Abstract
Lab-on-a-chip systems are increasingly used as tools for cultures and investigation of cardiac cells. In this article, we present how the geometry of microsystems and microenvironmental conditions (static and perfusion) influence the proliferation, morphology, and alignment of cardiac cells (rat cardiomyoblasts—H9C2). Additionally, studies of cell growth after incubation with verapamil hydrochloride were performed. For this purpose, poly(dimethylsiloxane) (PDMS)/glass microfluidic systems with three different geometries of microchambers (a circular chamber, a longitudinal channel, and three parallel microchannels separated by two rows of micropillars) were prepared. It was found that static conditions did not enhance the growth of H9C2 cells in the microsystems. On the contrary, perfusion conditions had an influence on division, morphology, and the arrangement of the cells. The highest number of cells, their parallel orientation, and their elongated morphology were obtained in the longitudinal microchannel. It showed that this kind of microsystem can be used to understand processes in heart tissue in detail and to test newly developed compounds applied in the treatment of cardiac diseases.
Introduction
Heart diseases are the leading causes of death around the world. According to the World Health Organization (WHO), about 17.5 million people die from cardiovascular disease (CVD) annually. 1 It is the cause of one-third of deaths globally. Additionally, modern lifestyles have also contributed to an increase in the number of patients with CVD. 2 Although there are various drugs used for CVD treatment, new therapies are needed. Preclinical studies of cardiovascular drugs are crucial during the development of new medicines. Drug toxicity is studied on both explanted heart tissue and animals—mainly rodents. In vitro tests are most often used at the first stage of research into new compounds. 3 In vitro tests require less time than in vivo tests; however, they are conducted under controlled conditions. 4 In recent years, the use of cardiomyocytes derived from human pluripotent stem cells (hPSC-CMs) has become more popular. hPSC-CMs exhibit a specific function of cardiac cells—a spontaneous rhythmic beating. Therefore, hPSC-CMs could be used as a great model for drug screening.5,6
Nowadays, microsystems (called lab-on-a-chip) are promising tools for the analysis of cells.7–9 These systems have a lot of advantages that are significant in biological studies, that is, the reduction of biological reagents, the shortening of analysis time, cost reduction, the possibility of real-time analysis, and the ability to mimic cell microenvironments.10,11 Poly(dimethylsiloxane) (PDMS) is flexible, easy to fabricate, biocompatible, water impermeable, gas permeable, optically transparent, and inexpensive.7,12,13 In addition to the advantages, PDMS also contains several limitations that potentially have negative effects on cell proliferation. Oxygen permeability through PDMS is three orders of magnitude higher than that through polystyrene (PS). Therefore, a hyperoxic microenvironment that can cause cell stress can be produced in a PDMS-based microsystem. 14 Moreover, the ability to allow water evaporation can significantly change the volume and concentration of culture solutions, which can affect cell proliferation. 15 The absorption of hydrophobic compounds and leaching of un-cross-linked oligomers are the next properties that could have a negative influence on cell viability.16,17 Despite these disadvantages, PDMS is the best-known and most useful material applied in the fabrication of microsystems used for cell culture. PDMS-based microsystems have been used to study culture, proliferation, and migration of various cell types.18,19 Moreover, studies of drug metabolism and cytotoxicity analysis have been performed in microscale. 20 Due to the fact that stem cells are increasingly utilized in regenerative medicine, new microfluidic devices for stem cell differentiation and culture have also been developed.21,22
Lab-on-a-chip systems are also used as tools for muscle tissue engineering, the investigation of muscle functions, and the creation of a heart model. Proliferation, as well as viability and contraction of cardiomyocytes under various conditions, was examined in microscale. In vivo cardiac cells are arranged parallel to each other and form muscle fibers. For this reason, it is important to create a cellular model most resembling the myocardial tissue. The medium flow is the factor that can stimulate the cells to orient themselves parallelly.23,24 Nguyen et al. 4 demonstrated a cardiac cell culture model (CCCM) platform, which mimics the work of the left ventricle. They tested the influence of the fluid flow on embryonic chick cardiomyocyte growth. Butcher et al. 25 presented a study on how a fluid flow environment influences the growth of porcine aortic valve endothelial cells (PAVECs) and porcine aortic endothelial cells (PAECs). The results of the static culture showed that both cell types are randomly arranged. During flow conditions, PAECs aligned parallel to each other and the flow direction, whereas PAVECs arranged parallel to each other and perpendicular to the medium flow. Annabi et al. investigated how hydrogel and tropoelastin influence the creation of heart tissue. 26 Single-cardiomyocyte behavior and the concentration of calcium ions inside the cell after stimulation with ionomycin were also examined. 27 Cheng et al. used electrochemical and optical measurements to explore cell metabolism. For this purpose, a single cardiomyocyte was stimulated by a transient electric field. 28 Grosberg et al. demonstrated a microtool for heart tissue structure–function and pharmacological studies. The contractile function of cardiac tissue was noticed after the usage of the drug. 29 Due to the fact that ischemia is the main heart disease, the effect of hypoxia on cardiac cells has also been studied in the microsystems.30,31
Although these existing microsystems were used for cardiac cell analysis, they still need to be improved in many aspects. In contrast to the studies described above, we examined the proliferation of H9C2 cells (in vitro cardiac cell model) in microsystems with three different geometries of culture microchambers. An external (i.e., mechanical) stimulation of cardiac cells could have an influence on their growth. 26 Therefore, we hypothesized that perfusion conditions and flow stimulation will improve H9C2 cell proliferation. In contrast to other studies, here we present how the geometry of culture microchambers and microenvironmental conditions (static and perfusion) influence cardiac cell proliferation, morphology, and alignment. Studies with verapamil hydrochloride, an L-type calcium channel blocker (CCB), were performed to check how culture conditions and the geometry of microsystems affect cell cytotoxicity. It was investigated which one of the developed microsystems could be useful for cardiac drug analysis. To the best of our knowledge, we examined H9C2 cell proliferation after continuous flow (72 h) of verapamil in different types of microsystems as a first.
