Abstract
F. tularensis is a Gram-negative coccobacillus that causes tularemia. Its LPS has nominal biological activity. Currently, there is controversy regarding the structure of the lipid A obtained from F. tularensis live vaccine strain (LVS). Therefore, to resolve this controversy, the purification and structural identification of this LPS was crucial. To achieve this, LPS from F. tularensis LVS was acid hydrolyzed to obtain crude lipid A that was methylated and purified by HPLC and the fractions were analyzed by MALDI-TOF MS. The structure of the major lipid A species was composed of a glucosamine disaccharide backbone substituted with four fatty acyl groups and a phosphate (1-position) with a molecular mass of 1505. The major lipid A component contained 18:0[3–O(16:0)] in the distal subunit and two 18:0(3-OH) fatty acyl chains at the 2- or 3-positions of the reducing subunit. Additional variations in the lipid A species include: heterogeneity in fatty acyl groups, a phosphate or a phosphoryl galactosamine at the 1-position, and a hexose at the 4′ or 6′ position, some of which have not been previously described for F. tularensis LVS. This analysis revealed that lipid A from F. tularensis LVS is far more complex than originally believed.
Introduction
LPS consists of the O-antigen, the core and the lipid A (lipophilic region) components, and is present in the outer membrane of Gram-negative bacteria. 1 It can trigger macrophages to produce inflammatory cytokines such as TNF-α and IL-1β, and other mediators such as nitric oxide, which, when produced excessively, may lead to inflammation and the development of shock. The complete structure of the enterobacterial lipid A, initially determined by Qureshi et al., consists of a β-(1→6)-linked glucosamine disaccharide backbone to which six fatty acyl groups are attached in a precise manner at the 2-, 3- and 2′- and 3′-positions. The hexaacylated enterobacterial lipid A contains two phosphates attached at the 1- and 4′-positions of the glucosamine disaccharide, which is responsible for stimulating the signaling cascade leading to inflammation, sepsis and septic shock. 1 – 6
F. tularensis is an intracellular Gram-negative bacterium that causes tularemia in humans.7,8 Our studies have focused on the live vaccine strain (LVS) subspecies that causes a similar disease in mice, but is attenuated in humans. The highly purified F. tularensis LVS LPS was previously found to be minimally stimulatory in primary murine macrophages and in HEK293T cells transiently transfected with TLR4/MD-2/CD14; however, the live F. tularensis LVS bacteria are highly stimulatory in murine macrophages and TLR2-expressing HEK293T cells. Several different structures have been proposed for F. tularensis lipid A. 9 – 13 The presence of a phosphate-linked galactosamine at the 1-position and a hexose at the 4′ position of the glucosamine disaccharide has been observed in lipid A from F. tularensis subspecies holarctica and F. novicida,10,12,13 but it has not been reported for the F. tularensis LVS lipid A. The structure of the lipid A from F. tularensis LVS has not been completely characterized, therefore, there remains a controversy associated with the structure of the lipid A component of F. tularensis LVS. 11 – 13
The objective of the present study was to qualitatively evaluate the structural differences between the lipid A structures of F. tularensis LVS, F. tularensis subspecies holarctica and F. novicida. 9 – 13 Therefore, we performed the first HPLC purification of methylated (at the phosphate) lipid A and carried out structural determination of the highly purified lipid A obtained from the LPS of F. tularensis LVS via MALDI-TOF MS. We also purified the unmethylated free lipid A (not bound to the LPS and normally present in F. novicida) by TLC. Our data revealed that the methylated lipid A is amenable to purification by reversed-phase HPLC (RP-HPLC) and can be fully characterized with respect to phosphates and fatty acyl groups using MALDI-TOF MS. The TLC-purified free lipid A was also analyzed by MALDI-TOF MS. We found via liquid-liquid extraction that the F. tularensis LVS lipid A in the aqueous phase contains the phosphate and the phosphate-linked galactosamine, but lacks the hexose attached to the 4′-position. However, the phenol layer contains all three substituents. The presence of the phosphate-linked galactosamine attached to the lipid A has not been previously described for F. tularensis LVS.
Materials and methods
Materials
The following HPLC-grade solvents were used: acetonitrile, chloroform, methanol and isopropanol were purchased from Burdick & Jackson (Muskegon, MI, USA). Silica gel H thin-layer plates were purchased from Analabs Inc. (North Haven, CT, USA) Dowex 50W-X8, Chelex 100, and Cellex-D were obtained from Bio-Rad as previously described (Bio-rad, Hercules, CA, USA). 14
Growth of bacteria and preparation of LPS
F. tularensis LVS cells were cultivated by List Biological Laboratories (Campbell, CA, USA). 7 The LPS was extracted using the modified Westphal/Jann protocol and the aqueous fraction was treated with RNase, DNase and proteinase K, and centrifuged at 3076 g to remove solids. The supernatant was dialyzed against 0.85% (v/v) NaCl solution using a 100 kDa-cutoff AGT hollow fiber cartridge, lyophilized and reconstituted in water. The LPS was re-extracted using a modified deoxycholate-phenol extraction and treated with RNase, DNase and proteinase K. 7 Finally, the LPS was purified from the free lipid A and phospholipids via a chloroform/methanol (2:1, v/v) extraction, as previously described. 8 This highly purified LPS was dialyzed against water and suspended in pyrogen-free saline. The phenol fraction from the initial water-phenol extraction was processed similarly, with the exception of the chloroform/methanol extraction step.
