Abstract
Conventionally, manual patch-clamp electrophysiological approaches are the gold standard for studying ion channel function in neurons. However, these approaches are labor-intensive, yielding low-throughput results, and are therefore not amenable for compound profiling efforts during the early stages of drug discovery. The SyncroPatch 384PE has been successfully implemented for pharmacological experiments in heterologous overexpression systems that may not reproduce the function of voltage-gated ion channels in a native, heterogeneous environment. Here, we describe a protocol allowing the characterization of endogenous voltage-gated potassium (Kv) and sodium (Nav) channel function in developing primary rat cortical cultures, allowing investigations at a significantly improved throughput compared with manual approaches. Key neuronal marker expression and microelectrode array recordings of electrophysiological activity over time correlated well with neuronal maturation. Gene expression data revealed high molecular diversity in Kv and Nav subunit composition throughout development. Voltage-clamp experiments elicited three major current components composed of inward and outward conductances. Further pharmacological experiments confirmed the endogenous expression of functional Kv and Nav channels in primary cortical neurons. The major advantages of this approach compared with conventional manual patch-clamp systems include unprecedented improvements in experimental ease and throughput for ion channel research in primary neurons. These efforts demonstrated feasibility for primary neuronal ion channel investigation with the SyncroPatch, providing the foundation for future studies characterizing biophysical changes in endogenous ion channels in primary systems associated with disease or development.
Keywords
Introduction
In a mature nervous system, ion channel activity is typically associated with intracellular signaling cascades that profoundly influence neuronal properties such as dendritic outgrowth, electrical connectivity, and synaptic transmission. 1 The expression and function of voltage-gated potassium (Kv) and sodium (Nav) channels are fundamental aspects of neural maturation and are subject to continual developmental modulation. 2 Given their importance in neuronal physiology and maintaining effective cellular communication, dysfunction in Kv and Nav channels can lead to profound pathological implications, such as epilepsy, ataxia, psychosis, multiple sclerosis, neurological disorders, and autism spectrum disorders.1,3 Consequently, these channels have been targeted for their therapeutic potential in treating these disorders and for pain management. However, the development of novel ion channel drug agents remains underexploited due to several factors. For one, the enormous molecular diversity in subunit composition leads to considerable ion channel heterogeneity with tissue-specific distribution in vivo, therefore making the identification of native conductances especially difficult in heterologous cell models. This is challenging during the early phases of drug discovery as many high-throughput screen (HTS) methodologies rely on recombinant homomeric expression systems that fail to reproduce the function of heteromeric voltage-gated ion channels in their native environment, such as the neuron.
The voltage-gated potassium channel family, for example, comprises a diverse group of protein complexes and is expressed in virtually all excitable cells, notably neurons. The pore-forming subunits of the Kv alone are encoded by up to 40 different genes.1,3 The opening and closing of the Kv channels controls ionic permeability, which polarizes and hyperpolarizes the membrane potential, ultimately impacting the propagation of voltage across the neuron. Mounting evidence supports the continual modulation of embryonic neuronal excitability throughout development. Developmental changes in excitability can be categorized into two periods: before and after formation of synaptic connectivity. These changes are in part regulated by K+ conductances elicited by Ca2+-activated Kv channels, transient K+ currents, and delayed rectifier K+ family channels that contribute to membrane potential repolarization in cell bodies that will support the diverse functions mediated by mature neurons within a dynamic system.4,5 On the other hand, the diversity of Nav channels is much less compared with Kv, with only 10 channel genes identified in mammals. 6 Nav channels influence action potential initiation and propagation in neuronal cells.7–10 In addition to promoting neural activity and embryonic cell health, Nav channels contribute to development processes such as axonal guidance, arborization, synapse formation, and refinement. 11 Through these mechanisms, Nav channels help establish and maintain functional neural circuits throughout maturation. Despite studies supporting an increase in Na currents in developing cortical neurons, 12 changes in their channel properties and molecular components are poorly understood.
Although the conventional manual patch clamp is the gold standard for assaying native ion channel function in neurons, these approaches are extremely labor-intensive, yielding low-throughput results, and are therefore not amenable for compound profiling efforts during the early stages of drug discovery. To bridge this gap, we describe a protocol allowing the characterization of endogenous neuronal voltage-gated Kv and Nav channel function in developing primary cortical cultures with the SyncroPatch platform, allowing investigations at a significantly improved throughput compared with conventional manual patch-clamp approaches. In this study, we compared the ontogeny of Kv and Nav channel expression and function in primary rat cortical neurons at different developmental time points using three complementary methods: (1) anatomical visualization of synaptic proteins and electrophysiological detection of network activity to confirm neuronal maturation and emergence of functional synaptic networks, (2) pharmacological manipulation of Nav and Kv channels using the SyncroPatch, and (3) quantification of ion channel mRNA expression levels by qPCR and assessment of their relationship relative to different stages of neuronal maturation in vitro.