Materials and Methods
Design and Fabrication of Microsystems
Three PDMS/glass microsystems with different culture microchamber geometries were used in our experiments. The first microsystem consisted of a microchamber with a circular shape (a diameter of 1 mm, a height of 50 µm, a volume of 3.91 × 10−2 μL, a culture chamber area of 0.78 mm2) ( Fig. 1A ). The second microsystem contained a microchamber with a longitudinal microchannel shape (a width of 300 µm, a length of 2 mm, a volume of 3.39 × 10−2 μL, a culture chamber area of 0.68 mm2) ( Fig. 1B ). The third microsystem had a microchamber with three parallel microchannels separated by two rows of micropillars (50 × 100 µm, gap 20 µm) ( Fig. 1C ). The main microchannel (2 mm long, 400 µm wide, 50 µm high, a volume of 0.19 μL, a culture chamber area of 1.91 mm2) was used for the cell culture, whereas two side microchannels (a width of 200 µm) were used for providing the culture medium.
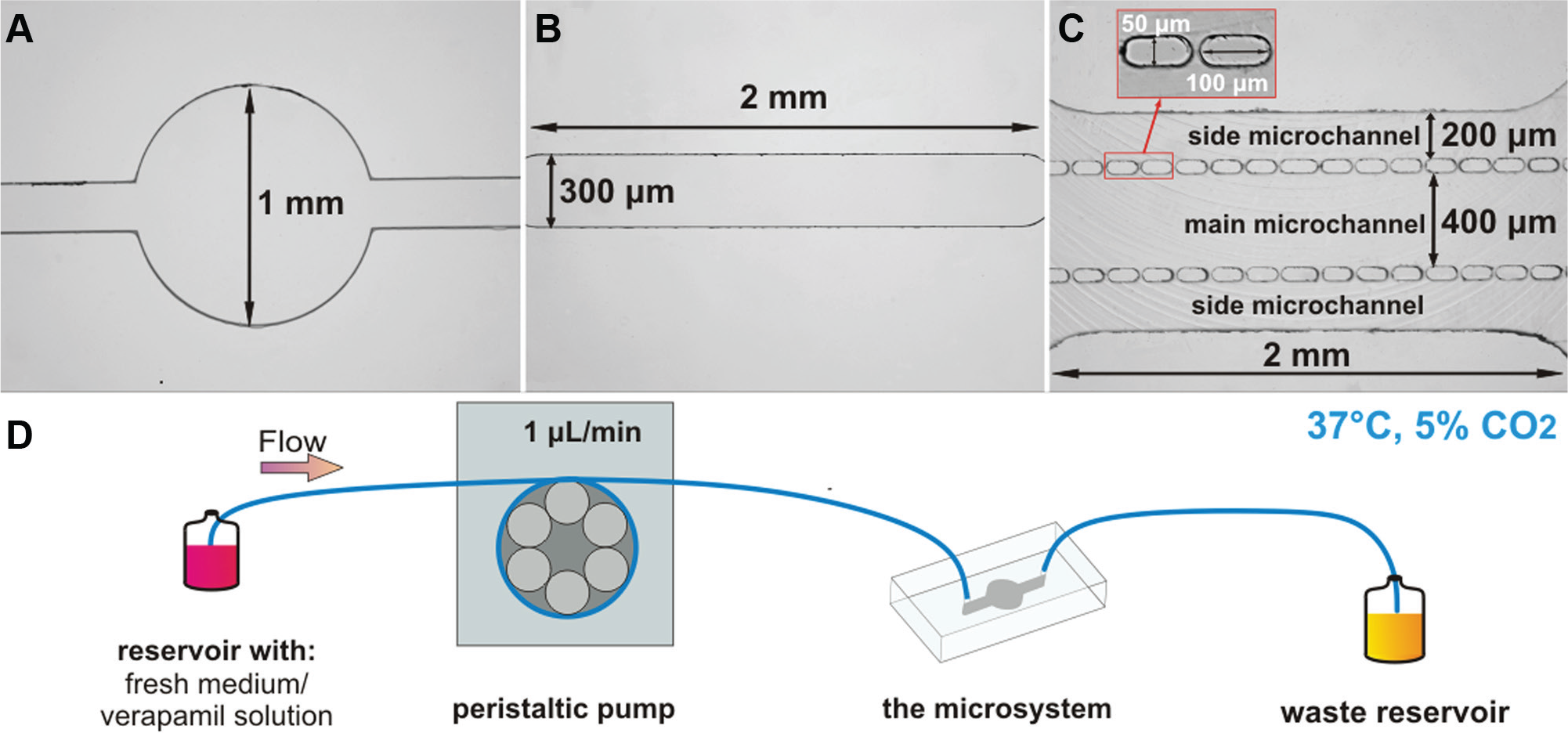
Geometries of the fabricated microchambers: (
The designed microsystems were composed of two layers: bottom, a glass slide (cat. no. 296.202.01, Chemland, Stargard, Poland), and top, PDMS plate (cat. no. 1272/2008, Sylgard 184, Dow Corning, Auburn, MI) with microstructures. The microstructures of the microsystems shown in Figure 1A , B were fabricated using photolithography and replica molding techniques.8,32 The microstructure of the third microsystem ( Fig. 1C ) was fabricated using micromilling and replica molding techniques. The micromilling technique was used for poly(methyl methacrylate) (PMMA) master fabrication. 33 The PDMS plates with microstructures were prepared by mixing a prepolymer and a curing agent with a weight ratio of 10:1. After degassing, the PDMS mixture was poured over stamps and cured at 70 °C for 1 h. The PDMS replica was peeled off from the stamps and holes were drilled. Next, the PDMS layers were bonded with glass slides using an oxygen plasma treatment (Plasma-Preen II 973, North Brunswick, NJ).
