Abstract
Cardiac safety testing of lead drug candidates is an important part of the drug discovery and development process. All new chemical entities need to be subjected to extensive preclinical assessment for cardiac liability, especially for a potentially fatal form of ventricular arrhythmia referred to as Torsades de Pointes. We have developed an innovative label-free, real-time system, the xCELLigence RTCA Cardio System, which is designed to monitor contractility of cardiomyocytes based on impedance measurement. The assay is performed using specially designed microtiter plates that are integrated with gold microelectrodes. The system was validated using mouse embryonic stem cell-derived cardiomyocytes, human-induced pluripotent stem cell-derived cardiomyocytes, and rat neonatal primary cardiomyocytes by applying a variety of tool compounds and drugs with known mechanisms of action. Our data show that the time resolution in the assay can provide important information about compound action. Furthermore, the impedance-based beating profile in response to compound treatment can provide mechanistic toxicity information regarding the target being modulated and may be able to flag pro-arrhythmic compounds. We believe the real-time and kinetic aspect of this technology combined with beat-to-beat measurement of cardiomyocyte contraction would make this instrument an important part of preclinical cardiac safety assessment.
Keywords

Introduction
Adverse drug side effects, such as cardiotoxicity, is a major concern in drug development and a major cause of drug withdrawal from the market. 1,2 Most of these drugs appear to interfere with the well-choreographed and elaborate movement of ions across cardiomyocytes membrane disrupting the electrical activity and beating of the heart. 1 However, drugs such as anthracyclines could also induce cardiotoxicity through direct structural or cellular damage. 3 Pharmaceutical companies therefore ensure that all lead compounds and drug candidates are subjected to a series of stringent screens to identify and possibly rectify potential adverse cardiac side effects before drugs enter into clinical trials and ultimately the market.
A number of in vitro and in vivo assays have been established to assess the effect of lead candidate compounds on cardiac function. 1 The in vitro assays are primarily designed to assess the interaction of compounds and drug candidates with the main ion channels known to be involved in maintaining the cardiac action potential. It is estimated that there are more than 70 types of ion channels that are differentially expressed across the repertoire of cardiomyocytes and involved in the overall cardiac current. 4 Preclinical cardiac safety studies have been mainly focused on about a dozen main ion channels with particular emphasis on human ether-a-go-go (hERG) channel. 1,5 hERG is mainly involved in repolarization current and its inhibition by drugs or compounds serves as a surrogate for QT (the time between the start of the Q wave and the end of the T wave) prolongation, which itself is a marker for Torsades de Pointes arrhythmia. 6 Most drugs withdrawn from the market appear to inhibit hERG channel activity by in vitro assays. 1,5 In addition to testing for hERG, lead compounds and drug candidates are also subjected to assays, which screen for direct damage to cardiomyocytes at the cellular or organellar level, such as mitochondria. 3,7 Anthracyclines, a widely used class of chemotherapeutic drugs, is known to induce heart damage by direct toxicity and their use is often hampered by their cumulative dose-limiting cardiotoxicity. 3,7,8
It is imperative to emphasize that while studying the effect of drug candidates on individual ion channels and cellular events may provide useful information, what is really needed is a functional assay system that can assess the integrated cardiac response to drug candidates. Currently, these types of studies are mainly performed in whole organ perfusion assays referred to as Langendorff heart assay or telemetry experiments using live animals. 1 Although these tests are considered more predictive, they are technically challenging, labor intensive, and not amenable for high throughput.
