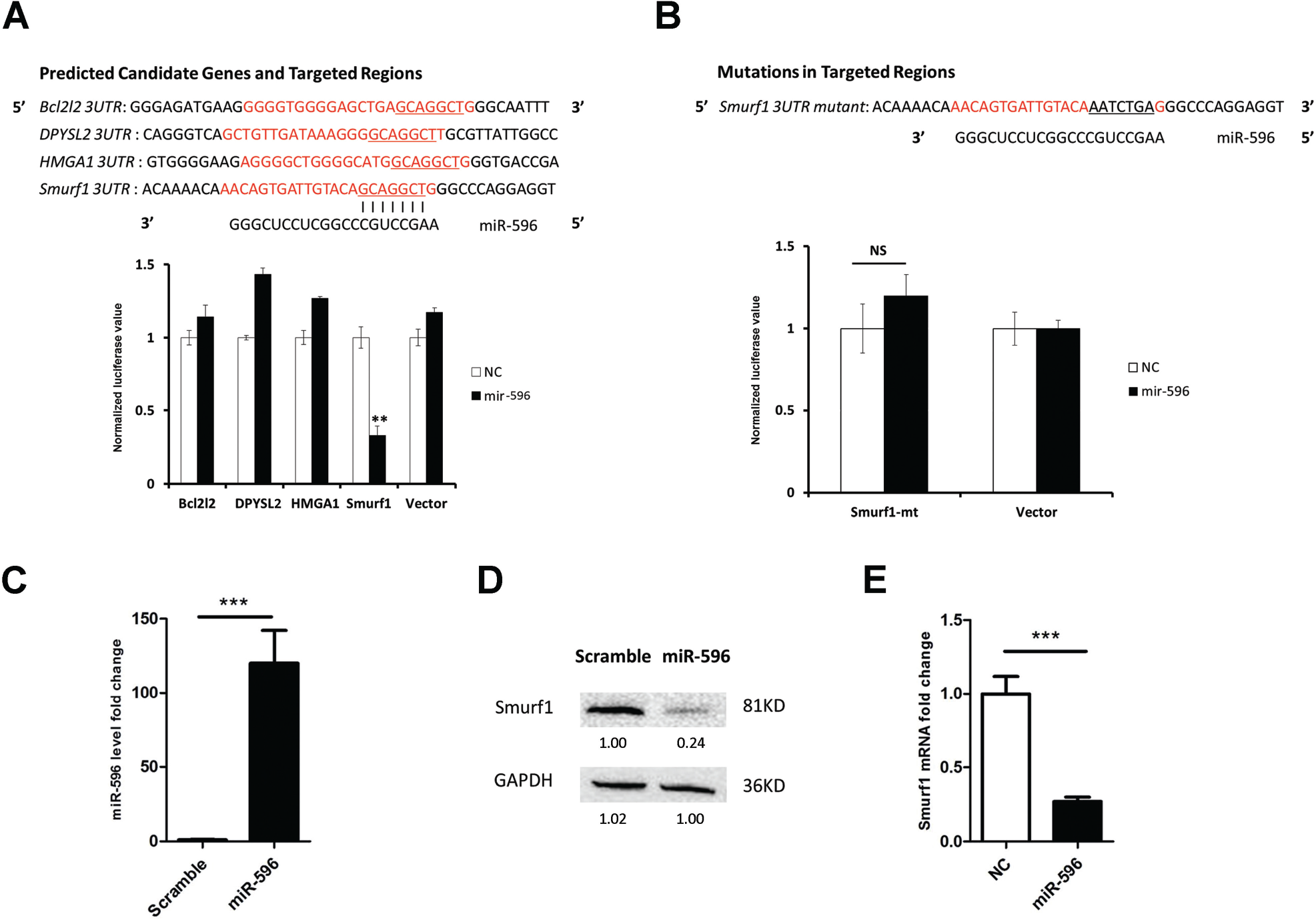
To investigate the mechanism involved in the apoptosis-inducing effect of miR-596, we analyzed target genes of miR-596 using an online software program (TargetScan) and obtained a list of candidates. We further narrowed down targets by searching whether they had potential links with p53. Four candidates were selected: Bcl2l2 is a ligand of Bcl2 that blocks apoptosis through direct interaction with p53. 13 DPYSL4 is a p53 tumor suppressor gene involved in the cell cycle and apoptosis. Its family member DPYSL2 may have a similar function; thus, we included it on our list. 14 HMGA1 is an architectural chromatin protein; recent research reveals that HMGA1 regulates p53 transcription. HMGA1 silencing enhances the p53 level in colon cancer cells. 15 Smurf1 is an HECT-type E3 ligase and regulates MDM2 by binding. MDM2 is an E3 ligase for p53 and directly promotes p53 degradation during the ubiquitinylation process. The ablation of Smurf1 enhances the p53 level and may lead to apoptosis via interaction with MDM2. 16
To determine the target for miR-596, candidate genes’ 3′UTR binding regions were cloned into the pGL3 luciferase vector, and the luciferase experiment was conducted ( Fig. 3A ). It was demonstrated that luciferase activity was strongly inhibited for Smurf1 3′UTR. None of the