Abstract
Cardiomyocytes (CMs) derived from induced pluripotent stem cells (iPSCs) provide an in vitro model of the human myocardium. Complex 3D scaffolded culture methods improve the phenotypical maturity of iPSC-CMs, although typically at the expense of throughput. We have developed a novel, scalable approach that enables the use of iPSC-CM 3D spheroid models in a label-free readout system in a standard 96-well plate-based format. Spheroids were accurately positioned onto recording electrodes using a magnetic gold–iron oxide nanoparticle approach. Remarkably, both contractility (impedance) and extracellular field potentials (EFPs) could be detected from the actively beating spheroids over long durations and after automated dosing with pharmacological agents. The effects on these parameters of factors, such as co-culture (including human primary cardiac fibroblasts), extracellular buffer composition, and electrical pacing, were investigated. Beat amplitudes were increased greater than 15-fold by co-culture with fibroblasts. Optimization of extracellular Ca2+ fluxes and electrical pacing promoted the proper physiological response to positive inotropic agonists of increased beat amplitude (force) rather than the increased beat rate often observed in iPSC-CM studies. Mechanistically divergent repolarizations in different spheroid models were indicated by their responses to BaCl2 compared with E-4031. These studies demonstrate a new method that enables the pharmacological responses of 3D iPSC-CM spheroids to be determined in a label-free, standardized, 96-well plate-based system. This approach could have discovery applications across cardiovascular efficacy and safety, where parameters typically sought as readouts of iPSC-CM maturity or physiological relevance have the potential to improve assay predictivity.
Introduction
Adult myocardium is comprised at the cellular level predominantly of phenotypically mature cardiomyocytes (CMs) in close proximity to cardiac fibroblasts. 1 Induced pluripotent stem cell-derived CMs (iPSC-CMs) offer significant advancement toward modeling the adult myocardium. However, the phenotype is frequently more representative of an immature stage, with spontaneous pacemaker activity, lower force generation, and underdeveloped sarcoplasmic reticulum (SR) calcium (Ca2+) handling and sarcolemmal ion channel expression. 2
Numerous studies have demonstrated that the phenotypical maturity of iPSC-CMs in vitro is limited postdifferentiation by culture in simple 2D monolayers and can be enhanced by mimicking a variety of physiological cues, such as 3D matrices and structured scaffolds,3,4 co-culture with nonmyocyte supporting cells,5–7 electrical pacing of contractile behavior and mechanical conditioning upon elastomeric substrates,7–10 and utilization of appropriate energy sources.11,12 In parallel, a wide variety of technical approaches have been developed in order to report functional performance. For example, iPSC-CMs together with nonmyocyte supporting cells or extracellular matrix proteins may be cast into strips or loops and contractility tracked optically.6,9–11,13–15 Alternatively, the self-assembly of 3D structures termed “microtissues,” “embryoid bodies,” or “spheroids” promoted by the use of low-adhesion surfaces or culture in hanging droplets has been combined with videomicrography to report contractility.7,16–19 Dye-based or optogenetic reporters, and even highly manual approaches such as microelectrodes, 16 multielectrode arrays,20,21 or force transducers, 10 have also been applied to 3D iPSC-CM constructs. One limitation of many of these approaches is the requirement for complex handling procedures and apparatus, resulting in low-throughput data generation, and few offer significant throughput.11,22
It therefore seemed important to us to develop improved approaches addressing several of these known opportunities, which would permit efficient, scalable, high-throughput iPSC-CM studies. The simplest iPSC-CM 3D structures to obtain are spheroids/microtissues formed on low-adhesion surfaces or hydrogels.7,16 One challenge is the subsequent difficulty in handling the resulting free-floating, actively beating spheroids and applying detection technologies in a robust manner. We sought to implement a high-throughput and scalable spheroid-based approach by circumventing some of these technical challenges by assembling iPSC-CM spheroids together with nonmyocyte supporting cells and gold–iron oxide nanoparticles. This rendered the resulting spheroids magnetic and able to be temporarily directed and located by arrays of neodymium magnets to facilitate accurate positioning of actively beating spheroids. We used this approach to locate actively beating co-culture spheroids within 96-well sensor plates developed for label-free functional measurements of 2D iPSC-CM monolayers 23 to determine whether contractility (impedance) and extracellular field potentials (EFPs) could be recorded from the nanoparticle-containing 3D spheroids. Remarkably, both parameters could be detected, providing the first plate-based measurements of this form from a 3D spheroid iPSC-CM model and suggesting that the approach could be scalable in combination with ordinary liquid handling automation. Indeed, pharmacological responses were produced efficiently using a combination of a 96-well liquid handling platform and acoustically dispensed compound plates. To investigate whether a 3D iPSC-CM model could recapitulate more physiological aspects of the adult myocardium, factors such as fibroblast co-culture (both human primary fibroblasts and a mouse fibroblast line), extracellular buffer composition, and electrical pacing were investigated. The utility of our new method is validated by three key studies where we demonstrate (1) strikingly enhanced contractile amplitude; (2) measurement of actual positive inotropic responses rather than beat rate effects, which are typically observed in 2D assays; and (3) responses to BaCl2 and E-4031 as indicators of the repolarization mechanisms potentially in operation. These studies cover areas of interest to cardiovascular therapeutics as well as general in vitro safety profiling, suggesting that our new method could have applications across a range of discovery activities and provide a means to develop more physiologically relevant assays in these fields at a scale able to generate hundreds to thousands of data points.
Materials and Methods
Materials
Cells used in this study were iPSC-CMs (iCell 2 , Cellular Dynamics International, Madison, WI) and primary human cardiac fibroblasts (PromoCell, Heidelberg, Germany) together with suppliers’ recommended media, and mouse embryonic fibroblast (MEF) NIH-3T3 cells (ATCC, Manassas, VA). Gold–iron oxide nanoparticle reagent (NanoShuttle-PL) was obtained from n3D Biosciences (Houston, Texas). Ninety-six-well ultra-low-attachment microplates (cat. 7007) were supplied by Corning Life Sciences (Corning, New York) and 96-well sensor plates (NSP-96 CardioExcyte96 sensor plates 0.6 mm, cat. 201002) were supplied by Nanion Technologies GmbH (München, Germany). Fibronectin from bovine plasma was obtained from Sigma-Aldrich (St. Louis, Missouri). Compounds were provided by the AstraZeneca compound collection or purchased from Sigma-Aldrich or Tocris Bioscience (Bristol, UK), and stock solutions were solubilized in DMSO.