H9C2 Cell Cultures
Rat cardiomyoblasts, H9C2 (cat. no. ATCC CRL-1446, American Type Culture Collection, Manassas, VA), derived from rat embryos, were used in the experiments. H9C2 cells were maintained in Dulbecco’s modified Eagle medium (DMEM, cat. no. D5671, Sigma-Aldrich, Dorset, UK) supplemented with 10% v/v fetal bovine serum (FBS, cat. no. 26140079, Gibco, Grand Island, NY), 1% v/v 100 mM streptomycin-penicillin (cat. no. P4333, Sigma-Aldrich, Rehovot, Israel), and 1% v/v Glutamax (cat. no. 35050061, Gibco, Lyon, France). The cells were cultured in a humidified incubator (37 °C, 5% CO2, HERA-cell 150, Thermo Fisher Scientific, Waltham, MA).
Cell Cultures in 96-Well Plates
H9C2 cell suspensions (density of 104 cells per well) were seeded in 96-well plates and allowed to attach overnight. Cell growth was analyzed daily using commercially available AlamarBlue assay (cat. no. BUF012A, Bio-Rad, Hertfordshire, UK). The culture medium was removed from the wells and 200 µL of 10% AlamarBlue solution (in the culture medium) was added to each well. Next, 96-well plates were incubated at 37 °C for 1 h. The intensity of fluorescence was measured using a multiwell plate reader (Cytation 3, BioTek, Winooski, VT) at an excitation wavelength of 532 nm and emission wavelength of 590 nm. After the AlamarBlue assay, a fresh culture medium was added in each well. The growth of the cells was monitored for 96 h.
Cell Culture in the Microsystems
At the beginning of the experiments, the fabricated microsystems were sterilized using 70% v/v ethanol (cat. no. 396420113, Avantor Performance Materials Poland S.A., Gliwice, Poland) and ultraviolet (UV) light (Black Ray). Then, to provide appropriate culture conditions, the microsystems were filled with the culture medium (supplemented with FBS) and placed in an incubator for 2 h. H9C2 cell suspension (density of 106 cells/mL) was prepared and the cells were seeded in the culture microchambers of each fabricated microsystem with a flow rate of 5 μL/min. To enable cell adhesion to the glass substrate, the microsystems were placed in an incubator for the next 24 h. After that time, each type of microsystem was subjected to static and perfusion conditions. While performing the static culture, the microsystems were placed in an incubator for 96 h. The culture medium in the microchambers was exchanged periodically—every 24 h for 10 min (a flow rate of 1 μL/min). The perfusion setup (system), used for continuous introduction of the culture medium (and verapamil), consisted of a reservoir with a fresh culture medium, a peristaltic pump, the microsystem, and a waste reservoir ( Fig. 1D ). During the perfusion culture, a fresh medium was continuously provided for 72 h (from 24 to 96 h; a flow rate of 1 μL/min). To ensure proper culture conditions (37 °C, 5% CO2), the sterilized setup was placed in an incubator. Furthermore, studies of cell growth using verapamil (verapamil hydrochloride, cat. no. ab120140, Abcam Biochemicals, Cambridge, UK) were performed under perfusion conditions. For this purpose, a 2 mM stock solution of verapamil hydrochloride in distilled water was prepared. Then, the stock solution was diluted to a final concentration of 10 µM in DMEM. Next, verapamil solution was steadily introduced in the microsystems for 72 h (from 24 to 96 h; a flow rate of 1 μL/min).
Viability Assay
The viability of H9C2 cells was performed 96 h after cell seeding for both static and perfusion (with and without verapamil exposure) cultures. For this purpose, fluorescent dyes were utilized: the acetoxymethyl ester derivate of calcein (calcein-AM [CAM], cat. no. 17783, Sigma-Aldrich, UK) and propidium iodide (PI, cat. no. P4864, Sigma-Aldrich, St. Louis, MO). The use of these dyes allows the distinguishing of live (using CAM) and dead (using PI) cells. The viability tests were performed according to the previously described method. 34
Data Analysis
Cell population changes, morphology, and arrangement, under both static and perfusion cultures, were monitored in the microsystems using an inverted microscope coupled with a charge-coupled device camera (CCD camera) (Olympus IX71). For data acquisition and analysis, cellSens image analysis software (Olympus) was used. Cell population changes were determined by a daily count of the cell number in each fabricated microsystem. The alignment of the cells was analyzed using Directionality plugin ImageJ software. The percentage of the cell at each angle of alignment was graphed, with an angle of 0° being perfect alignment. Additionally, data on the percentage of alignment cells (between −5° and 5°) were graphed for the perfusion culture. The cell viability in the microsystems was determined by counting the number of green (live cells) and red (dead cells) objects. The experimental data were presented as the mean ± standard deviation (SD) from at least four (for cell population changes) and at least three (for viability) experiments. One-way analysis of variance (ANOVA) was used to compare the differences in cell proliferation in the following days of cell culture. Test for variance (F test) and T test were used to compare differences between static and perfusion conditions, as well as to compare the difference between verapamil-treated and nontreated conditions. p values of less than 0.05 were considered statistically significant (asterisk indicates p < 0.05).
Results and Discussion
Fabricated Microsystems
The first proposed microsystem had a circular culture microchamber ( Fig. 1A ). We decided to use this geometry, due to the fact that it is the most popular geometry of culture microchamber in the microsystems described in the literature. 32 In vivo cardiac muscle cells are arranged in parallel to each other to form muscle fibers. We hypothesized that the next type of microsystem, with the longitudinal channel, would enhance the parallel arrangement of cardiac cells ( Fig. 1B ). The microsystems described above were designed in such a way that the medium could be introduced in the same place where the cells were cultured. In the third microchamber, with three parallel microchannels separated by two rows of micropillars ( Fig. 1C ), the cells were cultured in the main microchannel. In this microchamber, the culture medium was supplied through the side microchannels and diffused to the cells between micropillars. Different geometries of the microchambers and methods of supplying the culture medium enabled us to examine the influence of the medium flow on the morphology and arrangement of the cells.