Materials and Methods
Cell Culture
Rat cortical cultures were prepared from E18 rat embryos (BrainBits, Springfield, IL) dissociated by trituration after papain digestion as per the manufacturer’s protocol. The cells were resuspended in the NBActiv1 media (BrainBits) supplemented with 5% fetal bovine serum and 20 µg/mL laminin (Sigma-Aldrich, Natick, MA). Neurons were seeded at 80,000 cells/well on a 48-well microelectrode array (MEA) plate (Axion BioSystems, Atlanta, GA) covered with adhesion promoting molecules (PEI). For experiments on the SyncroPatch 384PE, dissociated neurons were plated in 100 mm of poly-
Solutions and Compound Preparation
Tetrodotoxin (TTX) was purchased from Tocris; all other chemical reagents were purchased from Sigma-Aldrich. The internal recording solution for Nav experiments was as follows (in mM): 110 CsF, 10 NaCl, 10 CsCl, 10 EGTA, 10 HEPES, 0.2 ATP, and 2 GTP (pH 7.2, adjusted with CsOH and osmolarity adjusted to 280 mOsm). Kv experiments were done with an internal solution containing (in mM) 110 KF, 10 NaCl, 10 KCl, 10 EGTA, 10 HEPES, 0.2 ATP, and 2 GTP (pH 7.2, adjusted with KOH and osmolarity adjusted to 280 mOsm). The external recording solution for Nav pharmacology consists of (in mM) 140 NaCl, 4 KCl, 5 CaCl2, 1 MgCl2, 5 glucose, and 10 HEPES (pH 7.4, adjusted with NaOH and osmolarity adjusted to 298 mOsm). One hundred nanomoles of TTX was added to the external solution to block Nav currents in Kv pharmacological experiments. Seal enhancer solution was prepared by supplementing the external solution with 5 mM CaCl2. Chip fill solution for all experiments includes the following composition (in mM): 140 NaCl, 4 KCl, 3 MgCl2, 5 glucose, and 10 HEPES (pH 7.4, adjusted with NaOH and osmolarity adjusted to 290 mOsm). TTX was made as 1 mM stock solution in 20 mM sodium citrate and tetracaine in DMSO. Stock compound solutions were serially diluted into their corresponding vehicle, and further diluted to the desired final concentrations in the external solution. The final vehicle content is 0.1%. Tetraethylammonium (TEA) and 4-Aminopyridine (4-AP) were prepared directly in external recording solution to the appropriate concentrations prior to experiments. All recording solutions used in the current–voltage experiments were similar to Kv recordings without TTX addition.
MEA Recordings
On days in vitro (DIVs) 9, 14, and 21, plates were placed into the Maestro amplifier and allowed to equilibrate for at least 10 min. After this, the spontaneous network activity was recorded for a period of 10 min. Recordings were made from 48 wells across 16 electrodes/well. Features relating to firing rate, bursts, and network bursts were extracted in the AxISMetric plotting tool and organized into a well-level data set. Bursts consist of a densely packed spike train, defined as a minimum number of spikes (i.e., 10) within a single burst and the maximum distance (i.e., 100 ms) between two spikes belonging to the same burst. Network bursting activity of the cultures was detected by averaging the number of bursts over all the active sites every minute throughout the course of the recordings (10 min).
Immunocytochemistry
Cortical cell cultures grown in 96-well plates were fixed in a solution containing 4% paraformaldehyde and 4% sucrose in phosphate-buffered saline (PBS, pH 7.4) at 37 °C for 15 min at room temperature (100 µL/well). Fixed cells were gently washed three times with 100 µL/well PBS and stored sealed and protected from the light at 4 °C in 200 µL of PBS until all time points were collected. Fixed cells were permeabilized with 0.1% Triton X-100 in PBS at room temperature for 5 min (100 µL/well). Cells were next blocked with 3% bovine serum albumin (BSA) in PBS for 1 h at 37 °C. Cells were incubated with primary antibody mix for 1 h at 37 °C in 1% BSA, 0.05% Tween-20, and PBS. Primary antibody mix included chicken anti-MAP2 (1:2000, Novus Biological NB300-213, Littleton, CO), rabbit anti-synapsin-1 (1:500, Novus Biological NB300-104), and mouse anti-PSD95 (1:500, Novus Biological NB300-556). After primary antibody incubation. cells were gently washed three times with 100 µL/well PBS before the addition of Alexa Fluor secondary antibodies (1:500, Thermo Fisher Scientific) goat anti-mouse 488, donkey anti-rabbit 546, and goat anti-chicken 647, again 1 h at 37 °C in 1% BSA, 0.05% Tween-20, and PBS. Cultures were gently washed three times with 100 µL/well Hoechst 33342 (Thermo Fisher Scientific), 2 µM in PBS, and stored sealed and protected from the light in the same.