Self-Assembly of iPSC-CM Co-Culture Spheroids
iPSC-CMs were resuspended from frozen vials according to supplier protocols to generate a suspension of 2000 cells/50 µL in CDI Plating media. Fibroblasts were routinely cultured according to supplier recommendations and resuspended at the required density (20–500 cells/50 µL in order to generate the ratios displayed in
Transfer of Co-Culture Spheroids to Sensor Plates
After assembly, nanoparticle-impregnated spheroids were transferred to sensor plates, using a custom-made neodymium magnet array located underneath the sensor plate to magnetically guide positioning of the spheroids onto the central sensing electrode located in the well base. Before transfer, a 50% media change was performed in assembly plates. In a sterile laminar-flow hood, a sensor plate (previously coated with 10 µg/mL fibronectin) was aligned with the custom-made neodymium magnet plate (
Detection of Spheroid Contractility and EFP
Impedance and EFP signals were typically acquired 2 days after transfer into sensor plates. Before signal acquisition, a complete media change using 37 °C media ensured volumes of 250 µL/well in all wells. After a minimum 3 h equilibration period, sensor plates were loaded onto a CardioExcyte96 platform 23 (Nanion Technologies GmbH) for signal acquisition, typically with the platform located inside a tissue culture incubator. Unless otherwise stated, all measurements were performed using CDI Maintenance media as the incubation buffer. Signals were recorded in 20 s sweeps and typically low-pass-filtered at 10 and 400 Hz (impedance and EFP, respectively) before analysis.
Whole-Plate Compound Dosing Using Automated Liquid Handling
Experiments were designed where all wells in a sensor plate would be dosed with compounds simultaneously, either as one-off additions or by multiple whole-plate additions. A compound source plate was created in a 384-well V-bottomed microplate using a 384-to-96 quadrant mapping. Compounds were dispensed into the required quadrants of the compound source plate using acoustic dispensing and Echo Cherry Pick software (Echo 555 platform, Labcyte, USA), creating a 250 nL/well dispense of 100% DMSO stock solution (1000× final concentration). A liquid handling platform (CyBio FeliX, Analytik Jena AG) equipped with a 96-pipettor head was used to perform whole-plate dosing. Briefly, the sensor plate was removed from the CardioExcyte96 platform and positioned on the stage of the FeliX. Fifty microliters per well of warm media was aspirated from the sensor plate, dispensed into the appropriate quadrant of the 384-well compound source plate, aspirated, and dispensed (off-center) back into the sensor plate. Compound dosing was achieved in less than 20 s, and the sensor plate returned to the CardioExcyte96 platform.
Electrical Pacing and Modified Tyrodes Conditions
Electrical field stimulation via the CardioExcyte96 platform (typically 1%–5% power) was adjusted to capture spheroid beating while preventing pacing artifacts. Experiments were performed at 1 Hz pacing frequency unless otherwise stated. For reduced external Ca2+ experiments, warmed Tyrodes solution (145 mM NaCl, 4 mM KCl, 1 mM MgCl2, 11.1 mM glucose, 10 mM HEPES, pH 7.4) containing the required amount of CaCl2 was substituted in place of the culture media by a 50% substitution, followed by 100% substitution, over the course of 30 min, with a subsequent 1 h equilibration period before further additions.
Optical Flow Analysis of Contractility
Video clips (20 s) were captured via bright-field illumination on an inverted microscope equipped with a 10× objective directly from the clear plastic assembly plates. Video files were converted to 8-bit grayscale Z stacks in ImageJ and cropped to 250 pixels per side (sufficient to encompass the complete spheroid). Optical flow was calculated using particle image velocimetry (ImageJ), determining the vector magnitudes per Z-stack image. Z-axis histograms were generated to display average vector magnitude against time.
Histopathology Processing
Spheroids were processed as formalin-fixed, paraffin-embedded blocks and sections stained with H&E as previously described. 25
Data Analysis
Impedance and EFP recordings from a single sensor plate experiment were processed using Nanion CardioExcyteControl software. Beat detection and determination of average parameters 23 in impedance mode (amplitude, beat rate, pulse width 50%, rising time, and falling time) or EFP mode (FPDmax) were calculated for each sweep. Manual quality control was performed to exclude individual wells with unstable beat detection or unstable electrical pacing. Further analysis was performed in RStudio and Microsoft Excel. Data are presented as mean ± SD, and statistical significance was calculated using the Student t test or Kruskal–Wallis test with Dunn’s post hoc test as indicated. A p value <0.05 considered significant.