other three candidates showed any inhibitory effects. The mutation of the Smurf1 3′UTR binding site abolished the inhibitory effect ( Fig. 3B ), demonstrating that miR-596 binds Smurf1 3′UTR in vitro.
Smurf1 was identified as a direct target of miR-596. (
We then determined the effect of miR-596 overexpression on the endogenous levels of Smurf1 mRNA and protein in HCT116 cells using qPCR and Western blot, respectively ( Fig. 3C ). Specifically, miR-596 attenuated the Smurf1 protein and mRNA level by 74% and 70%, respectively ( Fig. 3D , E ). The interaction of miR-596 with the Smurf1 3′UTR was in a classic 7-mer way. The 7 nt seed binding region was located at 3084–3090 nt of Smurf1 mRNA. The silencing of Smurf1 using siRNA could significantly induce apoptosis ( Suppl. Fig. S1 ). Thus, these results demonstrated that miR-596 negatively regulates the downstream target gene Smurf1.
miR-596 Enhanced p53 Protein Level via Targeting Smurf1
Apoptosis is closely related to p53 status and is determined by its expression level and integrity. 17 To investigate the correlation between miR-596 and p53, we conducted the following experiments. Forty-eight hours after miR-596 transfection, the cells were collected and Western blotting was performed. It was demonstrated that miR-596 overexpression significantly elevated the p53 protein level by 2.7-fold in HCT116 p53 wild-type cells ( Fig. 4A ).