Preparation of lipid A
Aqueous phase lipid A
The F. tularensis LVS LPS aqueous fraction (78 mg, Figure 1A, Lane 1) was suspended in 0.02 Thin-layer chromatography was performed on silica gel GHL plates with a solvent system composed of chloroform/methanol/water/ammonia (50:25:4:2, v/v/v/v). 1A illustrates the TLC analysis of the aqueous phase purified LPS of F. tularensis (20 µg, Lane 1, origin) and lipid A bound to LPS of F. tularensis (Lane 2). Band A (Lane 2) contains the unphosphorylated lipid A species and band B (Lane 2) represents the galactosamine phosphate lipid A species. 1B illustrates the TLC analysis of the F. tularensis live vaccine strain (LVS) LPS (Inzana group) prepared before and after the chloroform/methanol (2:1,v/v) extraction. Lane 1 represents the free lipid A species and LPS (origin) and Lane 2 is the purified LPS mixture. Band B is composed of the galactosamine phosphate lipid A species and was present predominantly in the aqueous layer. In contrast, Band C contains the phosphorylated lipid A species with both a galactosamine and hexose residue and was only present in the phenol layer.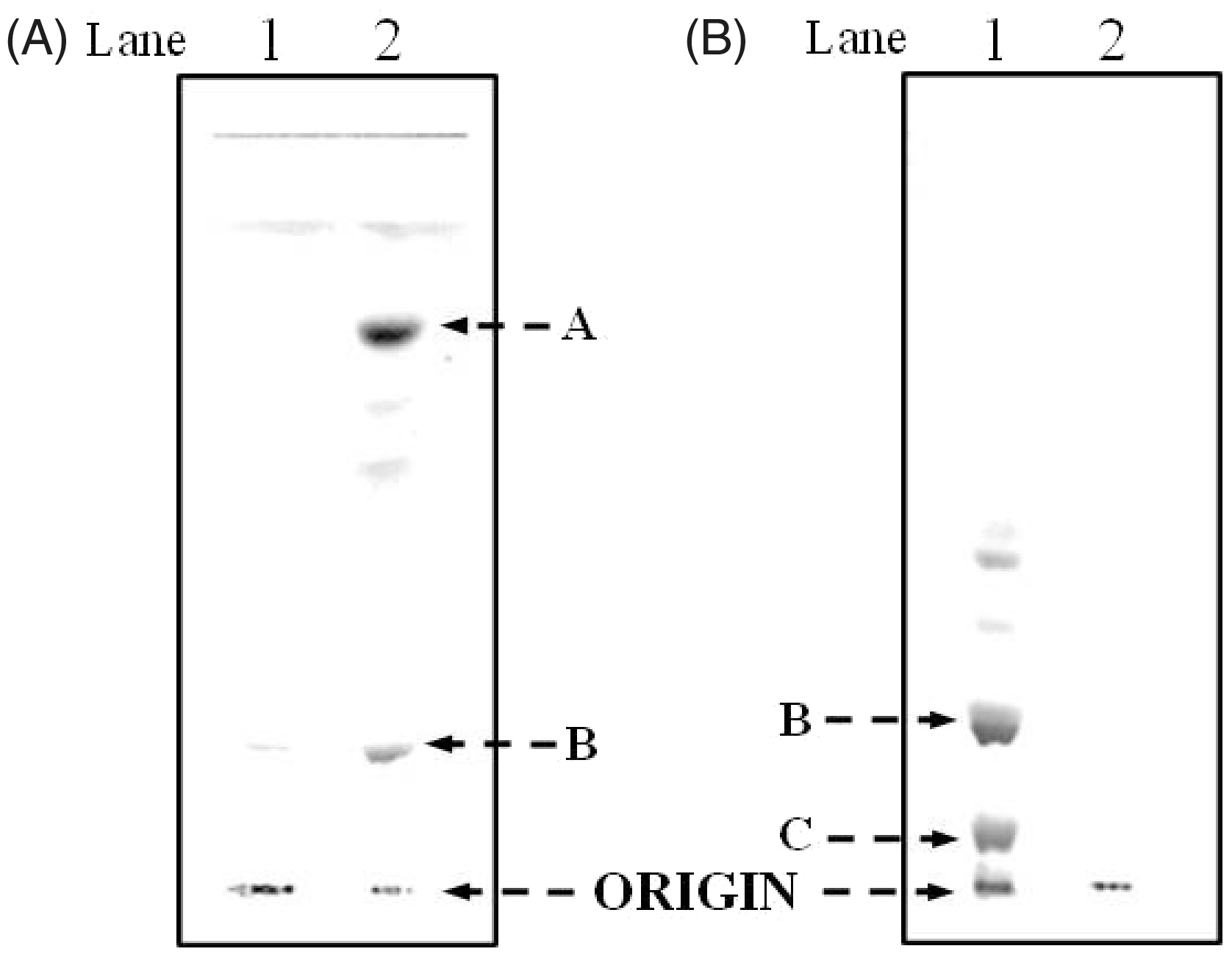
Phenol phase lipid A
The F. tularensis LVS LPS phenol phase was analyzed by MALDI-TOF MS without prior chloroform/methanol (2:1, v/v) extraction. The TLC analysis of the phenol phase showed the presence of the LPS and the phosphorylated lipid A species containing both galactosamine and hexose (TLC data not presented).
Preparation of F. tularensis live vaccine strain LPS
F. tularensis LVS (obtained from Center for Disease Control, Atlanta, GA, USA) LPS was prepared by the Inzana group, according to the method of Vinogradov et al. 9 ; however, instead of separating the water/phenol layers during the LPS extraction, the two layers were extensively dialyzed with water to remove the phenol, as previously described. 17 The free lipid A, which is present in the F. novicida LPS and not the F. tularensis LVS LPS, was extracted from the crude LPS mixture via chloroform/methanol (2:1,v/v). Following removal of the free lipid A, the LPS (249 mg) was extracted twice with chloroform/methanol (2:1,v/v) and concentrated to dryness using a rotary evaporator. The free lipid A (30 mg; Figure 1B, Lane 1) was purified using the silica gel GHL plates developed with chloroform/methanol/water/ammonia (50:25:4:2,v/v/v/v). The bands (band B, Figure 1B) were identified using iodine vapors and the lipid A species were eluted from the band with a chloroform/methanol (2:1,v/v) mixture containing 4% water. 4 The eluted species were analyzed by MALDI-TOF MS.