To address these challenges, we (ACEA Biosciences and Roche Applied Science) have codeveloped the xCELLigence RTCA Cardio System, which can measure the contractility of cardiomyocytes. The system uses noninvasive impedance readout for continuous monitoring of cardiomyocytes beating in the wells of specially designed microelectronic plates. The system has been validated using both stem cell-derived cardiomyocytes and primary cardiomyocytes and is able to sensitively and quantitatively detect the effect of ion channel and nonion channel modulators of cardiac function in real time. Furthermore, we found that pro-arrhythmic compounds produce a characteristic beating profile, which maybe reflective of the risk of arrhythmia. In addition, the ability to use the time resolution in the assay in conjunction with the physical beating of cardiomyocytes ensures the RTCA Cardio System could easily detect the effect of certain compounds missed by electrophysiological readouts. Finally, dynamic monitoring of the periodicity of beating over prolonged intervals of time, allows for detection of both short-term and long-term responses. Overall, taking into consideration the sensitivity, predictivity, real-time data acquisition, measurement of periodicity of beating over both short and prolonged window of time, and throughput makes this technology well suited for early preclinical safety assessment of compound.
Principle and Work Flow
Principle of Detection
The xCELLigence RTCA Cardio System uses an impedance readout to noninvasively quantify cardiomyocytes status in real time. 9,10 The cells are seeded in E-Plate Cardio 96, which is specially designed 96-well microtiter plates that are integrated with gold microelectrode arrays in the bottom of each well. Application of a low voltage (less than 20 mV) alternating current signal leads to the generation of an electric field between the electrodes, which interacts with the ionic environment of the growth medium inside the wells and is differentially modulated by the number of cells covering the electrodes, the morphology of those cells, and the strength of cell attachment (Fig. 1 A). Because cardiomyocytes contraction involves substantial cyclical modulation of cell morphology and adhesion, impedance technology can be applied for dynamic monitoring of cardiomyocytes contraction and beating, which is the ultimate functional manifestation of the heart (Fig. 1B).
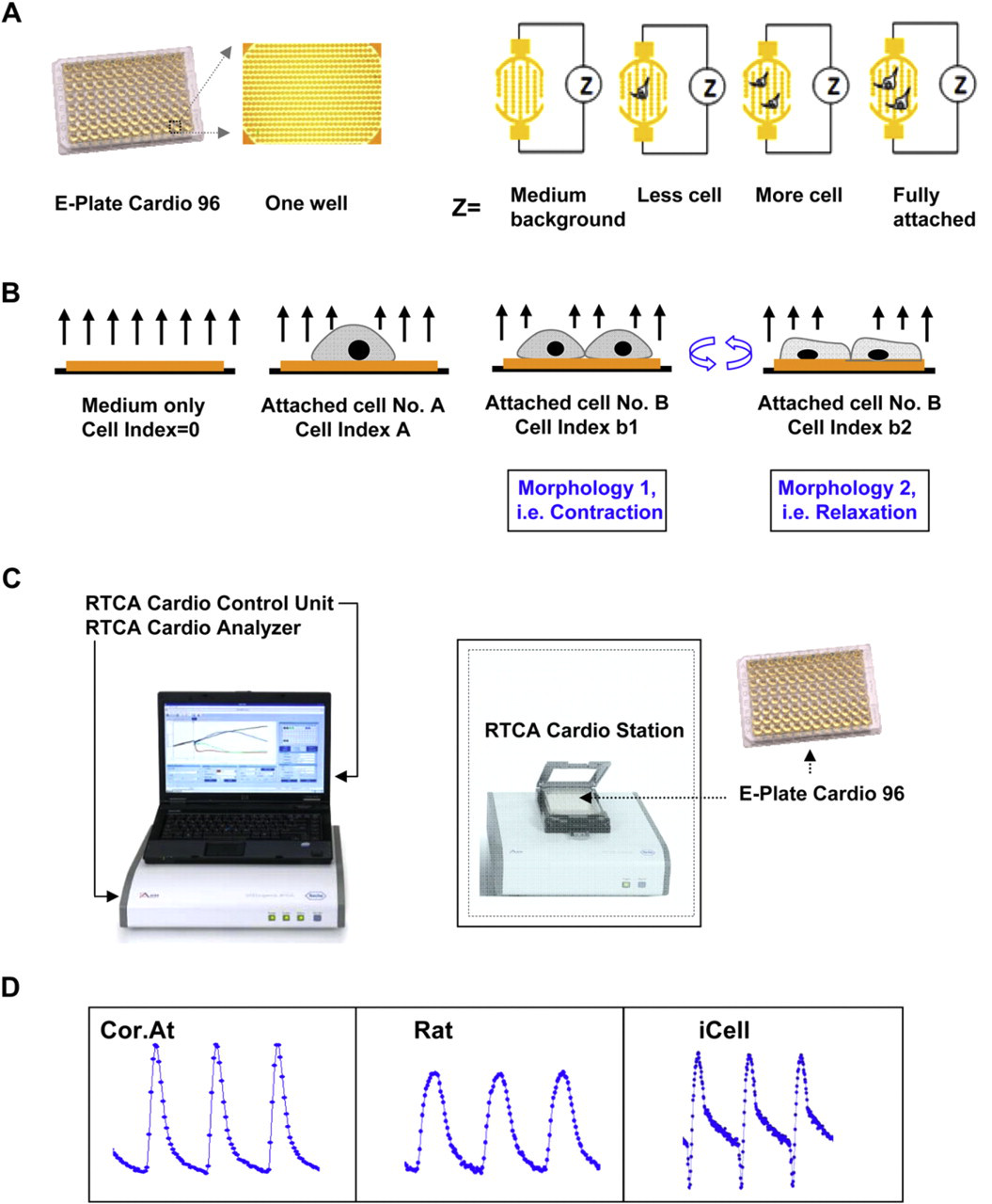
