Abstract
Membrane proteins are involved in different physiological functions and are the target of pharmaceutical and abuse drugs. Xenopus laevis oocytes provide a powerful heterologous expression system for functional studies of these proteins. Typical experiments investigate transport using electrophysiology and radiolabeled uptake. A two-electrode voltage clamp is suitable only for electrogenic proteins, and uptake measurements require the existence of radiolabeled substrates and adequate laboratory facilities.
Recently, Dictyostelium discoideum Nramp1 and NrampB were characterized using multidisciplinary approaches. NrampB showed no measurable electrogenic activity, and it was investigated in Xenopus oocytes by acquiring confocal images of the quenching of injected fluorophore calcein.
This method is adequate to measure the variation in emitted fluorescence, and thus transporter activity indirectly, but requires long experimental procedures to collect statistically consistent data. Considering that optimal expression of heterologous proteins lasts for 48–72 h, a slow acquiring process requires the use of more than one batch of oocytes to complete the experiments. Here, a novel approach to measure substrate uptake is reported. Upon injection of a fluorophore, oocytes were incubated with the substrate and the transport activity measured, evaluating fluorescence quenching in a microplate reader. The technique permits the testing of tens of oocytes in different experimental conditions simultaneously, and thus the collection of significant statistical data for each batch, saving time and animals.
The method was tested with different metal transporters (SLC11), DMT1, DdNramp1, and DdNrampB, and verified with the peptide transporter PepT1 (SLC15). Comparison with traditional methods (uptake, two-electrode voltage clamp) and with quenching images acquired by fluorescence microscopy confirmed its efficacy.
Introduction
Regulated transport across the membrane is fundamental for cell physiological functions. Membrane proteins represent the largest class of drug targets, but many mechanisms of their activity are still unknown.1–4 The aim of membrane transporter research is to understand the relation between structure and function and to investigate the steps of their transport cycle to identify the substrates and more selective and specific drugs. Extensive studies have analyzed transporter activity by using different methodologies, among them heterologous expression in cell lines or in Xenopus laevis oocytes. These cells are a well-known tool for functional expression. They are particularly useful to increase the number of proteins in their membrane, usually low in tissues of origin, for studying their activity. The Xenopus oocytes are cells of large size (>1 mm diameter), easy to microinject, and very suitable for heterologous expression of transport proteins.5,6 Oocytes can be easily collected and maintained in a low-cost medium in vitro, and they are able to express and accurately reconstruct complex membrane protein. In fact, in the presence of the appropriate eukaryotic promoters, functional expression of proteins from different organisms, from plants to humans, has been achieved in Xenopus oocytes, and they have been widely used in single-cell studies7,8 as well as in high-throughput screening applications. 9 Moreover, the heterologous proteins are expressed in an isolated system because their amount is usually higher than any other endogenous protein, due to the peculiar state of the full-grown oocytes. 10
All these characteristics and their high translational capacity offer an optimal system for functional characterization of solute carriers (SLCs), that is, membrane proteins mediating the transport of a large variety of charged and uncharged organic molecules or inorganic ions. 11
Typical experiments investigate transporter function in Xenopus oocytes using electrophysiology and radiolabeled uptake. A two-electrode voltage clamp is, however, suitable only for electrogenic proteins, and uptake measurements require the existence of radiolabeled substrates and adequate laboratory facilities. In recent years, many institutions have chosen to support radioactive-free experiments because working with radiolabeled compounds, when available, has high costs, has high risks for the workers and the environment, and requires a specifically controlled and isolated working area. In the literature, there are only a few papers that suggest alternative approaches to study transporter proteins, such as the use of fluorescence probes in transporter assays for investigating DMT1 transport using Phen Green SK 12 or exploring the function of c-Nramp1, c-NrampB, and rDMT1 using calcein 13 and, similarly, membrane–metal nanoparticle interaction,14–16 or using fluorimetry and ion-sensitive microelectrodes. 17 In all these examples, the fluorophore quenching was studied with confocal or classical microscopy, one oocyte at a time, limiting the number of samples collected for each batch. To our knowledge, only in Kottra et al.’s 18 study was the transport of a fluorescent compound measured by microplate reader in many oocytes simultaneously, but in the case reported, a fundamental requirement was the capacity of the transporter to move large fluorescent molecules across the membrane. Recently, we tested the iron transporters DdNramp1 and DdNrampB (formerly Nramp2) from Dictyostelium discoideum,13,19 using a multidisciplinary approach. While DdNramp1 is electrogenic, DdNrampB did not elicit detectable inward current, and its activity was investigated in Xenopus laevis oocytes by acquiring confocal images of the injected fluorophore calcein, which undergoes quenching upon binding iron and few other transition metals. 13 Both DdNramp proteins belong to the Nramp (SLC11) family, which is fundamental for the homeostasis of divalent transition metal ions from prokaryotes to humans. 20
The role of Nramp1 in innate immunity is an ancient evolutionary function, as Nramp gene expression and function after infectious stimuli have been reported in many eukaryotic organisms: vertebrates, invertebrates, plants, and the amoeba Dictyostelium discoideum, an animal model to study professional phagocytes.19,21–24 The other protein of the mammalian SLC11 family is DMT1 (SLC11A2, Nramp2, DCT1); this transporter is ubiquitously expressed, mainly in duodenum, immature erythroid cells, brain, placenta, and kidney. 25 DMT1 plays a key role in iron absorption at the apical membrane of enterocytes. 26 Moreover, DMT1 is also involved in iron recycling from the hemoglobin destruction after macrophage phagocytosis of senescent erythrocytes. 25 The importance of DMT1 for iron metabolism appears evident since mutations impairing DMT1 function cause congenital microcytic hypochromic anemia and serum and hepatic overload in humans. 27 Functional studies in Xenopus laevis oocytes on rat DMT1 and yeast Smf1 and mammalian cells expressing DMT1 demonstrated a proton-dependent metal ion transport with a symporter mechanism.27–30
In the genome of Dictyostelium discoideum there are two Nramp genes, encoding Nramp1 and NrampB (formerly Nramp2).13,19 DdNramp1, like the mammalian ortholog, is expressed only in phagosomes and macropinosomes. DdNramp1 was shown to be a symporter for H+ and divalent metal ions out of the phagolysosome; this action removes iron and other metals fundamental for intracellular pathogens to activate their virulence genes.13,19,22 DdNrampB is localized as a membrane protein in the contractile vacuole, an organelle that regulates osmolarity and presumably acts as a divalent metal sink/store. The presence in different compartments of Nramp1 and NrampB suggests that both transporters jointly play a role in iron homeostasis. 19 DdNramp1 and DdNrampB differ strongly in their amino acid sequence, and in our previous functional study of Xenopus oocytes, 13 we collected many functional data about DdNramp1, but the absence of significant electrogenic activity limited the characterization of DdNrampB. Both Nramp proteins can be functionally expressed in Xenopus laevis oocyte plasma membranes if opportunely modified in their C- and N-terminal region, but due to the evolutionary distance between the social amoeba and the Xenopus laevis proteins, synthesis is partially limited by the different codon usage. 31 We also showed that assays based on confocal imaging of fluorophore calcein quenching can be applied to study metal ion transport even if the transporter is electroneutral. Our results on DdNramp1 demonstrated that it behaved similarly to mammalian DMT1, while the little functional information about DdNrampB confirmed an evolutionary divergence of the two Dictyostelium discoideum Nramp genes. 13
A drawback of the confocal imaging assay is that only single oocytes can be studied at a time; thus, the assay, while reliable, is time-consuming before a statistically significant number of oocytes are analyzed. In this paper, we have developed a new assay, based on calcein quenching but using a microplate reader, that allows the simultaneous analysis of several oocytes. By applying this new assay, we have collected new functional information about the DdNrampB transporter determining its transport activity, ion selectivity, and pH dependence using a new methodology.