Analytical procedures
Samples for the glucosamine assay were hydrolyzed in 3
MS of lipid A species
The mass spectra of the lipid A species were acquired in both the negative ion and positive ion linear mode using a MALDI-TOF mass spectrometer (AXIMA-CFR; Kratos, Manchester, UK), equipped with a 337-nm nitrogen laser, a 20-kV extraction voltage and time-delayed extraction. Before MALDI-TOF analysis, the samples were dissolved in chloroform-methanol solution (4:1, v/v) and deposited in a 1:1 ratio with a saturated matrix solution of 6-aza-2-thiothymine in 50% acetonitrile and 10% tribasic ammonium citrate (9:1, v/v) on the stainless steel MALDI target plate. The samples were dried at 25°C before mass analysis. Each spectrum represented the average of 100 laser shots and Escherichia coli lipid A (Sigma, St Louis, MO, USA) was used as the external calibrant.
Results
Characterization of the lipid A obtained from the LPS of F. tularensis live vaccine strain
The F. tularensis LVS lipid A from the aqueous phase layer was analyzed by silica gel GHL TLC as shown in Figure 1A. Lanes 1 and 2 show the migration of the purified F. tularensis LPS (remained at the origin) and lipid A, respectively. Band A (Lane 2) corresponds to the tetraacyl lipid A without any phosphates and the slower moving bands represent lipid A species with differing types of fatty acyl chains. The more polar band B, close to the origin, corresponds to the tetraacyl monophosphoryl lipid A with a galactosamine. After methylation, the lipid A species from band B migrated close to the solvent front, thus suggesting the presence of a phosphoryl group attached to the lipid A (data not presented). The lipid A was methylated with diazomethane, as described previously
3
