Abstract
In the present study, we aimed to evaluate the possible effects of methylphenidate on rat testes. Forty-two Wistar rats were randomly distributed into three experimental groups of 14 rats each. For 90 days, each group via gavage received the following: group 1 = tap water (control group), group 2 = 5 mg/kg/day of ritalin (methylphenidate, MPH), and group 3 = 10 mg/kg/day of ritalin. After sacrificing the animals, the body weights as well as the absolute and relative testicular weights were measured. Testes were sampled, fixed, and processed and, by histopathological examination, quantitative morphometric analysis of Sertoli cells, spermatocytes, and spermatids was performed in stages II, V, and XII. Immunohistochemistry was performed for transforming growth factor (TGF)-β1 and p53, and the apoptotic index was assessed through the TUNEL method. Group 2 had a reduction of round spermatids in stage II. Group 3 had reduction in both stage II and stage V spermatids, as well as lower testicular weight. The p53 expression was increased in group 3. In groups 2 and 3, the TGF-β1 expression was reduced and the apoptotic index by TUNEL was increased. Body weights remained stable on either group. Our results showed that methylphenidate might negatively affect spermatogenesis not only by reducing testicular weight and amount of round spermatids but also by increasing apoptotic death and p53 activation. The findings of the study, however, must be cautiously interpreted.
Introduction
Methylphenidate (MPH) is a widely prescribed and effective treatment of attention-deficit/hyperactivity disorder (ADHD), which is one of the most common psychiatric diseases in childhood.1,2 Despite its demonstrated efficacy, tolerability, and safety in most children with ADHD on the short term, the long-term effects of MPH on the brain and other body systems remain a major concern. Possible adverse effects of MPH on growth and development is generally agreed to be modest and transient and tends to disappear in the long term, while the ultimate adult growth parameters do not seem to be affected.3,4 However, the effects of continuous treatment from childhood to adulthood is largely unknown. 5 Regarding the effects of MPH on the male reproductive system, scientific data is limited and all from animal studies. It was shown to have trophic effects on spermatogenesis in one study, 6 and the presence of relevant receptors on spermatogenic cells has also been reported.7,8 In the present study, we aimed to evaluate MPH effects on body weight and testis in rats by applying histopathological analysis, immunohistochemistry for the expression of transforming growth factor (TGF)-β1 and p53, as well as an assessment of the apoptotic index to further evaluate the cellular events in rat spermatogenesis under MPH effect.
Materials and methods
All experimental procedures were carried out in accordance with the European Communities Council Directive of 24 November 1986 (86/609/EEC) and approved by the Animal Care Committee of Gazi University.
Animals
A total of 42 prepubertal, male Wistar rats (age range: 21–24 days; weight range: 48.2–55.9 g) from the Gazi University Medical Faculty animal laboratory were used in this experimental study. All animals were group-housed in plastic cages at constant room temperature (21−22°C) and in a 12-hour light/12-hour dark cycle (lights on at 07:00 a.m.). They were given free access to food and water.
Experimental procedure and dosing
Rats were randomly distributed into three experimental groups each consisting of 14 rats. The procedures were allocated as follows: Group 1 (control group) was subjected to vehicle (tap water) via gavage; group 2 (low dose MPH group: MPH-LD) received 5 mg/kg/day of ritalin (Ritalin® 10 mg/tablet, Novartis, Istanbul, Turkey); group 3 (high dose MPH group: MPH-HD) received 10 mg/kg/day of ritalin. Ordinary water was used as the control solution. Drug dosages in treatment groups 2 and 3 were defined according to the rats' weights; ritalin was prepared in 10 mL of the vehicle and the doses were adjusted accordingly.
There were no specific restrictions in the diet contents and amount administered to the animals in our study. Standard rat feeding diet as routinely applied in our centre of Animal Experiments Laboratory in Gazi University was used. The feeding of the animals was performed under control by the veterinary doctors in the laboratory. The control and drug-administered groups consumed the same feeding material. The rats in all groups were given free access to food and water.