High-Content Imaging
Image acquisition was performed using a CellInsite CX7 HCS Reader (Thermo Scientific). Images were acquired using an Olympus 40× LUC Plan FL objective (0.6 NA) and Photometrics X1 CCD camera with 2 × 2 pixel binning. For each well examined, nine unique fields of view were sampled. Within each field, matched fluorescent images of Hoechst-labeled nuclei, MAP2-Alexa Fluor 647-immunolabeled neurons, synapsin-Alexa Fluor 546-labeled presynapse marker, and PSD95-Alexa Fluor 488-labeled postsynapse marker were acquired. In each channel, CCD camera exposure times for each channel were determined by surveying wells across multiple time points. Exposure times were held constant for all time points.
SyncroPatch Recordings
All recordings were conducted in the whole-cell configuration using the SyncroPatch 384PE incorporated into a Biomek FX pipetting robot (Beckman Coulter, Jersey City, NJ). Medium- and high-resistance, one-hole-per-well chips with varying catch pressure (–140 to –200 mBar) were tested to determine the optimal recording condition and to achieve whole-cell configuration. Voltage protocols were constructed using PatchControl 384 (Nanion Technologies, Livingston, NJ). Currents were evoked using a voltage step protocol from the holding potential of –90 mV for 10 ms to the test potential of –30 mV for 10 ms for Nav or +50 mV for Kv. The sweep interval was set to 1 s and the protocol was run for 240 s of the compound incubation time. One concentration of compound was applied to each cell and the concentration–response curve was calculated across the whole plate. Data were analyzed and IC50 values were calculated from average fits from the individual plots using DataControl 384. For current–voltage plots, points were plotted as mean peak amplitude ± SEM.
qPCR Analysis
Cortical neurons were harvested at days 7, 14, and 21 and total RNA was extracted with the RNeasy Mini Kit (Qiagen, Germantown, MD) according to the manufacturer’s protocol. Fifty nanograms of RNA was reverse transcribed and qPCRs were carried out with the iTaq Universal SYBR Green One-Step Kit (Bio-Rad Labs) by using the Bio-Rad CFX-96 Touch Real-Time PCR System (Bio-Rad Labs, Portland, ME). This material was used for qPCR against a curated predesigned panel of neuronal ion channel (SAB Target List) gene expression assay 10034376 (Bio-Rad Labs;