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
Results
Self-Assembly of Co-Culture Spheroid Clusters and Production of Plate-Based Arrays
Diffuse aggregates of iPSC-CMs together with primary human cardiac fibroblasts or MEFs visible after seeding in assembly plates (
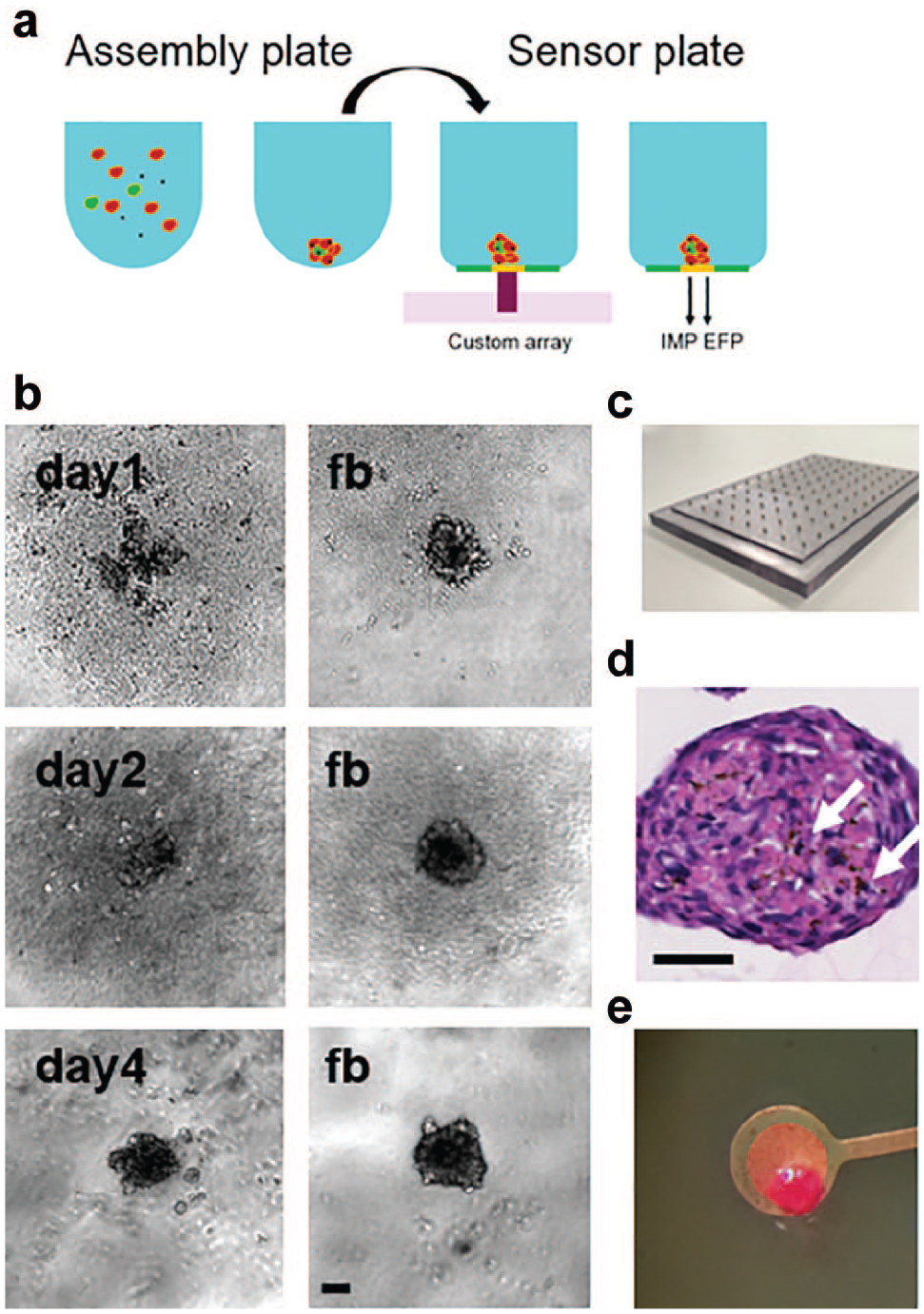
(
Incorporation of gold–iron oxide nanoparticles into the iPSC-CM spheroids during assembly (
Proof-of-Concept Detection of Contractility Parameters by Impedance Sensing in iPSC-CM 3D Spheroids
Spontaneous contractile activity of iPSC-CM spheroids could be detected by impedance sensing (
Comparison of Beat Characteristics in Impedance Recording of iPSC-CM Spheroids Assembled with or without Gold–Iron Oxide Nanoparticles and Fibroblasts.
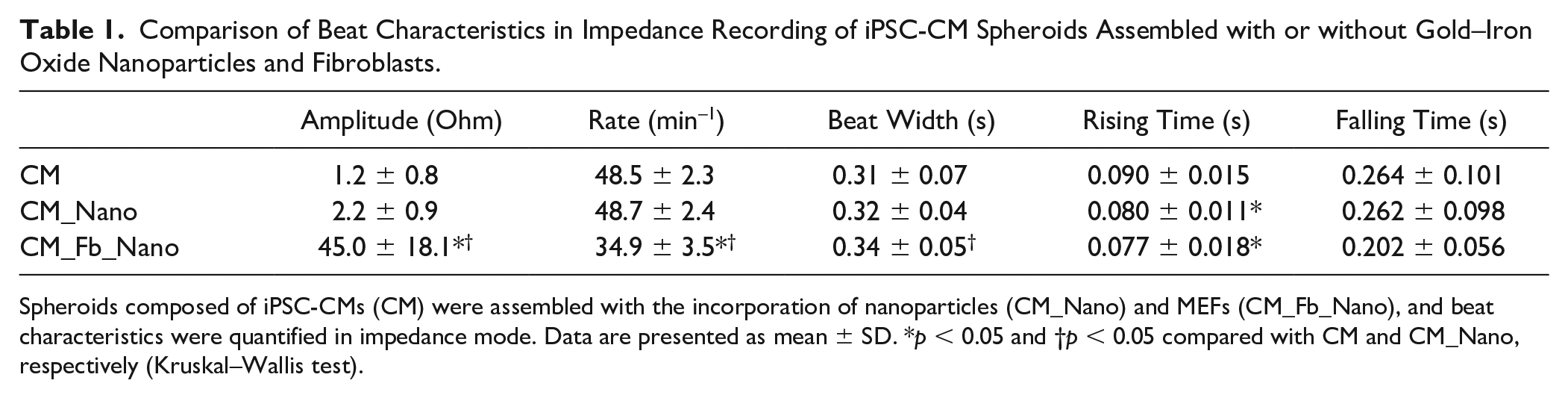
Spheroids composed of iPSC-CMs (CM) were assembled with the incorporation of nanoparticles (CM_Nano) and MEFs (CM_Fb_Nano), and beat characteristics were quantified in impedance mode. Data are presented as mean ± SD. *p < 0.05 and †p < 0.05 compared with CM and CM_Nano, respectively (Kruskal–Wallis test).
