Abstract
Matrix-assisted laser desorption/ionization imaging mass spectrometry (MALDI IMS) is becoming an important technology to determine the distribution of drugs and their metabolites in the tissue of preclinical species after dosing. Interest in IMS is growing in the ophthalmology field, but little work to this point has been done to investigate ocular drug transit using this technology. Information on where and how a drug is distributing through the eye is important in understanding efficacy and whether it is reaching the desired target tissue. For this study, ocular distribution of brimonidine was investigated in rabbits following topical administration. Brimonidine has been shown to lower intraocular pressure and is approved to treat glaucoma, the second leading cause of blindness in the world. We have developed IMS methods to assess transit of topically administered brimonidine from the anterior to the posterior segment of rabbit eyes. Using IMS, brimonidine was detected in the cornea, aqueous humor, iris, and posterior segments of the eye. The distribution of brimonidine suggests that the route of transit following topical administration is mainly through the uvea-scleral route. This study demonstrates that IMS can be applied to assess ocular transit and distribution of topically administered drugs.
Introduction
In drug discovery and development, matrix-assisted laser desorption/ionization (MALDI) imaging mass spectrometry (IMS) is growing in importance as a technology for determining the localization of drugs and their metabolites in animal tissue after dosing. In this context, MALDI IMS has been used to help answer questions of drug distribution, concentration, metabolism, and toxicity.1–7 Unlike other common analytical techniques used in drug discovery and development for the determination of drug distribution such as autoradiography, IMS has an advantage of not requiring a label. IMS has also been gaining interest in the ophthalmology field, with studies designed to (1) assess the relative distribution of endogenous molecules in various ocular anatomical structures, (2) determine posttranslational modifications of ocular proteins, and (3) examine the distribution of exogenous compounds dosed in the eye.8–15 However, little work to this point has been done to investigate ocular drug transit using this technology.
In this study, we used MALDI IMS to examine the distribution of topically administered brimonidine, a drug used to treat glaucoma. Glaucoma represents a group of diseases that can cause damage to the optic nerve and, if left untreated, ultimately result in vision loss and blindness. A primary risk factor for glaucoma is increased intraocular pressure, and as such, it is a major focus in the treatment of glaucoma. 16 Brimonidine is a selective α2-adrenergic receptor agonist that is used in the treatment of glaucoma as a topical eye drop medication. It has been shown to reduce intraocular pressure by both reducing aqueous humor production and increasing outflow through the trabecular meshwork. 17 Thus, by reducing pressure, it would have a protective effect on retinal ganglion nerve cells. 16 Indeed, studies using 14C-brimonidine demonstrated that brimonidine reached posterior tissues of the eye at nanomolar concentrations after topical administration in preclinical species. 18
The eye is a complex organ that has many unique anatomical features. It contains two main divisions: the anterior segment and the posterior segment. The anterior segment includes the cornea, sclera, anterior chamber, iris, ciliary body, and lens, while the posterior segment includes the posterior sclera, choroid, retinal pigment epithelium, retina, optic nerve, and vitreous humor. These anatomic and physical barriers pose challenges to ocular drug delivery, especially to the posterior regions of the eye. Common approaches to ocular drug delivery include systemic administration, periocular and intravitreal injections, and topical application. Drug delivery to the posterior segment of the eye after systemic administration is constrained by the blood-retinal barrier. Intravitreal and topical administration offer the advantage of being local delivery methods that allow a smaller total dose of a drug to be administered while maintaining high local concentrations. Of these, topical application is more advantageous in that it is less invasive but has the challenge that multiple barriers need to be crossed for a drug to transit the eye. Previous studies have shown that there are three main routes of transit following topical administration: the trans-vitreous route, the uvea-scleral route, and the periocular route.19,20 Understanding drug distribution after topical dosing and how it transits the eye is important to know both if the drug is reaching its target and how it is cleared. It is of special importance in the design and study of drugs to treat posterior segment eye diseases including age-related macular degeneration, endophthalmitis, proliferative vitreoretinopathy, and diabetic macular edema. 21
Materials and Methods
Materials
High-performance liquid chromatography (HPLC)–grade acetonitrile, HPLC-grade methanol, trifluoroacetic acid (TFA), alpha-cyano-4-hydroxycinnamic acid (CHCA), and sodium carboxymethylcellulose (CMC) was purchased from Sigma-Aldrich (St. Louis, MO, USA). Ultrapure water was prepared with a Purelab Flex system.