Materials and Methods
c-NrampB Construction and cRNA Preparation
To achieve the expression and localization of NrampB and Nramp1 on the Xenopus laevis oocyte membrane, chimeric constructs were generated, as described in Buracco et al. 13 For cRNA transcription, the cDNA encoding c-NrampB, Nramp1, and DMT1 and the cDNA encoding PepT1-FLAG 32 transporter were linearized with NotI, and cRNA was synthesized in vitro in the presence of 10 mM Cap analog and 200 U of T7 RNA polymerase. 33 All enzymes were supplied by Promega (Milan, Italy).
Oocyte Culture, cRNA, and Fluorophore Injection
Oocytes were obtained from adult females of Xenopus laevis (Envigo, Bresso, Italy). The frogs were anesthetized in MS222 1 g/L (tricaine methansulfonate; Sigma, Milan, Italy) solution in tap water, and portions of the ovary were removed through an incision on the abdomen. The oocytes were treated with 0.5 mg/mL collagenase type IA (Sigma) in ND96 calcium-free solution for at least 30 min at 18 °C to remove follicular cells and debris. Collagenase activity was stopped with repeated wash steps in ND96 calcium-free solution and NDE (ND96 plus 2.5 mmol/L pyruvate and 0.05 mg/mL gentamycin sulfate). After 24 h at 18 °C in NDE, the healthy-looking oocytes were injected with cRNA diluted in sterile water (0.5 µg/µL), using a manual microinjection system (Drummond Scientific Company, Broomall, PA). The oocytes were then incubated at 18 °C for 3–4 days in NDE before electrophysiological or different transport studies. 33 The experiments were carried out according to institutional and national ethical guidelines (permit no. 1011/2015PR for Elena Bossi and 815/2016PR for Michela Castagna).
For monitoring the transport of metal or dipeptide in Xenopus laevis oocytes by fluorescence quenching, control oocytes and oocytes transfected with cRNA encoding for the specific transport protein were injected with 50 nL of fluorophore dissolved in intracellular solution.
Calcein (λex = 470 nm, λem = 510 nm; Sigma) was used for quantifying divalent metal ions and FITC-dextran 35 (35 KDa, λex492 = nm, λem518 = nm; Sigma) for quantifying the pH decrease as a consequence of PepT1 activity; fluorophores were injected at a concentration of 25 µM or 12.5 µM as indicated, according to the method used. The nominal volume of a 1.2 mm diameter oocyte is 1 µL; therefore, a 50-nL injected drop will be diluted 20 times. The expected equilibration time constant in the oocyte cytoplasm is a few hundred seconds. 6
Finally, to determine the K0.5 of calcein quenching by CoCl2, the oocytes were co-injected with calcein 12.5 μM and the concentration of CoCl2 was increased from 0.1 μM to 3 mM in intracellular solution at two different pH values, 5.5 and 7.6.
Electrophysiology and Data Analysis
Electrophysiological studies were performed using the two-electrode voltage clamp technique (Oocyte Clamp OC-725B; Warner Instruments, Hamden, CT). Intracellular glass microelectrodes were filled with 3 M KCl and had tip resistances between 0.5 and 4 MΩ. Agar bridges (3% agar in 3 M KCl) connected the bath electrodes to the experimental chamber. The recording was conducted at a fixed voltage, and the holding potential (Vh) values were −40 mV for SLC11 transporters (rDMT1, c-Nramp1, and c-NrampB) and −60 mV for SLC15 (PepT1). Representative traces of transport current were recorded perfusing the substrate at the indicated concentration; the mean of the transport-associated currents reported in the histograms was determined by subtracting the currents in the absence of divalent metal ion (external control solution) from the corresponding ones in its presence. Clampex 10.2 (Molecular Devices, Sunnyvale, CA) was used to control and record the experiments. Data were analyzed using Clampfit 10.2 (Molecular Devices).
Monitoring Transport by 55Fe2+ Uptake
Uptake experiments were performed 4 days after cRNA injection in Xenopus oocytes. Noninjected oocytes were considered as a control of transporter expression. The uptake solution contained 100 µM 55FeCl2, 100 mM NaCl, 1.8 mM KCl, 0.6 mM CaCl2, 0.6 mM MgCl2, and 0 mM MES at pH 6.5. Ascorbic acid (100 µM, freshly prepared) was added to maintain iron in the reduced form. Inhibition experiments were performed in the presence of 1 mM cold CoCl2. Groups of 8–10 oocytes were incubated for 60 min in uptake solution, washed in ice-cold uptake solution devoid of FeCl2, dissolved in 10% sodium dodecyl sulfate (SDS) solution, and counted in a liquid scintillation counter. 34 DMT1-, c-Nramp1-, and c-NrampB-induced uptake was calculated as the difference between the mean uptake measured in cRNA injected oocytes and the mean uptake measured in noninjected oocytes. For statistical analysis, the Student t test was applied.
Monitoring Transport by Fluorophore Quenching and Confocal Microscopy
From 10 to 30 min from the injection of 25 µM calcein or FITC-dextran, the oocytes were placed in Na solution at the indicated pH containing or not divalent metals at a final concentration of 0.1 mM or 10 mM Gly-Gln (GQ) and observed at the confocal microscope equipped with a 5× Plan Neofluar 0.15 objective. Every 30 s, for a total of 10–15 min, images of single oocytes were acquired, by using excitation at 488 nm and emission at 505–550 nm. For F/F0 quantification, the F0 fluorescence intensity at time 0, and F at the subsequent times, was calculated in the whole area of the oocytes using ImageJ (National Institutes of Health, Bethesda, MD). Changes in fluorescence intensity in the entire oocyte or in selected spots were proportionally linear with time, as previously reported. 12
Monitoring Transport by Fluorophore Quenching and Microplate Reader (XLOS)
To determine the concentrations of FeCl2 and CoCl2 needed to quench 50% of calcein fluorescence (K0.5), the fluorescence was measured in a group of 10–30 oocytes injected with 50 nL per oocyte of 12.5 µM calcein in the presence of increasing concentrations of CoCl2 (from 0.03 µM to 1 mM as the final cytoplasmatic concentration) in intracellular solution at two different pH values (5.5 and 7.6). After 15 min of incubation at room temperature in rotating plates, the oocytes were treated for Xenopus laevis oocyte smoothie (XLOS) methods. The method is summarized in Figure 1 . In brief, after washing three times in ND96, the oocytes were homogenized by pipetting up and down in the presence of 150 µL per oocyte of ND96 + 10% SDS, centrifuged for 5′ at maximal speed, and 100 µL of clear supernatant was transferred in a 96-well plate and the amount of fluorescence was measured on a Infinite 200 microplate reader (Tecan Group Ltd., Männedorf, Switzerland) at λ485 nm for excitation and λ535 nm for emission.
