Abstract
Lack of efficacy and poor safety outcomes are deemed to be the greatest causes of clinical failure of novel therapeutics. The use of biomarkers that give accurate information on target engagement, providing confidence that pharmacological activity in the target organ is being achieved, is key in optimizing clinical success. Without a measurement of target engagement, it can be very difficult to discern the basis for any lack of efficacy of a drug molecule within the pharmaceutical industry.
Target engagement can be measured in both an in vitro and in vivo setting, and in recent years imaging measurements have been used frequently in drug discovery and development to assess target engagement and receptor occupancy in both human and animal models.
From this perspective, we assess and look at the advancements in both in vivo and ex vivo imaging to demonstrate the enormous potential that imaging has as an application to provide a greater understanding of target engagement with a correlative therapeutic impact.
Introduction
The year 2018 was very productive for the pharmaceutical industry. A record 59 drugs were approved by the Food and Drug Administration (FDA) in 2018, 1 and data 2 suggest that success rates in phase III trials have increased from less than a one-in-two chance at the beginning of the decade to an almost two-in-three chance. There has been a lot of research into the reasons for clinical failure and a strategy for improvement.
The lack of safety and efficacy is thought to be the greatest cause of clinical failure. A review performed by AstraZeneca with a goal to understanding the factors impacting the progression of their projects created a “5R framework.” 3 This framework and the implementation of it helped them to change their approach toward the research and development process. The 5R framework encompasses the need to focus on the right target, the right safety, the right patients, the right tissue, and the right commercial potential. It has been claimed that the use of this concept as a guide in drug discovery and development has significantly increased the probability of clinical success. 4 In addition to the 5R framework developed by AstraZeneca, Pfizer also performed a review to uncover the systematic learnings that can be applied to improve compound survival. 5 Not only was it found that most failures were caused by a lack of efficacy, but also in a large number of cases (43%) it was not possible to conclude whether the mechanism had been tested adequately. A key finding was that an integrated understanding of the fundamental pharmacokinetic/pharmacodynamic principles of exposure at the site of action, target binding, and expression of functional pharmacological activity determines the likelihood of candidate survival. 6
Biomarkers enable a direct correlation between target engagement and measurements of drug efficacy and/or toxicity. For instance, if full target occupancy is confirmed for a drug in vivo and the drug fails to produce an expected therapeutic effect, then the target and mechanism would be properly tested and invalidated for the intended clinical indication. Without a measurement of target engagement, however, it can be very difficult to discern the basis for a drug’s lack of efficacy. 7 Questions arise as to whether the target was invalid or if the drug candidate simply failed to fully engage the target in vivo. Therefore, the use of robust and sensitive biomarkers not only is a key element in ensuring the clinical success of novel therapeutic agents, but also is vital in preclinical studies where they can provide information that is critical in understanding the biological effects of the candidate compound.
The process of understanding the target engagement of a therapeutic compound is often first performed in in vitro cell culture models. This is measured traditionally using a simple substrate–product assay. 8 These traditional methods can often become problematic when the molecules that are being measured are not uniquely modified by the target enzyme of interest. Radioligand assays, for example, have historically been run to confirm ligand binding to receptors in cells, 9 and more recently, several chemoproteomic methods have been shown to measure target engagement in cells. These are particularly used where targeted therapies and kinases have become a major focal point of drug discovery research and a chemoproteomic approach can be used to directly measure inhibitor–kinase interactions in cells. 10
In addition to understanding the cellular activity of therapeutics in discovery in an in vitro setting, it is also important to understand their function in an in vivo setting where additional factors, such as biodistribution, tissue penetration, and environmental context, come in to play. Recently, advances in whole-body animal imaging, both in vivo and ex vivo, have enabled noninvasive formats for measuring target engagement. From this perspective, we discuss the opportunities of in vivo imaging and the important role it can play in assessing target engagement, as well as the complementary ex vivo assessment with further imaging technologies.