To verify the flow of the culture medium in the microchambers, a computer modeling simulation using the microelectromechanical systems (MEMS) simulations module of COMSOL Multiphysics software was performed. The simulation was conducted for 1 µL/min ( Fig. 2A ) and 5 µL/min ( Fig. 2B ) flow rates at the inlet. A similar fluid trajectory was observed in the microsystems for both analyzed flow rates. We noticed that in the circular microchamber, the fluid spread around the chamber. It was also observed that the fluid distributed with different flow velocities in the microchamber. The highest value of a flow velocity was obtained in the center of the chamber. In the longitudinal channel, the fluid distributed parallel to the chamber walls, with a steady flow velocity in the whole microchamber. In the microchamber with micropillars, the fluid flow was parallel to the chamber walls; however, the value of flow velocity was very low. Moreover, to identify the value of the wall shear stress in various regions of the microchambers, the distribution of this parameter in the microchambers was simulated ( Fig. 2C ). The wall shear stress generated in the microchambers was directly dependent on the microchamber geometry and the fluid trajectory. Comparison of the wall shear stress in different microchambers (for the same flow rate) showed that the highest wall shear stress was in the longitudinal channel. The usage of a 5 µL/min flow rate generated a nearly five to six times higher wall shear stress than the flow rate of 1 µL/min. To ensure the proper growth of the cells and to prevent their stress, we decided to use a flow rate of 1 µL/min in the following experiments. Simulation results were confirmed by the experimental method performed in the fabricated microsystems (see Supplementary Materials).
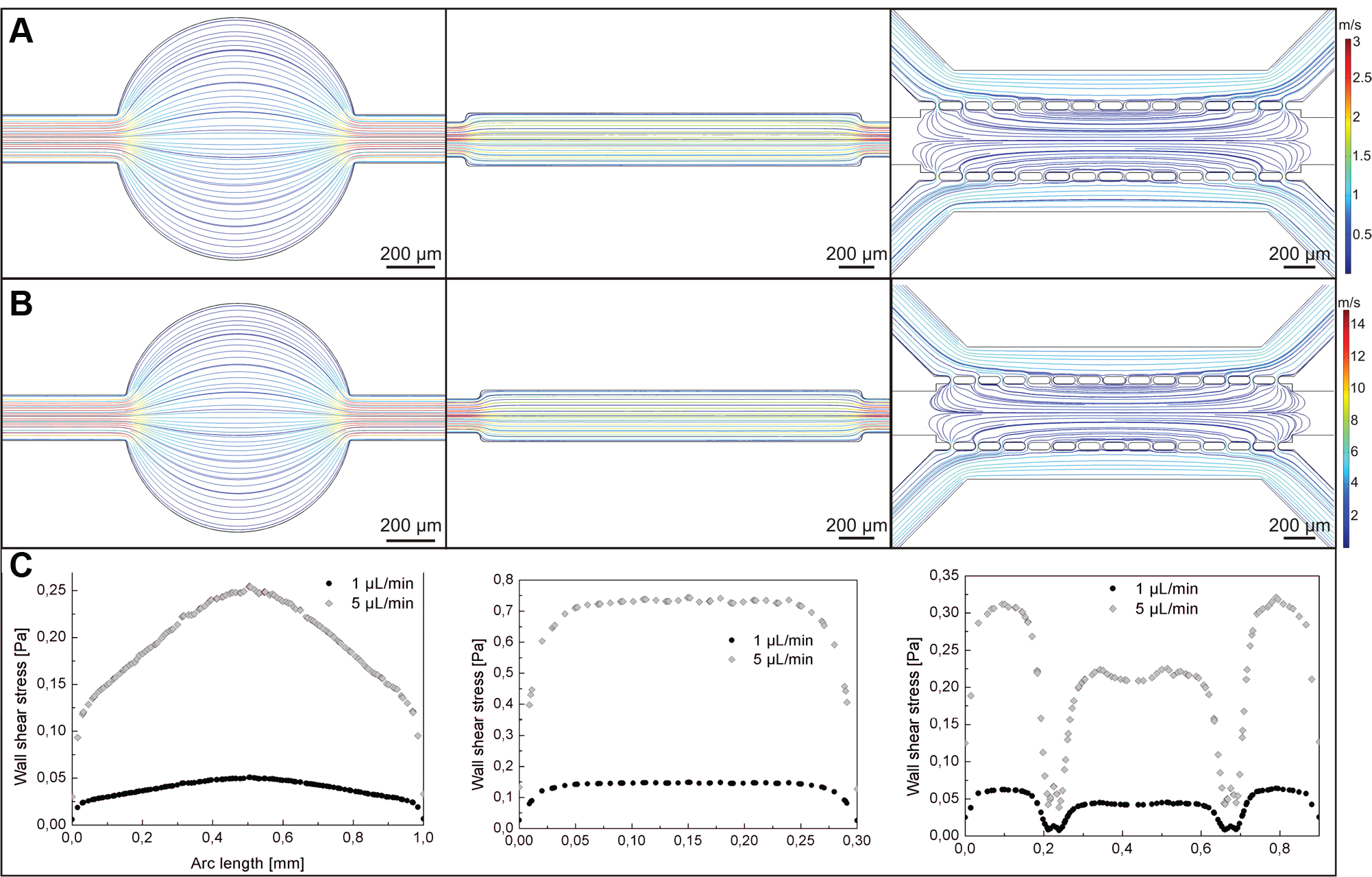
Fluid flow modeling in microsystems with circular chamber, longitudinal channel, and three parallel microchannels separated with two rows of micropillars at (
H9C2 Cell Culture
Rat cardiomyoblasts (H9C2 cell line) are characterized by a spindle shape, sugar residues of the cell surface, and parallel arrangement. An important feature of these cells is their ability to differentiate the mononuclear myoblast into multinuclear myotubes. Moreover, H9C2 cells have voltage-dependent Ca2+ channels, which are a characteristic feature of cardiac cells.35,36 H9C2 cells are most often used as a cardiac cell model. Therefore, we decided to use them in our research. First, to enable cell adhesion, all microsystems and 96-well plates were placed in an incubator (in static culture) after cell seeding. We observed that 24 h after seeding, H9C2 cells adhered to the substrate very well. At this time, in each type of microsystem two kinds of cultures were started: under static and perfusion conditions. Ninety-six-well plates were used only for the static culture. Due to the fact that culture conditions were changed 24 h after cell seeding, the results were normalized to the cultures at this moment. We assumed that the cell population at this time equaled 100%. Cell population changes in the following days were monitored relative to 24 h cultures.
