Abstract
The storage of screening collections in DMSO is commonplace in the pharmaceutical industry. To ensure a high-quality screening collection, and hence effective and efficient high-throughput screening, all compounds entering the GlaxoSmithKline (GSK) screening collection undergo a liquid chromatography–mass spectrometry (LC-MS) quality control (QC). It is generally accepted that even under optimal conditions, a small percentage of these compounds are unstable after prolonged storage in DMSO. This article presents how these QC data can be mined using a data-driven clustering algorithm to identify chemical substructures likely to cause degradation in DMSO. This knowledge provides new structural filters for use in excluding compounds with these undesirable substructures from the collection. This information also suggests an efficient, targeted approach to compound collection clean-up initiatives. Stability studies are also designed to maintain a high-quality screening collection. To define the best practice for the storage and handling of solution samples, GSK has undertaken stability experiments for two decades, initially to support the implementation of new automated liquid stores and, subsequently, to enhance storage and use of compounds in solution through an understanding of compound degradation under storage and assay conditions.
Introduction
DMSO is a well-known solvent that is commonly used in the laboratory. DMSO is a polar, aprotic solvent with low volatility that easily dissolves many substances that have poor solubility in water. The storing of screening collection compounds in DMSO solution is commonplace and facilitates the dispensing of small amounts of large numbers of compounds for high-throughput screening (HTS). The success of biological screening depends on the quality of corporate collections, which can be affected by compounds that have degraded because of their storage conditions. Throughout the past two decades, much research has been put into finding the best handling procedures and storage conditions for screening collections.1,2,3 This work has shown that some compounds are unstable after prolonged storage in DMSO, even when the solutions are frozen at −20 °C. For ensuring and maintaining a high-quality screening collection, it is therefore important to be able to identify unstable compounds in DMSO and to validate conditions for storage and automation of compound supply for HTS. Being able to identify unstable compounds in DMSO would allow new structural filters to be constructed, to add to those already in use, to prevent the addition of labile compounds to the screening collection. It would also help to efficiently target future collection clean-up efforts with a view to removing degraded compounds.
Published work covering chemical instability in DMSO has been limited,4–8 and attempts to build a predictive model have been only partially successful.9,10 This is not surprising, considering the range of chemistries involved. Degradation may occur via hydrolysis, oxidation, decarboxylation, or substitution reactions. Decomposition is a relatively rare event under optimized storage conditions. This means that, without prior knowledge, data would have to be collected on a very large number of compounds for a meaningful model to be constructed. Factors other than the structure of the parent compound can influence the rate of decomposition. For example, minor impurities may catalyze the reaction, or the salt form may be influential. 11 These observations led us to consider a pragmatic approach based on data mining. Since the creation of the GlaxoSmithKline (GSK) liquid store and its subsequent automation more than 20 years ago, millions of compounds have been acquired. As well as using a set of structural filters to exclude problem compounds, including classes known to be unstable, GSK uses a liquid chromatography–mass spectrometry (LC-MS)-based quality control (QC) process for all purchased compounds entering its HTS collection. The correct identity (molecular weight) and purity [≥80% by ultraviolet (UV)] are the inclusion requirements. 12 Even from the more reliable suppliers, a small percentage (2–5%) of compounds fail on purity. QC purity failure can occur for several reasons, such as poor purification, contamination during handling, and so on. It was our hypothesis, however, that these failing compounds were likely to contain a higher proportion of DMSO-unstable moieties. The multiple reasons for QC purity failure mean that the dataset is likely to be noisy. Hence, data-mining techniques are ideally suited for its analysis. In this article, it is shown how a data-driven algorithm has been successfully used to mine QC data to identify chemical substructures that are likely to render compounds unstable after long-term storage in DMSO.