Established and Emerging Methods: In Vivo Imaging
In vivo imaging can be used to visualize biological functions changed by drugs, which can accelerate disease understanding and drug development. In vivo imaging can be used to observe changes at the organ, tissue, cellular, or molecular level. 11 Broadly speaking, the methods can be categorized either to assess anatomical changes and morphology or for molecular imaging at a functional level. Techniques such as ultrasound, magnetic resonance imaging (MRI), and computed tomography (CT) are used for anatomical imaging,1,12 and optical bioluminescence and fluorescence, positron emission tomography (PET), and single photon emission computed tomography (SPECT) are used for molecular visualization.11,13 With a novel therapeutic compound, each imaging modality can encompass many solutions, such as demonstration of organ accumulation and compound distribution, target expression and engagement, biological activity, toxicities, and use as a surrogate for a clinical endpoint. Specifically, in the target engagement space in vivo imaging is demonstrating wide utility for being able to assess compound distribution and in assessing metabolic activity and the downstream cellular response to treatment.
Anatomical and Morphological Imaging: Ultrasound, MRI, and CT
The advantages of ultrasound include high spatial resolution and contrast in soft tissue, fast and uncomplicated protocols, and portable and economical equipment. These enable the technique to become a competitive imaging modality for preclinical studies.14–16 It can be used across therapy areas, for example, in cardiovascular, oncology, toxicology, developmental, neurology, abdominal (assessing renal function and gastrointestinal systems), or inflammatory disease in quantifying perfusion and monitoring cell surface biomarker expression. High frame rates and techniques such as Doppler echocardiography have achieved wide utilization in the cardiovascular space 17 and in murine cancer research. 18 The development of novel contrast agents is demonstrating widespread use in preclinical applications. The most common ultrasound contrast agents in clinical use are microbubbles. As their name suggests, their diameters range between 1 and 10 µm. This size limits the application of microbubbles to the intravascular system to assess functional parameters like vascularity, perfusion, blood flow velocity, and angiogenicity, or to characterize vasculature molecularly by using targeted microbubbles. In this area, it has shown its potential to be informative in identifying the target engagement of candidate compounds. 19 Targeting ligands that have been attached to the surface of a microbubble have been used across disease areas. The flexibility of the platform and the ability to be able to use a variety of targeting ligands make this a flexible approach that can be used widely. VEGFR2 targeting and assessment of tumor angiogenesis remains a popular and well-used approach, particularly as it is of high importance as a biomarker in oncology research. In addition to attaching specific proteins and antibodies, it is also possible to attach therapeutic targets and carry bioactive substances for site-specific delivery. 20 Longitudinal ultrasound imaging has been shown to identify changes in VEGFR2 expression postantiangiogenic therapy and demonstrate drug binding and a therapeutic response to therapy. 21
Although frequently used as a stand-alone approach, ultrasound can also be used in combination with photoacoustic imaging, demonstrating added value. Photoacoustic imaging delivers exquisitely detailed images of biological tissue by listening to the sound that light makes. 22 This technology, in combination with ultrasound, is exciting clinicians and researchers alike as it provides a level of detail not previously possible. Photoacoustic imaging benefits from the advantages of wavelength-dependent absorption selectivity in optical excitation and acoustic resolution of ultrasound detection. As with ultrasound, axial resolution depends on the ultrasound transducer frequency and bandwidth. The absorption of short pulses of light generates broadband photoacoustic signals. 