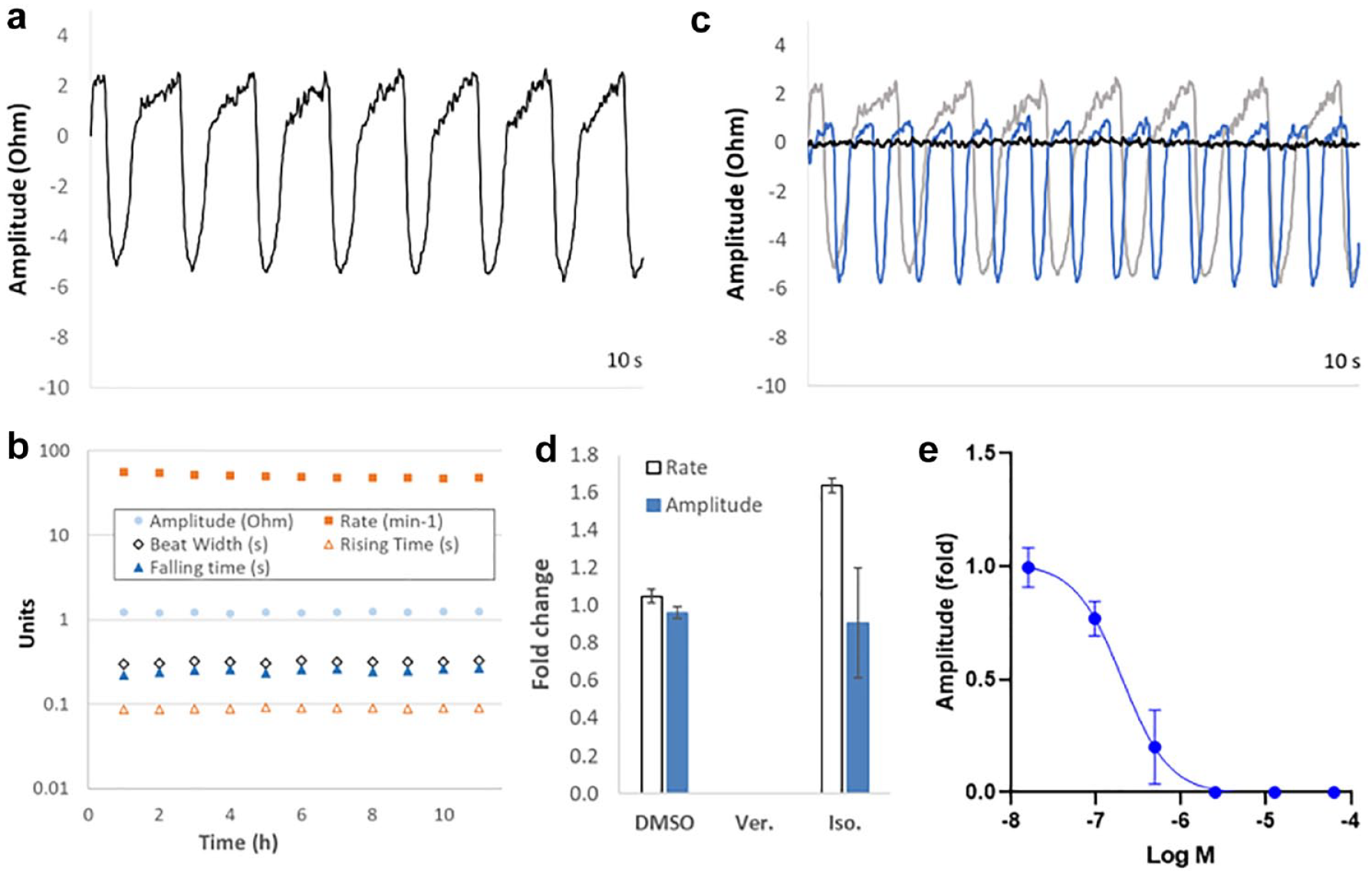
Detection of contractility parameters by impedance sensing in iPSC-CM spheroids. (
An efficient approach to compound addition was developed based on whole-plate experiments using an automated 96-well liquid handling platform and compound source plates prepared by acoustic dispensing of DMSO stock solutions. Warm media was aspirated from the recording plate, rapidly mixed with the 250 nL DMSO stock solution volumes within the source plate, and then dispensed back into the recording plate. This approach avoided the preparation of additional warmed compound dilutions for dosing, minimized experimental times, and maintained final DMSO concentrations at 0.1% (or lower if required) to prevent vehicle artifacts. Rapid compound dosing (less than 20 s for automated media transfer), coupled with large well volumes (250 µL), maintained sensor plate temperature, which was stable to within 0.5 °C during subsequent recordings 3 min after compound addition. Effects consistent with known pharmacology were observed following the addition of 1 µM verapamil (L-type Ca2+ channel inhibitor) or 400 nM isoproterenol (β-adrenoceptor agonist), resulting in cessation of beating activity or an increase in beat rate, respectively (
Fibroblast Co-Culture Dramatically Modulates Intrinsic Contractility
Self-assembly of iPSC-CM spheroids together with fibroblasts of primary human cardiac or MEF origin resulted in co-culture spheroids with significantly altered functional characteristics. Most apparent was a dramatic increase in contractile amplitude, with fibroblast co-culture resulting in an approximately 15-fold (mouse) or 6-fold (primary human cardiac) increase in beat amplitude (representative traces shown in