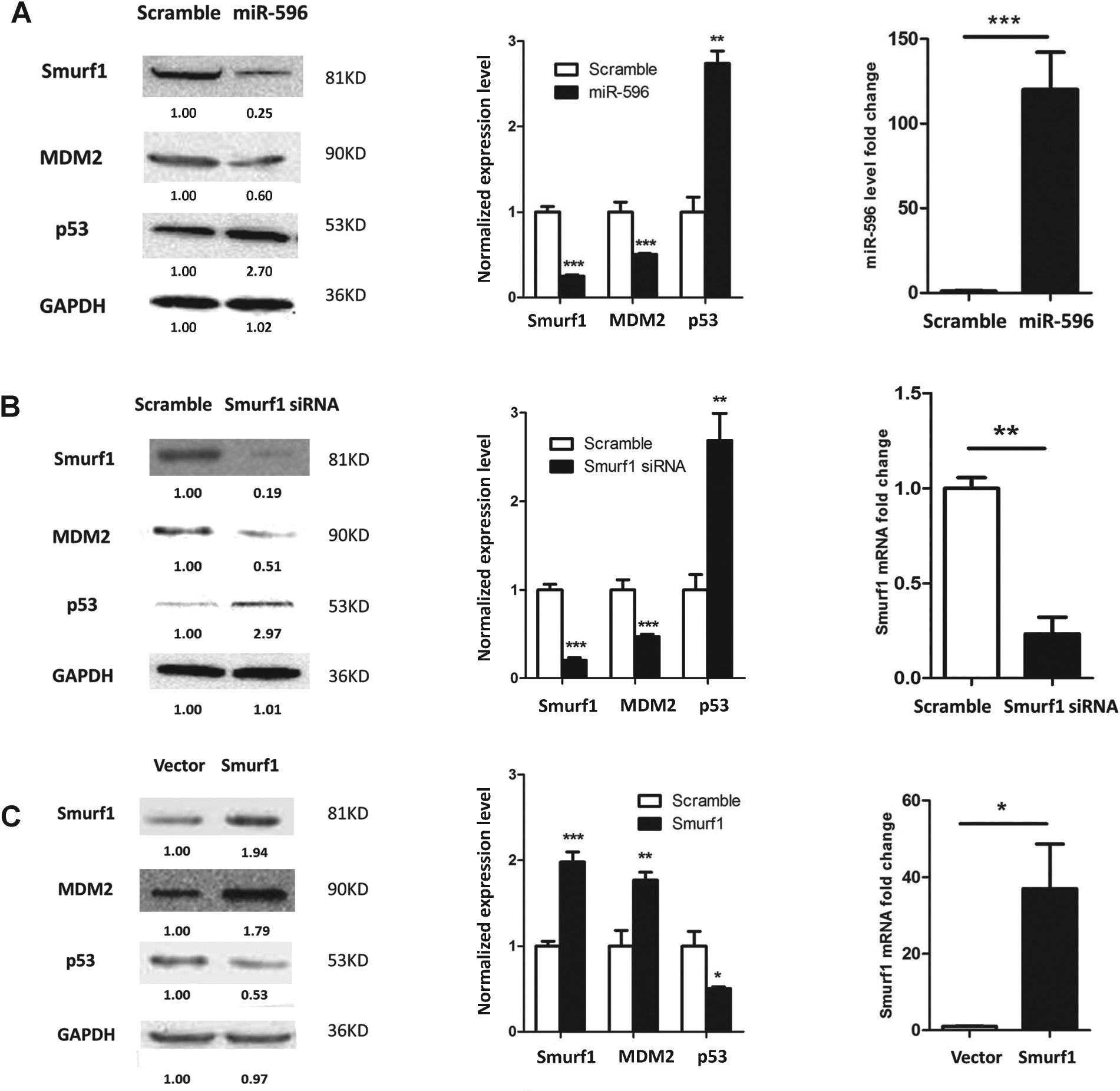
miR-596 enhances p53 via targeting Smurf1. (
Previously, it was reported that Smurf1 binds and stabilizes MDM2, which catalyzes p53 degradation. Smurf1 significantly extends MDM2 half-time and thus enhances the MDM2 protein level. Thus, Smurf1 promotes p53 degradation. 16 We assumed that miR-596 probably elevated p53 via the Smurf1/MDM2 axis. To demonstrate this, Western blotting was performed for Smurf1, MDM2, and p53. miR-596 attenuated both the Smurf1 and MDM2 levels to 26% and 62%, respectively, while the p53 level was increased by 2.72-fold ( Fig. 4A ). Silencing of Smurf1 led to a similar effect ( Fig. 4B ), and overexpression of Smurf1 reversed this effect ( Fig. 4C ).
Some miRNAs bind the 5′UTR of mRNAs and increase their expression level. 21 To exclude the possibility that miR-596 directly increases p53 levels by 5′UTR binding, we detected the p53 mRNA level by qRT-PCR. It was demonstrated that the overexpression of miR-596 did not affect the p53 mRNA level. There was no binding affinity between the miR-596 and the p53 5′UTR region ( Suppl. Fig. S2 ), indicating that miR-596 does not interact and increases p53 directly. Thus, it was demonstrated that miR-596 increased p53 via targeting Smurf1 ( Fig. 5 ).