23 Photoacoustic imaging has many promising biomedical applications in tomographic imaging of the skin and other superficial organs by laser-induced microscopy. 24 This offers critical advantages over current high-resolution optical imaging modalities of deeper depth and absorption penetration. A further clinical application to be applied preclinically is that of early breast cancer detection, alongside early response to treatment. 25 Particularly in the small-animal imaging space, laser-induced photoacoustic imaging measures unique optical absorption contrast, can give unique signatures of important biochemical information posttreatment response, and provides better resolution in deep tissues than optical imaging. 26
The principal strengths of MRI are its noninvasiveness and high spatial resolution for rodent studies. 27 A major limitation is its low sensitivity, which significantly constrains the possible roles of the technique in pharmaceutical research. In general terms, the role of in vivo MRI in pharmacological research and in target engagement is in assessing the effects of the drug on tissue morphology, physiology, and biochemistry. For quantifying tumor function in early treatment response, it has been used in assessing target engagement, and there are various examples of its use, particularly using functional MRI. Dynamic contrast-enhanced MRI (DCE-MRI) derives quantitative physiological information from sequential images following an injection of contrast agent. 28 Over the past decades, there has been much progress in the development of MRI contrast agents; the most commonly used are the gadolinium-based contrast probes, with at least nine formulations approved for human use. In inflammatory diseases such as rheumatoid arthritis, DCE-MRI enables serial changes in functional status and abnormalities in tissue function. It has been used to assess early response to therapy and target engagement. In one study, a change in the DCE-MRI parameter Ktrans was observed prior to changes in synovial fluid. 29 In oncology, DCE-MRI has also been extensively used to evaluate the drug efficacy of monoclonal antibodies and antivascular agents; the ability to detect a vascular response has been a key focus for measuring changes in vascular permeability within the tumor, especially when targeting with an antiangiogenic therapy. In one study, a decrease in Ktrans of more than 60% was observed post-linifanib therapy. 30 In addition to this, another review documents several examples of where DCE-MRI can be used to demonstrate functional effects at an early time point posttreatment. 31
MicroCT provides high-resolution images and rapid data acquisition and is a cost-effective means for detecting soft tissue structures and skeletal abnormalities in small animals. 32 Accuracy in images is determined by x-ray dose, with a higher dose giving higher resolution and better imaging quality. X-ray dose and the amount you can give is a concern of in vivo microCT imaging. Too high a dose would risk burns to the animal or, in the case of tumor imaging, could risk a radiotherapy effect. Careful assessment and optimization needs to be performed to balance dose against imaging quality. There are examples of the use and applicability of microCT imaging in assessing target engagement across disease areas. 33 Due to the issues that are often encountered with low-dose CT imaging, the use of ex vivo microCT imaging to gain high-resolution image quality is a popular option. In addition to this, iodinated CT contrast agents are frequently used to improve image quality and enhance weak endogenous contrast between soft tissues. This has shown potential in both in vivo 34 and ex vivo therapeutic studies. 35
Molecular Imaging at a Functional Level: Bioluminescence, NIR, and PET
Bioluminescence imaging is a technique that has been developed over the last decade, utilizing the native light emission from several living organisms that bioluminesce. Three such species are the North American firefly, the sea pansy, and the bacterium Photorhabdus luminescens. Proteins in these bioluminescent species are capable of generating light in an enzymatic reaction that consumes ATP.