All drugs were dissolved in saline and orally administered 7 days/week for 3 months with 12-hour intervals in two divided doses via gavage, the metal tip of which was bent and coated with silicone. During the study period, two rats in group 1, three rats in group 2, and two rats in group 3 were excluded or lost due to various reasons (mainly due to disease and death).
Rats were weighed and then euthanized by taking 6−8 mL intracardiac blood under anesthesia. The right and left testes were removed, weighed, and fixed in buffered 10% formalin for histological examination. The absolute testicular weight was the weight of the organ on a sensitive scale. The relative weight was the ratio of the absolute testicular weight to the body weight of the rat. Tissue sections 4-μm thick were stained with hematoxylin-eosin. Morphological descriptions of rat spermatogenetic cells and stages were used as a guide, while the quantitative analysis of spermatogenetic cells was applied as described in or similarly to previous studies.9–14 Each slide was analyzed for three selected stages of the spermatogenic cycle (stages II, V, and XII). For each stage, three separate seminiferous tubules were randomly selected and in these, total cell counts were assessed for spermatogonia, pachytene spermatocytes, zygotene spermatocytes, round spermatids, and Sertoli cells. Relevant cells specific to each stage were included. 9 The ratio of the spermatogenetic cells to the Sertoli cells was calculated.
Indirect immunohistochemical staining (using p53 and TGF-β1 primary antibodies) and apoptosis evaluation were performed on 5-μm-thick paraffin tissue sections.
Determination of apoptosis
The terminal deoxynucleotidyl-transferase-mediated dUTP-biotin nick end labeling (TUNEL) staining method was applied to determine the number of apoptotic cells. 15 The testes sections were stained with the in-situ cell-death detection kit (ApopTag Plus Peroxidase In Situ S7101, Lot: 0603025277). Five-μm-thick sections were prepared with an automated microtome device from the paraffin blocks. The slides were incubated at 60°C overnight. Sections were deparaffinized in xylene for an hour and rehydrated with reduced alcohol series (100%, 95%, 85%, 70%, and 60%). They were incubated with 20 μg/mL proteinase K for 10 min. Washing with phosphate-buffered saline (PBS) with pH 7.4 was performed at each stage. Endogenous peroxidase activity was blocked with 3% H2O2. After PBS wash, the sections were incubated with equilibration buffer for 10–15 min and terminal deoxynucleotidyl-transferase (TdT) enzyme (77 μL reaction buffer + 33 μL TdT enzyme mix, 1 μL TdT enzyme) for 60 min at 37°C. Working strength stop/wash buffer (1:10) was applied for 10 min at room temperature and slices were incubated with anti-digoxigenin conjugate for 30 min. After three PBS washes for 5 min, the sections were stained with 3,3’-diaminobenzidine (DAB) components to detect TUNEL positive cells and then counterstained with Methyl green. Two independent observers blind to the study groups evaluated the TUNEL-positive cells.