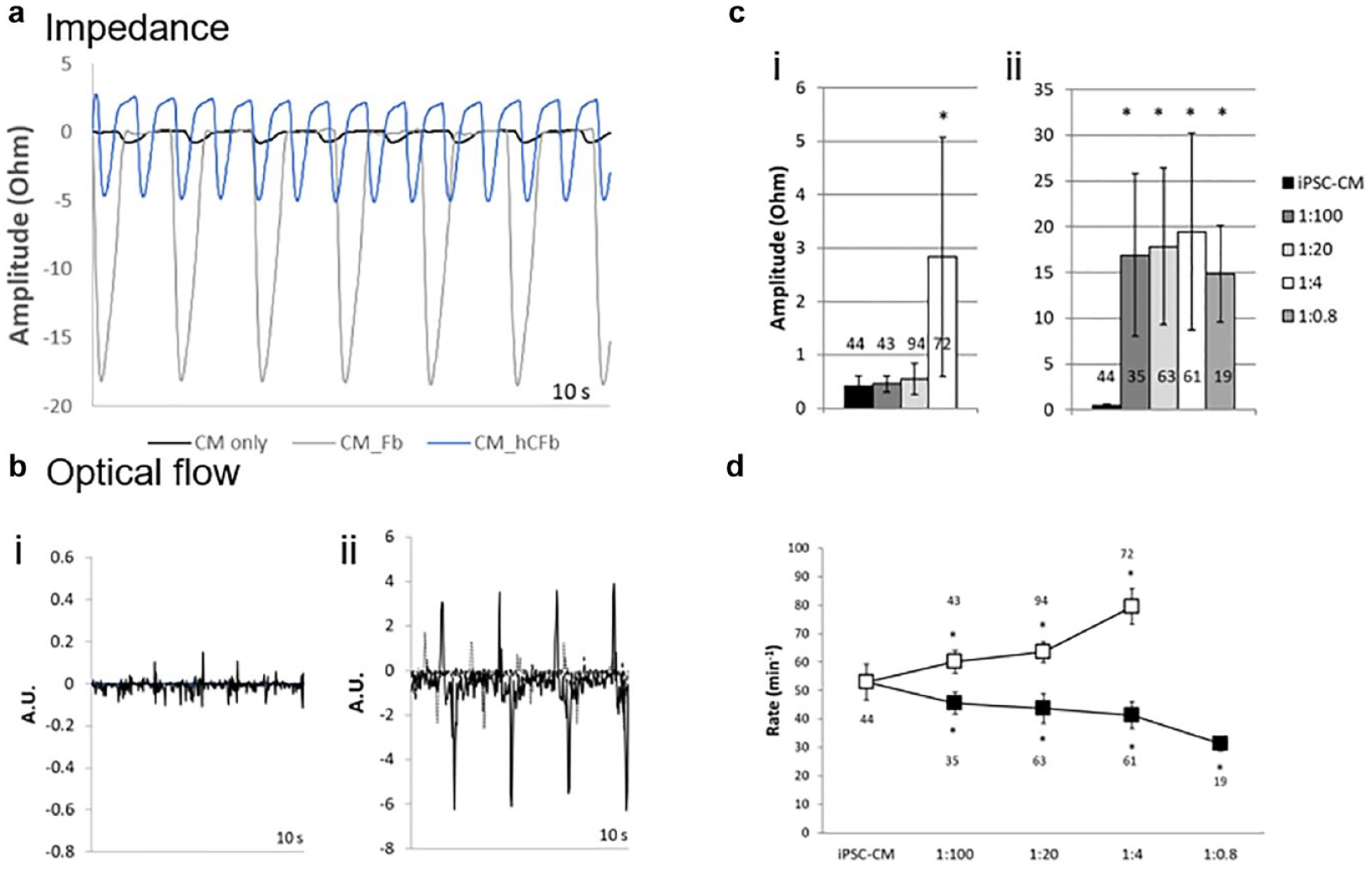
Fibroblast co-culture dramatically modulates intrinsic contractile behavior. (
Primary human cardiac fibroblasts required an initial seeding ratio of at least 1:4 to significantly increase beat amplitude, although MEF seeding ratios as high as 1:100 induced a dramatic effect on analysis on day 10 (
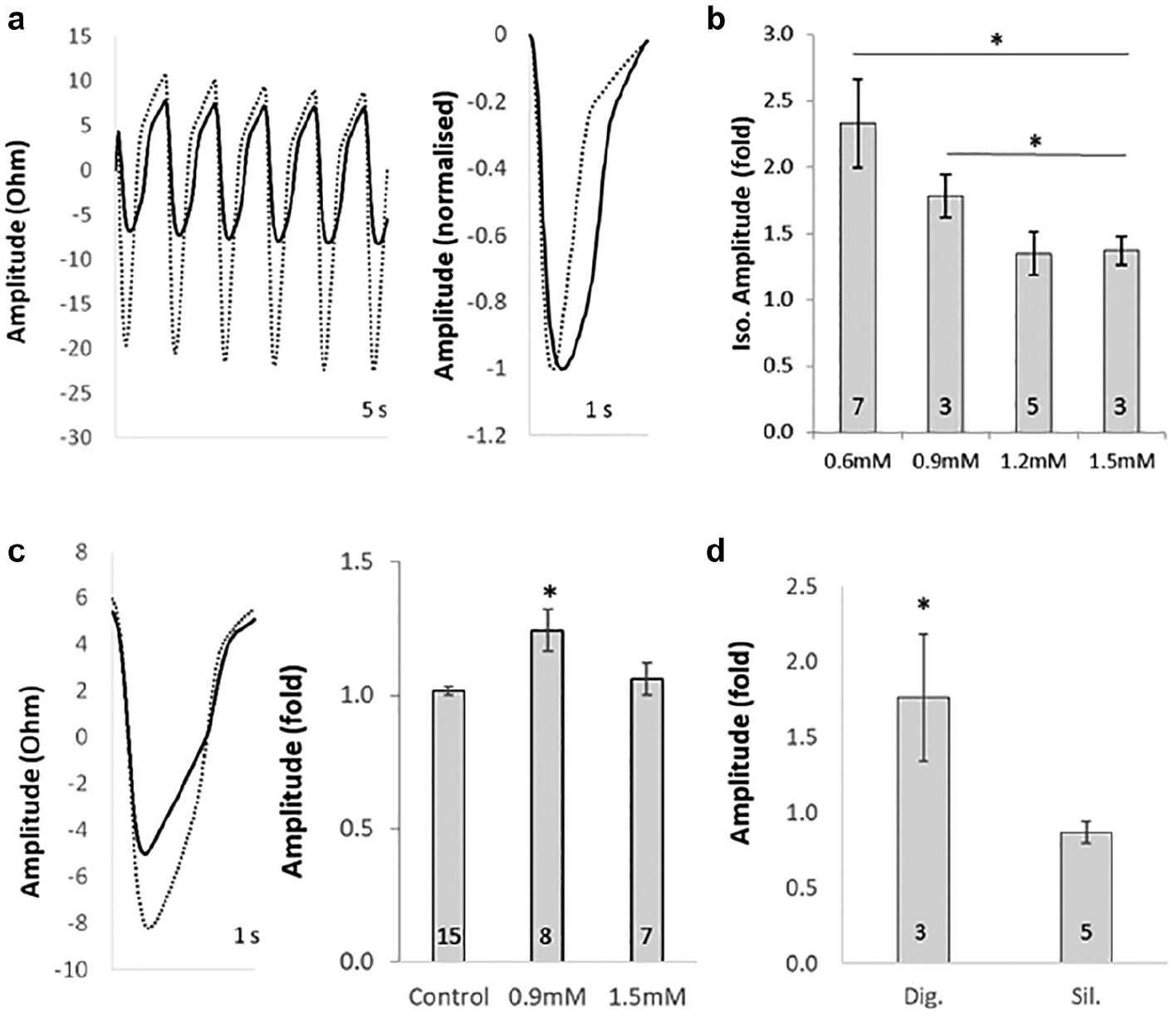
Inotropic responses are modulated by extracellular Ca2+ concentrations and electrical pacing. (
Inotropic Responses Revealed by Electrical Pacing with Reduced Sarcolemmal Ca2+ Influx
Positive inotropes such as isoproterenol typically evoke chronotropic rather than inotropic responses in iPSC-CMs (e.g.,
Similar to isoproterenol, neither digoxin (Na+/K+ ATPase inhibitor) nor sildenafil (phosphodiesterase type 5 inhibitor) elicited increased beat amplitude responses under basal conditions (data not shown). However, under paced/reduced Ca2+ conditions, digoxin evoked a significant inotropic response (1.9- ± 0.5-fold increase in beat amplitude), while sildenafil was without a positive inotropic effect (0.8- ± 0.1-fold). Hence, these conditions confer a degree of mechanistic translation to the native adult CM, given that digoxin is a positive inotrope in adult ventricular myocytes by a non-β-adrenergic mechanism, 26 and that sildenafil is only chronotropic via HCN (hyperpolarization-activated and cyclic nucleotide-gated channels) pacemaker channel activation in isolated myocytes. 27
Detection of EFP in Actively Beating 3D Spheroids
EFP could be recorded from actively beating co-culture spheroids (
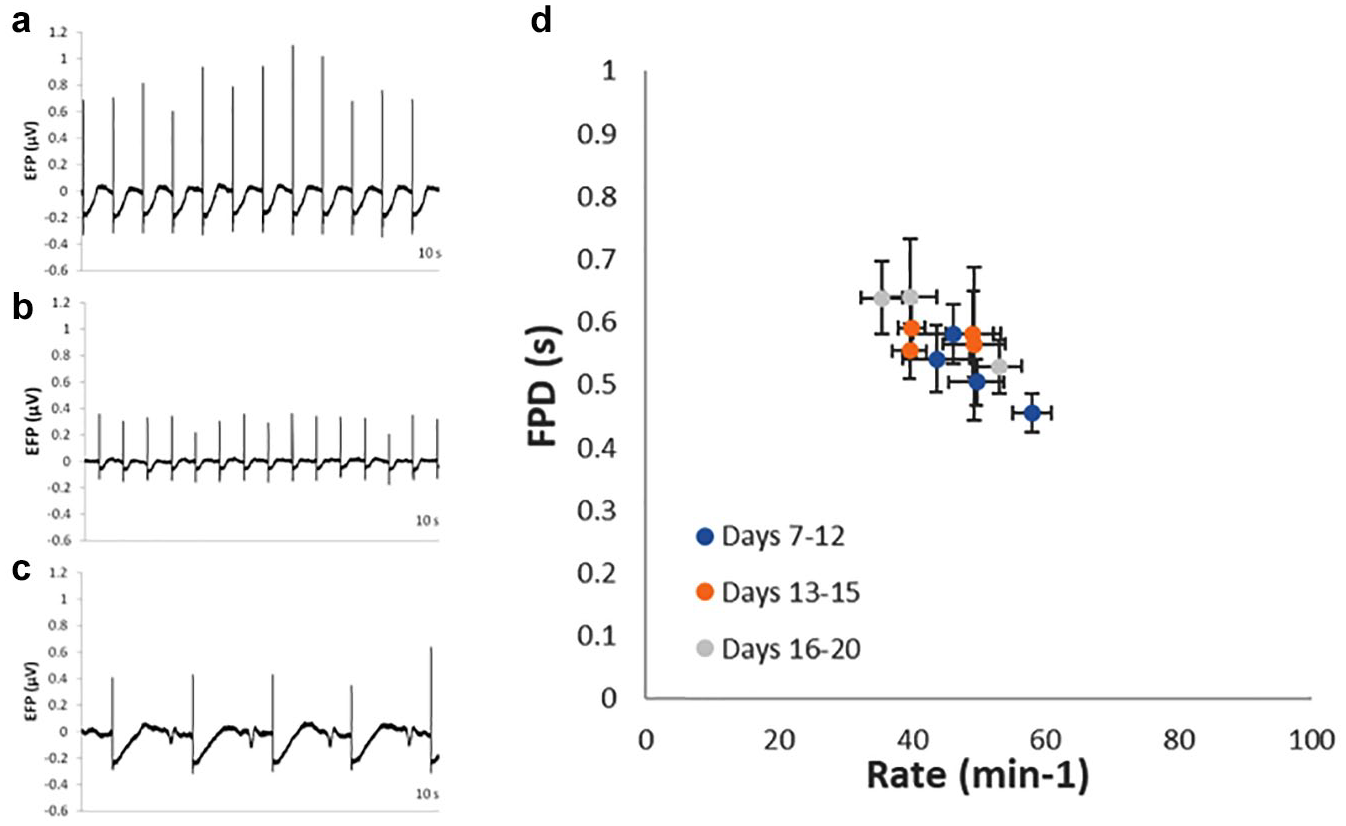