to confirm the presence of a phosphate group, obtain optimal fractionation on a reversed-phase column and enhance fragmentation for MALDI-TOF MS analysis. The resulting methylated lipid A was then fractionated into three major fractions by RP-HPLC (Figure 2). The major and minor lipid A species, which vary in the number and type of fatty acyl chains, from the HPLC fractions are described in detail in Tables 1–3 (for details see below). It is essential to methylate the phosphate, otherwise the lipid A will be retained on the RP column.
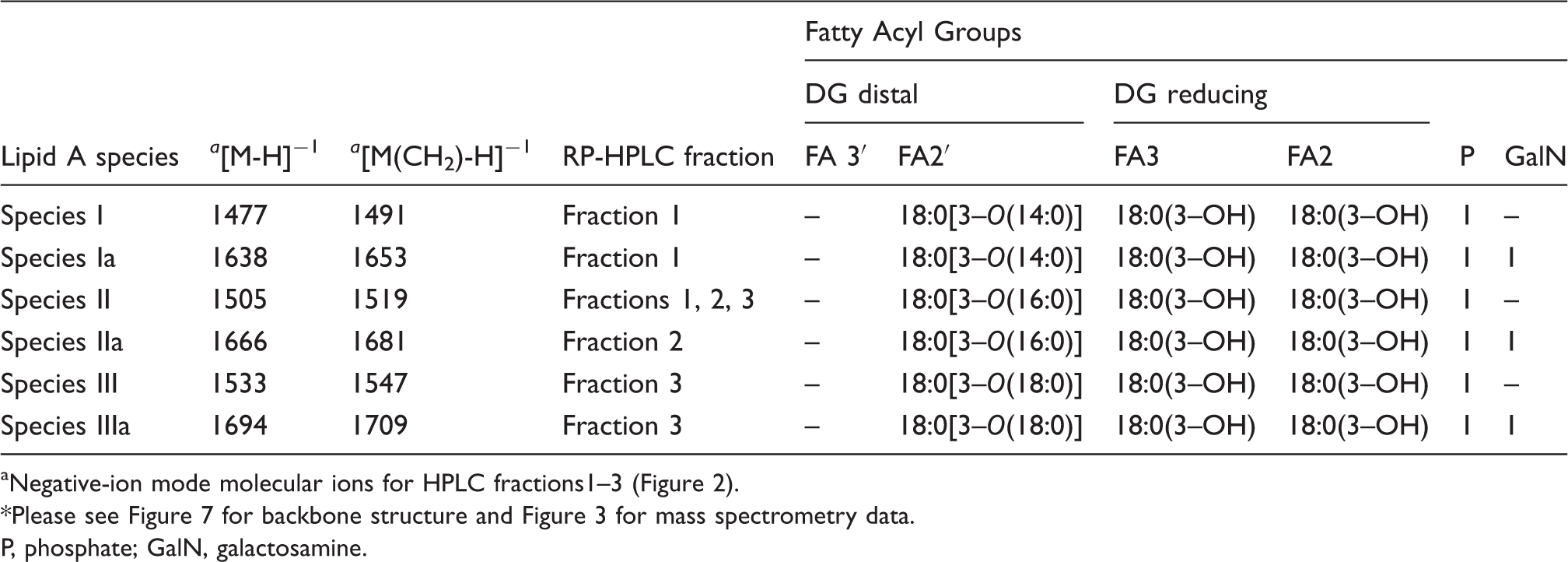
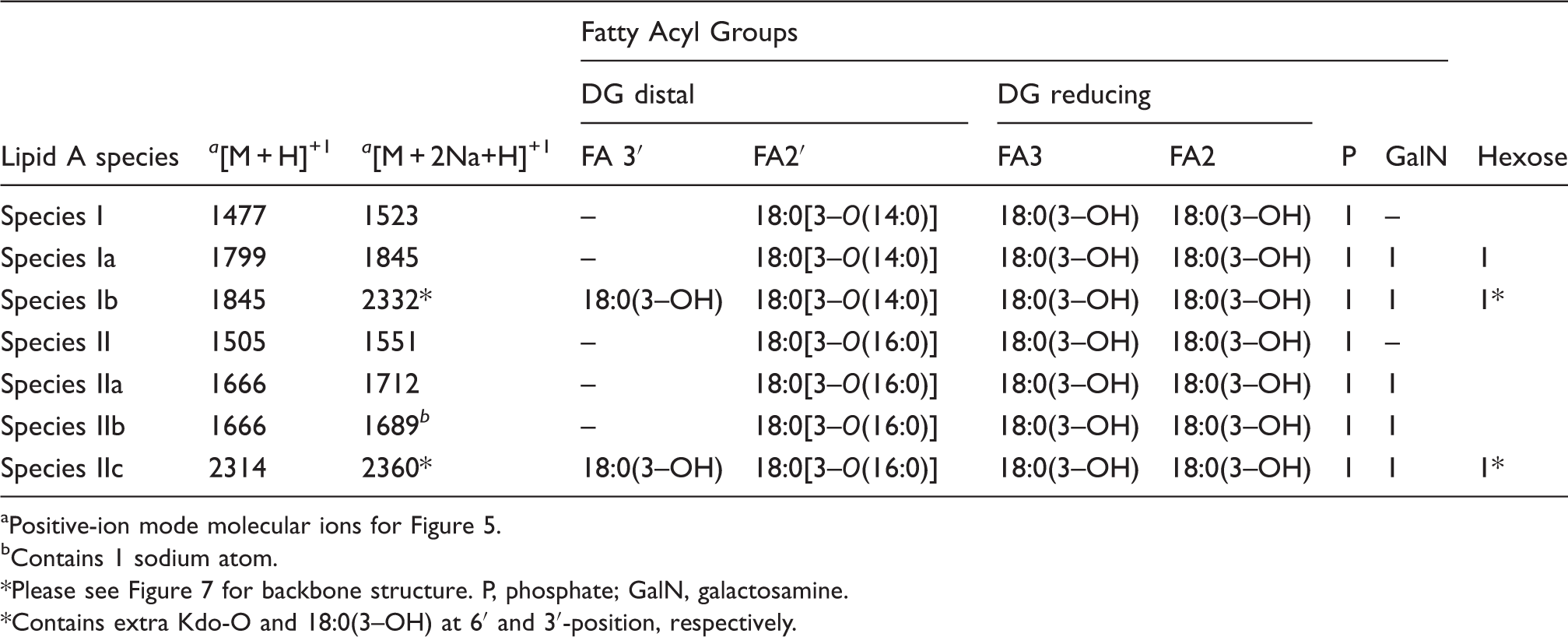
Reversed-phase-HPLC analysis of the methylated lipid A from F. tularensis live vaccine strain (LVS). Proposed fatty acyl distribution of methyl-lipid A species of HPLC fractions 1–3 aNegative-ion mode molecular ions for HPLC fractions1–3 (Figure 2). *Please see Figure 7 for backbone structure and Figure 3 for mass spectrometry data. P, phosphate; GalN, galactosamine. Proposed fatty acyl distribution of methyl-lipid A species of HPLC fractions 1–3 Positive-ion mode molecular ions for HPLC fractions 1–3 (Figure 2). Proposed fatty acyl distribution for lipid A species of LPS and free lipid A present in the phenol phase; or in the chloroform/methanol washes of aqueous plus phenol phase LPS (Inzana Prep) Positive-ion mode molecular ions for Figure 5. bContains 1 sodium atom. Please see Figure 7 for backbone structure. P, phosphate; GalN, galactosamine. *Contains extra Kdo-O and 18:0(3–OH) at 6′ and 3′-position, respectively.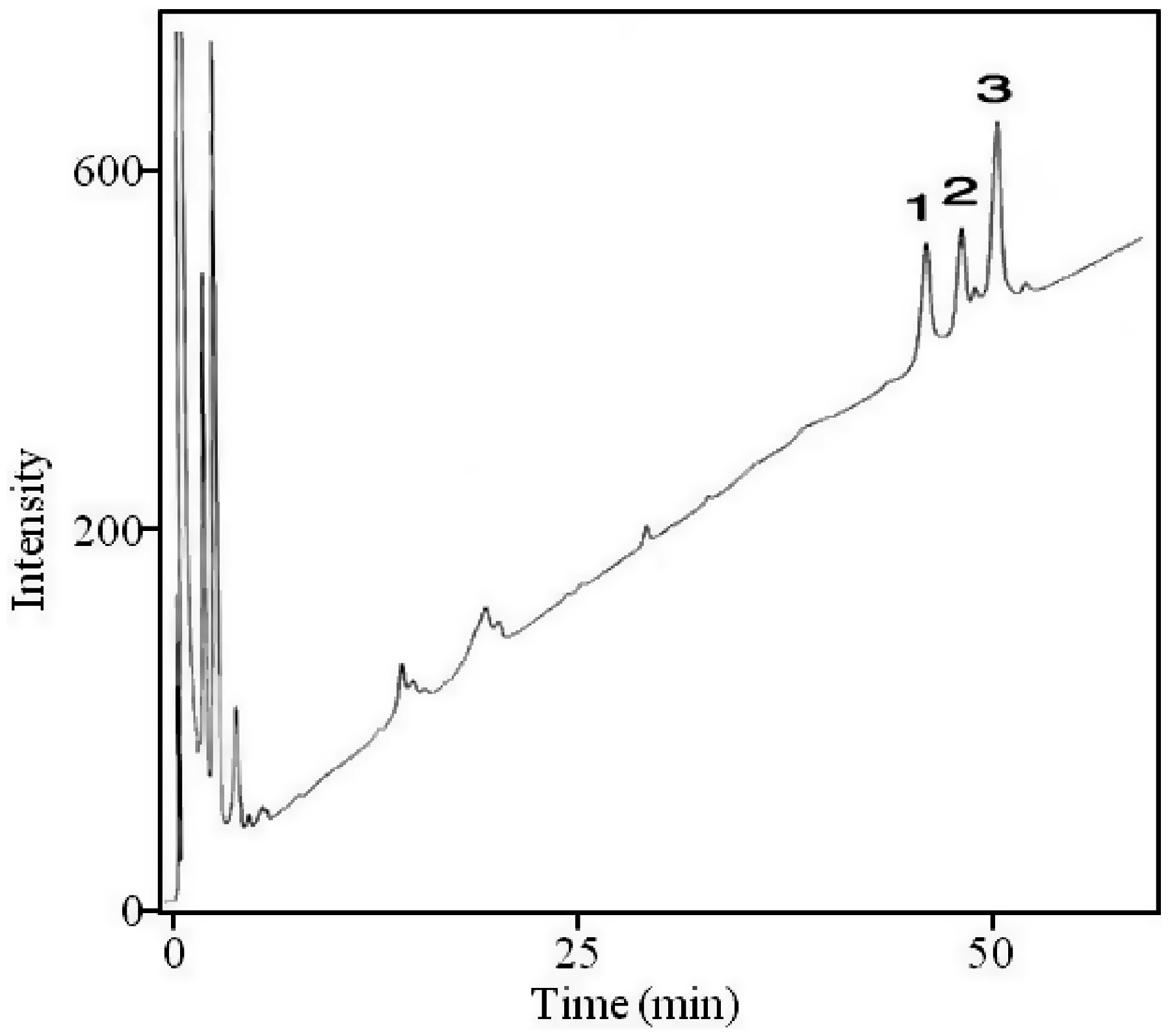

MS of the HPLC-purified methylated lipid A obtained from F. tularensis live vaccine strain LPS
Methylation of a phosphate in the free acid form leads to dimethylation of the lipid A as described by Qureshi et al.