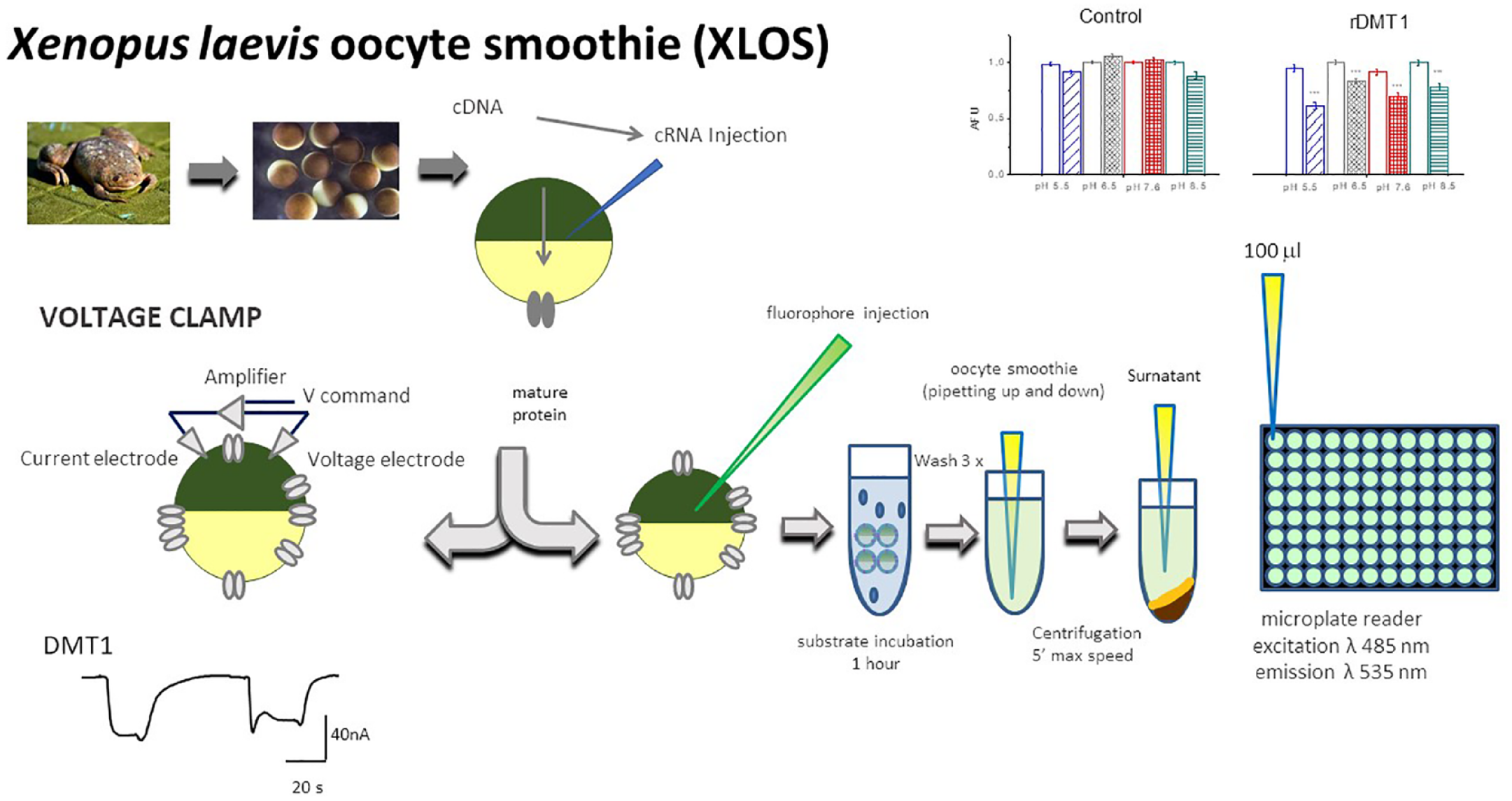
Schematic representation of XLOS technique. Steps required to measure the quenching of cytoplasmatic fluorescence on plate reading and the two electrode voltage clamp (TEVC). For the classical TEVC and XLOS, the oocytes were injected with mRNAs in vitro transcribed from the cDNAs coding for the proteins of interest. After 48–72 h, the function of the protein was usually tested. In the XLOS technique, after fluorophore injection, the oocytes were incubated for 1 h in buffered solution at the desired pH in the presence or absence of substrate. After the incubation, the oocytes were washed three times and then mechanically homogenized by pipetting up and down and centrifuged, and clear supernatant was transferred in the plate for the fluorescence reading.
Data Collection, Analysis, and Graph Preparation
To verify the divalent metal transport across membranes of Xenopus laevis oocytes expressing c-Nramp1, c-NrampB, or rDMT1, control oocytes were injected with 50 nL of 12.5 µM calcein.
To prove the cytoplasmic acidification due to the coupled transport of dipeptide and proton in oocytes expressing rPepT1, oocytes were injected with 20 µM FITC-dextran. Ten minutes after the injection of the fluorophore, oocytes were incubated for 1 h in the indicated external solution at the specified pH, in the presence or absence of one divalent metal ion (CoCl2 100 µΜ), or for rPepT1, in the presence or absence of 10 mM GQ. After wash three times in ND96, oocytes were treated as described above ( Fig. 1 ).
All the experimental fluorescence values collected by the plate reader were determined by subtracting the intrinsic fluorescence of the oocytes 35 of each batch from the calcein or FITC-dextran fluorescence values. After subtraction of the mean fluorescence values of the control oocytes, oocytes not expressing the transporters but injected with calcein incubated in saline solution at pH 7.6 were used for each batch to normalize the arbitrary units of fluorescence value collected for each oocyte.
The mean of the residual fluorescence was plotted for the different transporters and conditions. Figures and statistical analysis were prepared with Origin Pro 8.0 (Microcal Software Inc., Northampton, MA). Significance was verified by one-way analysis of variance (ANOVA) and the Bonholm test.
Solutions and Reagents
The ND96 and NDE solutions used during oocyte preparation and culture had the following compositions (in mM unless otherwise noted): ND96: 96 NaCl, 2 KCl, 1 MgCl2, 1.8 CaCl2, 5 HEPES (pH 7.6); and NDE: 96 NaCl, 2 KCl, 1 MgCl2, 1.8 CaCl2, 5 HEPES, 2.5 pyruvate, 250 µg/mL gentamicin. The external control solution for electrophysiological studies and confocal and XLOS experiments is as follows (in mM): NaCl or tetramethylammonium (TMA) Cl 98 buffer solution: 1 MgCl2, 1.8 CaCl2, HEPES, MES, PIPES, or TAPS, depending on the pH condition. The final pH values of 5.5, 6.5, 7.6, and 8.5 were adjusted with NaOH or TMAOH, respectively. Intracellular solution had the following composition (in mM): 130 KCl, 4 NaCl, 1.6 MgCl2, 5 EGTA, 10 HEPES, 5 glucose, pH 7.6.