(A) Principle of impedance readout. The Cardio Plate 96 is a microtiter plate, which is integrated with gold microelectrodes in the bottom of each well. The impedance signal is generated by the application of low-voltage signal, which creates a current between the electrodes. The interaction of cells with the electrodes impedes the current and generates the impedance signal (Z value), which is proportional to the number of cells inside the well and the morphologic and adhesive characteristics of the cells. The impedance signal is displayed as an arbitrary unit referred to as Cell Index, which is a ratio of the change in impedance to background impedance. (B) The detection of cardiomyocytes beating signal is based on the rhythmic changes of cell attachment and morphology due to contraction and relaxation of cardiomyocytes, which modulates the impedance signal accordingly. Due to fast data acquisition rate of the xCELLigence RTCA Cardio System, the beating of cardiomyocytes can be recorded and resolved with high temporal resolution. (C) The RTCA Cardio System is composed of four main components, which include the E-Plate Cardio 96 and plate station, which fit inside the CO2 incubator, the electronic analyzer, control unit, which includes a computer and a software. (D) Typical beating waveforms recorded from Cor.At, rat neonatal primary cardiomyocytes, and human iPS cardiomyocytes.
xCELLigence RTCA Cardio Instrument
The xCELLigence Real-Time Cell Analysis (RTCA) Cardio System is composed of four main components (Fig. 1C); the 96-well electronic microtiter plate (E-Plate Cardio 96); the RTCA Cardio Station, which accommodates the Cardio Plate and is placed inside a tissue culture incubator; the RTCA Control Unit, which operates the software and continuously acquires and displays the data; and the RTCA Analyzer, which sends and receives the electronic signals between the control unit and the station. The analyzer connects to the station through a ribbon cable that can pass through the sealed door of any standard tissue-culture incubator. The xCELLigence RTCA System measures impedance signal and then processes and displays the data by converting impedance value into a Cell Index (CI) value. 9,10 CI is an arbitrary unit and is a reflection of cell number, morphology, and attachment. The main feature of the xCELLigence RTCA Cardio System is the fast data acquisition rate, 12.9 ms for entire 96-well Cardio Plate, which allows high temporal resolution of cardiomyocytes beating cycles. Because impedance measurement is noninvasive, the millisecond data acquisition rate can be combined with longer-term monitoring to study both the short-term and long-term effect of compounds on cardiomyocytes.