Topical Drug Application
Dutch Belted rabbits were dosed topically in the right eye with 30 µL of a 3% suspension of brimonidine or vehicle (0.05% Tween 80 in phosphate-buffered saline). Animals were sacrificed at 15 min or 60 min after administration, and eyes were collected. Nondosed eyes were also collected.
MALDI IMS Analysis
After enucleation, rabbit eyes were snap-frozen in liquid nitrogen and then embedded in a 2.7% w/v solution of CMC to facilitate sectioning. Eyes were sectioned at 12 µm on a CM3050 S cryostat (Leica Biosystems, Wetzlar, Germany). Sections were thaw mounted on steel target plates for MALDI IMS or glass slides for hematoxylin and eosin (H&E) staining. Plates for MALDI IMS were coated with CHCA matrix using a TM-Sprayer instrument (HTX Technologies, Chapel Hill, NC, USA). The matrix was prepared at 5 mg/mL in 50% acetonitrile with 0.1% TFA. The TM-Sprayer was programmed to spray eight passes of matrix at 70 °C in a criss-cross pattern with a 3 mm track spacing. A matrix flow rate of 0.150 mL/min and a stage speed of 1200 mm/min were used. After matrix coating, the sample plate was rehydrated in a petri dish containing 1 mL of 70% methanol for 2 min. MALDI MS was performed on a Sciex (Framingham, MA, USA) 5800 MALDI TOF/TOF instrument. Mass spectrometry (MS) spectra were collected in positive reflector mode from m/z 250–900. An external calibration was performed just prior to the imaging experiment. Imaging resolution was set to 80 µm. MS/MS spectra were acquired using a collision energy of 2 kV with the metastable suppressor on. TissueView software (Sciex) was used for the visualization and analysis of the MALDI IMS data. Regions of interest designating the cornea and the iris were selected, and the mean intensities of each region were extracted. Each time point included three separate rabbit eyes. Two sections from each eye were analyzed. Data were expressed as means ± SEM, and statistical analysis was performed using Student’s t test for unpaired samples. Coregistration of H&E images with brimonidine ion images was done to assess drug localization in specific anatomical structures of the eyes.
Results
Detection of Brimonidine in Rabbit Eye
Optimization of MALDI MS sensitivity for brimonidine on ocular tissue involved the evaluation of several matrices. CHCA matrix was found to give the best MALDI sensitivity on tissue. This matrix also gave a clean background in vehicle-dosed tissue in the molecular ion region of brimonidine at m/z 292. Brimonidine ( Fig. 1A ) was then measured in the eye using MALDI IMS after a single topical dose. The full scan MS signal of brimonidine detected from dosed tissue ( Fig. 1B ) shows characteristic isotope peaks at m/z 292 and m/z 294 due to the bromine atom in brimonidine. MS/MS fragmentation resulting in fragments specific to this drug further confirmed the identity of this peak ( Fig. 1C , D ).