Cell Culture under Static Conditions
It was found that the number of H9C2 cells decreased in the microsystems during static cell cultures ( Fig. 3A ). In the circular microchamber, a slight decrease of 81.9 ± 18.8% (n = 5) of the H9C2 cell population was observed 48 h after cell seeding. In the following hours, the cell number decreased to 64.5 ± 7.5% (n = 5). The number of the cells in the microsystem with the longitudinal microchannel was 75.1 ± 24.6%, 65.4 ± 14.1%, and 61.4 ± 13.1% (n = 5) for 48, 72, and 96 h after cell seeding, respectively. Similarly, a decrease in the cell population was noticed during a culture performed in the microsystem with three michrochannels separated by two rows of micropillars. No statistical differences were observed between microsystems with different geometries.
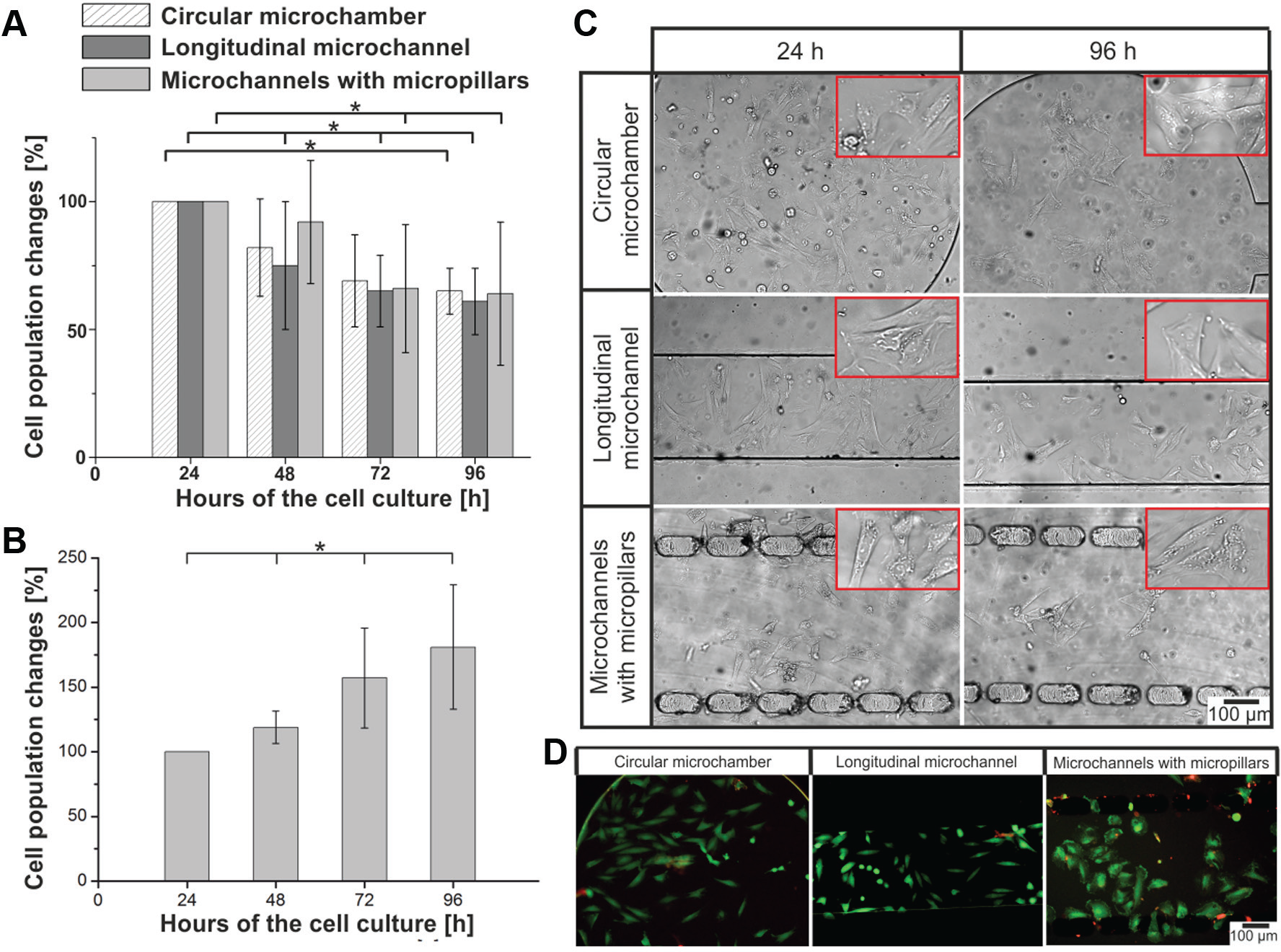
H9C2 cell culture under static conditions. (
Figure 3C shows the morphology and arrangement of H9C2 cells cultured 24 and 96 h after cell seeding. The morphology and arrangement of H9C2 cells in the microchambers were similar in each microsystem, at each stage of the cell culture. The cells adhered to the substrate, forming flattened and irregular shapes. Moreover, the cell arrangement in each microchamber was random. High viability (green objects) of the cells was observed at the end of cultures ( Fig. 3D ). Contrary to the results obtained in the microsystem, it was observed that a static cell culture in macroscale (96-well plates) led to an increase of H9C2 cell numbers ( Fig. 3B ). Differences between micro- and macroscale could result from the different culture environments (i.e., culture volume, properties of culture surface, and culture medium distribution into the cells). A high volume of fresh culture medium can enhance the growth of cardiomyoblasts in 96-well plates. A low cell number in microsystems could result from a high ratio of surface area to volume (SAV) and a low culture volume. Static cultures in the microsystems with similar geometry and microchamber volume, as in our research, have been presented in the literature. In many reports, a high proliferation of various cell types, that is, A549, MRC-5, MCF-7, HepG2, and HT-29 cells, was described.7,11,20,32,34 As presented in the reports above, growth and proliferation of the cells in the microsystems are slower than in 96-well plates. Based on the literature, we concluded that not only culture volume had an influence on the proliferation of H9C2 cells cultured in the microsystems. We supposed that low growth of H9C2 cells cultured under static conditions in the microsystems could also result from such factors as the nature of the tested cells and the fact that the cardiomyoblasts cultured in microscale require an external (i.e., mechanical) stimulation for proper growth and division.