35
The light emitted from bioluminescence is not detectable by the human eye, but ultrasensitive charge-coupled device (CCD) cameras can be used to image the bioluminescence signal. Different substrates are employed to trigger the signal depending on what species the luminescent protein is derived from. For example, firefly luciferase, which is perhaps the most commonly used organism in bioluminescence imaging, requires the use of
Another similar technique is that of fluorescence imaging, which is attractive in part due to its operational simplicity. The near-infrared (NIR) window is particularly suitable for in vivo investigations. The optical window is wide; however, imaging in the NIR wavelength range between 700 and 900 nm takes advantages of the low absorption of biological molecules in this region. This allows deep biological penetration and good signal to noise. 36 Both luminescence and fluorescence imaging have demonstrated huge potential as a novel in vivo imaging approach to measure target engagement. This includes the monitoring of different biological processes in immunology,37,38 oncology,39,40 virology,41,42 and neuroscience.43,44
Receptor ligand engagement in vivo and quantification of intracellular receptor ligand binding can be achieved with whole-body macroscopic lifetime-based Förster resonance energy transfer (FRET) imaging (NIR MFLI FRET). 45 MFLI FRET provides a direct measurement of systemic delivery, target availability, and intracellular drug delivery in preclinical studies. 46 MFLI has been used to measure FRET longitudinally in intact and live animals. MFLI FRET is well suited for guiding the development of targeted drug therapy in heterogeneous tumors in intact, live small animals. 47 Fluorescence lifetime imaging microscopy (FLIM) quantifies FRET occurrence by estimating the reduction of the fluorescence lifetime of the donor fluorophore when it is near the receptor fluorophore. Previously, FLIM FRET was confined to microscopic techniques alone, but as we can see from the examples above, the utility of MFLI FRET has also been extended for use as an in vivo tool for the longitudinally measured quantification of receptor binding. The authors demonstrated for the first time that NIR MFLI FRET was able to directly assess intracellular ligand accumulation in heterogenous breast cancer xenografts. Importantly, the results demonstrated that a high level of receptor expression may not guarantee the efficacy of drug delivery. 47
Building on this technology and as a further approach to monitor ligand binding in living cells is a recent study developing a bioluminescence energy transfer (BRET) assay. 48 The nanoBRET target engagement assay measures compound binding at select target proteins within intact cells. The BRET assay approach is similar to the use of fluorescent ligands but overcomes potential issues whereby fluorescent uptake into cells can often lead to high levels of nonspecific binding; the use of bioluminescent tags mitigates this. One study using a luciferase-tagged G-protein-coupled receptor (GPCR) demonstrated binding of propranolol in real time. 49 In addition to its potential as a cellular assay, it also has recently been shown that it is possible to translate this into an in vivo setting. In a triple-negative breast cancer model, it was shown that it is possible to visualize ligand binding in vivo. 50 In the study the authors demonstrated ligand binding to a GPCR. A caveat of this, however, was also assessed; the resolution of the nanoBRET in this context was limited to the detection of established tumors only. This in vivo nanoBRET method is also applicable to monitoring target engagement in animal models for other cell surface receptors, such as receptor tyrosine kinases, 51 and for intracellular kinases. 52