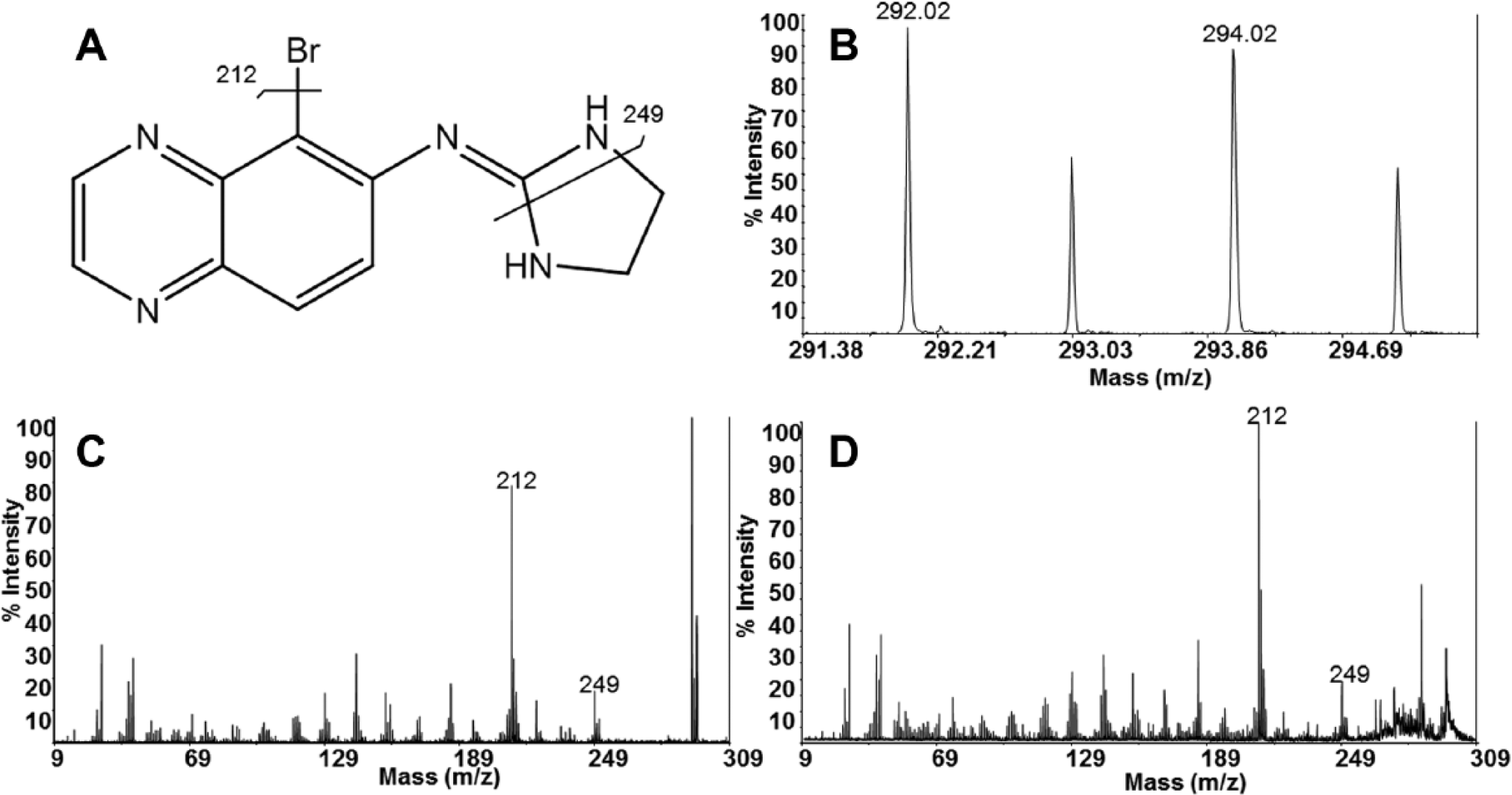
Detection of brimonidine in dosed eyes. (
IMS of Eye Section Showing Localization of Brimonidine
After confirming that brimonidine could be detected in a topically dosed rabbit eye, we performed full scan imaging across the entire cross section of an eye. The resulting MALDI image shows the localization of brimonidine ( Fig. 2 ). Other ion signals corresponding to endogenous molecules specific to the lens and the ciliary body are shown for visualization purposes. It can be seen that the brimonidine signal is concentrated in the front of the eye at 15 min postdose. Coregistration of IMS with histology reveals that brimonidine was localized to the cornea, anterior chamber, and iris and may have reached the posterior segment of the eye.