Cell Culture under Perfusion Conditions
In perfusion cell cultures, the cell number increased significantly compared with the 24 h cultures ( Fig. 4A ). In the microsystem with the circular microchamber, 48 h after the cell seeding, the cell number reached 128.6 ± 31.1% (n = 6). At the end of the culture, the cell population was almost twice as high as 24 h after cell seeding. The H9C2 cell population in the microsystem with the longitudinal michrochannel also increased significantly in the following days. The largest cell division was observed between the third and fourth days of the culture. Finally, the number of H9C2 cells was circa 3.5 times higher than in the initial results (24 h after cell seeding). The curve of the cell population changes in the longitudinal microchannel increased exponentially, similar to the model of cell growth. Another tendency of H9C2 cell growth was observed in the microsystem with three microchannels separated by two rows of micropillars. The cell number was maintained at the same level, 107% ÷ 126% (n = 5), during the whole culture. To summarize, the perfusion culture enhanced cell growth in each tested microchamber. However, the value of the increase was different depending on the type of microsystem (longitudinal channel > circular chamber > three parallel microchannels separated by two rows of micropillars). Contrary to the static cultures performed in the microsystems, the obtained results showed that the perfusion cultures significantly enhanced H9C2 cell division ( Fig. 4A ). The highest differences of the cell number between perfusion and static cultures were noticed in the longitudinal microchannel. We supposed that it can result from regular flow rate and high wall shear stress in this microchamber, which in turn could have a large influence on the rate of cell division.
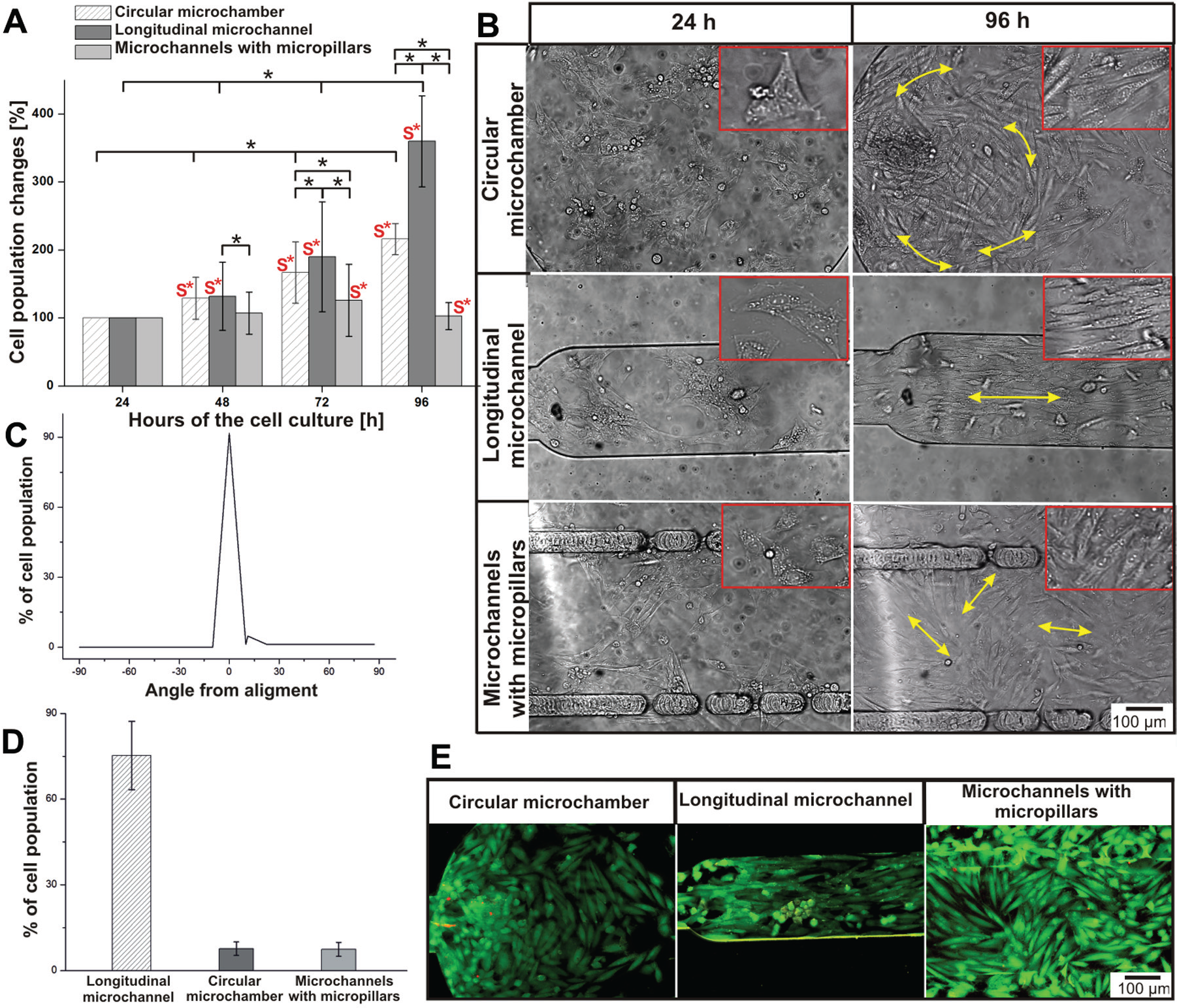
H9C2 cell culture under perfusion conditions. (
It was also noticed that the flow of the culture medium determined cell alignment and cell orientation, depending on the geometry of the microsystem ( Fig. 4B ). In the circular microchamber, a part of the cardiomyoblast was arranged parallel to the others and to the medium flow. In Figure 4B , H9C2 cell orientation was marked using yellow arrows. As is shown in Figure 2 , the flow rate of the culture medium is different in various places in the circular microchamber. It may influence the reduction of regularity in the cells’ arrangement The parallel orientation of the cells was observed in the microsystem with the shape of the longitudinal channel in which the medium flowed above the cultured cells. The cardiomyoblasts were elongated and strictly adhered to each other. H9C2 cell alignment mimicked the cell orientation in myocardium tissue. In the microsystem with micropillars, the cells were the most flattened and randomly oriented. There are some places (clusters) in which the cells were arranged parallel to each other (yellow arrows). Alignment analysis ( Fig. 4C ) showed that H9C2 cells aligned parallel to the others and to the direction of flow medium in the longitudinal microchannel within 72 h of perfusion. Data on the percentage of alignment cells (between −5° and 5°) in the microsystems with 96 h cultures were graphed in Figure 4D . Cell alignment in the circular microchamber and microsystem with micropillars equaled 8.7% ± 2.1% and 8.1% ± 3.3%, respectively, whereas for the longitudinal microchannel it was 76.7% ± 13.6% (n = 3). The above analysis showed that the microchamber geometry determines the alignment of H9C2 cells cultured under perfusion conditions. The high viability of the cells (96.0 ± 2.6%, 94.5 ± 2.1%, and 97.7 ± 1.8%, n = 4) was noticed at the end of the cell cultures ( Fig. 4E ). A comparison of the morphology and orientation of the cells cultured in the microsystems under static and perfusion conditions showed that flow conditions determine cardiac cell alignment, especially in the longitudinal microchannel.