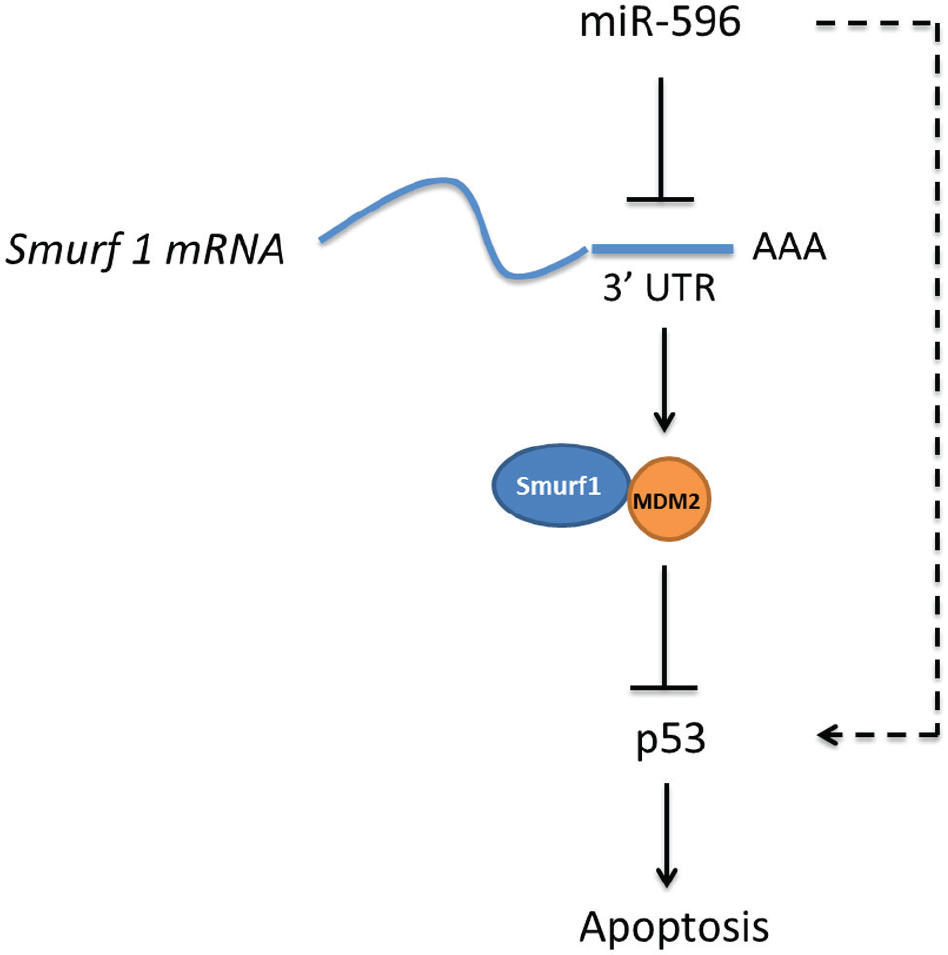
miR-596 induces apoptosis by enhancing p53 via the Smurf1/MDM2 axis. miR-596 binds to the 3′UTR of Smurf1 mRNA and inhibits its translation. Downregulated Smurf1 leads to lower levels of MDM2. As a result of the slower speed of p53 degradation, the p53 protein level is increased. Thus, miR-596 indirectly enhances p53 and induces apoptosis.
miR-596 Is a Clinical Indicator of Survival in Cancer Patients
To evaluate the clinical significance of miR-596, we assessed the correlations among miR-596 expression and clinical pathological factors based on data from GEO datasets. The expression level of miR-596 was significantly lower in the colorectal cancer tissue than in the adjacent nontumor tissue ( Fig. 6A , N = 40, p = 0.019). Moreover, the expression level of Smurf1 was significantly higher in colorectal cancer tissue than in the adjacent nontumor tissue ( Fig. 6B , N = 17, p = 0.049). Additionally, the OS time of patients with a low miR-596 expression level was significantly shorter than that of patients with a high expression level ( Fig. 6C , N = 69, p = 0.036). The OS time of patients with a high Smurf1 expression level was slightly shorter than that of patients with a low expression level ( Fig. 6D , N = 177, p = 0.039). These results are in accordance with miR-596’s targeting of Smurf1 as described above. Therefore, miR-596 has a survival benefit and could be a clinical indicator of survival in cancer patients.