Since the introduction of automated liquid stores approximately 20 years ago, GSK has undertaken stability experiments to validate conditions for storage and automation of compound supply for HTS and to understand compound degradation under storage conditions. The concept design focused on long-term storage at −20 °C in sealed 96 deep-well blocks. A long-term stability study was initiated in January 2000 on 309 structurally diverse compounds stored in the liquid store with flexibility to select time points based on the emerging patterns and stability data produced. All these compounds had initially passed QC. The aim was to establish how long the integrity of compounds could be maintained at −20 °C in sealed blocks. These compounds were analyzed throughout a 6-year timeframe (LC-MS and water content), and the results were published in 2009. 13 Less than 10% of the compounds had degraded throughout that time period. Degradation was determined using the UV purity difference between the time-zero purity and the purity values at the subsequent time points. Compounds showing a purity difference of ≥10% were defined as having degraded, because this value considers any measurement variation within the analysis. Degradation of individual compounds was not linear throughout time, with the greatest degradation occurring in the early months. Those compounds that showed degradation in the early months also showed significant degradation after prolonged storage. Encouragingly, 89% of compounds were still suitable for entry into the screening collection (≥80% pure and correct) after 6 years compared with 100% at time zero. These results predicted an acceptable level of degradation when extending the storage lifetime beyond 6 years, and extrapolation of the data corroborated this (12% degradation after 10 years). This article presents QC data for this set of 309 compounds, having extended the storage time to 20 years (QC time points at 9, 12, 16, and 20 years).
Materials and Methods
Long-Term Stability Study
A list of 309 compounds was selected based on having >50 mg available from the solid-compound store and randomly selected based on structural diversity. A solid amount of 40 µmol (±10%) was solvated with DMSO (4 mL) to give a concentration of 10 mM. The 10 mM dissolved samples (2 mL in duplicate) were transferred to a 96 deep-well plate and sealed with a Beckman Coulter foil seal (Beckman Coulter, Brea, CA). To monitor their long-term stability, the plates were stored at −20 °C in the liquid store and analyzed at various time points. LC-MS analysis was used to measure the degradation of the compounds throughout time, taking the measurements in duplicate. For the LC-MS experiments, 50 µL of 10 mM DMSO solution was diluted to 150 µL in a 96-well microplate to give a concentration of 3.33 mM. The LC-MS system comprised an Agilent 1100 high-performance liquid chromatography (HPLC) system (Agilent, Santa Clara, CA) fitted with a Supelcosil ABZ+ 3 µm column (3.3 cm × 4.6 mm; Sigma-Aldrich, St. Louis, MO) held at 25 °C, an Agilent 1100 binary LC pump, an Agilent 1100 autosampler (1 µL injection volume), and an Agilent 1100 UV diode array detector (DAD) scanning between 210 and 350 nm at 2 nm intervals. For the 20-year time point, an Agilent 1200 HPLC system was used, keeping all conditions the same. The mobile phase was A: 0.1% aqueous formic acid + 10 mM ammonium acetate, and B: 90% acetonitrile + 0.07% formic acid + 10 mM ammonium acetate. The run time was 9 min using gradient elution (100% A to 0% A). A Waters ZQ mass spectrometer (Waters, Milford, MA) was used as the mass detector. The instrument was set up using both positive and negative electrospray ionization, a capillary temperature of 100 °C, and a capillary voltage of 3.2 kV. Full-scan mode was used for detecting ions between 100 and 1000 Da at a scan rate of 0.5 s. The system was controlled using MassLynx software (Waters). The LC-MS instrument was validated prior to each experiment using repeat injections of a system suitability test mix12,14 to check the reproducibility. During the experiments, system suitability samples were analyzed at the beginning and at the end of a batch of samples to ensure correct functioning of the instrument. The diode array peak area (sum of all wavelengths) was used to determine the purity with respect to other peaks present in the chromatogram. The water measurements were carried out via well-fluid acoustic impedance using the Echo 555 (Labcyte, Sunnyvale, CA). This is a fast, nondestructive technique and enabled the sample to be used for other measurements following the water analysis.