Immunohistochemical evaluation
Tissue samples from each group were fixed in neutral formalin for 72 hours and processed for paraffin embedding. Four to five μm thick sections were processed for polylysin microscope slides. For immunohistochemical examination, the slides were stored on a microwave oven in a 0.01 M citrate buffer (Lot: 90030610D, Cat: AP-9003-500, LabVision, Fremont, California, USA). Endogenous peroxidase activity was blocked in 3% hydrogen peroxide (Lot: AHP40114, Cat: TA-125-HP, LabVision). Epitopes were stabilized by applying a serum-blocking solution (Lot: 41080981, Cat: 85-9043, Zymed, South San Francisco, USA) and then two groups of slides were prepared from each block. First group was incubated with p53 (FL-93; Rabbit Polyclonal IgG, Lot: A1806, Cat: SC-6243, Santa Cruz Biotechnology, California, USA), second group was incubated with TGF-β1 (v) (Rabbit Polyclonal IgG, Lot: A1306, Cat: SC-146, Santa Cruz Biotechnology) and diluted in UltraAb Diluent (Lot: 50481612, Cat: 00-3118, Zymed) for 60 min at room temperature. After the incubation, the secondary antibody (Broad spectrum Histostain Plus, Lot: 41080981, Cot: 85-9043, Zymed) was applied. Afterwards, 3-amino-9-ethylcarbazole (AEC; Lot: 50481594, Cat: 00-2007, Zymed) for TGF-β1 antibody and DAB-tetrahydrochloride (DAB-4HCL; Lot: 40980870, Cat: 00-2020 Zymed) for p53 antibody were used as chromogen. Then, the slides were counterstained with Mayer’s hematoxylin and analyzed with Olympus BH2 photo-light microscope. Serial sections were examined and immunolabeling patterns were compared. Two observers blind to the samples evaluated the immunolabeling scores. The labeling intensity was graded semi-quantitatively and the HSCORE was calculated using the following equation: HSCORE = ∑ Pi (I + 1), where i = labeling intensity with a value of 1, 2, or 3 (weak, moderate, or strong, respectively) and Pi = percentage of labeled spermatogenic cells for each intensity, varying from 0% to 100%. 16
Statistical analysis
Data were analyzed using the application Statistical Package for the Social Sciences (SPSS). All data were expressed as mean ± standard deviation. The comparisons between groups were tested using one-way analysis of variance (ANOVA) or Kruskal Wallis variance analysis, followed by Tukey’s multiple comparison test or Mann Whitney U test. Statistical significance was set at p < 0.05.
Results
Testicular and body weights
In the MPH-LD group, both testicular and body weights did not show significant difference compared to control group. The absolute and relative testicular weights of the MPH-HD group were significantly lower than those of the control group (p < 0.001 and p < 0.01, respectively). The body weights and the absolute and relative testicular weights of the rats in the three groups are presented in Table 1 .
Body weight and absolute and relative testicular weight on day 90 of the experiment in rats in the control, MPH-LD, and MPH-HD groups
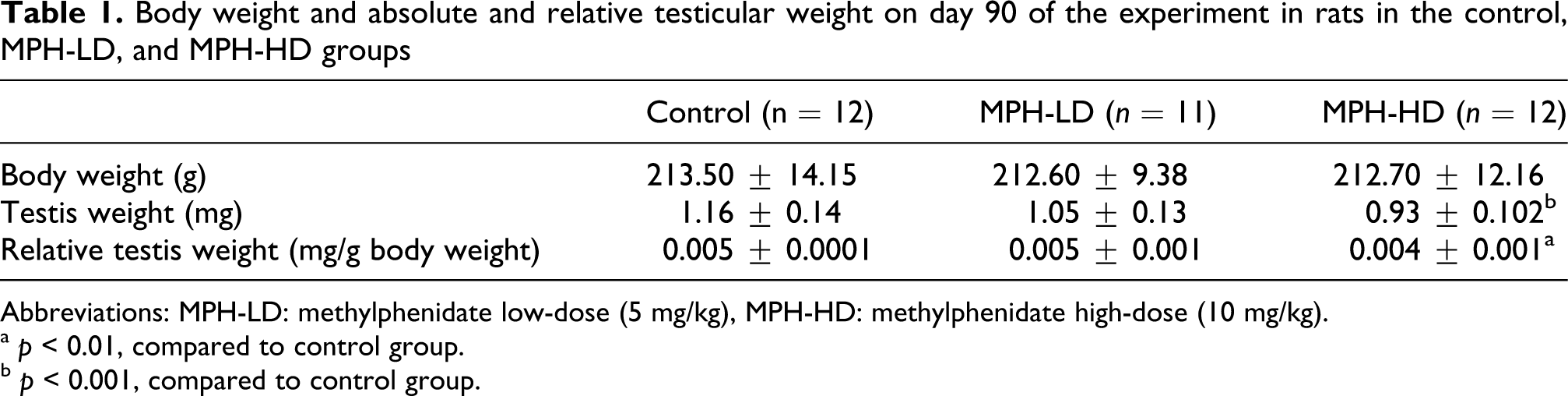
Abbreviations: MPH-LD: methylphenidate low-dose (5 mg/kg), MPH-HD: methylphenidate high-dose (10 mg/kg).
a p < 0.01, compared to control group.
b p < 0.001, compared to control group.