Results
Dictyostelium discoideum c-NrampB Membrane Expression
In our previous work, we first expressed the unmodified cDNA coding for DdNramp1 or DdNrampB wild-type that resulted in a mostly undetectable current when divalent metal ions were perfused with a two-electrode voltage clamp. This result was not surprising as both proteins are not plasma membrane proteins.
19
Replacing the N- and C-termini of the original proteins with those rDMT1,
13
thus creating the chimera proteins c-Nramp1 and c-NrampB, was the strategy used to express them on the plasma membrane of oocytes—the same approach previously used for yeast metal transporter Smf1p.
36
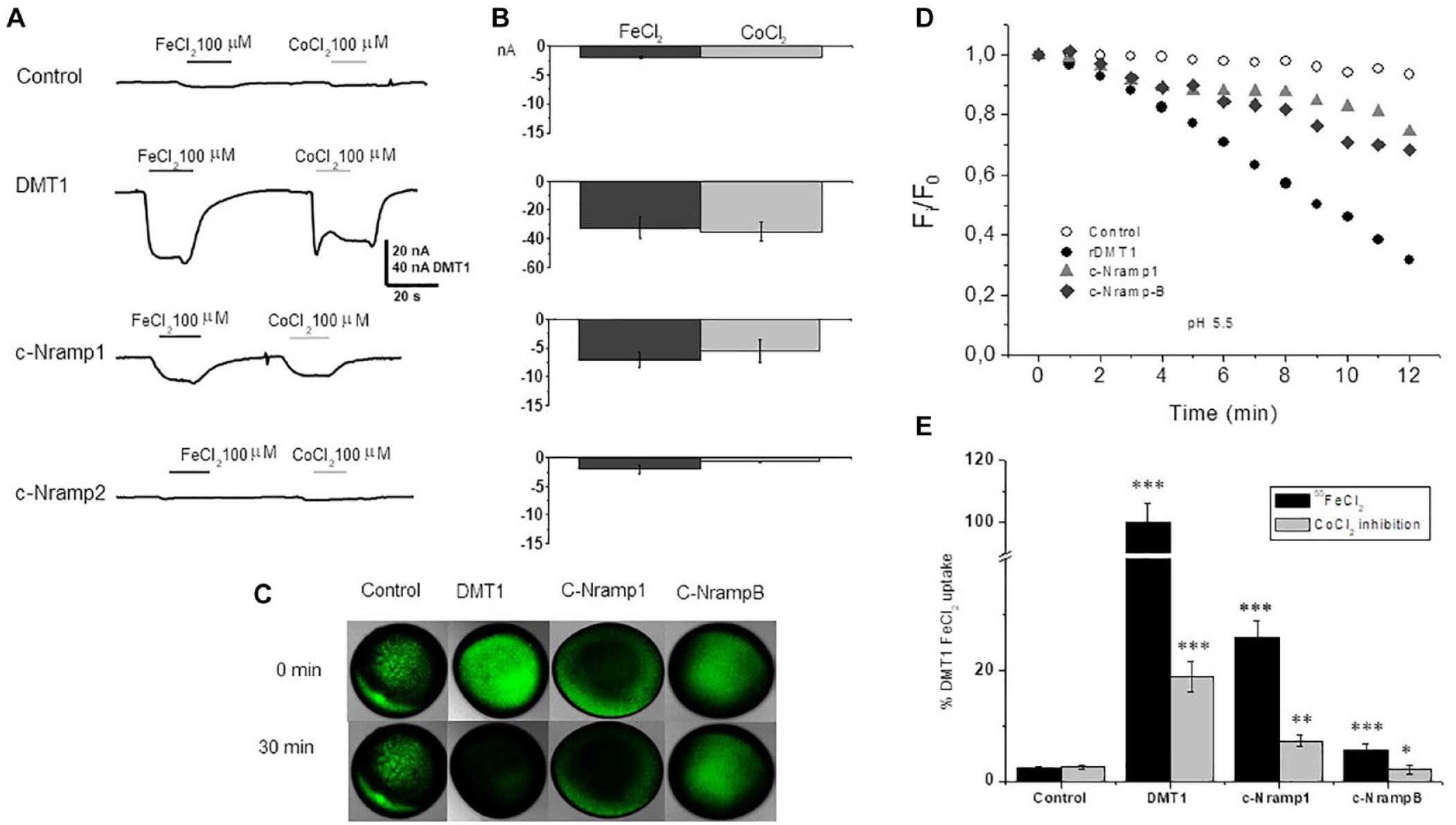
It was expected that DdNrampB, when inserted in the appropriate construct, c-NrampB, should show, if electrogenic, detectable membrane currents in the presence of divalent metals. Nevertheless, when the transporter function was investigated by electrophysiology, c-NrampB did not elicit any measurable transport current in either the sodium or TMA solution, independently from the substrates (divalent metal), holding potential, and pH of the testing solution (the traces recorded at pH 7.6 and at different holding potentials are not shown because they are like those reported in
Fig. 2A
). Conversely, the same protocol applied to c-Nramp1 or rDMT1 elicited a current of tens of nanoamperes (
(
Determination of Transport Activity in Oocytes Expressing c-NrampB by Fluorophore Quenching in Multiwell Assay
The confocal microscopy assay is very reliable, but it can be applied to single oocytes that need to be followed for 30–60 min to get a 50%–100% quenching when the metal transporter is efficient and its functional expression level is appreciable and the oocytes are healthy. c-NrampB and c-Nramp1 appear to be less efficient in comparison with rDMT1 (
We therefore developed a new method called XLOS to functionally characterize c-Nramp1 and c-NrampB and to collect data from a significative number of oocytes without using radiolabeled divalent metal ions, in a time- and money-saving approach. Changes in the intracellular calcein fluorescence caused by the interaction with metal ions entering the cell were monitored by injecting the fluorophore, exposing the oocytes to different testing conditions for 1 h. After the incubation time, the oocytes were homogenized and centrifuged to separate the yolk protein from the cytoplasmic components, and finally, the fluorescence of the clear supernatant was measured (
The technique was applied to characterize c-NrampB. The experiments were also conducted with oocytes expressing rDMT1, as the reference transporter, and c-Nramp1. Moreover, PepT1, the peptide transporter, was tested because it works with a completely different substrate and allows us to test a different fluorophore to validate the technique.