Results
Developmental Characterization of Acutely Dissociated Primary Rat Cortical Neurons: Immunocytochemical (ICC) Staining, MEA Recordings, and Voltage-Gated Ion Channel Expression (qPCR)
Primary cortical neurons were immunocytochemically characterized to evaluate the expression of neuronal markers across different developmental stages (
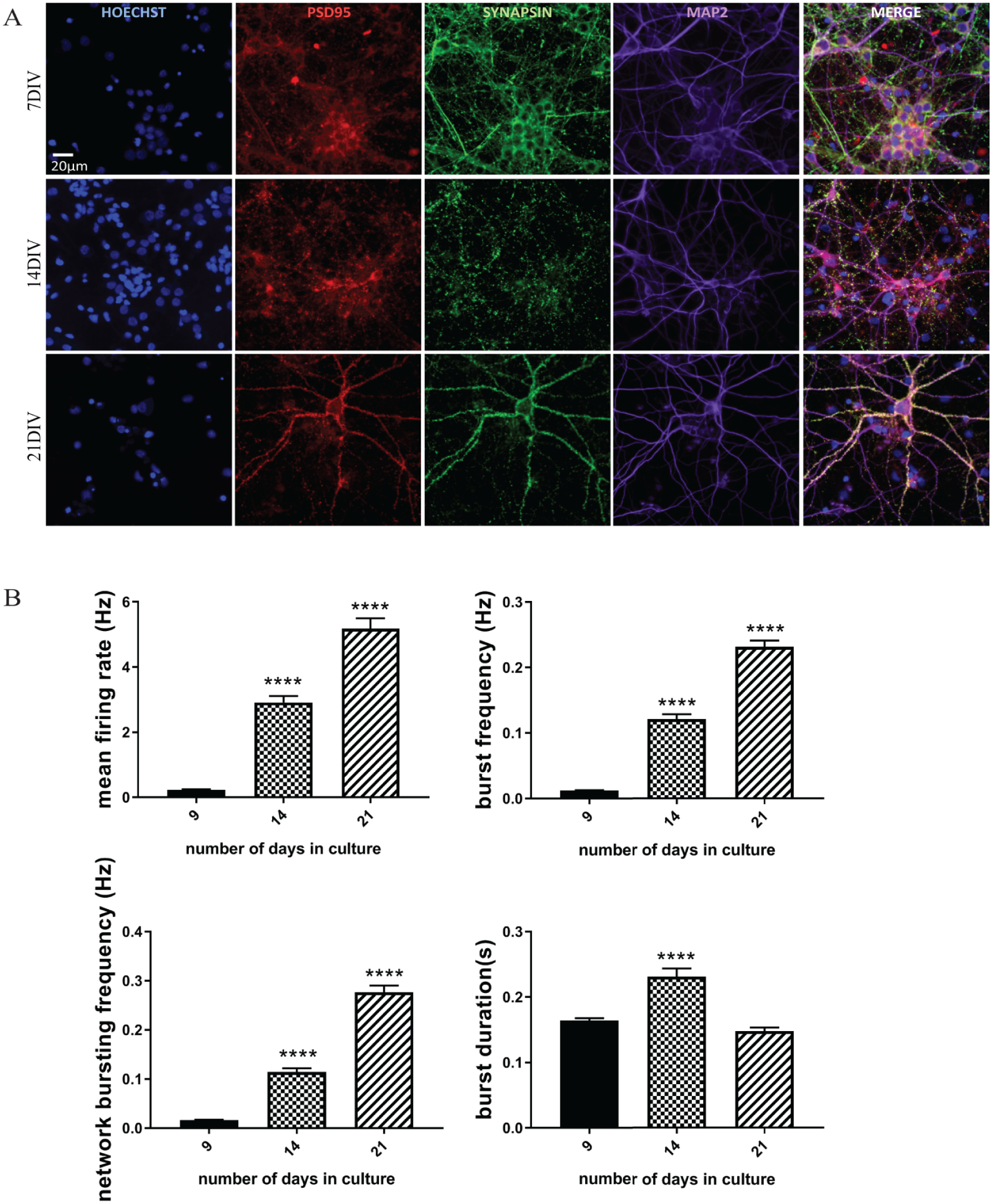
Developmental profile of synaptic marker expression and electrophysiological activity in cortical cultures. (
Recording Nav and Kv Currents from Primary Cortical Neurons at DIVs 7, 14, and 21
To assess the potential relationship between ion channel function and in vitro neural development, automated patch-clamp technology was applied to dissociated neurons derived from rat cortex at different culture time points. While the SyncroPatch has been shown to reliably record Nav and Kv channel activity in recombinant systems,
16
to our knowledge its application to primary cortical neurons has never been reported before. In this study, the optimal parameters for cell catching and whole-cell break-in were identified and 384-well chips with varying resistance as determined by cell-catching hole sizes were tested. A hold pressure of –50 mBar, V catch of –20 mV, and two 1000 ms negative suction pulses of –180 and –160 mBar in a medium-resistance chip (5–8 MΩ) resulted in successful patching of primary cortical neurons at a density of 4000 cells/well. For quality control criteria, several parameters were established, including cell-catching seal resistance (RCATCH), seal stability (RSEAL), cell capacitance, and baseline peak current amplitude (
Table 1
). To achieve high-quality recordings, whole-experiment RSEALs were set at >200 MΩ. Cortical neurons recorded under these conditions showed robust voltage-activated Na+ and K+ currents in our I-V experiment, with a success rate of ~40% for cells that have a seal resistance of >200 MΩ, cell capacitance of >10 pF, and catch resistance of >10 MΩ at the end of the experiment. Representative raw traces taken on cortical DIV 14 are shown in a color-coded overview of all 384 wells (
Quality Control Criteria for Automated Patch Clamp.

Acceptance criteria for automated patch clamp.