Cardiomyocytes
One of the main challenges in preclinical cardio-safety assessment has been the lack of a predictive and biologically relevant model system available in sufficiently high quantities to be used for screening of cardiotoxic and pro-arrhythmic drugs, especially during the hit to lead or lead optimization stage. 11 Although, primary cardiomyocytes from human and rodent systems can be used, technical difficulties in obtaining sufficiently pure cardiomyocytes in high enough yield has been an obstacle to wider adoption. 12 The recent advances in stem cell technology and particularly in differentiating embryonic or induced pluripotent stems cells have created a unique opportunity for providing physiologically relevant and disease relevant model systems for preclinical safety assessment of compounds. 11 –15 In particular, the ability to use defined culture and media conditions for selective differentiation of desired lineages such as cardiomyocytes, coupled with the ability to genetically engineer stem cells for enrichment and selection of pure populations of differentiated phenotypes makes this a powerful approach for pharmacology and toxicity studies. 11 –15 We have tested the utility of xCELLigence RTCA Cardio System with primary rat neonatal cardiomyocytes, mouse embryonic stem cell-derived cardiomyocytes (Cor.At; Axiogenesis), and human-induced pluripotent stem cell-derived cardiomyocytes (iCells; Cellular Dynamics International). As shown in Figure 1D, the xCELLigence RTCA Cardio System can detect the beating activity of all three cardiomyocytes.
Work Flow
A typical work flow (Fig. 2 A) starts with coating of the surface of the Cardio Plate 96 with an extracellular matrix protein, such as fibronectin at 37 °C degree for 3 h. Stem cell-derived cardiomyocytes such as Cor.At cells or iCells, which are typically shipped frozen, are thawed, assessed for extent of viability, and then seeded at the density of 30,000–60,000 cell per well. Alternatively, primary rat neonatal cardiomyocytes can be freshly harvested according to standard protocols and seeded into the wells of the Cardio Plate 96 as discussed above. Immediately before culturing cardiomyocytes in Cardio Plate, fresh culture medium is added to Cardio Plates and the background impedance of the media is determined. Once cardiomyocytes are seeded the recording of beating activity in terms of interval between each recording and the duration of each recording is programmed into the RTCA Cardio software and therefore all recordings are performed automatically once the experiment is initiated. Depending on the cardiomyocytes and the seeding density, it can take from 1 to 5 days to observe consistent and reproducible beating profile. The observation of beating activity is determined by both physical and electrical coupling of cardiomyocytes in the bottom of the well, which would result in synchronous beating. The culture medium may need to be changed on daily basis. Figure 2B shows the overall impedance signal and beating activity of Cor.At cells as measured by the xCELLigence RTCA Cardio System. Compound treatment is initiated once the beating activity of cardiomyocytes, measured by beating rate and amplitude, becomes stable with low coefficient of variation across the wells in the Cardio Plate. Although compound treatment procedure can be determined and designed based on the users' specific requirements, we typically start with baseline recording of beating activity for 10–30 min before compound addition. Subsequent to compound addition, the beating activity is recorded at preprogrammed schedule typically about 24 h. Longer treatment times can also be performed; however, it is worth noting that the recommended procedure for culturing cardiomyocytes may require media change every 24 h. Figure 3 A shows the typical beating profiles of Cor.At cells in response to treatment of compounds with well-defined mechanisms. Treatment of Cor.At cells with tetrodotoxin, a voltage-gated sodium-channel inhibitor results in slowed beating rate. Isradapine, a voltage-gated calcium-channel inhibitor leads to a significant effect on both beating rate and amplitude. The hERG channel inhibitor, E4031 results in inconsistent beating profile or waveform. As a matter of fact, all hERG channel inhibitors results in a similar profile (Y Abassi, JBP in press). Therefore, the beating profile can be predictive of mechanism of compound toxicity. The well-known inotropic and chronotropic agent, isoproterenol, results in increased beating rate of Cor.At cells within short time of treatment. Importantly, the xCELLigence RTCA Cardio System allows for both the time- and dose-dependent measurements of compound activity on cardiomycyte beating as shown for isoproterenol in Figure 3B.