2
However, if a sugar is attached to the phosphate, then it is monomethylated. The methyl groups can also be removed during mass spectrometric analysis. The HPLC fractions 1–3 were analyzed via MALDI-TOF MS in the negative ion mode. The mass spectrum of the lipid A HPLC Fractions 1–3 (Figure 3, A–C) contained an abundant molecular ion [M-H]−1 at m/z 1519 and 1533, which is consistent with the mono- and dimethylated tetraacyl phosphorylated lipid A Species II (Table 1) with 18:0[3–O(16:0)] in 2′-position on the distal subunit. The molecular ion observed at m/z 1491 (1519-28) in fraction 1 (Figure 3A) contains 18:0[3–O(14:0)] at the 2′-position on the distal subunit and the phosphate is mono-methylated, creating a mass shift of 14 Da (Species I, Table 1). The cluster of peaks averaged out at m/z 1653 (1491 + 162) in fraction 1. A peak of similar intensity was also observed at m/z 1652 (1491 + 161). We realize that the problems with mass accuracy cannot distinguish between galactose and galactosamine, but as the presence of galactose has never been reported in any Francisella lipid A, Species Ia therefore represents the addition of the phosphate-galactosamine at the 1-position (Table 1, Figure 3A), based on the structures proposed earlier for F. novida and F. tularensis subspecies holarctica.10,12 However, this peak has not been previously observed for lipid A from F. tularensis LVS. Similarly, the ion observed in Fraction 2 at m/z 1681 (1519 + 162) represents the mono-methylated form of Species IIa with the addition of phosphate-galactosamine at the 1′-position (Table 1, Figure 3B). Similar to the m/z 1653 ion, this lipid A species has not been previously reported for the LVS strain. The ion observed in Fraction 3 at m/z 1709 (1547 + 162) represents the mono-methylated form of Species IIIa with the addition of phosphate-galactosamine (Table 1, Figure 3C). No unphosphorylated lipid A species were identified by negative MALDI-TOF mass spectrometry.
Negative-MALDI-TOF MS spectra of HPLC fractions 1–3 (A–C).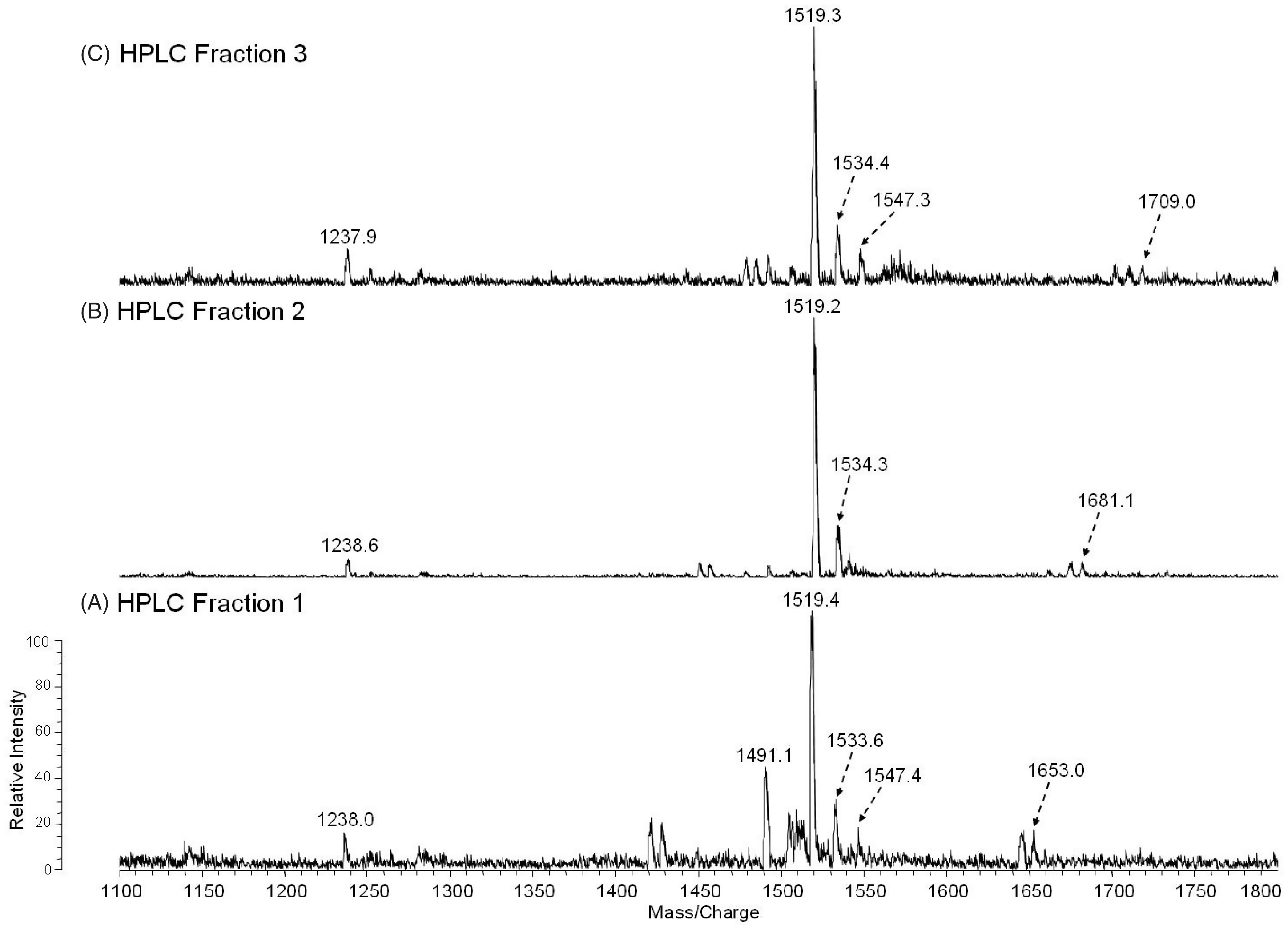
To investigate the positions and types of fatty acyl groups attached to the lipid A species, we analyzed the HPLC fractions 1–3 by positive MALDI-TOF MS (Figure 4, A–C, Table 2). The mass spectrum of the fraction 1 in Figure 4A contained an abundant molecular ion [MH + Na]+1 at m/z 1390, that is consistent with the mono-methylated lipid A containing 18:0[3–O(14:0)] at the 2′-position on the distal subunit and 16:0[3-OH] at the 2-position at the reducing subunit (Species IV, Table 2). To further confirm the position of the fatty acyl chains, the oxonium ion of the non-methylated Species IV, with a molecular mass of 1369, was observed at m/z 653 (Table 2). This ion resulted from the cleavage of the distal glucosamine from the reducing glucosamine at the oxygen bond. The most abundant ion in the fraction 2 mass spectrum at m/z 1418 is consistent with the mono-methylated lipid A containing 18:0[3–O(14:0)] at the 2′-position in the distal end and 