Identification of Unstable Chemical Moieties in DMSO
DMSO solutions (10 mM) were held in the liquid store at −20 °C. Each sample was in a 1.2 mL Abgene tube fitted with a pierceable cap (Thermo Fisher Scientific, Waltham, MA). DMSO solutions were dispensed into Greiner 384-well polypropylene plates (25 µL; Greiner Bio-One, Monroe, NC) prior to analysis. The LC-MS instrument used for the collection clean-up work was a Waters Acquity ultra-performance liquid chromatography (UPLC) system with an Acquity UPLC bridged ethylene hybrid (BEH) C18 1.7 µm column (2.1 × 50 mm) held at 40 °C, an Acquity DAD (210–350 nm scanning range), and a Waters single quadrupole detector (SQD) mass spectrometer (100–1000 m/z range, in both positive and negative ionization modes). The mobile phase was A: 100% water with 0.1% formic acid, and B: 100% acetonitrile with 0.1% formic acid. The run time was 2 min using gradient elution (97% A to 0% A), and the injection volume was 0.2 µL. The system was controlled using MassLynx software.
The data mining was performed using a data-driven clustering algorithm.15,16 Here, a set of ~950,000 compounds that had passed the QC process on purity was used as the stable set, and a set of ~35,000 compounds that had failed the QC process on purity was used as the unstable set. Because this dataset is binary in its response (i.e., stable or unstable), a binary-response version of the data-driven algorithm was used.
Results
Long-Term Stability Study
The analysis of the 309 compounds selected for the long-term stability study shows that after 20 years of storage at −20 °C in sealed plates, only 15% of the compounds have degraded (
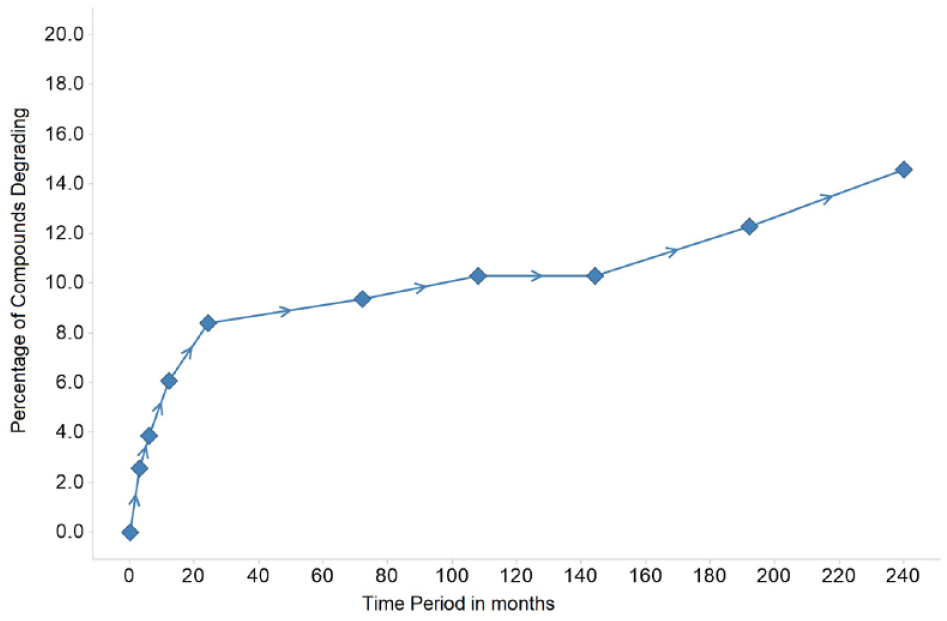
Percentage of compounds degrading throughout time in the 20-year stability study.