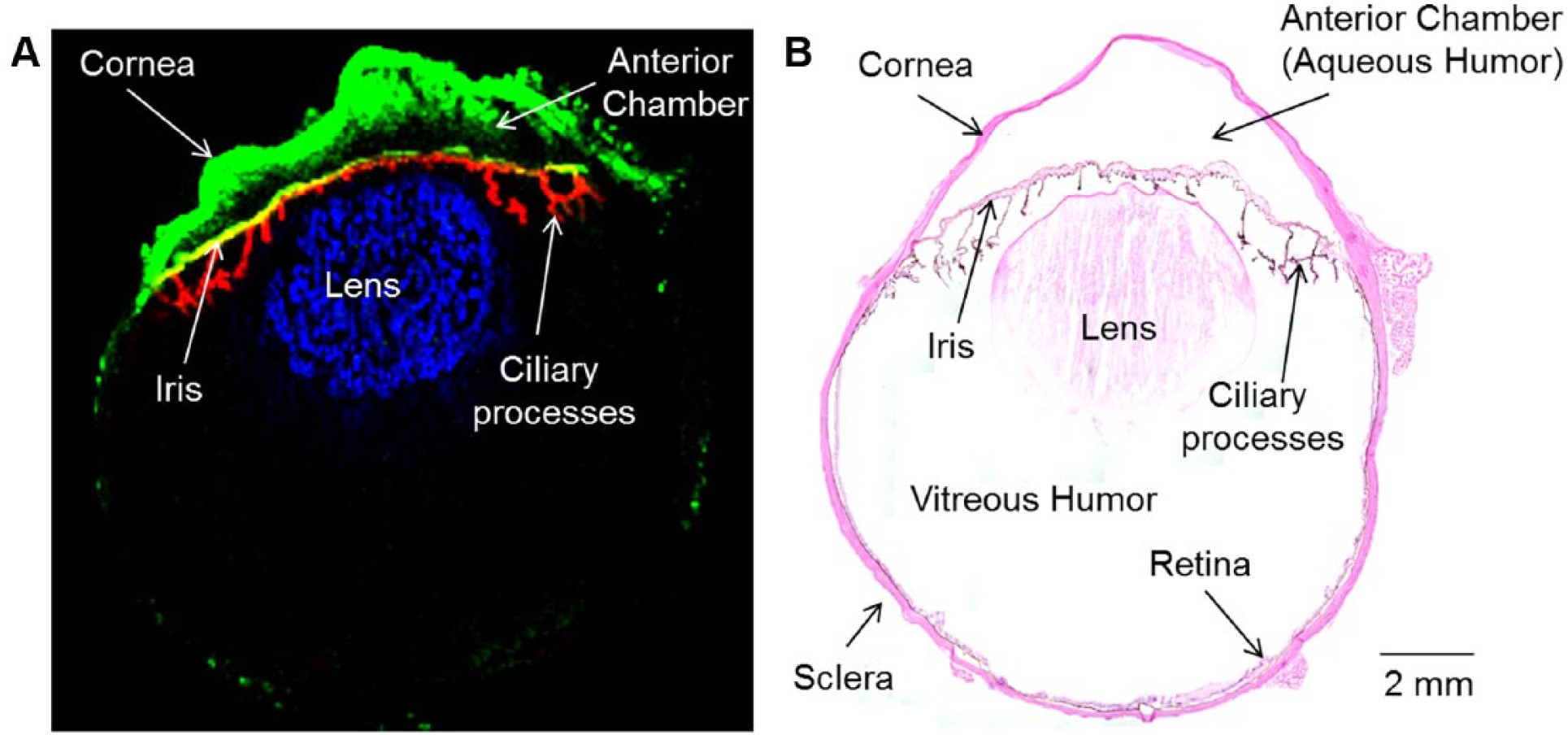
Localization of brimonidine in eye at 15 min after topical dose. (
Transit through Eye at Different Time Points
Having established localization, we next sought to track the transit of brimonidine by using rabbit eyes at both 15 min and 60 min after the topical dose. At these two early time points, most of the brimonidine is still in the anterior portion of the eye ( Fig. 3A ). However, notable differences in distribution can be observed. For example, more brimonidine is localized in the cornea at 15 min compared with 60 min, indicating drug penetration into the eye and clearance over time. In addition, brimonidine levels were observed to increase in the iris from 15 to 60 min ( Fig. 4 ). Imaging data across the multiple animal eyes reproducibly demonstrated that brimonidine was localized in the cornea, aqueous chamber, and iris. While at these early time points brimonidine is not distributed to a large degree in the back of the eye, some brimonidine was detected in the posterior portion of the eye ( Fig. 3B ). Brimonidine signal was not detected in the nondosed contralateral eye (not shown). Furthermore, no metabolites of brimonidine were detected at either time point examined.