18:0[3-OH] at the 2-position at the reducing end (Species I, Table 2). The oxonium ion observed at m/z 653 again suggests the presence of the 18:0[3–O(14:0)] in the distal subunit of the glucosamine disaccharide. The most abundant ions observed in the fraction 3 mass spectrum, m/z 1446 and 1474, are consistent with the mono-methylated form of the lipid A species with molecular mass of 1425 and 1453, respectively (Figure 4C). The oxonium ions observed at m/z 681 and 709 suggest the presence of a 18:0[3–O(16:0)] on the distal subunit of lipid A Species II and 18:0[3–O(18:0)] on the distal subunit of lipid A Species III, respectively (Figure 4C, Table 2). The species containing the phosphates are not detectable in the positive ion mode due to the presence of the net negative charge. Overall, the mass spectral data are consistent with the proposed structures of the lipid A HPLC fractions 1–3 as summarized in Tables 1–2.
Positive-MALDI-TOF MS spectra of HPLC fractions 1–3 (A–C).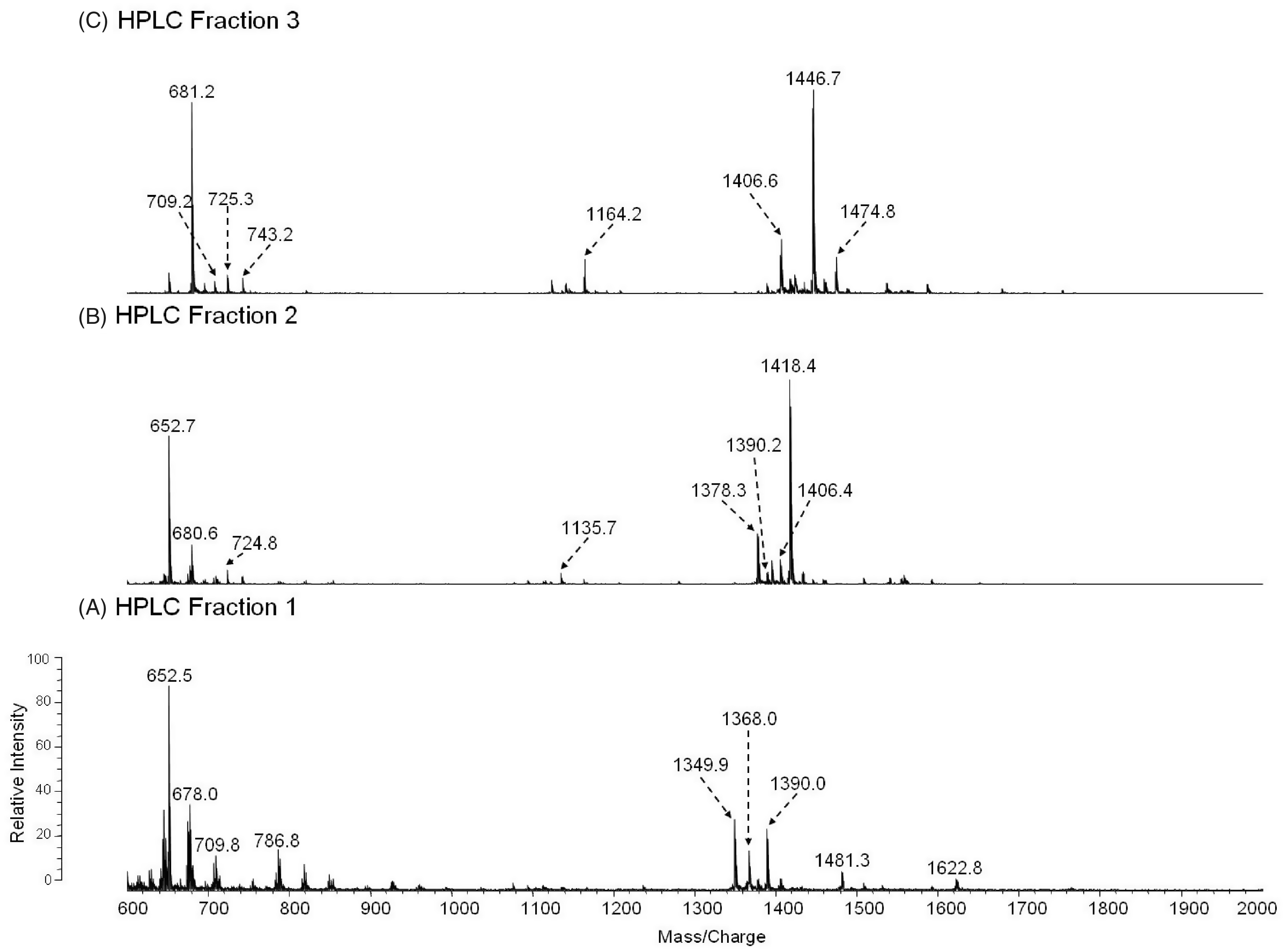
MS of the TLC-purified non-methylated lipid A obtained from F. tularensis live vaccine strain (Center for Disease Control) LPS
The major fractions of free lipid A were extracted from crude LPS with chloroform/methanol (2:1,v/v) and purified by TLC without acid hydrolysis or methylation to minimize any loss of fatty acyl groups. The positive MALDI-TOF MS analysis of the TLC band B (Figure 1B, Lane 2) and the TLC band above band B (band not observed in Figure 1B) is illustrated in Figure 5A and B, respectively. The molecular ion observed in Figure 5A at m/z 1712 ([MH + 2Na]+1) is representative of the tetraacyl lipid A with the addition of galactosamine attached to the phosphate (Species IIa, Table 3). The peak observed at m/z 1551 was as a result of a loss of galactosamine (Species II, Table 3). The MALDI-TOF MS analysis of the TLC band above band B (Figure 1B, Lane 2) showed a major peak at m/z 1551 that contained the tetraacyl phosphorylated lipid A observed in Figure 5A with the absence of galactosamine (Figure 5B). These data revealed the presence of a phosphate and galactosamine in the F. tularensis LVS lipid A structure for the first time. In conclusion, the LVS lipid A is similar to that of other Francisella lipid A species. The LVS lipid A definitely contains a phosphate (that could be methylated) and a galactosamine, but the substitution with these components may be partial as compared to that observed in the lipid A species of other F. tularensis subspecies or in F. novicida.
12
Moreover, we did not observe hexose in the purified lipid A fractions obtained from the aqueous phase layer. However, in the MS analysis of the phenol phase LPS (List Biologicals and preparation by the Inzana group), a peak was observed at m/z 1846 ([MH + 2Na]+1), which represents Species Ia of Table 3 (Figure 6). Lipid A Species Ia contains a hexose at the 4′ or 6′-position, which is consistent with previous MS analysis of lipid A from F. novicida.
12