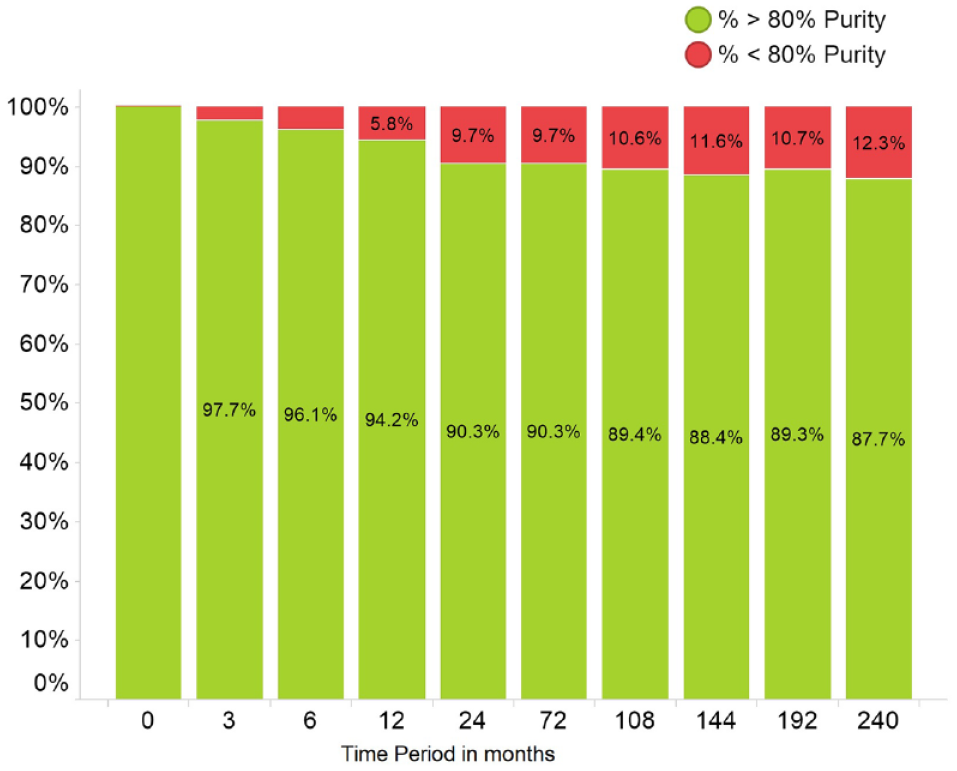
Purity versus time in the 20-year stability study.
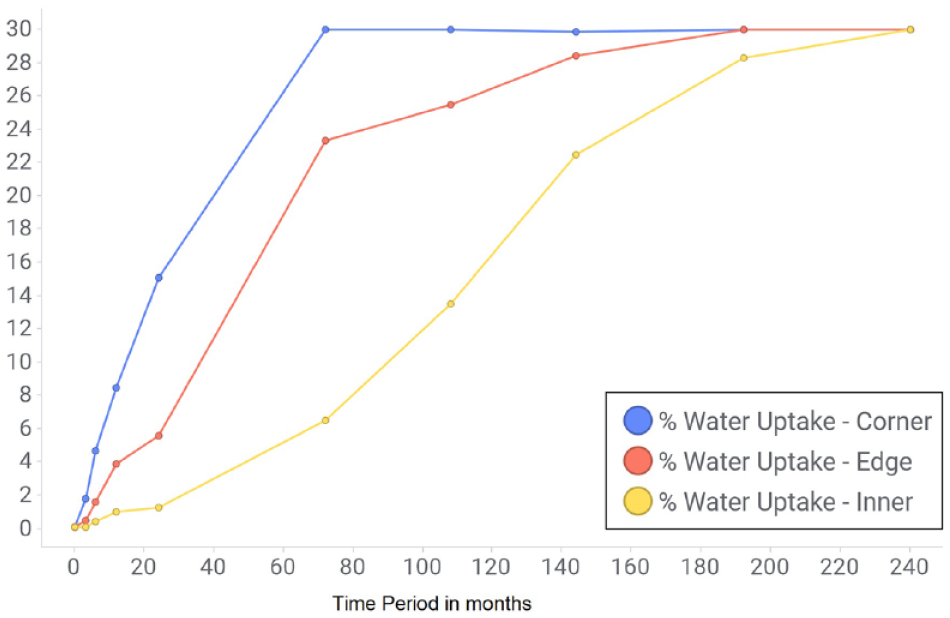
Water uptake versus time in the 20-year stability study.