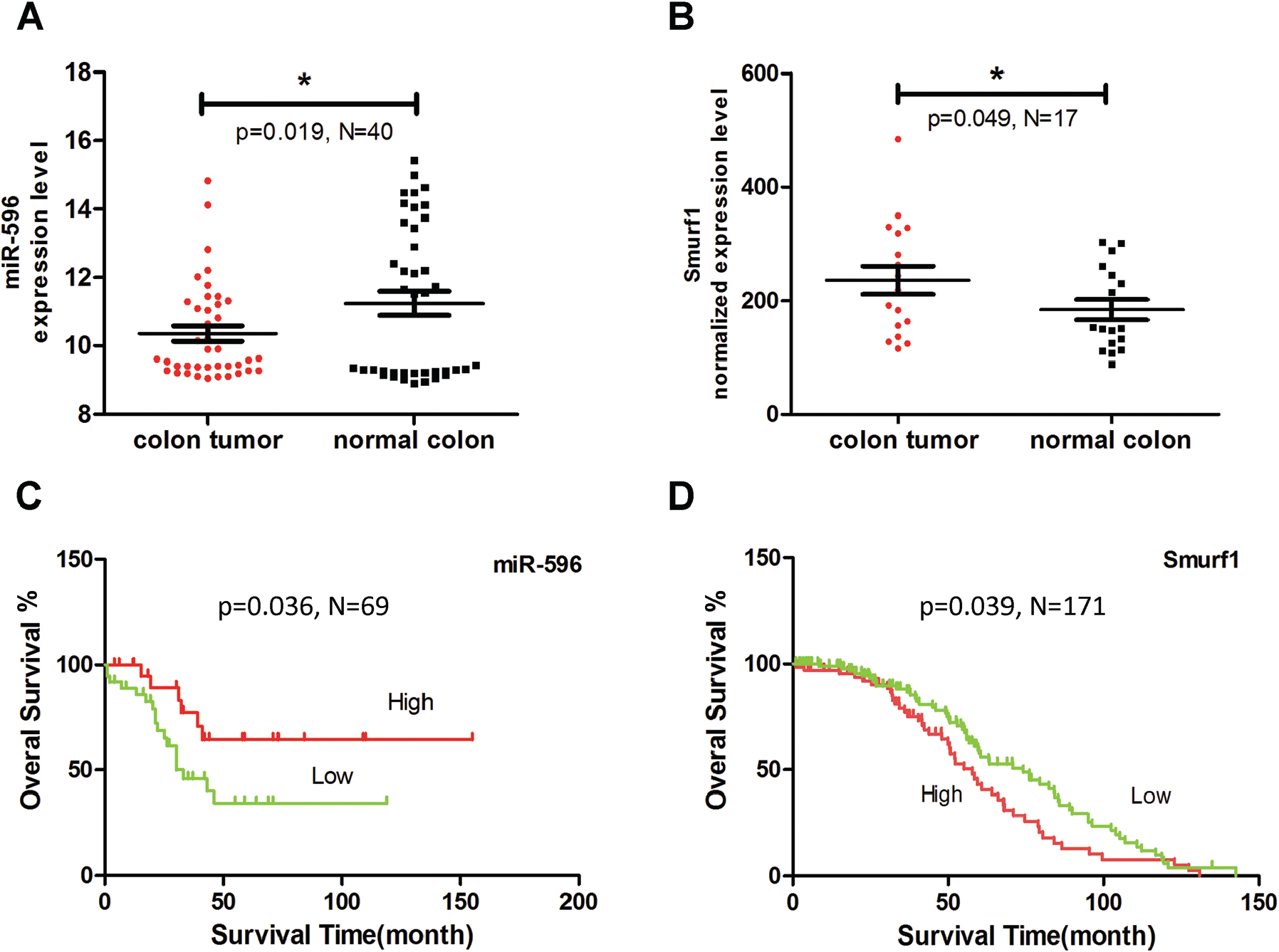
miR-596 is a survival indicator in cancer patients. (
Discussion
More than 1100 miRNAs have been annotated in the human genome. It is well established that miRNAs participate in carcinogenesis. However, it is essential to identify miRNAs with a potential therapeutic value. In this study, we systematically investigated the effects of individual miRNAs on apoptosis, and our screening covered 82% of the miRNA library. Our results showed that 11.6% of the miRNAs acted as proapoptotic factors. Among them, 25% have been reported as apoptosis inducers in previous studies, for example, miR-16, miR-145, and miR-197.22–24 For the remaining 75% miRNAs, our study annotated their clear roles in apoptosis for the first time. Consequently, this result will be helpful for future studies ( Suppl. Table S1 ).
We identified nine miRNAs that induced p53-related apoptosis through the second-round screening. Interestingly, miR-200a was among the hits. miR-200a has been reported to regulate p53 activity through Ypel2. 8 By ablation of Ypel2, which is a p53 inhibitor, miR-200a indirectly enhances p53 and induces apoptosis. We demonstrated that miR-596 functions in a manner similar to that of the miR-200 family. They both increase the p53 level through targeting p53 inhibitors and thus induce apoptosis. However, miR-200a might induce apoptosis in a more complex manner. We demonstrated that miR-200a was still able to induce apoptosis in p53 knockout cell lines, although this induction was much weaker than in p53 wild-type cell lines. This indicated that miR-200a might also play a role in p53-independent apoptosis.