First, the capability of Co2+ to reduce calcein fluorescence in the presence of yolk and cellular components of the oocytes was verified. Different concentrations of CoCl2 were injected together with calcein dissolved in intracellular solution at pH 5.5 and 7.6, and after 15 min of incubation, the oocytes were homogenized and the assay performed as described in Materials and Methods. These experiments proved the appropriate sensibility of calcein in the cytoplasmic yolk environment in the intracellular concentration range expected for transport activity for the divalent metal ion of interest (
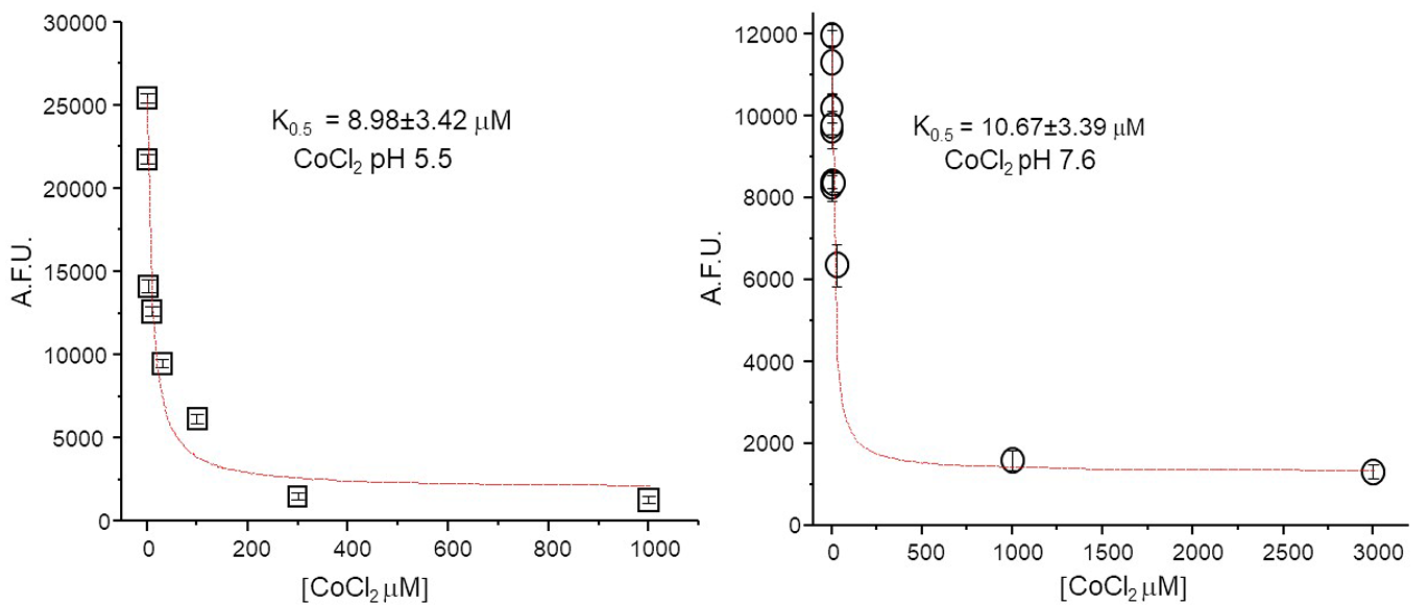
Quantification of K0.5 of calcein in the cytoplasmic environment. The decrease of calcein fluorescence in relation to the amount of CoCl2 present in the cytoplasm. The concentrations reported in the graph are the final concentrations in the oocytes (considering the 50 nL injected and the nominal volume of 1.2 µL of oocytes) and ranged from 30 nM to 3 mM. The two different pH values are those the intracellular solution injected (left, pH 5.5; right, pH 7.6).
The data allowed us to determine the K0.5 of calcein for cobalt (K0.5 Co2+ = 8.98 ± 3.42 µM at pH 5.5 and 10.67 ± 3.39 µM at pH 7.6) in the physiological condition. As expected, these values are slightly higher than those previously recorded by spectrofluorimetric measurement in saline solution, 16 but in agreement with those calculated for other divalent cations in the same conditions.14–16
Control oocytes; oocytes expressing c-NrampB, c-Nramp1, or rDMT1 transporters microinjected with calcein; and control oocytes not injected with the fluorophore were incubated for 1 h in buffer solutions (Na or TMA based) in the presence or absence of 100 µM FeCl2 or CoCl2. Then they were homogenized and centrifuged. and 100 µL of their clear supernatant was collected to quantify the fluorescence intensity as described. Fe2+ and Co2+ transported in the cytoplasm of the oocytes quenched the fluorophore injected. The fluorescence values after subtraction of autofluorescence (value of fluorescence of the cytoplasmic extract from oocytes of the same batches not injected with the fluorophore) and normalized as indicated in Materials and Methods were reported in
Figure 4
. Data were collected in control oocytes, oocytes not expressing exogenous transporters but injected with the fluorophore, and oocytes expressing the indicated transport protein at different pH values in the presence or absence of metal. The fluorescence in all SLC11-expressing oocytes was reduced when exposed to FeCl2 or CoCl2; the residual fluorescence differed statistically significantly between oocytes expressing the same transporter but not exposed to the metal ions and the control oocytes tested in the same condition. Differences are present for all the expressed transporter proteins for the two metal ions and the two pH values tested (
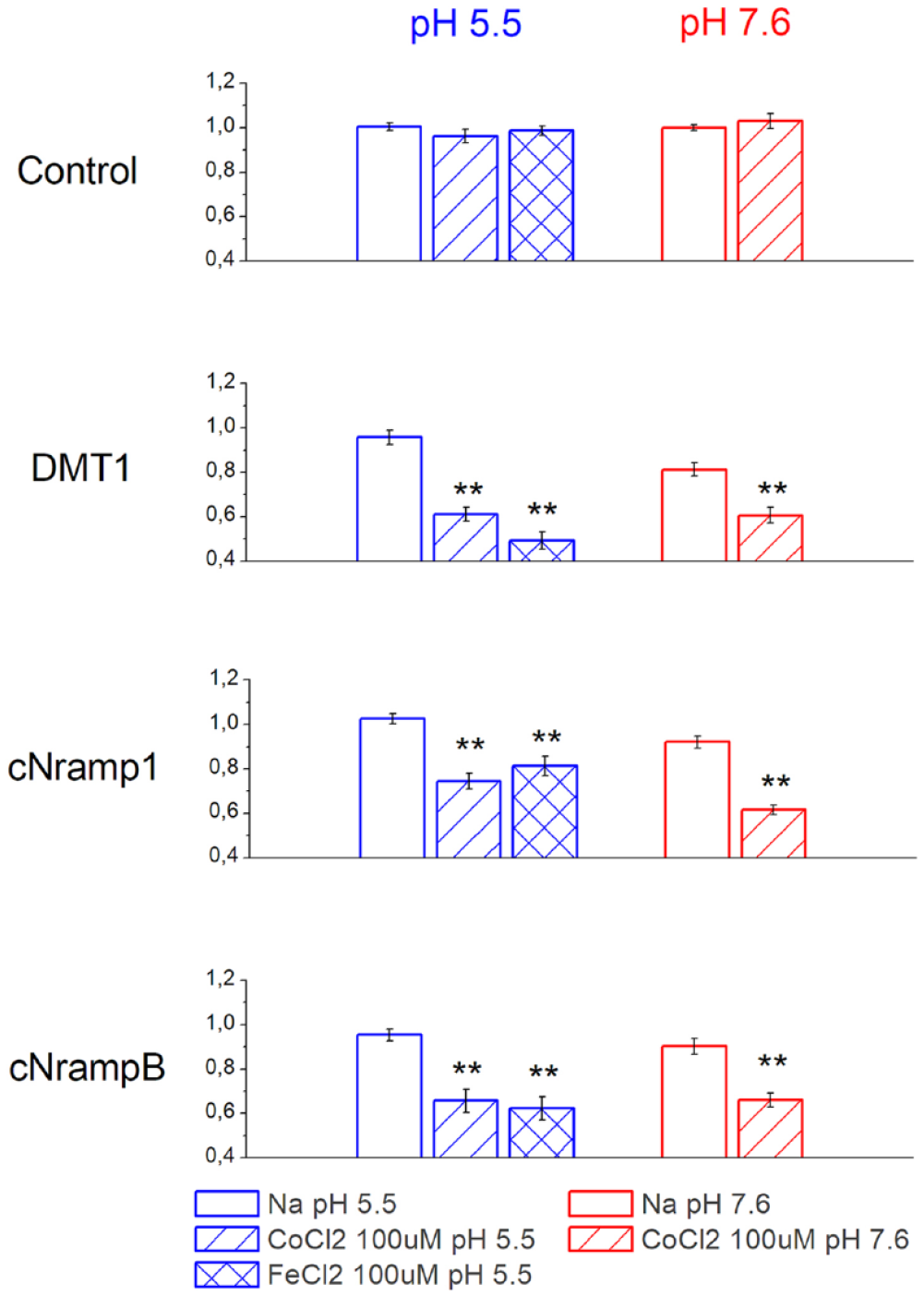
Divalent metal transport in Xenopus oocytes measured by calcein quenching. Residual fluorescence obtained as specified in the text in Xenopus laevis oocytes expressing rDMT1, c-Nramp1, and c-NrampB determined by XLOS methods. Fluorescence of calcein in control oocytes and oocytes expressing rDMT1, c-Nramp1, a nd c-NrampB in the absence or presence of 100 µM CoCl2 or FeCl2 in sodium solution at pH 5.5 (blue, left) or of 100 µM CoCl2 in sodium solution at pH 7.6 (red, right). Quenching in oocytes expressing rDMT1, c-Nramp1, and c-NrampB incubated for 1 h in the indicated metal ion is significantly different from that of oocytes expressing the same transporter and incubated in the same solution without CoCl2 or FeCl2 (***p > 0.001, **p > 0.01, one-way ANOVA, Bonholm test; mean ± SE of 24–205 oocytes, 5–15 batches).