PET is a nuclear medicine functional imaging technique that is used to observe metabolic processes in the body. PET produces images of the body by detecting the radiation emitted from a positron-emitting radioligand that is introduced into the body on a biologically active molecule. Preclinical studies using small laboratory animals demand high spatial resolution provided by dedicated PET systems. 53 PET is gaining utility as a target engagement assay, either through the labeling of a novel therapeutic or through the use of probes as biomarkers. 54
One area showing and demonstrating immense promise is the use of immuno-PET imaging to assess target engagement, and applications of this have been demonstrated both clinically 55 and preclinically. 56 One study validates the use of immuno-PET to directly visualize drug disposition for the measurement of target engagement at the site of action, offering the potential for dose selection in the clinic. 57 Dose-dependent inhibition of tumor uptake of 89Zr-GSK2849330 confirmed the target engagement of mAb to the HER3 receptor. This approach is applicable across therapy areas; for example, an 89Zr CD4 antibody fragment was used for noninvasive imaging of CD4+ T cells in mice with induced colitis, offering the potential to guide the development of antibody-based imaging in humans with irritable bowel disease. 58 Another example, with an immune checkpoint therapy, use of the PET radiotracer 64Cu WL12, showed peptide-based PET as a promising tool for optimizing a dose and therapeutic regimen of PDL1 antibody therapy. 59
PET can also be used to quantify target engagement and enable dose finding of some targeted small-molecule therapeutics. Fasting glucose, insulin, and C-peptide levels in plasma, and glucose uptake using 18F-FDG PET have all been evaluated in the determination of target engagement and pathway modulation. 60 Given the difficulties related to the acquisition of tumor tissue for the evaluation of PD markers, such as clinical usefulness, technical issues resulting in poor reproducibility and interpretability, and analytical issues leading to poorly defined cutoff values, functional imaging has emerged as a novel method and powerful tool for evaluating the PD effects of PI3K pathway drugs. 61 Functional imaging biomarkers have the potential to quantify biological characteristics of tumors and measure on- and off-target effects that allow serial, noninvasive assessments of whole tumor, which is particularly important in the context of intra- and interpatient tumor heterogeneity. Additionally, functional imaging could be used to guide the assessment of both the optimal biological dose and drug schedule ( Fig. 1 ). 62
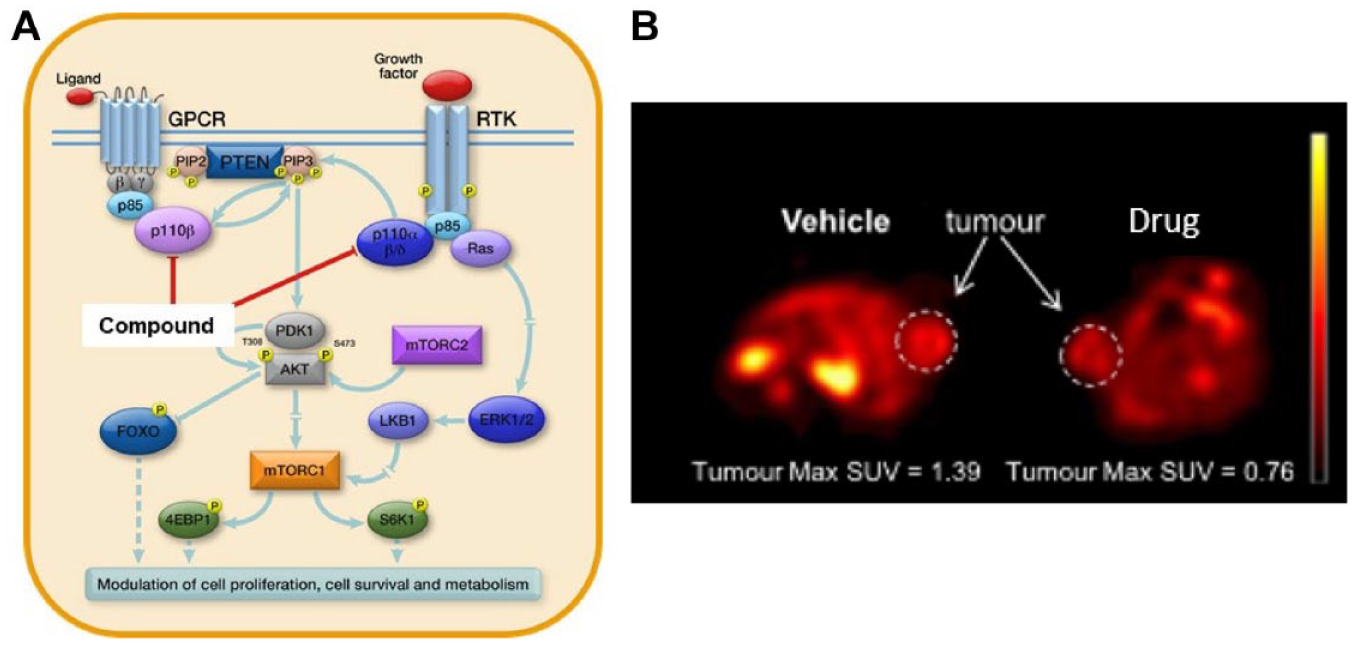
18F-FDG PET to measure tumor engagement following the administration of a PI3K inhibitor 2 h postadministration.