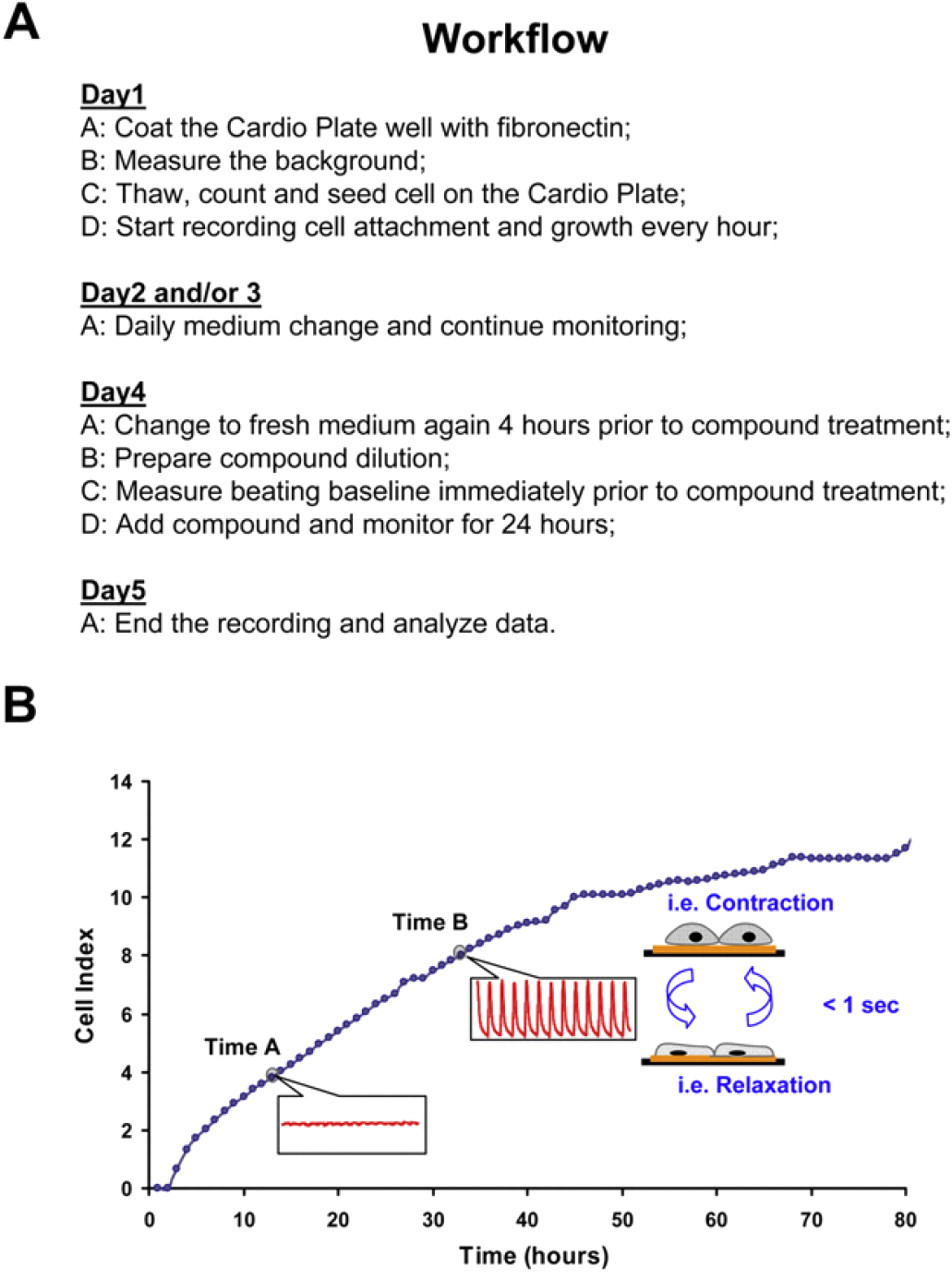
(A) Typical work flow for culturing, growing, and recording beating of cardiomyocytes using the xCELLigence RTCA Cardio System. (B) Dynamic monitoring of attachment, growth, and beating activity of mouse embryonic stem cell-derived cardiomyocytes (Cor.At) using the xCELLigence RTCA Cardio System. In the example at the given time A and B, the Cardio System measures impedance of Cor.At cardiomyocytes and transforms the values to Cell Index (CI) values, which reflect changes in beating activity (shown in red). To obtain the overall CI at time A and B (darkened gray circles), which reflects the cell number, attachment, and beating activity, the average CI values for each recording period (20 s for both time points of A and B) is displayed.