Another ion observed at m/z 2333 corresponds to Species Ib (Table 3) and is composed of a pentacyl LPS containing Kdo minus O (–16) and an 18:0[3–OH], which has not been previously described for the LVS strain (Figure 6). Ions observed at m/z 1551 (Species II) and m/z 1523 (Species I) result from the addition of two sodium atoms during MS analysis to m/z 1505 and m/z 1477, respectively (Table 3, Figure 6). The oxonium ion peak at m/z 681 was not observed; however, a peak at m/z 887 (681 + 161 + 2Na) was observed in the phenol phase layer, which is consistent with a hexose attached to the lipid A species. This would then suggest that during the initial aqueous/phenol extraction of the crude LPS, the purified LPS and free lipid A are present in both phases. Therefore, the absence of the various lipid A species in previous analysis could possibly be due to the fact that various investigators used different phases for analysis.
Positive-MALDI-TOF MS spectra of free lipid A extracted from crude LPS of F. tularensis from the TLC band B (Figure 1B, Lane 1), which was fractionated into two fractions 1 (A) and 2 (B). Positive-MALDI-TOF MS spectrum of LPS obtained from the phenol phase (List Biologicals) before the chloroform/methanol wash.
Discussion
The lipid A of F. tularensis LVS has not been fully characterized because of difficulties associated with purifying the free lipid A species. Such issues include solubility and the absence of the 1′-phosphate, which is removed during the hydrolysis of the free lipid A from LPS. In the present study, we present the structural determination of F. tularensis LVS lipid A that is different from the lipid A structures biosynthesized by Enterobacteriaceae, Rhodobacter, Chlamydia, Brucella, Aquiflex, Neisseria, and Caulobacter with respect to the number of phosphates, fatty acyl chain length, and the presence of a galactosamine phosphate at the 1′-position.2,15,20– 26 Our initial objective was to study the lipid A bound to LPS. Therefore, we utilized a chloroform/methanol (2:1,v/v) extraction to purify LPS from the aqueous phase. This purification was necessary to remove free lipid A (not bound to LPS) because of the large amount of free lipid A present in the crude LPS mixtures, as reported previously for F. novicida lipid A. 12 As a result of the presence of phosphate on a portion of the lipid A species, the mixture was methylated following hydrolysis of the purified LPS to enhance resolution and recovery from the RP-HPLC column. Fatty acid analysis after acid hydrolysis coupled with MS analysis of the fatty acids revealed the presence of predominantly 18:0[3–OH], 14:0, 16:0 and 18:0 fatty acyl chains. The backbone also contained two glucosamine residues that are normally present in the lipid A of Enterbacteriaceae, Neisseria gonorrhoeae and R. sphaeroides.2,4,20,15 The MALDI-TOF MS analysis of the purified HPLC fractions (fraction 1–3) revealed that we were able to purify and analyze the F. tularensis lipid A species, despite problems associated with purification of the lipid A species.