Detection of EFP in actively beating iPSC-CM co-culture spheroids. Representative traces showing EFP signals detected in (a) control, (b) 100 nM verapamil-treated, and (c) 1 µM Bay-K8644-treated co-culture spheroids. FPD (b) shortening and (c) lengthening were apparent, together with (c) DADs in Bay-K8644-treated spheroids. Data from 1037 fibroblast co-culture spheroids, between days 7 and 20 postseeding in 12 separate recording plates, detected in both EFP and impedance modes, demonstrating baseline FPD and beat rate apparent during that period. Mean ± SD, n, and %CV values of these parameters are presented in
Repolarization Mechanisms Differ in iPSC-CM and Co-Culture Spheroids
Co-culture using MEFs significantly affected beat rate and FPD, with reduced beat rate and marginally increased FPD compared with iPSC-CM-only spheroids (BR: 41.3 ± 4.7 bpm vs 52.9 ± 6.4 bpm; FPD: 0.53 ± 0.06 s vs 0.50 ± 0.07 s). These differences were further examined using short (3–10 min) applications of either vehicle, 30 nM E-4031 (hERG channel inhibitor), or 100 µM BaCl2 (inhibitor of several K+ channels, including Kir2.1). Responses to E-4031 were time dependent and arrhythmogenic (
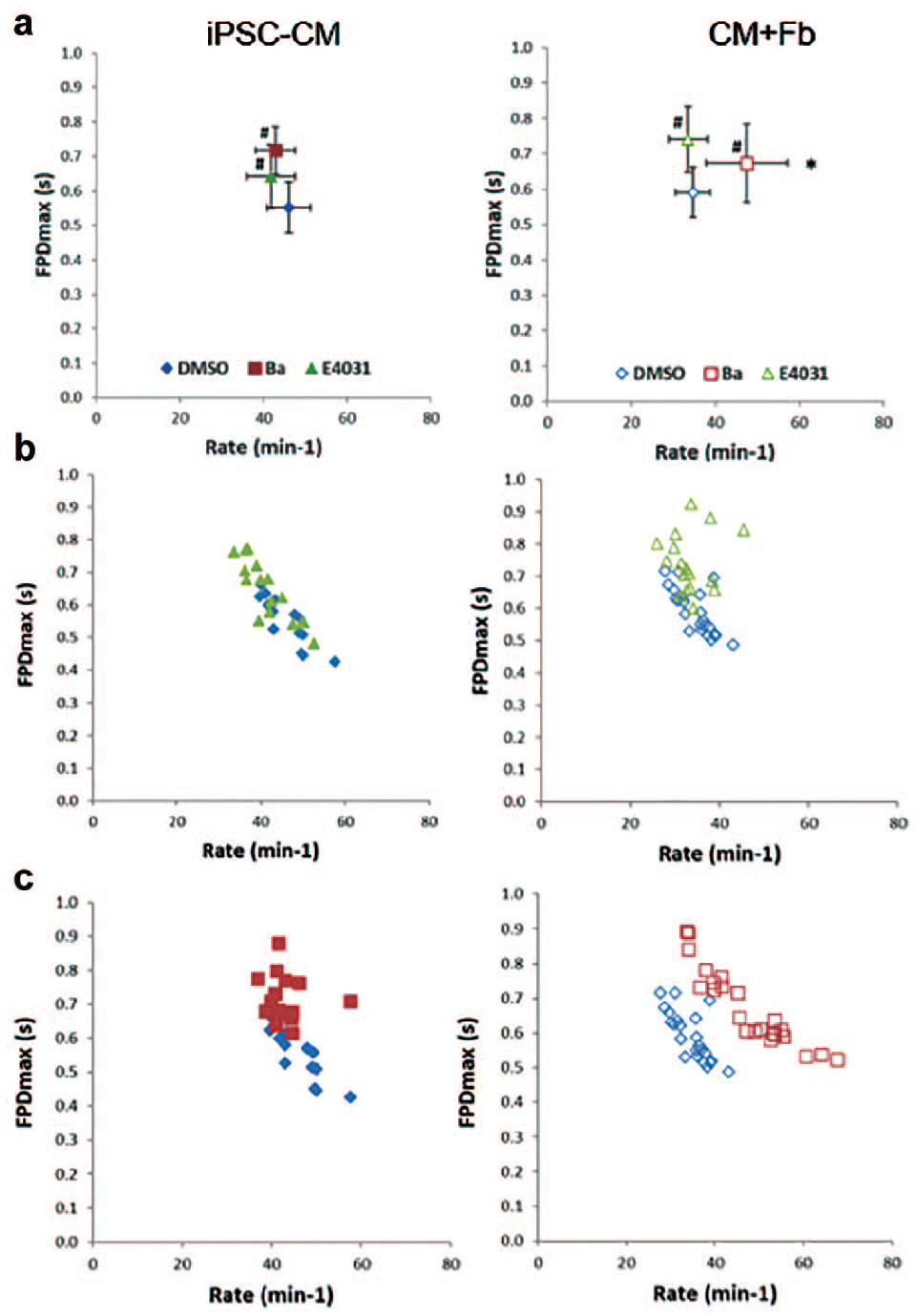
Repolarization mechanisms in iPSC-CM-only and co-culture spheroids. FPD versus beat rate relationships of iPSC-CM-only spheroids (iPSC-CM, closed symbols) and MEF co-culture spheroids (CM+Fb, open symbols) following dosing with DMSO vehicle (blue symbols), 30 nM E-4031 (green symbols), or 100 µM BaCl2 (red symbols) to examine repolarization mechanisms. (
Discussion