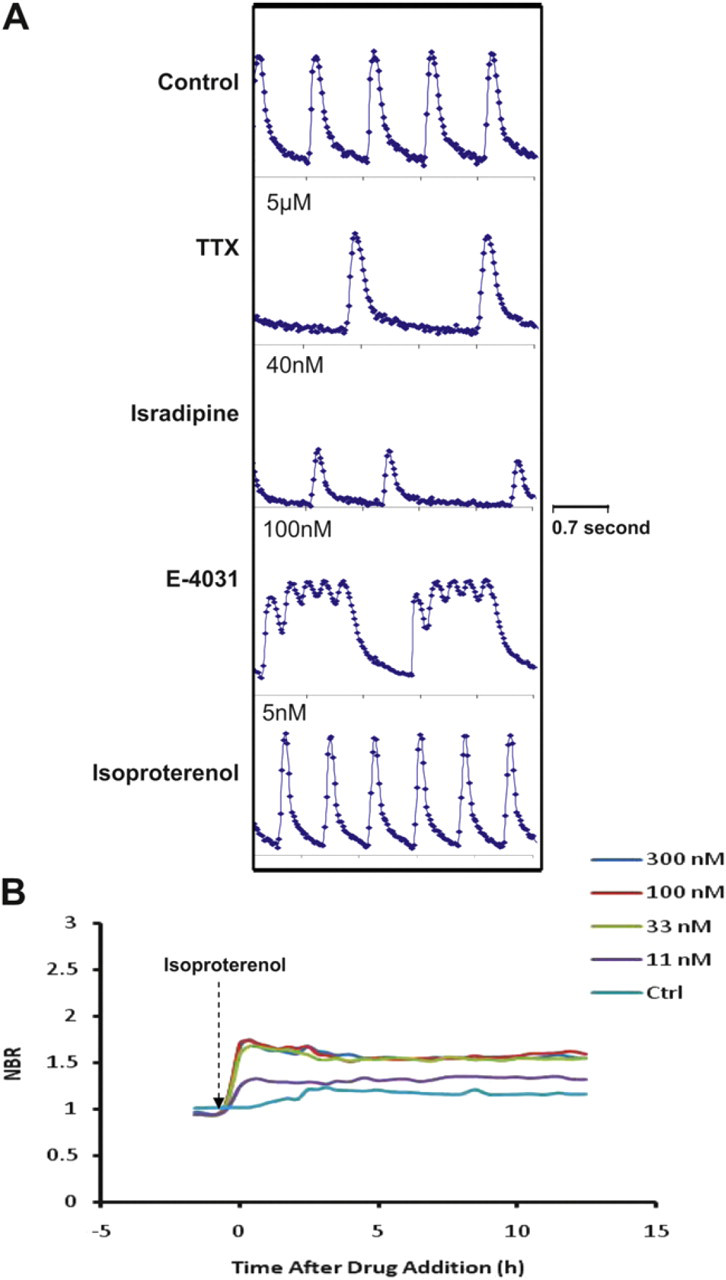
(A) Modulation of Cor.At beating activity by ion channel modulators and G protein-coupled receptor agonists. Tetrodotoxin is a voltage-gated sodium channel inhibitor; isradipine is a voltage-gated calcium channel inhibitor; E4031 is a potent hERG channel inhibitor; and isoproterenol is a β2-adrenergic receptor agonist and ionotropic/chronotropic agent. Data recorded for about 3.5 s for each treatment. (B) Dynamic monitoring of normalized beating rate (NBR) of Cor.At cells in response to increasing doses of isoproterenol.