Histopathological evaluation
There was no necrosis, obvious depletion, disorganization or exfoliation of germ cells, tubular atrophy, inflammatory reaction, or apparent pathological alteration in Leydig cells in any of the slides examined. Sertoli cells were analyzed and counted in all of the evaluated tubules. No vacuolation, swelling, or cytoplasmic apoptotic bodies or phagocytized spermatogenic cells were noted in the Sertoli cells. Figure 1 demonstrates the seminiferous tubules and spermatogenetic stages evaluated in the control animals.

Low-power view of seminiferous tubules and detailed views of spermatogenetic stages II, V, and XII are shown in A, B, C, and D, respectively. Samples belong to the control group. A Sertoli cell (large arrow head), spermatogonia (small arrow heads), pachytene spermatocytes (thin arrows), round spermatids (thick arrows), and a zygotene spermatocyte (hollow arrow) are indicated. Hematoxylin and eosin; original magnification ×100 in A, ×400 in B, C, and D.
The ratio of round spermatids to Sertoli cells in stage II of the spermatogenic cycle was significantly lower in the MPH-LD and MPH-HD groups compared to the control group (p < 0.01 and p < 0.001, respectively; Table 2 ). The ratios of spermatogonia, pachytene spermatocytes, or round spermatids to Sertoli cells in stage V of the MPH-LD group were not different from those of controls. The ratio of round spermatids to Sertoli cells was significantly lower in the MPH-HD group compared to controls (p < 0.001; Table 3 ). On the other hand, the ratio of spermatogonia, pachytene spermatocytes, or zygotene spermatocytes to Sertoli cells in stage XII did not show significant difference among the groups (Table 4 ).
Ratios of spermatogonia / Sertoli cells, pachytene spermatocytes / Sertoli cells, and round spermatocytes /Sertoli cells in stage II
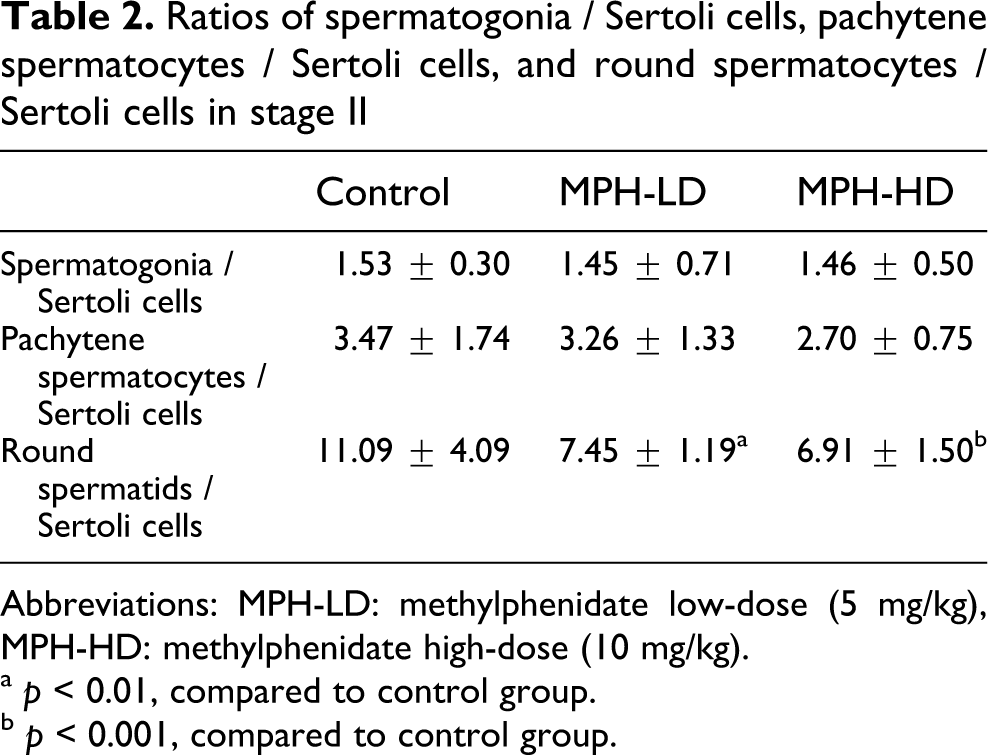
Abbreviations: MPH-LD: methylphenidate low-dose (5 mg/kg), MPH-HD: methylphenidate high-dose (10 mg/kg).
a p < 0.01, compared to control group.
b p < 0.001, compared to control group.