SPECT is a nuclear medicine imaging technique that uses gamma rays. It is similar to conventional planar imaging using a gamma counter but is able to provide 3D information. SPECT radionuclides are characterized by relatively long half-lives. They return to the ground state by the emission of single rays, compared with two in PET. In a similar way to PET, SPECT is used to collect physiological information and measure the biodistribution of radioactive substances. It is not as sensitive or as detailed as PET, but it is less expensive. Several studies have assessed biodistribution and target engagement using SPECT tracers and demonstrated similar potential to that seen with PET. 63 One interesting area is the ability of dual labeling, that is, taking a particle/therapeutic and radiolabeling it with two distinct radionuclides. One study describes the use of dual-labeled nanoparticle SPECT probes for bioimaging. 64 Here a gold nanoparticle was radiolabeled and then used as a multispectral SPECT imaging contrast agent to differentiate tumors with differing MMP9 expressions. This holds huge potential for tapping into and understanding pharmacodynamic activity and target engagement binding.
In vivo imaging methodologies provide an enormous amount of longitudinal, real-time, and 3D information for crucial target engagement and other studies. As with any set of techniques, there is also much benefit to be gained from the implementation of other complementary approaches. There exists a number of interesting ex vivo or endpoint-based imaging techniques that add an additional angle to the data to be gained for the improved investigation of target engagement. This article provides some focused examples of where mass spectrometry imaging (MSI) in particular can add further insight, not only as a stand-alone technique but also when combined with other imaging modalities. The best scientific approach for answering a given question may ultimately require the implementation of several technological methodologies.
Established and Emerging Methods: Mass Spectrometry Imaging
MSI involves the generation of mass spectra from the sampling of molecular ions or elements from surfaces in a dynamic manner. This allows for the recording of spatial information alongside the MS data, in turn allowing for the reconstruction of ion images showing analyte distribution across the said surface. Some of the earliest examples employed matrix-assisted laser desorption ionization (MALDI)-MSI for the imaging of peptides and proteins in biological samples, 65 and secondary ionization mass spectrometry (SIMS) for elemental distribution across different surfaces. 66 Brief descriptions and useful references to obtain extra information on each of the MSI techniques discussed in this paper are given in Table 1 . When compared with some of the in vivo imaging techniques, the use of MSI for target engagement is in its infancy. However, MSI approaches have been developed to cover the detection of a wide range of molecules (e.g., metabolites, lipids, proteins, peptides, and drug compounds) from biological samples.65–74 This has resulted in the ability to not only detect the distribution of a dosed drug in tissue sections but also detect the local and downstream impact of the said drug and its metabolites on biomolecular pathways, providing an indirect measurement of target engagement. 74 Multiple MS ionization techniques are now available for performing MSI (e.g., MALDI, desorption electrospray ionization [DESI], time-of-flight SIMS [TOF-SIMS], and laser ablation–inductively coupled plasma–MS [LA-ICP-MS]), and these can be coupled to a range of mass analyzers, providing different levels of resolution, sensitivity, and detection capability. Between the MSI instrumentation available, the lack of need for labeling, the broad range of analytes detectable, and the spatial resolution achievable, MSI is a valuable and complementary tool alongside and subsequent to in vivo, preclinical imaging and other ex vivo surface analysis techniques.
A Brief Explanation of the MS Techniques Discussed within This Article, Including Some Informative Publications in Which Each of the Techniques Is Described in Further Detail.