Identification of Unstable Chemical Moieties in DMSO
Data-driven clustering was applied to the QC data for hundreds of thousands of compounds to identify sizeable structural clusters enriched in potentially unstable compounds, all sharing a common structural motif. A presumed stable set of ~950,000 compounds was added to a potentially unstable set of ~35,000 compounds. The stable set was drawn from those purchased compounds that had passed the QC process on purity and were thus in the GSK screening collection. The unstable set contained compounds that had failed QC on purity, some of which probably due to instability. When applied to our dataset, the data-mining algorithm returned ~1000 clusters, each represented by a different structural motif. In total, they contained ~50,000 compounds, ~17,000 of which were from the potentially unstable set. The next step was to check if the structural motifs contained compounds unstable after long-term storage in DMSO. The most appropriate way to do this was to re-analyze compounds containing these structural motifs that were in our screening collection. To interpret the results of the proposed re-analysis exercise, it was first necessary to define the underlying rate of instability within our screening collection. The results of the re-analysis by LC-MS of ~1000 compounds randomly selected from the screening collection suggested a rate of instability of approximately 3.6% (i.e., 3.6% of the samples were ≥10% less pure than when first analyzed and hence categorized as degrading).
It was not feasible to check all the clusters returned by the algorithm because the number was so large. The algorithm output ranked the clusters in an order in which the first cluster returned is deemed to be the most significant. Therefore, to reduce the numbers, it was decided to focus on the top 350 clusters. This number was further reduced by selecting only the motifs in which the compounds failing QC were acquired from more than one source. It was felt that this would reduce the possibility that the QC failures were due to an inadequate purification procedure or other handling issues specific to one supplier. A subset of 137 motifs was thus selected. To quickly evaluate a significant number of motifs, it was decided to re-analyze no more than 60 samples from each of the motifs selected. Further samples were then looked at only if, for a motif, more than 10% of the re-analyzed samples did not pass QC on purity or more data were needed to render the result statistically significant. Accordingly, 59 motifs were found to be unstable, and 37 motifs were classified as stable. A total of 41 motifs were not classified. This was because there were fewer than 30 compounds containing these motifs in the GSK screening collection that had both suitable time-zero data and re-analysis data. The chemical structures of the most unstable motifs are given in Figure 4 . Typically, the samples that