The data were collected at two different pH values because rDMT1 is known to have maximal activity (higher transport current and higher uptake rate) at pH 5.5,12,30,37 and the data reported in Buracco et al. 13 suggested that c-Nramp1 also has the best activity when the external solution is slightly acidic, whereas for c-NrampB they suggested that this transporter increased its functionality at neutral pH; the quenching value of FeCl2 was collected only at pH 5.5 because of the possible transport at pH 7.6 of Fe3+ ions by endogenous proteins.
The fluorescence in oocytes expressing rDMT1, c-Nramp1, and c-NrampB incubated for 1 h in the indicated metal ion is significantly reduced if compared with that of oocytes not exposed to the metal or to control oocytes in the same conditions.
In oocytes expressing rDMT1, Fe2+ and Co2+ that are translocated in the cytoplasm caused calcein quenching of about 50% and 40% for Fe2+ and Co2+, respectively, at pH 5.5 (residual fluorescence after iron exposure, 0.491 ± 0.039; after cobalt exposure, 0.610 ± 0.030). At pH 7.6, the capability of Co2+ to quench the fluorophore was similar and the residual fluorescence after 1 h of uptake was about 70% (0.697 ± 0.029). These data agree with data collected on the transport-associated currents measured in the presence of Mn2+ by a two-electrode voltage clamp. 13 c-Nramp1-expressing oocytes tested by XLOS after incubation with CoCl2 or FeCl2 reduced the calcein fluorescence by about 20% at pH 5.5 (residual fluorescence in the presence of cobalt, 0.744 ± 0.034; in the presence of iron, 0.812 ± 0.044), demonstrating that both ions were transported and able to reduce calcein fluorescence intensity with ion selectivity: Co2+ > Fe2+. At pH 7.6, the reduction of fluorescence increased to 40% (residual fluorescence, 0.615 ± 0.021). c-NrampB-expressing oocytes at pH 5.5 showed that Fe2+ and Co2+ (about 35% fluorescence quenching) were transported and quenched the calcein inside the cell, suggesting similar ion transport in all tested conditions (residual values of fluorescence at pH 5.5 for cobalt, 0.656 ± 0.051; at pH 5.5 for iron, 0.622 ± 0.051; and at pH 7.6 for cobalt, 0.659 ± 0.031). Thus, c-NrampB transports divalent metal ions, confirming the preliminary 55Fe uptake data. Moreover, ion dependence was tested substituting NaCl with TMACl in the incubation solution at the two pH values (5.5 and 7.6) (data not shown). The data collected showed similar activity in all the conditions, indicating that c-NrampB is a sodium-independent transporter, like other members of the SLC11 family.
pH Dependence