Current–voltage experiment using primary cortical neurons on DIV 14 on the SyncroPatch 384PE. (
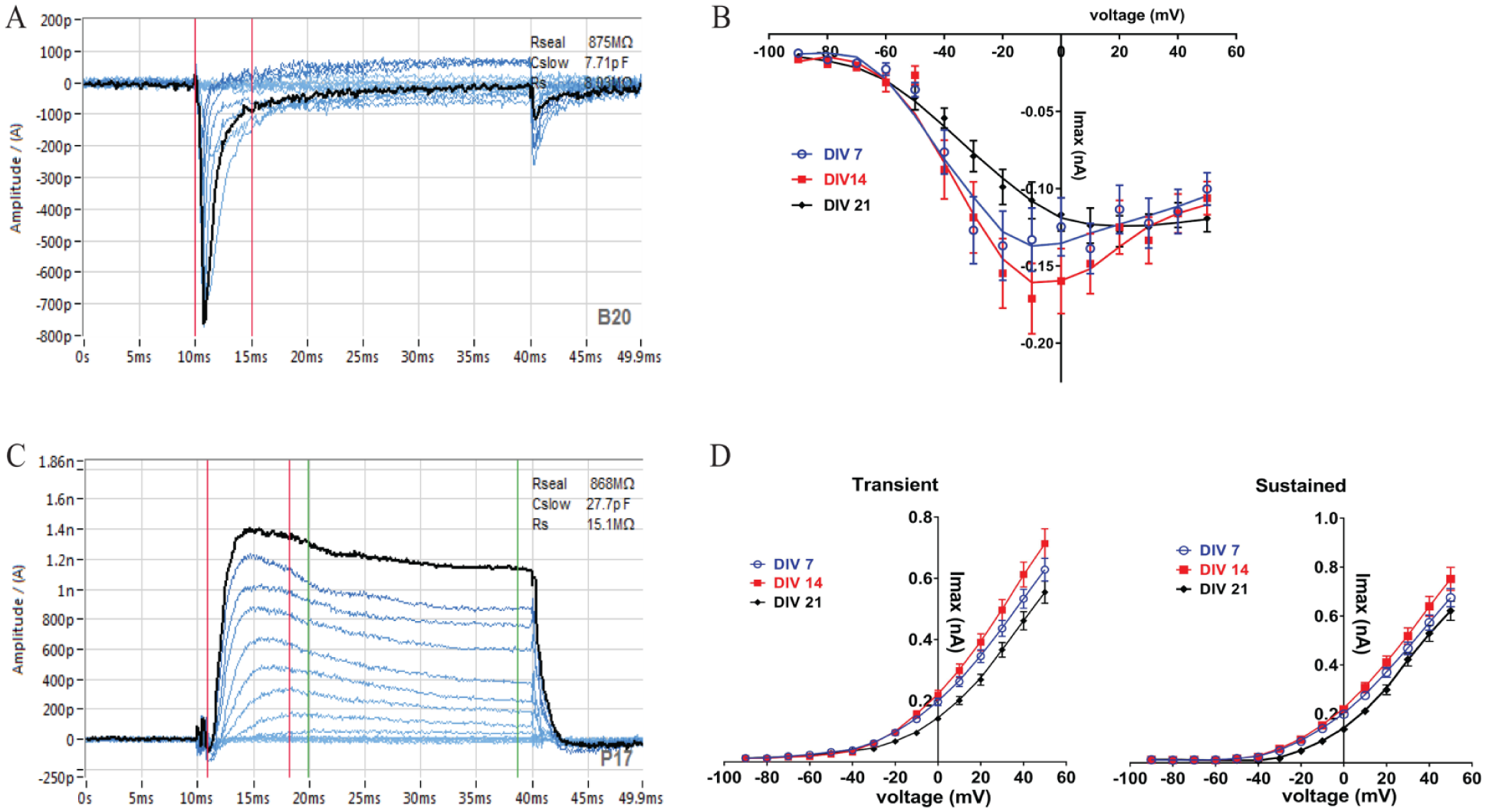
Current–voltage relationship of Nav and Kv currents recorded on the SyncroPatch 384PE. Currents were evoked using a voltage step protocol from the holding potential of –90 to +50 mV with 10 mV increments. Representative traces show inward Na+ (
Modulation of Nav and Kv Channels with Reference Inhibitors
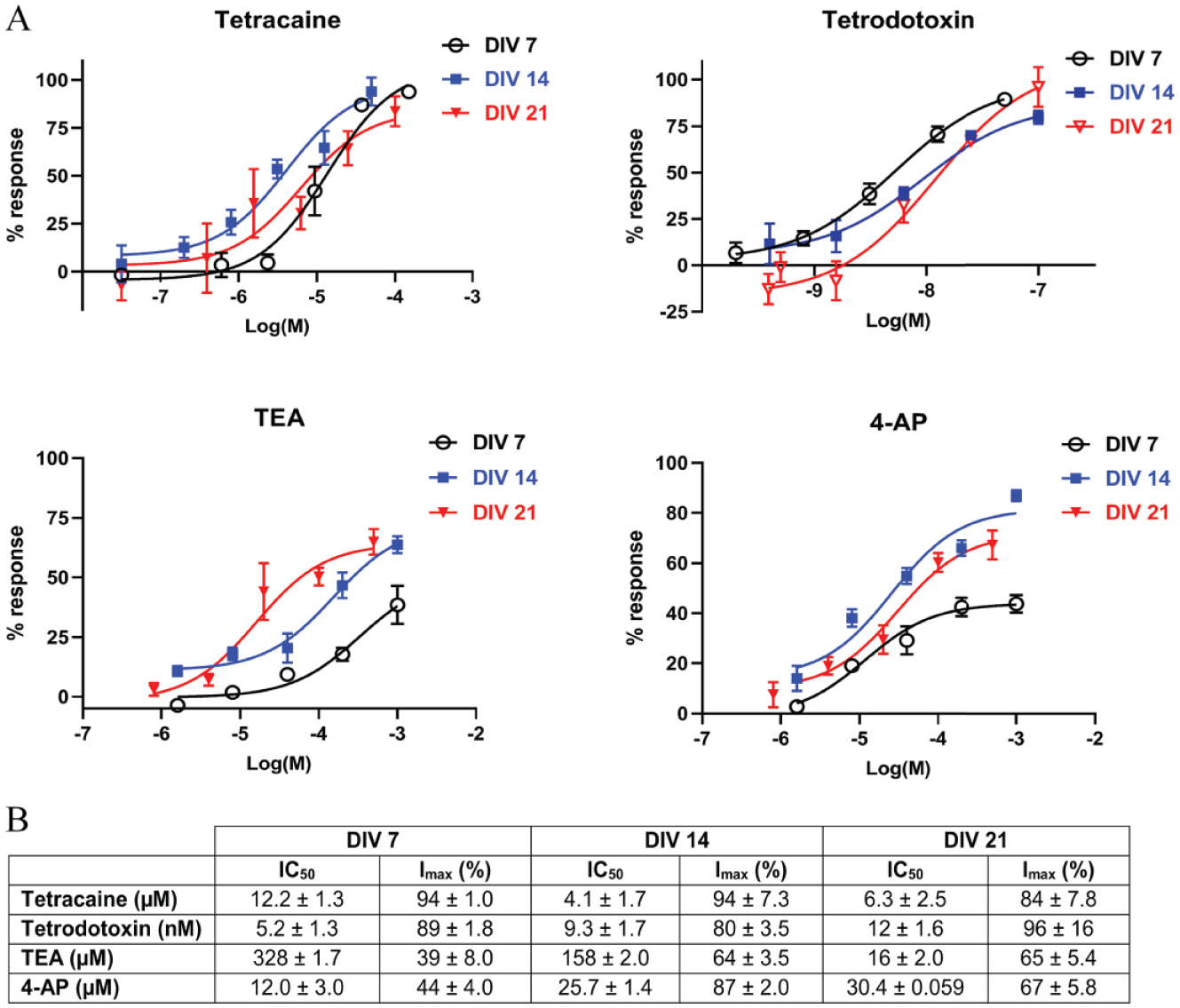
Gene expression data revealed high molecular diversity in Kv and Nav subunit composition across all time points (
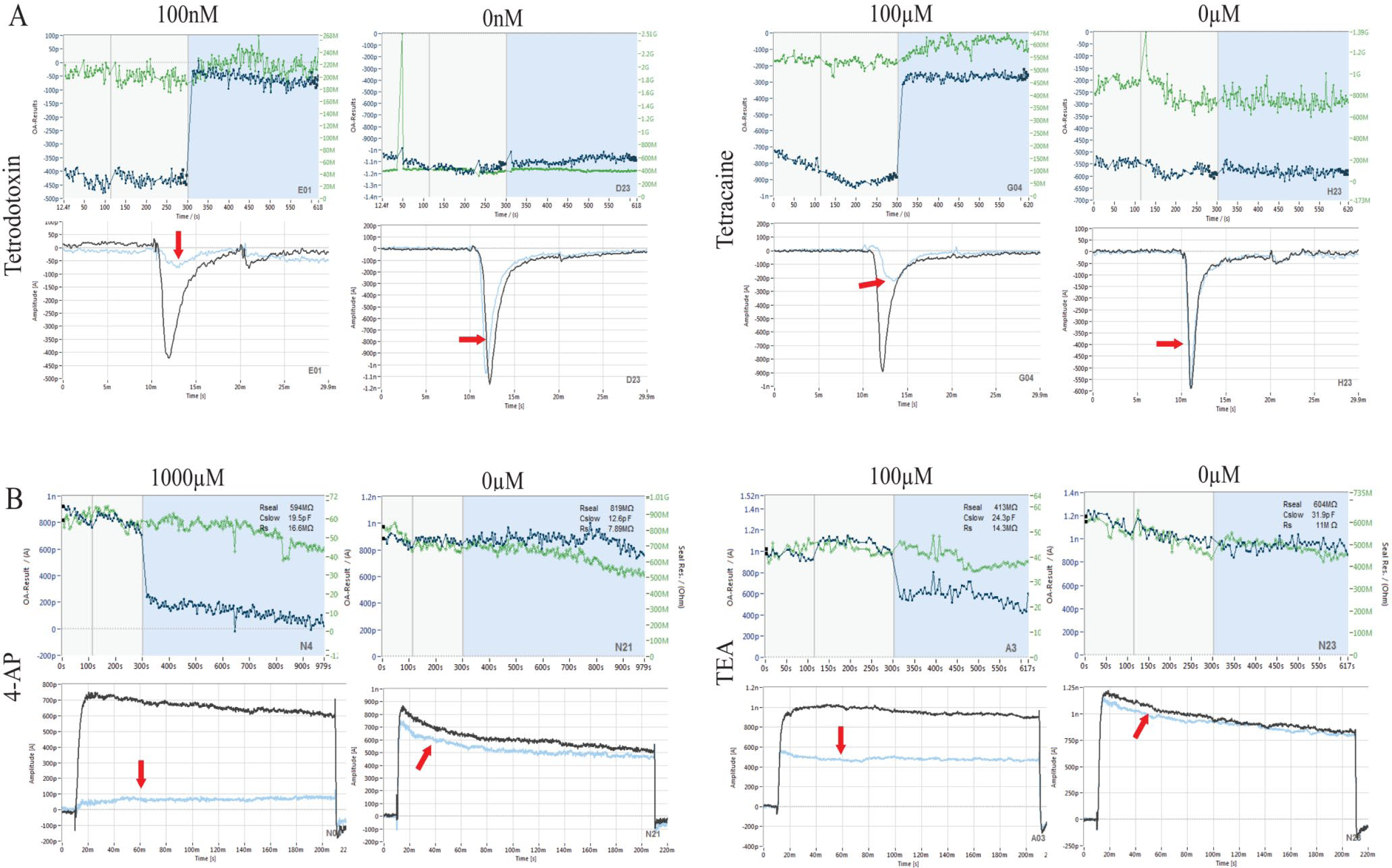
Analysis of TTX/tetracaine block of Nav currents (
(
Discussion
Our findings are in agreement with the literature that the expression and localization of neuronal proteins such as PSD95 and synapsin indicate synaptic connections and dendritic development, correlating well with neuronal maturation. 17 In addition to extensive morphological and cytological differentiation, a hallmark of an adult nervous system is the development of neuronal excitation. 18 MEA recordings demonstrated random spiking activity beginning around days 7–9 in culture, developing into strong spiking and burst activity up to DIV 21, indicative of organized synaptic connectivity present in mature cultures. Next, we aimed to identify and characterize some of the different neuronal Kv and Nav conductances endogenously expressed over time in these mature, cultured cortical neurons. Voltage-clamp