had degraded had been in solution for several years, although in a few cases it was as little as 3 months. There were some compounds from the original stable set containing the identified unstable motifs, although the overlap was small, and they were found to degrade with time when retested. When viewed as a compound collection clean-up exercise, the re-analysis of compounds from the screening collection containing motifs classified as unstable yielded a failure rate of 27% (2578 out of 9647). This is between seven- and eightfold greater than that observed when compounds were randomly selected for re-analysis.
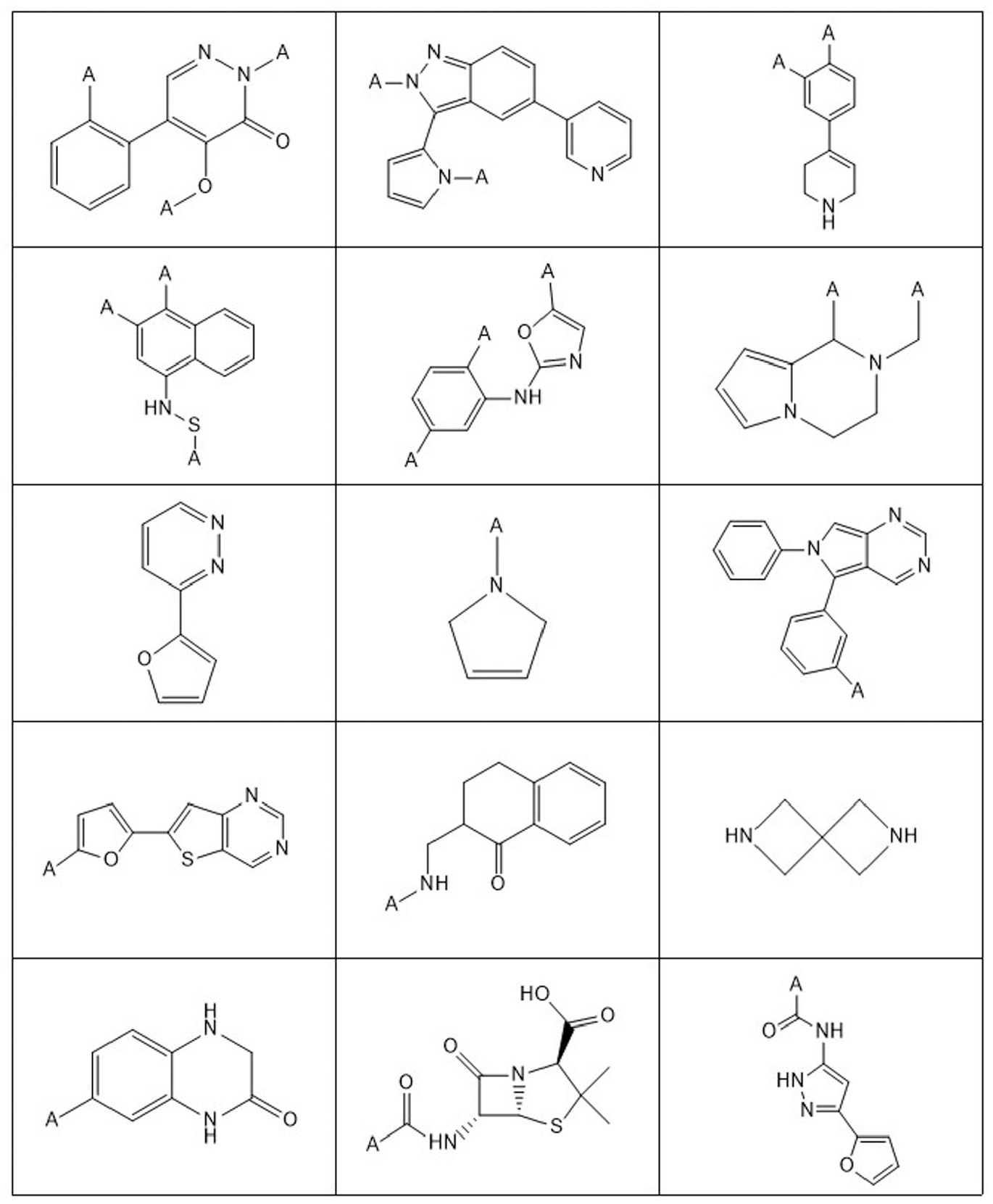
Chemical moieties that were identified as unstable in DMSO by applying a data-driven clustering algorithm to quality control (QC) data.
Discussion
It has been shown that applying a data-driven clustering algorithm to QC data is an effective way to rapidly identify many potentially unstable chemical substructures in a screening collection. Re-analysis of compounds containing these substructures then needs to be carried out. This serves to distinguish those substructures in which some of the initial samples were simply impure from those substructures that were really degrading. So far, >40% of the substructures identified by data mining and followed up in this way have proven to be genuine indicators of potentially unstable compounds. The substructure filters identified through this work are being used to avoid the purchase or synthesis of compounds for the screening collection. Therefore, prioritization of resources away from compounds that are potentially unstable in DMSO can be undertaken. Knowledge of these potentially labile substructures is also useful in the drug discovery process so that informed decisions can be made based on previous experience. When collection clean-up resources are limited, QC effort should be focused on samples containing substructures identified as unstable. This is between seven and eight times more efficient at identifying failing samples than selecting compounds at random for QC. This approach to the identification and removal of failing compounds provides a way of maintaining the overall quality of the screening collection.
The article published in 2009 contained QC results for the first 6 years of the long-term stability study.
13
The current article includes QC results taken at time points after 9, 12, 16, and 20 years, showing a linear increase in the percentage of compounds degrading during those years (
Footnotes
Acknowledgements
We thank Stefan Senger for his guidance on computational chemistry. Natalie Warren-Godkin was an industrial placement student at the time of the project.
We thank every member of the sample management, analytical chemistry, and computational chemistry departments in GSK, who throughout the years have contributed one way or another to this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