A comparison of the results obtained in microsystems under perfusion conditions with the standard culture method (static culture in 96-well plates) proved that flow conditions have an influence on cardiac cell growth. It was investigated that H9C2 cell division in the longitudinal microchannel is twice as high (on the last day of cell culture) as in cultures performed using the standard method. The cell population was a little higher in the circular microchamber than in the 96-well plates. In turn, in the microchamber with micropillars, the cell number was lower than in the 96-well plates. In vivo cardiomyocytes are under constant physical stimulation (pressure, stretch, and shear stress) and mechanical stress during contraction and relaxation. 37 Fluid flow and shear stress influence cell proliferation and phenotype. Cell orientation is an important factor in cardiomyocyte signaling (random cell arrangement does not mimic physiological conditions). The highest parallel orientation of the cells was obtained in the longitudinal microchannel because the cells could mimic in vivo cardiac cells better than in other microsystems. This indicated that perfusion culture can enhance cardiac cell growth; however, the value of the flow rate and the fluid trajectory greatly help to determine cardiac cell orientation. It was observed that if the cells adhere to each other parallelly, cell division is strongly enhanced.
Cell Culture with Verapamil
The influence of an additional factor (cardiac drug) on H9C2 cell proliferation was also investigated in our research. Calcium ions play a significant role in the physiological processes of myocardial cells, that is, impulse formation and conduction, and excitation–contraction coupling. 38 CCBs are the most frequently used drugs in the treatment of CVD. CCBs inhibit Ca2+ flow through L-type calcium channels; the conduction of impulses through an atrioventricular node decrease, causing a drop in blood rate. 30 Based on the literature, we decided to use a low toxic concentration (10 μM) of cardiac drug—verapamil hydrochloride.39,40 It is an L-type CCB, used in the treatment of ischemic heart disease, hypertension, and cardiac arrhythmia. Due to the fact that in the microsystems only a perfusion culture enhanced H9C2 cell growth, we used this kind of culture for analysis of cell population changes after verapamil exposure. The obtained results showed that continuous flow of verapamil caused no significant changes (compared with the 24 h cultures) of cell proliferation during the following days ( Fig. 5A ). A comparison of perfusion cultures with and without verapamil showed significant differences in cell proliferation for each microsystem for the 96 h culture ( Fig. 5A ). The highest differences were noticed in the longitudinal microchannel. This could be because of the high influence of the medium flow on cell orientation and as a consequence of cell division in this type of microchamber. It was shown that the continuous flow of verapamil inhibited cell division and growth. It could be possible that verapamil had a toxic influence on the cells. Therefore, viability tests at the end of the cultures were performed. A high viability of H9C2 cells was noticed on the last day of the culture ( Fig. 5B ). The point of the viability measurements was that dead cells, which