Ratios of spermatogonia / Sertoli cells, pachytene spermatocytes / Sertoli cells, and round spermatocytes / Sertoli cells in stage V

Abbreviations: MPH-LD: methylphenidate low dose (5 mg/kg), MPH-HD: methylphenidate high dose (10 mg/kg).
a p < 0.001, compared to control group.
Ratios of spermatogonia / Sertoli cells, zygotene spermatocytes / Sertoli cells, and pachytene spermatocytes / Sertoli cells in stage XII

Abbreviations: MPH-LD: methylphenidate low-dose
Immunohistochemical expression of p53 and TGF-β1
The p53 immunoreactivity of primary spermatocytes in the seminiferous tubules is shown in Figure 2 . The number of cells immunoreactive for p53 was similar in the MPH-LD and control groups, and significantly increased in the MPH-HD group compared to controls (Table 5 ). With TGF-β1, significantly lower number of cells was found in the MPH-LD and MPH-HD groups compared to controls (p < 0.001 and p < 0.0001, respectively; Table 5). Figure 3 shows the TGF-β1 immunoreactivity of primary spermatocytes in the seminiferous tubules.

p53 immunoreactivity of primary spermatocytes in seminiferous tubules. It is weak in control group (A), moderate in methylphenidate low dose (MPH-LD) group (B), and strong methylphenidate high dose (MPH-HD) group (C). (°): ×400.
Apoptotic cell counts and p53 and TGF-β1 expression in controls, MPH-LD, and MPH-HD groups

Abbreviations: MPH-LD: methylphenidate low dose (5 mg/kg), MPH-HD: methylphenidate high dose (10 mg/kg).
a p < 0.001, compared to control group.
b p < 0.0001, compared to control group.
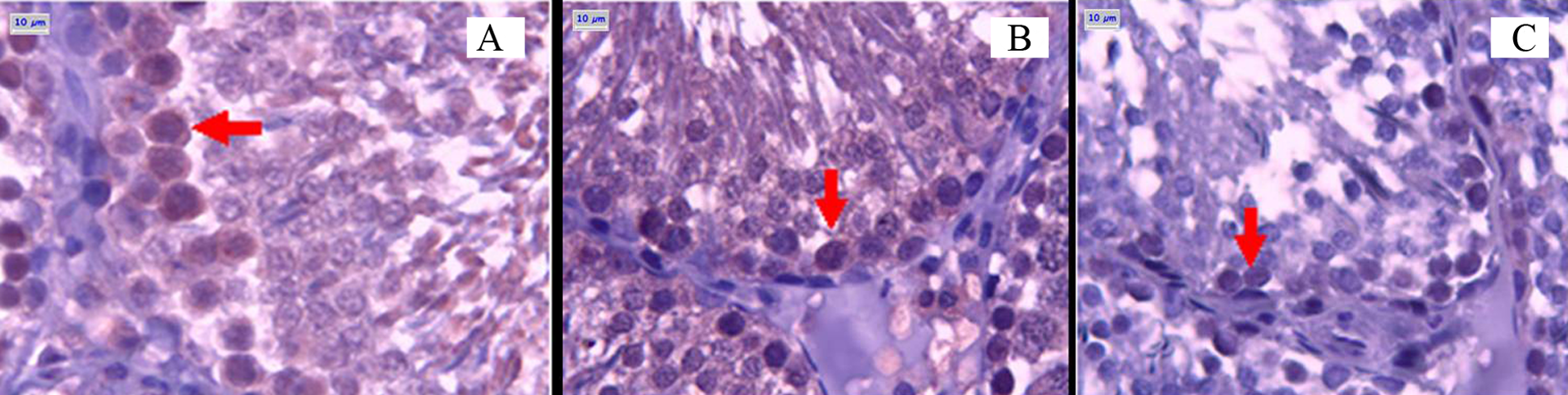
TGF-β1 immunoreactivity of primary spermatocytes in seminiferous tubules. It is strong in control group (A), weak in MPH-LD (B), and weaker in MPH-HD group (C). (°): ×400.