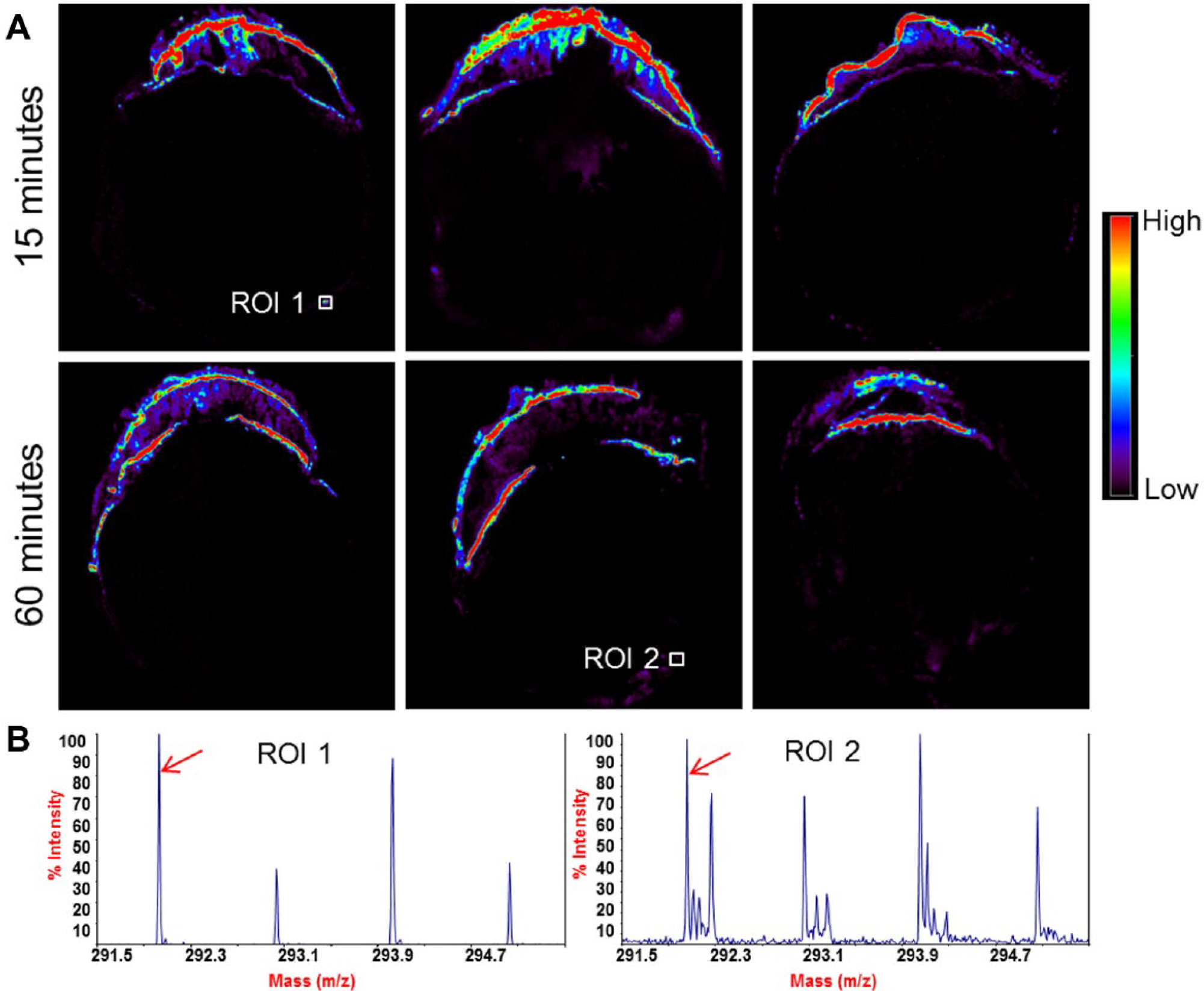
Detection of brimonidine at different time points. (
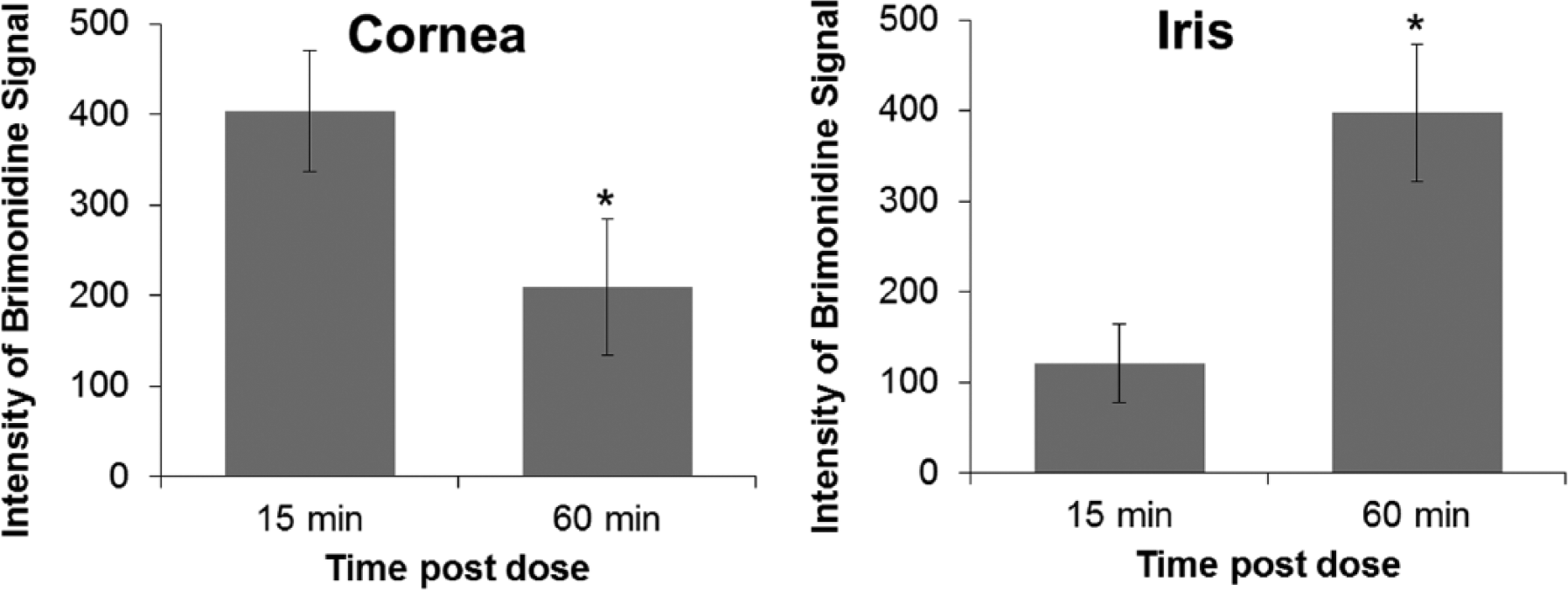
Relative quantitation of brimonidine signal in ocular tissues. Regions of interest containing the cornea and the iris were selected, and the intensity of the brimonidine signal in each region was extracted. Each bar graph represents the mean intensity ± SEM. *p < 0.05 (two sections per eye were analyzed, n = 3 eyes per time point).