might possibly affect the results of cell population changes, could be found in the culture. As shown in Figure 5C , only live cells were the microsystems. Cultures with verapamil were performed under perfusion conditions; therefore, if H9C2 cells cultured with verapamil died, they would have detached and washed out from the microsystems. This could be the reason why low proliferation of the cell was observed at 96 h. A decrease of H9C2 cell population may also have been caused by a verapamil overdose (the drug was administrated over 72 h). The blocking of L-type calcium channels by verapamil reduced the intracellular Ca2+ concentration, which led to hemodynamic shock, dilatation of cardiac cells, and impairment of their function. 41
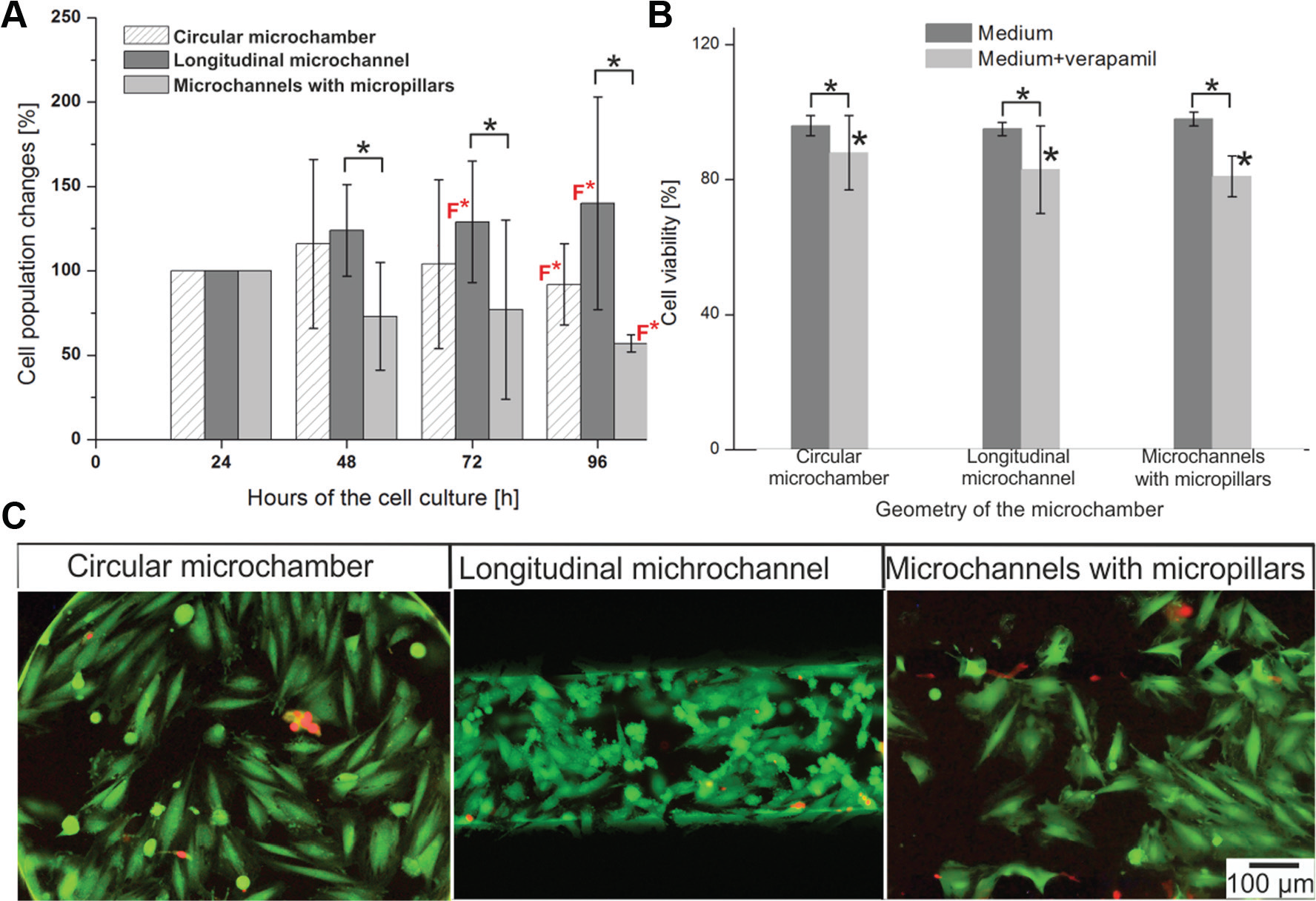
H9C2 cell culture under perfusion conditions after verapamil exposure. (
In contrast to other works,24–26,42 here we have presented how the geometry of the culture microchamber may influence cardiac cell arrangement. The alignment of various cells, that is, embryonic chick cardiomyocytes, PAVECs, PAECs, H9C2 cells, and primary neonatal rat cardiomyocytes, under perfusion conditions was investigated. However, H9C2 cell alignment was investigated only by a few research groups. In studies presented in the literature, the influence of flow medium in one type of the geometry—circular microchambers 26 or parallel channels 24 —was investigated. Here, we tested the arrangement of the H9C2 cells in three different microsystems under both static and perfusion cultures. Additionally, the proliferation of the H9C2 cells after continuous flow (72 h) of verapamil in different types of microsystems was investigated as a first. Our research has allowed us not only to find appropriate H9C2 cell culture conditions, but also to select the suitable geometry of the culture microchamber. The obtained results showed that the microsystem with the longitudinal microchannel can be used to mimic cardiac cells’ microenvironment and most likely to analyze the therapeutic effect of other cardiac drugs.
Conclusion
In this study, we have demonstrated how the geometry of microsystems (circular chamber, longitudinal channel, and three parallel microchannels separated by two rows of micropillars) and microenvironmental conditions (static and perfusion) influence H9C2 cell proliferation, morphology, and alignment. The obtained results support our hypothesis that the external stimulation (continuous medium flow) enhanced both the growth and parallel orientation of cardiac (H9C2) cells. Moreover, it was noticed that the usage of verapamil, an L-type CCB, inhibited H9C2 cell proliferation in cultures performed under perfusion conditions. We observed that these parameters depend on the geometry of the culture microchamber. Additionally, it was noticed that flow and shear stress could mimic the physiological conditions of heart tissue and play an important role in cardiac cell proliferation orientation and division. The highest cell proliferation, parallel orientation of the cells, and the highest influence of verapamil exposure were obtained in the longitudinal microchannel. Therefore, this microsystem can be used to understand processes in heart tissue in detail and to test newly developed compounds used for the treatment of cardiac diseases.
Footnotes
Acknowledgements
The authors thank Dr Patrycja Ciosek (Warsaw University of Technology) for statistical analysis consultation.
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Centre for Research and Development within a frame of LIDER program no. LIDER/026/573/L-4/12/NCBR/2013.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