In this study, we have profiled more than 1000 iPSC-CM co-culture spheroids in a simple, scalable approach that returned label-free measures of contractility and EFP. An efficient process for dosing with pharmacological agents was demonstrated using a combination of an acoustically dispensed compound source plate and an automated liquid handling platform. Furthermore, these methods are amenable to profiling the 3D co-culture phenotype of iPSC-CMs and reveal that iPSC-CM phenotypes under these conditions can share multiple maturity-associated features usually found in more complex and low-throughput approaches. To validate our approach in relevant areas of CM biology, we demonstrate that spheroid co-culture of iPSC-CMs with fibroblasts of diverse origin increased beat amplitudes, by up to an order of magnitude in some conditions. Inotropic responses usually absent from iPSC-CMs could be revealed by reducing sarcolemmal Ca2+ flux and electrically pacing the spheroids to control beat rate, and the same effect was seen with inotropes of diverse mechanisms. Finally, action potential repolarization apparently involved divergent ion channel ensembles in iPSC-CMs compared with co-culture spheroids, including a BaCl2-sensitive component distinct from the E-4031 response. These results clearly demonstrate that the iPSC-CMs respond phenotypically to the 3D co-culture spheroid environment, and the system provides the throughput to profile further optimizations, such as cell type, long-term optogenetic or electrical pacing, and extracellular matrices. This novel and simple approach recapitulates multiple maturation-associated features of iPSC-CM physiology usually attributed to more technically complex and low-throughput approaches and facilitates further development of spheroid co-culture models for applications as diverse as disease modeling, iPSC differentiation, the cardiovascular therapy area, or drug safety.