From the current analysis and previous nuclear magnetic resonance (NMR) data
12
, we can describe the structure of the F. tularensis LVS lipid A, as shown in Figure 7. The predominant lipid A species of F. tularensis has a diglucosamine backbone containing four fatty acids with a molecular mass of 1425 Da. The major component contains 18:0[3–O(16:0)] at the 2′-position on the distal subunit and two 18:0(3–OH) at the 2- and 3-positions on the reducing subunit. The 16:0 fatty acyl in the acyloxyacyl linkage can be substituted by either 14:0 or 18:0. Additional tetraacyl lipid A species containing either a phosphate or galactosamine phosphate attached at the 1-position were also observed, as shown in Figure 3 and Tables 2–3. The phosphate-linked galactosamine has not been previously observed in the lipid A of F. tularensis LVS. In contrast to previous studies, we did not observe high concentrations of the lipid A species containing a hexose attached at the 4′-position in the RP-HPLC fractions.
12
Proposed structure of the lipid A derived from the LPS of F. tularensis. The locations of the fatty acyl groups attached to the backbone are shown in Tables 1 and 2. Some of the lipid A species lacked the galactosamine phosphate moiety at the 1-position and heterogeneity was observed in the fatty acyl groups regarding the chain lengths. The presence of a hexose was observed only from the phenol phase LPS extract and this hexose was present at the 6′ position as reported earlier (12). An additional 18:0(3-OH) at the 3′-position was also observed.
Moreover, use of mild acid hydrolysis to remove lipid A bound to LPS also removes a portion of the phosphate and galactosamine, which increased the difficulty in structural determination of the lipid A species. Therefore, to circumvent this problem, we used the free lipid A present in large concentrations in the crude LPS preparations. 12 Mass spectrometry analysis of the crude LPS showed only one major peak at m/z 1505; however, we obtained more detailed mass spectral data from the TLC fractions. The TLC fractions also showed the presence of both galactosamine and phosphate attached to the free lipid A of F. tularensis LVS (Figure 5 and 7). Our results are consistent with those obtained by Vinogradov et al. via NMR analysis in the initial characterization of the F. tularensis LVS lipid A structure. 9 However, the lipid A structure determined from previous studies on F. tularensis LVS did not contain a phosphate attached to galactosamine. Moreover, further purification of the extracted LPS was not conducted in previous studies, which enhanced the complexity of the lipid A species.
The first structural analysis of F. tularensis LVS lipid A was carried out by Vinogradov et al. 9 They showed a partial MALDI-TOF mass spectrum of the molecular ions and suggested that the major lipid A structure was composed of the diglucosamine backbone substituted with four fatty acyl groups. 9 Nuclear magnetic resonance spectroscopy was utilized to determine the position of the fatty acyl groups, which consisted of one 18:0[3–O(14)] in the distal subunit and two 16:0 (3–OH) and 18:0(3–OH) fatty acyl chains at either the 2- or 3-positions of the reducing subunit. 9 However, they did not identify phosphate or sugars at the reducing end. In contrast, Phillips et al. reported only one lipid A for F. tularensis LVS that lacked a galactosamine or a hexose; however, the F. tularensis subspecies holarctica strain 1547–57 lipid A contained a phosphate-linked galactosamine at the 1-position. 10 The major lipid A structure was comprised of the glucosamine disaccharide backbone substituted with four fatty acyl groups. Wang et al. and Gunn and Ernst characterized the F. novicida lipid A structure, which showed the presence of galactosamine attached to the phosphate at the 1-position and a hexose at the 4′ position; however, the F. tularensis LVS lacked both the phosphate and galactosamine. 11 – 13
Analysis of our current data revealed that the lipid A extracted from the F. tularensis LVS LPS contains either no phosphate, one phosphate at the 1-position of the glucosamine disaccharide or a 1-phospho-galactosamine. The extracted free lipid A contained both the 1-phosphate and the 1-phospho-galactosamine. Analyses by NMR spectroscopy of the free lipid A from F. tularensis has already been carried out by Vinogradov et al. 9 and Wang et al. 12 The LPS and the free lipid A obtained from the phenol phase contained lipid A species with one phosphate and a galactosamine phosphate, both at the 1-position, a glucose attached to either the 4′ or 6′-position 12 and in some cases an extra 18:0(3-OH) was also observed. There were a few variations in the fatty acyl groups observed, for example 16:0(3-OH) instead of 18:0(3-OH), but these species were present in small amounts. These structures are consistent with those described earlier for the various strains of F. tularensis 9 – 13 ; however, the galactosamine phosphate and glucose substituents reported previously 12 have not been observed in the lipid A obtained from the F. tularensis LVS strain previously.
In summary, multiple species of both LPS-bound and free lipid A were observed in the F. tularensis LVS aqueous and phenol phase preparations. Further analysis of the purified LPS and lipid A revealed that the species was biologically inactive as far as cytokines are concerned. 27 Interestingly, F. tularensis LVS LPS induces antigen-specific B-1a antibodies that provide long term protection against F. tularensis LVS challenge. 28 The diminished activity of intact F. tularensis LPS may potentially be a consequence of the unusual lipid A structure or a combined effect of the absence of the 4′-phosphate and the presence of galactosamine, fewer fatty acyl groups or the longer fatty acyl groups attached to the lipid A component of LPS.
Footnotes
Acknowledgements
This research was supported by NIH grants GM50870 (NQ) and NIH NIAID Mid-Atlantic Regional Center of Excellence grant U54 AI-15571168 (NQ, SNV, TJI), and grants to RJC. We thank David Morrison and Robert Ernst for helpful suggestions with the manuscript.