System Software and Data Processing
The xCELLigence RTCA Cardio System software encompasses features for the operations, data acquisition and processing and analysis functions. The software allows for real-time recording and display of beating activity across the entire 96 wells of the Cardio Plate (Fig. 4 A). The analysis function of the software resides within the “Data Analysis” tab and contains a variety of algorithms designed to retrieve important and relevant information from the waveform of the beating profile. Figure 4B and Table 1 show the definition of main parameters. In theory, each measured beating cycle corresponds to one excitation–contraction coupling of the cardiomyocytes. The beating profiles are composed of a sequence of positive peaks (+P) and negative peaks (–P). The CI value difference between one negative peak to the following positive peak is defined as amplitude. The time between each positive peak is defined as beating period and the beating rate is calculated based on each beating period to derive how many beatings occurred in 1 min. Three time-related parameters, rise time T r, falling time T f, and half-amplitude widthT IBD50, resolve the temporal beating characteristic as illustrated. For data analysis, the related parameters are calculated for every beating within one recording period and the average and standard deviation are derived correspondingly. To compare the effect of tested compounds, beating rate or amplitude after compound treatment are normalized to the same time point before compound treatment to obtain the normalized beating rate or normalized amplitude. To evaluate the degree of arrhythmia, the beating rhythm irregularity index is derived based on the coefficient of variation (i.e., standard deviation divided by average) of the beating period during one record period. Another index, beating pattern similarity, is the overall beating waveform comparison indicative score includes the change of amplitude as well.
Brief description of the main parameters for data analysis provided in the RTCA Cardio software

Note: N/A means not available.
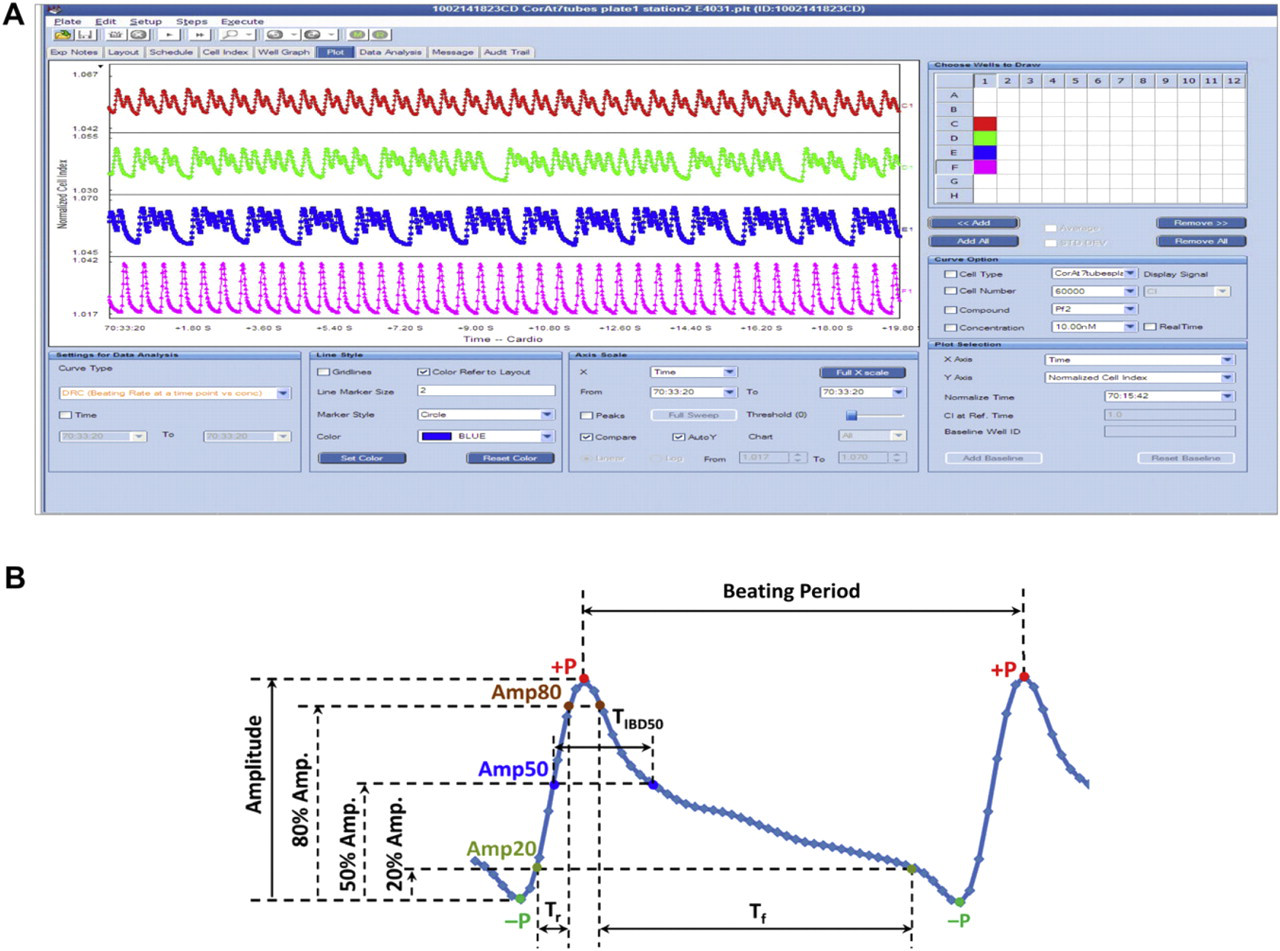