To better investigate the pH dependence of c-NrampB, we measured the functionality of the transporter with the XLOS technique in a larger range of pH values, from 5.5 to 8.5, comparing the behavior with the pH dependence of rDMT1. 26 To compare the difference between the two transporters, the data here were reported as the percentage of residual fluorescence after cobalt exposure with respect to the fluorescence of oocytes expressing the same transporter at the same pH, but in the absence of metal.
The data collected by XLOS in the presence of CoCl2 demonstrated for rDMT1 a Co2+ internalization significantly higher at pH 5.5 when compared with all the pH conditions tested (
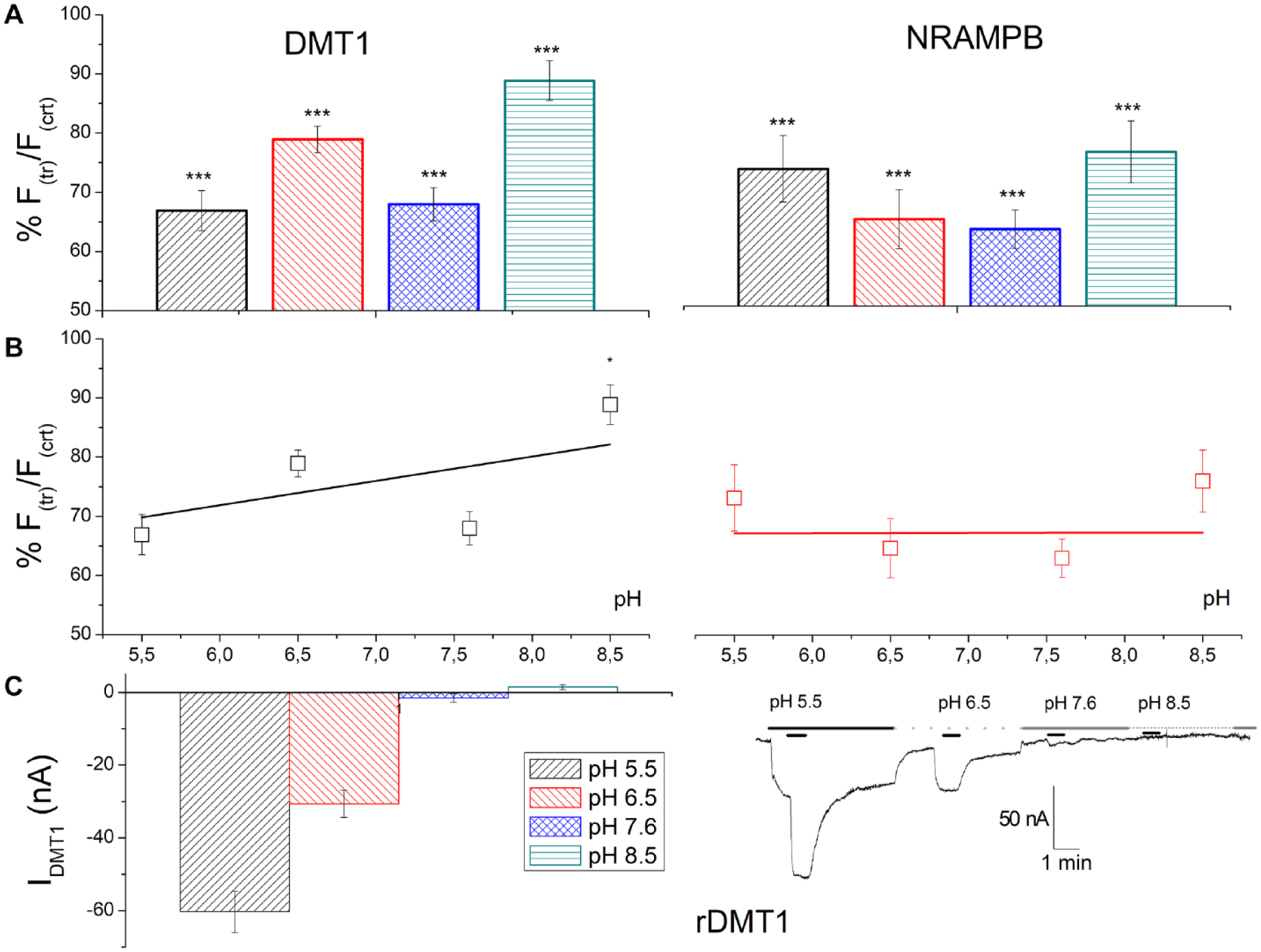
Effects of pH of the external solution on the transport of CoCl2. (
Otherwise, c-NrampB showed similar activity in all conditions, with a residual fluorescence that ranges from 63% to 76%, with the best values for pH 6.5 and 7.6 (
In

Transport current–calcein quenching relationships. The percentage value of quenching calculated using XLOS methods was plotted versus the transport current recorded in the same oocyte. The values were collected for four oocytes of two different batches. The current was recorded by a two-electrode voltage clamp at the holding potential of −40 mV in Na solution at pH 5.5 in the presence of 100 µM CoCl2. The oocytes were perfused for 5 min with the substrate (gray bar in the inset). Traces of the lowest and the highest recorded currents are reported in the inset. The data were linearly fitted (gray line).
Calcein fluorescence remained stable in control oocytes or oocytes expressing the transporters in the absence of divalent metal ions for at least 4 h. Moreover, in the presence or absence of transported substrates (FeCl2 or CoCl2), the calcein fluorescence value in oocytes not expressing exogenous transporters was also constant, suggesting that divalent metal ions were nonpermeating and, consequently, were not able to quench calcein inside the cell in the range of pH values tested (data not shown).
Monitoring Activity of Peptide Transporter PepT1 by XLOS Experiment
We performed XLOS experiments using a different fluorophore to confirm the applicability of XLOS with different transport proteins. FITC-dextran is pH sensitive and thus quenchable inside the oocyte following acidification. We analyzed oocytes expressing the rabbit proton-coupled peptide transporter (PepT1). XLOS applied on oocytes expressing PepT1 gave results comparable to those of traditional techniques, namely, current recorded by a two-electrode voltage clamp (
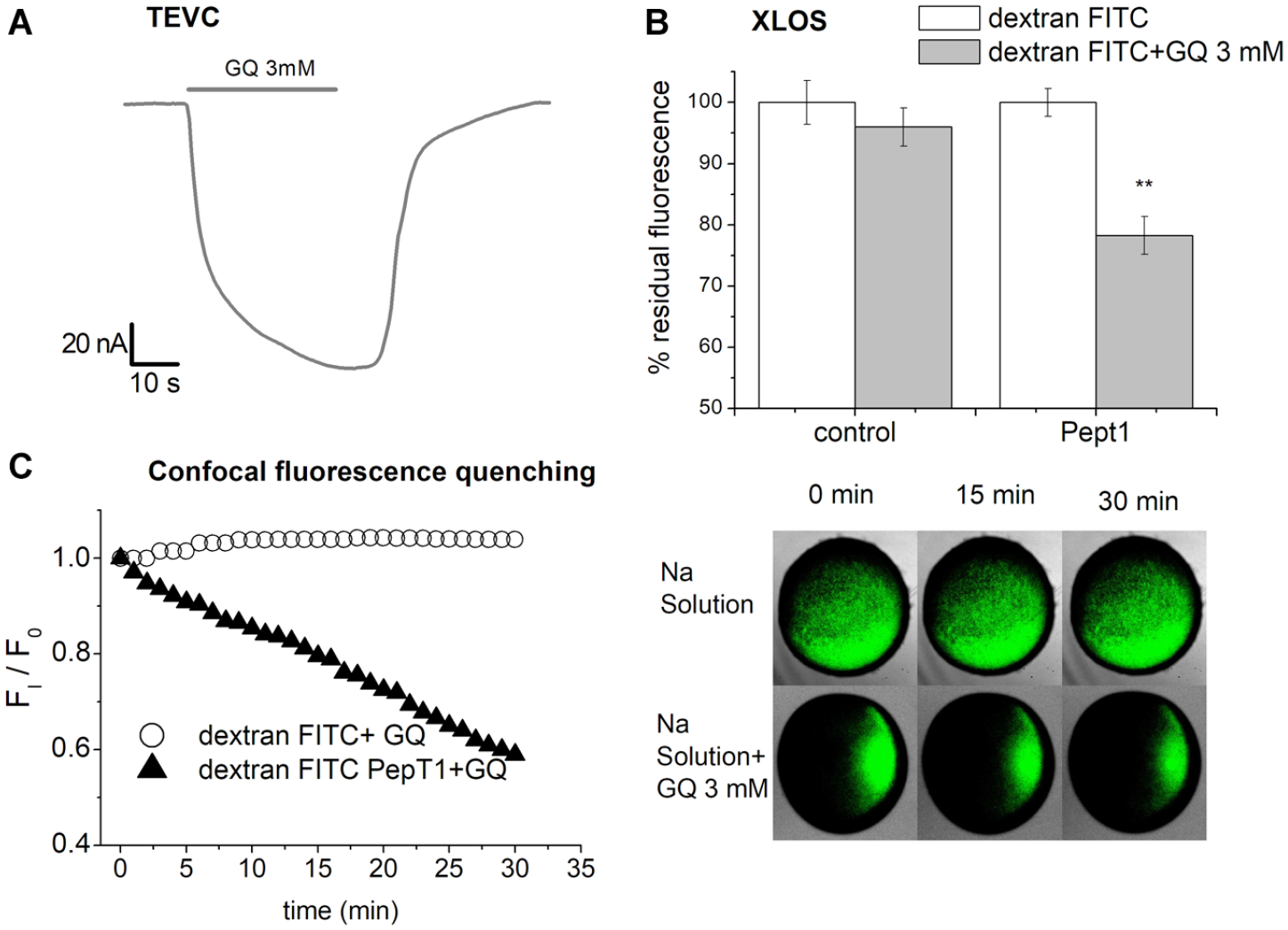
Dipeptide transport in Xenopus oocytes measured by FITC-Dextran quenching. (
All together, these experiments demonstrated the validity, sensitivity, and specificity of the XLOS method and support the applicability of studying metal ion transporters.