Studies such as that of Vallianatou et al. 75 illustrate the ability of DESI-MSI to measure the penetration of drugs through the blood–brain barrier. In this particular case, the study showed the potential reduction of Loperamides’ blood–brain barrier permeability in the presence of Elacridar. 75 Using MSI, it is possible to obtain highly resolved images of drug compounds, showing penetration into discrete structures of brain and other tissues. Importantly, the distribution is of the drug alone, as opposed to with the addition of a label or any change in chemical structure. While this study is MS based, we already see the importance of performing H&E staining alongside MSI to give anatomical relevance to the drug localization. Another study using MALDI-MSI for target engagement again employs H&E staining to support the results of the MSI. In this case, the posttranslational modification of histones in situ, in both whole cells and tissue sections of a mouse model of gastric cancer, was used to infer target engagement of histone deacetylase inhibitors. 74
With the majority of the focus placed on the benefit of MSI as a technique not requiring the use of a label, it is also worth highlighting that label-based workflows are in some cases beneficial. For example, isotope tracers have been used to monitor the activity of 11β-hydroxysteroid dehydrogenase in situ in brain. 76 In this case, MALDI-MS was shown to be beneficial due to the localization information that was gained while providing the added benefit over autoradiography of differentiating between drug compounds of interest and their metabolites, in this case facilitating the inference of enzyme activity. 77
There are several recent review articles and studies that have described the implementation of multiple complementary MS technologies.78,79 Some such examples have highlighted the use of TOF-SIMS. TOF-SIMS has been implemented extensively in academia and has the benefit of improved spatial resolution over DESI and MALDI-MSI, achieving a lateral resolution of <500 nm in some cases. Bruinen et al. describe the combined use of TOF-SIMS MS and tandem MSI (imaging of the instrumentally induced fragments of the molecules sampled) for the intracellular localization of α-tocopherol. 79 This provided simultaneous identification and localization of the molecule of interest in a 3D manner, utilizing the depth profiling capability of SIMS and potential for high-resolution imaging.
Laser ablation–inductively coupled plasma (LA-ICP)–MS is relatively new to its application in drug discovery as an MSI technique but is a complementary offering to most other ionization techniques with its ability to detect elements and display the localization of these in tissue. LA-ICP-MS is particularly useful in showing the distribution of chemotherapy treatments, as the metal in the metal complexes can be readily detected using this technique. 80 LA-ICP-MS has been used, for example, in the imaging of the platinum component of cisplatin in order to correlate drug distribution with localized evidence of nephrotoxity. 80 Approaches such as this may be easily employed for the indirect confirmation of target engagement. This technology has recently been developed further to create a system for performing imaging mass cytometry whereby proteins are labeled with metal-conjugated antibodies, in situ on a tissue section. In one study, this has allowed for the detection of the binding of platinum from cisplatin treatment to collagens in different tissues. 81
In summary, the multiple MSI techniques undoubtedly enable the detection of a wide range of analytes at varying levels of spatial resolution; when combined with histology images, it is possible to view the engagement and impact of drugs on target tissues and molecules in much greater depth, while using as few subjects and samples for analysis as possible. At Medicines Discovery Catapult, we are implementing in vivo imaging techniques, MSI, and other imaging methodologies (not discussed in this paper), with the aim of using each of the approaches to gain complementary information.
Future Perspectives: Multimodal Imaging
As imaging technology develops, it can be easy to focus on each modality individually. However, there is much benefit to be gained from combining advances in the in vivo imaging field with those of the ex vivo techniques such as MSI. Over recent years several studies have been performed whereby MRI and MSI have been used as complementary approaches. Aichler et al. used MSI to investigate the specificity and kinetic behavior of gadolinium-based contrast agents subsequent to their use in MRI, 82 with another study using the MRI data as a frame on which to overlay 3D MSI data. 83
Some specific examples of multimodal imaging have also been applied to examine biodistribution and associated kinetics of drug compounds ( Fig. 2 ). MRI and MSI combined with IHC were used to study the long-acting drug cabotegravir, its biodistribution and associated pharmacokinetic properties following intramuscular or subcutaneous administration. 84 MALDI-MSI was used to provide improved spatial resolution, allowing for ion images of cabotegravir to be overlaid with microscopy images, correlating drug distribution with specific cell populations, and thus allowing for more in-depth and biologically relevant conclusions to be drawn from the study than would necessarily be possible were only one imaging modality was employed.