depolarization experiments elicited at least three major current components. The current response showed rapidly activating inward Na+ currents followed by outward K+ conductances that consist of a transient and maintained component. Na+ and K+ current averages across wells differ from previous reports from single-cell manual patch-clamp recordings with a noticeable variability in amplitude and pharmacological properties (blocker IC50 values).19–22 These observations are expected as ion channel densities, as well as maximal conductances of ion currents, can be highly variable in a heterogenous neural population consisting of different neuronal and glial cells. 23 To elaborate, the indiscriminate patching in the SyncroPatch, combined with the presence of astrocytes shown to express voltage-dependent conductances, 24 may lead to contaminating currents from nonneuronal cells, therefore confounding the analysis of neuronal Kv and Nav currents. The inclusion of antimitotic drugs such as cytosine arabinoside has been shown to keep glial cell proliferation to a minimum. Therefore, an important next step will be to repeat these pharmacological experiments under these culture conditions to examine potential differences in ion channel physiology in a purer neuronal culture. Additional explanations for these discrepancies might be due to differences in the experimental setup between the platforms. This includes potential differences in cell preparation protocols, patching configuration/holding potential selection, and the dosing paradigms (within vs across wells). Manual patch-clamp recordings are done on selected adherent cells in which the larger contact area between the cell and the platform may lead to an increase in cell mass, as opposed to cells in suspension that randomly attach to the substrate with suction on the SyncroPatch. Assuming that channel density remains the same, cells with a larger surface area from the manual patch recordings will produce larger peak whole-cell currents. Additionally, the achievement of gigaseals (>1 GΩ) in the manual approach minimizes background noise, which may lead to higher-resolution recordings. Nonetheless, given the delicate nature of these primary neurons, assigning a less stringent seal resistance cutoff of >200 MΩ is congruent with previous SyncroPatch studies investigating human induced pluripotent stem cell (iPSC)-derived cardiomyocytes. 3 Despite a noticeable decrease in success rates in these studies compared with experiments in stable cell lines, our data are consistent with previous reports looking at primary T cells (~12%) using automated platforms. 16 Nonetheless, this is an exceptional improvement in throughput from single-cell patch recordings of primary neurons. To elaborate, an overall average of ~40 primary neurons can be successfully recorded from a single run with an experiment time of 30–40 min for a 5-point concentration–response curve, which may take up to a month to complete in manual patch-clamp approaches.