Numerous studies seek to enhance the contractility of iPSC-CM constructs into the range of native human myocardium. Engineered tissue approaches frequently incorporate nonmyocyte cells derived from the progenitor cell differentiation protocol as support cells that aid the formation of the tissue constructs, enhance the ventricular phenotype, and modulate contractility both positively and negatively according to their relative proportions.5,7,14,15 Inclusion of additional fibroblasts into these constructs can be associated with increased action potential duration, reduced propagation (conduction velocity), and altered ion channel repertoires.3,6,16,29 In the current study, contractility was profoundly enhanced by primary human fibroblast co-culture (
A key aspect of CM contractility is the precise organization and function of SR Ca2+ handling apparatus. The (lack of) inotropic responses of iPSC-CMs reflect the relative immaturity of the intracellular SR/T-tubule system and a dominant effect of sarcolemmal ion fluxes.
2
However, a degree of functional integration between sarcolemmal and SR Ca2+ pathways was confirmed in co-culture spheroids by the appearance of DADs following Bay K-8644 (
The ensemble of expressed ion channels mediating action potential characteristics is a significant aspect of the mature iPSC-CM phenotype. Repolarization of cardiac action potential involves a number of ionic fluxes, notably IKr (hERG channel) and the contribution of IK1 (Kir2.1 channel) to resting membrane potential. Comparing the effects 30 nM E-4031 (selective hERG blocker) with 100 µM BaCl2 (which also blocks hERG channels at high concentration but is relatively selective for IK1 at concentrations of 50–100 µM
39
) demonstrated unique effects of BaCl2 in the fibroblast co-culture spheroids, specifically an increase in the spontaneous beat rate consistent with depolarization of the resting membrane potential. This effect was apparent in both primary human cardiac fibroblast co-cultures (
Use of iPSC-CMs in a spheroid format of approximately 2000 cells is an efficient approach compared with engineered strips or loops requiring 1 × 10E6 cells per construct, with additional Matrigel and collagen gels.6,14 Spheroids formed by self-assembly of iPSC-CMs, fibroblasts, and gold–iron oxide nanoparticles are mechanically robust and reliably initiate spontaneous beating within 2 days of seeding, which is not always the case for other HT approaches, such as 384-well 2D monolayers. Using automated liquid handling platforms and acoustically dispensed compound source plates facilitated robust experimental design and maintained low DMSO concentrations, a process amenable to generating IC50 data via cumulative dosing. Gold–iron oxide nanoparticles did not interfere with sensor readout or influence beat characteristics. Understanding how the field potential of a beating (moving) spheroid relates to effects of intercellular conduction and individual ionic conductances remains to be determined, although empirical data in this study and others,20,21 including simulations, 44 confirm the validity. Stable EFP measurements were detected at approximately half the rate of impedance measurements, which may relate to these factors. Further developments of the current approach could involve use of automated liquid handling platforms to perform the transfer of assembled spheroids into sensor plates, which may improve the reproducibility of spheroid EFP measurements. Additionally, long-term pacing of the spheroids to further enhance maturity,7,9,10 perhaps via an optogenetic approach implemented in fibroblasts rather than iPSC-CMs, 45 could be explored.
Overall, these novel methods offer a high-throughput, scalable approach to understand the iPSC-CM 3D spheroid functional phenotype and develop diverse applications in tissue engineering, drug discovery, and disease modeling.
Supplemental Material
sj-pdf-1-jbx-10.1177_2472555220975332 – Supplemental material for A Scalable Approach Reveals Functional Responses of iPSC Cardiomyocyte 3D Spheroids
Supplemental material, sj-pdf-1-jbx-10.1177_2472555220975332 for A Scalable Approach Reveals Functional Responses of iPSC Cardiomyocyte 3D Spheroids by Matthew P. Burnham, Rachel Harvey, Rebecca Sargeant, Niels Fertig and Malcolm Haddrick in SLAS Discovery
Footnotes
Acknowledgements
We thank Sonja Stölzle-Feix, Leo Dörr, and Ulrich Thomas for their assistance with the CardioExcyte96 platform.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.P.B., R.S. and M.H. are/were employees of AstraZeneca and potentially own stock and/or stock options in the company. N.F. is founder and CEO of Nanion Technologies GmbH.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