Previously, miR-596 was reported to target LGALS3BP in oral cancer. 20 LGALS3BP is responsible for the phosphorylation of ERK1/2 and can induce cell proliferation. By targeting LGALS3BP, miR-596 strongly inhibits cell proliferation and thus inhibits cancer progression. Here, we identified Smurf1 as another target of miR-596. Smurf1 directly binds MDM2, which facilitates p53 degradation. By binding with MDM2, Smurf1 increased MDM2 half-time and thus increased the amount of MDM2. Consequently, this led to p53 degradation. By targeting Smurf1, miR-596 was able to slow p53 degradation and thus increase the p53 amount.
We also demonstrated that the cross talk of miR-596 with p53 occurred in an indirect manner rather than a direct manner. Overexpression of miR-596 did not affect p53 mRNA level, and there was no binding affinity between miR-596 and p53 5′UTR. These results excluded the possibility that miR-596 binds to p53 mRNA 5′UTR and thus increases the p53 level directly.
The expression level of miR-596 was lower in tumor tissues and in most cancer cell lines (data not shown). One explanation is that miR-596 is located at fragile genome loci, which are easily lost in cancer cells.18,19 Another explanation is that its upstream methylation status affects the miR-596 expression level in cancer cells. 20
Our functional and mechanistic analyses revealed that miR-596 bears wide antitumor characteristics, such as inducing apoptosis and inhibiting cell proliferation and migration ( Suppl. Fig. S3 ). High expression of miR-596 confers survival benefits in cancer patients. Thus, it may act as an antitumor factor in future clinical applications.
In summary, this study has important significance in furthering our understanding of the role of miRNAs in apoptosis. We believe that these findings will provide new insights into the physiological and therapeutic importance of miRNAs in cancer.
Footnotes
Acknowledgements
The authors wish to thank Hanshuo Zhang and Juan Li for providing guidance and making suggestions during experiments. The authors also wish to thank Professor Huang for providing HCT116 p53 wt/ko cells.
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.