(A) A snapshot of the display screen of the RTCA Cardio software. The beating activity of cardiomyocytes for the entire 96 wells can be displayed in real time. (B) Analysis and dissection of cardiomyocytes waveform. The software can automatically identify and quantify parameters such as amplitude, beating rate, rising time (T r), falling time (T f), and beat duration. These parameters can be used to generate dose–response curves for compounds.
Summary
The xCELLigence RTCA Cardio System is a new platform for monitoring the beating function of cardiomyocytes. The results presented in this article demonstrate that impedance-based readout applied to dynamic monitoring of cardiomyocytes beating provides a relatively high throughput and convenient way to screen for potential toxicities of compounds, which may impact heart function. Combined with potentially unlimited supply of physiologically relevant stem cell-derived cardiomyocytes, this technology would allow for screening of cardiotoxic liability of compounds much earlier in the drug discovery process. The system is able to sensitively and quantitatively detect the effect of ion channel and nonion channel modulators of cardiac function in real time. Furthermore, pro-arrhythmic compounds produce a characteristic beating profile, which maybe reflective of the risk of arrhythmia. In addition, the ability to use the time resolution in the assay in conjunction with the physical beating of cardiomyocytes, the effect of certain compounds missed by electrophysiological readouts can be detected. Overall, taking into consideration the sensitivity, predictivity, real-time data acquisition, measurement of periodicity of beating over both short and prolonged window of time, and throughput makes this technology well suited for early preclinical safety assessment of cardiotoxic compounds.
Footnotes
Acknowledgments
Competing Interests Statement: The authors certify that they have no relevant financial interests in this article and that any/all financial and material support for this research and work are clearly identified in the Acknowledgments section of this article.