Discussion
In drug discovery and development, the distribution of a drug after dosing is critical to understand efficacy, toxicity, and metabolism. Tissue homogenate liquid chromatography–mass spectrometry (LC-MS) and autoradiography are two commonly used techniques to determine this, but each has its drawbacks. LC-MS analysis of homogenates provides quantitative data for the concentration of a compound in dissected tissue; however, all spatial information within the tissue is lost. As an imaging technique, autoradiography preserves the tissue localization but requires a radiolabel and so is often used only for select compounds later in the development process. In addition, because total radioactivity is being detected, one cannot distinguish between parent drug and metabolites. MALDI IMS has emerged as an ideal way to determine the distribution of a drug after dosing. Because no label is required, these types of imaging studies can be done earlier in the drug discovery process. In addition, the mass spectrometric detection is able to differentiate both parent drug and metabolites. However, while quantitation is routine for LC-MS and autoradiography methods, this is more challenging with MALDI IMS, but progress is being made in this area.5,6,22,23
In this study, we were able to detect unlabeled brimonidine in ocular tissues. The localization determined with MALDI IMS in our study corresponds well with previous radiolabel studies carried out by Acheampong et al.18,24 In these studies, 14 C-brimonidine was dosed in rabbit and monkey eyes, and drug concentrations were determined after dissecting apart different regions of the eye and measuring total radioactivity. They found that brimonidine was rapidly absorbed into the eye after topical application, resulting in high concentrations in the aqueous humor and the iris-ciliary body, the presumed site of action for lowering intraocular pressure, 24 and demonstrated that brimonidine could reach the back of the eye at concentrations sufficient to activate α2-receptors in retina. 18 The drug distribution determined using MALDI IMS in our study lines up well with regions of the eye where high radioactivity was measured including the cornea, anterior chamber, iris, and some in the posterior segment of the eye. Furthermore, these previous studies noted high levels of brimonidine in pigmented tissues such as the iris, which was attributed to drug binding to melanin. Binding of brimonidine to melanin could act as a depot of drug and prolonged retention of the drug in the eye. Our study observed accumulation in pigmented tissues such as the iris as well. A drawback of our current MALDI IMS study is that it does not provide quantitative data for brimonidine in particular eye regions such as these other works. In addition, the sensitivity is likely lower as we did not detect any brimonidine metabolites.
Brimonidine has been prescribed because of its mechanism of lowering intraocular pressure. It has been found to decrease intraocular pressure in two ways: reducing aqueous humor production and increasing aqueous humor outflow, which would presume its site of action is in the aqueous humor and ciliary body.16,24 We were able to show brimonidine localized to this area in the same time frame as its intraocular pressure–lowering effect (~60 min after dosing). 25 Other studies have suggested a second mechanism, in the posterior portion of the eye, by which brimonidine can treat glaucoma by protecting retinal ganglion cells from damage. These studies showed that brimonidine could reach the back of the eye to have this effect.16,18,26 We were also able to suggest that some brimonidine reached the posterior region of the eye, which was observed at both time points studied ( Fig. 3 ). This is in agreement with radiolabel studies that showed that brimonidine was detected in the retina by 15 min postdose. 26 While three local routes of drug penetration have been described,19,20 a drug may reach the posterior segment of the eye through a combination of routes. Using IMS, we detected transcorneal distribution, presence in the anterior chamber, and drug surrounding and in the posterior region of the eye. This distribution of brimonidine suggests that the uveal-scleral route is the main route of transit of topically applied brimonidine. It is noted that the signal observed around and in the back of the eye is low. This may be due in part to the early time points used in this study, which were likely before the peak concentration time (Tmax) for brimonidine in the choroid/retina. 18 Also, additional tissue sections through the depth of the eye may better capture drug distribution in these structures. Inclusion of more time points and more tissue sections may help better define drug transit. Further, the low brimonidine signal may be due to inadequate sensitivity needed to detect brimonidine in the layers surrounding the eye. The current setup of MALDI IMS used in our study had a spatial resolution of 80 µm. With this limit, we were not able to differentiate between sclera, choroid, or retinal tissue in the eye. Improvements to the spatial resolution used in this study are needed to determine specifically which layers in the back of the eye contained brimonidine. Unique molecular localization in different layers in the back of the eye has been demonstrated in other studies by imaging at 10 µm on a commercially available MALDI system. 8
In summary, this MALDI imaging study showed that brimonidine was detected in the cornea, anterior chamber, and iris and reached the posterior segment of the eye. The distribution of brimonidine suggests that the route of transit following topical administration is mainly through the uvea-scleral route, consistent with other reports. 26 This study demonstrates that IMS can be applied to monitor distribution and ocular transit of topically administered drug.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