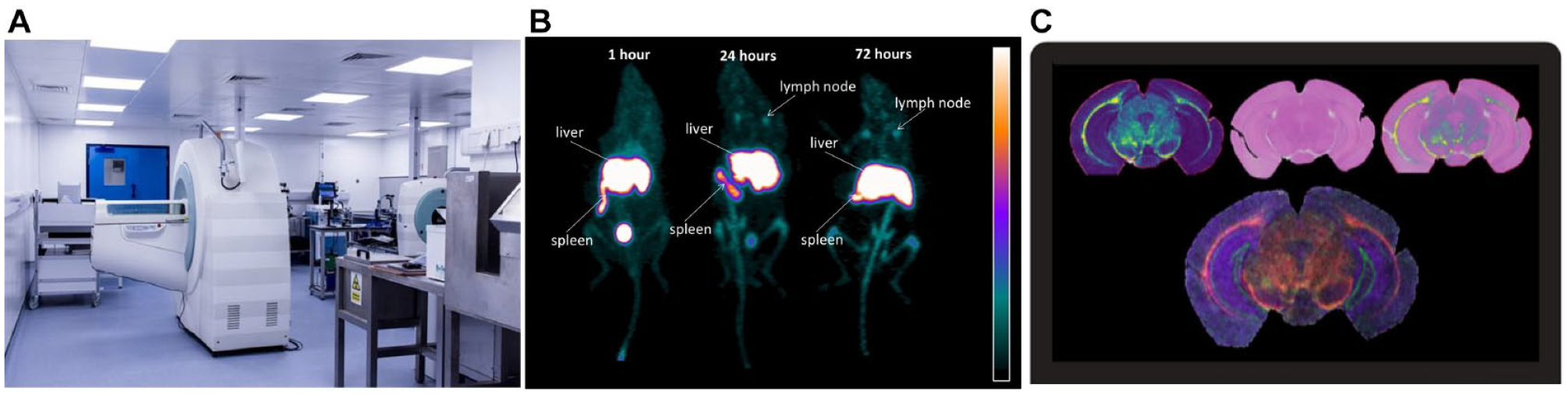
Multimodal imaging capabilities. (
The combining of methodologies to assess target engagement in both an in vivo and ex vivo setting offers enormous future potential. One possible example might include the noninvasive assessment of biodistribution using fluorescent imaging and NIR dyes to assess whole-body accumulation/distribution and therapeutic binding. Tissues could then be removed to allow for correlation of the in vivo imaging assessment with ex vivo, downstream MSI markers. The combination of in vivo PET and ex vivo MSI offers a much broader understanding of the action of the compound in the target organ, while also improving the achievable spatial resolution through MSI.
One aspect of these multimodal approaches that is still developing is the way in which the datasets are correlated, be that spatially or with regard to statistical or systems biology information gained. A separate review article discusses the co-registration and correlation of 3D clinical in vivo images. 85 This method of working is transferable to preclinical studies such as those discussed in this article. The conclusion of this review was that the use of fiducial markers is the current optimum for spatial co-registration of CT and MR images. This type of approach is obviously likely to be far more successful in tissue with well-definable anatomical features, such as neurological tissue. This can also be said with regard to co-registering MS and other imaging modalities where fiducial markers (clear markers that are consistently present across all images) or structural features are also used for manual co-registration of MSI with histology images in commercially available MSI software from various vendors. The ideal for the co-registration of imaging data would be an automated software tool that could perform co-registration without direct user intervention, thus reducing subjectivity and room for human error. It is encouraging to see that the development of such tools is being investigated for 2D ex vivo imaging 86 and for 3D in vivo/ex vivo imaging. 87
Conclusions and Future Perspectives
To conclude, it is known that through the use and implementation of imaging techniques, a greater understanding of target engagement and correlative therapeutic impact may be achieved. The application of analysis from multiple methods aims to increase the information gained on drug compounds or delivery mechanisms earlier in the drug discovery process to inform go/no-go decision making, save costs where compounds are not found to be efficacious by stopping development earlier in the process, and provide the data necessary to accelerate the development of successful compounds to provide earlier access to patients.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge Innovate UK for our funding.