TUNEL evaluation of apoptotic indices
The apoptotic cell counts of the MPH-LD and MPH-HD groups were higher than those of the control group (p < 0.0001; Table 5 and Figure 4 ).

Spermatogonium apoptosis with TUNEL staining. Seminiferous tubules of control group (A), MPH-LD group (B), and MPH-HD group (C). TUNEL-positive spermatogonium cells at ×400, inset: TUNEL-positive spermatogonium cells at ×100. TUNEL staining.
Discussion
In addition to the reports on humans,2,4,5,17–20 several studies in animals also linked MPH with temporary growth-restriction.21–24 The National Toxicology Program—Center for the Evaluation of Risks to Human Reproduction (NTP-CERHR) stated that there was limited but sufficient evidence for the reversible growth restriction in animals with postnatal MPH exposure. 4 Two studies on rats showed reversible growth restriction with subcutaneous administration of MPH at 35 mg/kg/day or more. 4 It was also noted that the growth restriction in these animals only occurred under high MPH doses received through a non-therapeutic route of exposure, which may not replicate the use of MPH in children with ADHD. We did not observe statistically significant reduction of body weight in any of the rat groups in our study, which may be connected to our smaller dosage of MPH and an oral route of drug administration compared to the above-mentioned studies. On the other hand, the MPH-HD regimen caused decreased testicular weight, which may be interpreted as an indicator of germ cell depletion. 25
MPH is believed to work by affecting dopamine transport and D2 receptors and also has minor effects on serotonin and noradrenalin.23,26 There is evidence that D2 and several α- and β-adrenergic receptors are expressed in the rat testis and spermatozoa of rats, mice and humans.7,8 Although the exact mechanism is not yet identified, these findings seemingly explain the route of MPH effects in the mammalian male reproductive tissues.
Spermatogenesis in the rat is a gradual transformation process of germ cells into spermatozoa over a period of 48–53 days. One spermatogenic cycle (from spermatogonium to meiosis to sperm release) takes 12–13 days and consists of 14 stages.10,11 In our study, the numbers of round spermatids in stage II in both low- and high-dose groups and in stage V in the MPH-HD group were lower than controls. Other cells that were assessed (spermatogonia, pachytene, and zygotene spermatocytes) were not affected in either group. No alterations were found in stage XII spermatids of either group. These findings indicate a cell-specific toxic effect of MPH on rat testes, which does not appear to be stage-specific as both stages II and V were affected in the MPH-HD group. Any cellular sensitivity to a specific toxic substance during spermatogenesis causes death of these cells by apoptosis and, in time, progressive depletion of the descendent cells—‘maturation depletion.’13,27 In this context, different interpretations are possible for our results. The preceding cells of round spermatids, spermatocytes, might have been the actually affected ones, but, based on the maturation depletion phenomenon, this would necessitate a more prominent depletion of round spermatids than in our study. This latter concept may also challenge the interpretation that the target cell was the round spermatids. More studies are required to clarify these issues.
To date, there have been few studies addressing MPH effects on sperm development in animals and, to our knowledge, none in humans. The NTP-CERHR Monograph in 2005 4 stated that there were insufficient data on the possible reproductive effects of MPH in humans and experimental animals. While one of the two NTP animal studies demonstrated reduced sperm motility in mice fed on diets containing 15 mg/kg/day MPH, the second study, including a different mouse strain and using a different design, did not replicate this result. 4 Using adolescent animals and a lower dose of MPH (2 mg/kg/day), Adriani et al. showed transient trophic effects of MPH on rat testes such as increased testicular weight and sperm quantity. 6 Our data showing negative rather than trophic effects might be reflecting the prepubertal age of the rats and the higher dose of MPH in our experiments.