Discussion
In this work, our aim was to develop a novel approach to measure substrate uptake in a nonelectrogenic transporter using a fluorescent signal. Recently, we have encountered difficulties in characterizing orthologs of the SLC11 family, NrampB cloned from Dictyostelium discoideum. The necessity of measuring the activity of the protein in a radiolabel-free laboratory suggested that an indirect method be used to determine the amount of substrate entering into the cell. In the paper of Buracco et al., 13 we have characterized Nramp1 of Dictyostelium discoideum and revealed the activity of NrampB. The characterization was achieved using multidisciplinary approaches, but due to the absence of relevant electrogenic activity in NrampB, its function was investigated in Xenopus oocytes by acquiring confocal images of the quenching of injected fluorophore calcein. The confocal imaging assay was reliable, but the data can be collected from only one oocyte at time, and the significant quenching usually requires more than 30 min; thus, it is time-consuming to obtain a statistically significant number of oocytes with this assay. Here, a novel method to measure uptake is reported.
After injection of a fluorophore, oocytes were incubated with the substrate and their transport activity measured by evaluating fluorescence quenching in a microplate reader. This new technique allows the testing of tens of oocytes at the same time by applying different experimental settings, with statistically significance reached in each batch tested, thus saving time and, more importantly, reducing the number of animals.
To better characterize chimeric DdNrampB protein, the construct previously prepared 13 was used. In the coding cDNA, the N- and C-termini of the protein were replaced with the N- and C-termini of rDMT1, to improve its expression on the plasma membrane and to allow functional characterization in Xenopus oocytes, as suggested by Chen et al. 36 Electrophysiological experiments in oocytes expressing the c-NrampB showed the absence of recordable transport-associated currents, advising that the transporter could be electroneutral and thus underlining the necessity of a different approach to study the function, avoiding the use of radiolabeled metal ions. Consequently, we developed a new assay, named XLOS, that could be utilized as a substitute to establish the radiolabeled uptake methodology with easy-to-use laboratory equipment and low-cost reagents. Our novel assay is an improvement of a previous confocal microscopy assay, based on calcein, a low-cost divalent metal indicator fluorophore, injected into the oocyte cytoplasm and undergoing quenching due to the imported divalent metals in a single oocyte. 13 XLOS was first applied to measure the metal transport of the well-characterized and electrogenic divalent metal ion transporter rDMT1, and then in c-Nramp1- and c-NrampB-expressing oocytes using FeCl2 and CoCl2 as substrates. c-NrampB, in particular, is a useful candidate to test XLOS. In fact, the difficulty of revealing the transport current and the data previously collected 13 demonstrating best functionality around neutral pH suggest that c-NrampB can transport metal in the absence of relevant transport current, as happens for DMT1 at neutral pH. 30 The presence of substrate translocation even in the apparent absence of transport currents in electrogenic transporter happens between SLC proteins;30,38 moreover, it is possible to find examples of transporters of the same family that move a different amount of charge per cycle, like the type 2 Na-phosphate transporters (SLC34).39–41 The data collected in Buracco et al. 13 and here with XLOS let us hypothesize that c-NrampB moves divalent cations inside the cytoplasm of the oocytes in an electroneutral manner because of the detectable entrance of divalent ion in the radiolabeled assay and in our XLOS approach, but does not elicit quantifiable currents.
To verify the possibility of applying the methods to measure transport in general, the same protocol was tested in oocytes expressing PepT1 utilizing FITC-dextran, commonly used as a cellular fluorescence tracer, 42 as a pH-sensitive probe. In PepT1, the transport of GQ is associated with temporary acidification of the cytoplasm; 43 thus, we measured the FITC-dextran quenching by the XLOS method as a signal of proton increase in the cytoplasm, that is, reduction of the pH due to the cotransport of H+/dipeptide (GQ). We have chosen this transporter because it is highly expressed in Xenopus oocytes and works well at neutral pH. Unfortunately, it was not possible to also test the transporters of the SLC11 family with FITC-dextran because the sensitivity of this fluorophore requires good transport of protons at pH 7.6 and none of the divalent metal transporters tested possess the specificity required. Moreover, at pH 5.5 FITC-dextran was also quenched in the absence of substrate and even in control oocytes.
Appling XLOS, we demonstrated the quenching action of metals entered in the oocytes by SLC11 transporters and the quenching action of protons entered together with GQ through SLC15a1 protein. The data obtained in rDMT1 and PepT1 studied with XLOS replicated the information obtained by electrophysiological characterization of the transporter function. For rDMT1, the metal ions’ ability to quench the fluorophore was higher at pH 5.5 and displayed a similar efficiency for the transport of the two ions utilized. Regarding c-Nramp1-expressing oocytes, Co2+ and Fe2+ at pH 5.5 were transported and able to quench calcein with a slight preference for Co2+. At pH 7.6, only data with Co2+ were collected to avoid potential interference at this pH of Fe3+ ions that can be transported by endogenous protein, thus altering the results. The data collected at pH 7.6 confirmed previous electrophysiological results. 13 This validation with known transporters lets us apply the method to study the electroneutral c-NrampB. The protein expressed in the oocytes showed that Fe2+ and Co2+ were transported and were able, once inside the cell, to quench calcein at a similar percentage for all pH values tested, suggesting pH independence of the transport. Moreover, c-NrampB showed metal ion uptake in the presence of both NaCl and TMA solution (data not shown), confirming that, like the other SLC11 members, is was sodium independent.
Until now, only a few works have been published on the characterization of transporters in Xenopus oocytes with fluorescent probes, namely, to investigate DMT1 transport using Phen Green SK 12 and in oocytes expressing c-Nramp1, c-NrampB, and rDMT1using calcein, 13 but in both cases, the fluorophore quenching was studied with confocal microscopy. In our laboratory, a similar approach, using calcein and conventional fluorescence microscopy, was also applied to investigate the crossing of the plasma membrane by the metal nanoparticles.14–16 In all these works, a single cell was studied, at times greatly limiting the number of samples collectable for each batch of oocytes. Only in Kottra et al. 18 was a plate reader approach applied to measure the transport of a fluorescent compound, but in this case, the protein studied could move across the membrane large fluorescent molecules.
Our data on XLOS, based on fluorophore quenching followed by microplate reader analysis, permitted us to obtain results from tens of oocytes in different experimental conditions at the same time, achieving significant statistical data from the same batch, reducing the number of animals used to collect oocytes, and last but not least, saving time for researchers.
Furthermore, the method permits us to collect data on electroneutral transporters, not suitable to be studied with the classical electrophysiology.
In conclusion, the method, although with some limitations regarding the substrate or the tested condition, has allowed us to characterize an electroneutral carrier, like the DdNrampB, and to confirm previous studies on DdNramp1 and rDMT1. Moreover, the data presented here provide significant support to the applicability of the XLOS method.
Footnotes
Acknowledgements
We thank Pamela Roccia for the help in the new experiments reported in this revision.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was financed with FAR 2017-2018 from the University of Insubria.