One of the aims of this recent work has been to establish a methodology that leverages the SyncroPatch, cell biology (ICC), and electrophysiological (MEA) and genetic (qPCR) approaches to provide a foundation to investigate the molecular components of voltage-gated channels on the developmental regulation of voltage-dependent K+ and Na+ currents in primary neurons. Although the largest and most prevalent inward and outward currents were recorded at DIV 14, these changes did not show a clear correlation with the mRNA expression levels of any particular Nav and Kv subunit. Functional confirmation studies with different broad-spectrum Nav blockers showed consistent IC50 values with partial to complete suppression of voltage-gated currents (~80%–95% reduction) at all culture time points. The noticeable increase in sensitivity to Kv blockers in older cultures compared with DIV 7 could be due to expression-level changes in TEA- and 4-AP-sensitive potassium channels across DIV. For example, Wulff and Zhorov described K+ currents generated by subunit arrangement in Kv1-type (Kcna1, Kcna2, and Kcna5), Kv2-type (Kcnb1 and Kcnb2), Kv3-type (Kcnc1 and Kcnc2), Kca-type (Kcnma1), and Kir-type (Kcnj5) potassium channels as highly sensitive to 4-AP and TEA. 25 Apart from Kcnj5 and Kcnc1, the current data indicate a developmental increase in mRNA expression for all the aforementioned Kv channels. This overall net increase in Kv subunit expression from DIV 7 to DIV 21 may explain the observed change in channel blocker sensitivity over time. Additionally, despite a noticeable increase in Nav current amplitude from DIV 7 to DIV 14, a reverse in this trend was observed at DIV 21. Interestingly, these results differed from rat cortical slice recordings taken at different postnatal ages, which demonstrated a significant increase in Nav currents with maturation. 12 Thus, to investigate these discrepancies, an important future work would be to apply the experimental framework described in this study with cortical neurons harvested from postnatal rats at different developmental stages.
Establishing a developmental link between ion channel function and neuronal maturation constitutes a considerable challenge as primary cortical cultures are heterogeneous in cell type (excitatory vs inhibitory) as well as in endogenous ion channel subunit assembly and composition, leading to broad channel diversity with unique biophysical and pharmacological properties. 25 Despite a clear correlation between network-level electrophysiological recordings in the MEA over time, there was a lack of developmental changes in Nav/Kv current densities from I-V experiments. This could be in part due to the prominent role of excitatory/inhibitory synaptic transmission, rather than Nav/Kv function, in the development of network bursting activity observed in MEA recordings. As such, it would be very important for future applications of this approach to focus on the developmental aspects of excitatory/inhibitory synaptic transmission in primary cortical neurons and how this may relate to developmental changes in glutamate and gamma-aminobutyric acid (GABA) ion channels. Furthermore, the limited availability of selective modulators for specific homotetramers/heteromultimers may prohibit the functional characterization of certain ion channels through pharmacological manipulations in a primary culture model. However, the ability to autopatch primary neurons allows for a higher level of physiological relevance as they innately recapitulate the molecular makeup consisting of diverse ion channel populations, receptors, second messenger expression, kinase modulation, and other accessory proteins that may be absent in recombinant systems. Additionally, the conservation of these important molecular characteristics provides a future opportunity to interrogate the multifaceted interaction between endogenous G-protein-coupled receptor (GPCR) signaling and ion channel function. Applying this technology to primary neurons is uncharted territory in GPCR–ion channel drug discovery that can potentially pave the way for new drug candidates and facilitate the exploration of novel chemical matter.
In summary, we hope that these efforts will provide the foundation for future studies characterizing the developmental roles of endogenous ion channels in primary neurons and facilitate new frontiers in ion channel target drug discovery and development.
Supplemental Material
Toh_Supp_Fig_1 – Supplemental material for Application of High-Throughput Automated Patch-Clamp Electrophysiology to Study Voltage-Gated Ion Channel Function in Primary Cortical Cultures
Supplemental material, Toh_Supp_Fig_1 for Application of High-Throughput Automated Patch-Clamp Electrophysiology to Study Voltage-Gated Ion Channel Function in Primary Cortical Cultures by May Fern Toh, Julie M. Brooks, Tim Strassmaier, Rodolfo J. Haedo, Corey B. Puryear, Bruce L. Roth, Kosalvisal Ouk and Sokhom S. Pin in SLAS Discovery
Supplemental Material
Toh_Supp_Table_1 – Supplemental material for Application of High-Throughput Automated Patch-Clamp Electrophysiology to Study Voltage-Gated Ion Channel Function in Primary Cortical Cultures
Supplemental material, Toh_Supp_Table_1 for Application of High-Throughput Automated Patch-Clamp Electrophysiology to Study Voltage-Gated Ion Channel Function in Primary Cortical Cultures by May Fern Toh, Julie M. Brooks, Tim Strassmaier, Rodolfo J. Haedo, Corey B. Puryear, Bruce Roth, Kosalvisal Ouk and Sokhom S. Pin in SLAS Discovery
Footnotes
Supplemental material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