To the best of our knowledge, this is the first animal study showing significant negative MPH effects in the quantity of spermatogenic cells. Our administered doses of MPH were much higher than the therapeutic doses for children with ADHD, which are at maximum 1 mg/kg/day or 60 mg/day. 28 However, this is also true for other animal studies on MPH. 21 Many drugs are generally administered to rats at doses up to 10-fold higher than humans due to the faster hepatic drug metabolism in rodents. 29 Negative effects on testicular morphology and sperm quantity were more evident in the 10 mg/kg/day group than the 5 mg/kg/day group, which indicates a dose-dependent toxicity of MPH on the male reproductive system.
Apoptosis is required during pubertal spermatogenesis to eliminate abnormal germ cells and to maintain a proper amount of maturing germ cells. Its control is crucial in order to achieve normal, mature spermatogenesis.30,31 While apoptosis is a massive event in the first pubertal spermatogenetic wave, in the adult testes it is a rare event that mainly affects spermatogonia. 32 In damaging states of mouse testes, apoptotic cells increase and the Fas/FasL system has been implicated as the initiator. 33 Additionally, the tumor suppressor protein p53 participates in the regulation of germ cells' apoptosis. 34 With the TUNEL method, we found that apoptotic indices were higher in both MPH groups while p53 expression was only increased in the MPH-HD group. However, we could not specify or count the positive cells in these methods due to the lack of characteristic cellular details in the slides. Still, in light of the previous studies and in parallel with our quantitative findings on round spermatid counts, MPH seems to damage spermatogenic cells and somehow, directly or indirectly, induce apoptosis.
The TGF-β is a pluripotent regulator of cell growth, apoptosis, motility, tumorigenesis, proliferation, differentiation, and gene expression. TGF-β1 is expressed in spermatogonia, spermatocytes, and round but not elongated spermatids of both 21 days old and adult Wistar rats. 35 TGF-β1 was reported to have both stimulatory and inhibitory effects on cellular proliferation.36,37 Similarly, it has dual effects on the apoptotic pathways that seem to depend on the cell type and context. 38 This dual activity also extends to the spermatogenic cells. Teerds and Dorrington 39 suggested that TGF-βs either promote the differentiation of germ cells in an autocrine fashion or maintain the necessary communication with adjacent Sertoli cells. TGF-β1 was found to specifically stimulate and increase the DNA synthesis level of preleptotene spermatocytes in the spermatogenic stage VIIa. 39 On the other hand, TGF-β1 was also shown to have an inhibitory impact on germ cell proliferation.40–42 While Olaso et al. 43 reported that TGF-β1 and -β2 hold a negative effect on the development of gonocytes, Konrad et al. 44 revealed that TGF-β isomers induce the apoptosis of pubertal male germ cells via the mitochondrial pathway. The significantly low levels of TGF-β1 in MPH-LD and -HD groups in our study do not parallel the high p53 expression and apoptotic counts considering its inhibitory effects and participation in the apoptotic pathways. Our results may instead indicate a TGF-β1’s loss of stimulatory effects. The p53 protein has a role in the TGF-β-induced growth arrest and may be the prominent regulator of TGF-β’s cytostatic activity. 45 Our low TGF-β1 expression levels could be due to its inhibition through different pathways involving p53. Finally, we must mention that these results for TGF-β1 cannot easily be applied to humans, as TGF-β1 was not detected in human seminiferous tubular cells. 46
In conclusion, we believe that our findings must still be cautiously interpreted and long-term prospective studies on animals with different ages and MPH doses must be conducted as studies on human testicular morphology, sperm development and fertility is somewhat difficult due to ethical factors. Comparison of adults with and without a history of long-term MPH use may disclose interesting results in terms of fertility, offering new approaches to future studies.
Footnotes
We thank Bayındır Hospital for their financial support.
