Abstract
Production of adenosine in the extracellular tumor microenvironment elicits strong immunosuppression and is associated with tumor progression. Thus, targeting adenosine-generating ectonucleotidases is a potential strategy to stimulate and prolong antitumor immunity. Because the reaction products of ectonucleotidases differ by a single phosphate group, selective detection in an assay format that is compatible with high-throughput screening (HTS) has been elusive. We report the development of biochemical assays capable of measuring the activity of ectonucleoside triphosphate diphosphohydrolase-1 (ENTPD1; also known as CD39) and ecto-5′-nucleotidase (CD73). Both assays leverage the Transcreener HTS Assay platform, which facilitates selective immunodetection of nucleotides with homogenous fluorescent readouts, fluorescence polarization or time-resolved fluorescence energy transfer. The Transcreener AMP2 Assay was used to measure CD39 activity, allowing detection of adenosine monophosphate (AMP) production (Z′ > 0.6) with subnanomolar amounts of CD39, allowing IC50 determination for tool compounds, consistent with previously reported values. To detect the production of adenosine by CD73, the Transcreener ADP2 Assay was coupled with adenosine kinase (AK); conversion of adenosine to AMP and adenosine diphosphate (ADP) by AK allows detection with ADP2 antibody. The Transcreener AMP2 Assay was used to screen a 1280 Library of Pharmacologically Active Compounds (LOPAC) library and a 1600-compound subset of a ChemBridge diversity library for CD39 inhibitors, allowing the identification of nine and eight candidate compounds from each library, respectively. The Transcreener ADP2 Assay was used to screen 1600 compounds from the ChemBridge diversity library for CD73 inhibitors and identified 14 potential candidates. HTS-compatible assays for ectonucleotidase activity may allow identification of purinergic signaling pathway inhibitors important for tumor-specific immune responses during tumor pathogenesis.
Introduction
Ectonucleotidases are plasma membrane–bound enzymes with externally oriented active sites that hydrolyze nucleotides to produce nucleosides. In purinergic signaling pathways, in which extracellular receptors sense levels of purines in the extracellular milieu, ectonucleotidases play a crucial role in maintaining immune homeostasis.
One key enzyme in this process is ectonucleoside triphosphate diphosphohydrolase-1 (ENTPD1), also known as CD39 or NTPDase1 protein. CD39 catalyzes the hydrolysis of γ- and β-phosphate residues of triphospho- and diphosphonucleosides to the monophosphonucleoside derivative. The enzyme is capable of hydrolyzing adenosine triphosphate (ATP), adenosine diphosphate (ADP), uridine triphosphate, and uridine diphosphate (UDP) with similar efficiency. Through its actions, it affects the amount of ligands available to P2 receptors, which are ligand-gated ion channels or G protein–coupled receptors. In addition to cancer, medically significant roles of P2 receptors include chronic pain perception and vascular complications of diabetes.1,2
The ability of CD39 to hydrolyze ATP to ADP to adenosine monophosphate (AMP) holds particular biological importance. AMP can serve as substrate for CD73 (also known as 5′ nucleotidase), which hydrolyzes AMP to adenosine. Extracellular adenosine has a significant impact on dozens of disease states. CD39 may directly affect cancer progression by regulating T-cell and natural killer cell activity and antitumor responses in general3,4 and immunosuppression of melanoma cells specifically. 5 During tumor pathogenesis, secreted adenosine acts as an immunomodulator, affecting tumor-specific immune responses. 4
In the extracellular matrix, the ratio of extracellular ATP to extracellular adenosine informs immune homeostasis. High levels of ATP results in immunoactivation by causing increased interleukin-1 (IL1) and IL18 levels in dendritic cells, increased proliferation of natural killer cells, increased maturation of macrophages, and increased interferon-β (IFNβ) and IL17 levels in T cells. 4 In contrast, high levels of adenosine elicits immunosuppression by reducing IL12 and increasing IL6, IL8, IL10, vascular endothelial growth factor, and tumor necrosis factor–α in dendritic cells, reducing cytotoxicity and IFNβ in natural killer cells, increasing IL10 and M2 macrophages while reducing tumor necrosis factor and IL12 and reducing proliferation and cytotoxicity of T cells. 4
Although US Food and Drug Administration–approved drugs targeting P2 receptors exist, such as clopidogrel, a P2Y12 receptor inhibitor used to reduce heart disease and stroke in patients at high risk, drug development efforts focusing on ectonucleotidases have taken a slower path. In large part, this has been due to lack of sufficiently sensitive and specific ectonucleotidase assays. For example, a conventional malachite green assay for ectonucleotidase activity detects phosphate and not the nucleoside reaction product. Because many biological reactions release phosphates, this results in high background and lack of signal specificity. In addition, such approaches are not compatible with high-throughput screening (HTS).
In this study, we investigated two biochemical assays for CD39 and CD73, respectively, and pursued pilot-scale HTS trials to identify CD39 and CD73 inhibitors. Both assays leverage the Transcreener HTS platform, which facilitates selective immunodetection of nucleotides with fluorescent readouts such as far-red fluorescence polarization (FP) and time-resolved fluorescence energy transfer (TR-FRET).
Materials and Methods
Enzymes, Substrates, and Reagents
Recombinant human CD39/ENTPD1 protein was obtained from R&D Systems (Minneapolis, MN, catalog No. 4397-EN). CD39 inhibitors polyoxometalate-1 (POM1) and PSB069 and CD73 inhibitor PSB12379 were obtained from Tocris Bioscience (Minneapolis, MN, catalog No. 2689 and 2573). Recombinant human CD73 was obtained from R&D Systems (catalog No. 5795-EN). Recombinant human adenosine kinase-1 was obtained from Novo CIB (Lyon, France, catalog No. E-Nov5). Transcreener AMP2 Assay and Transcreener ADP2 Assay kits were obtained from BellBrook Labs (Madison, WI). ATP and ADP were obtained from Sigma Aldrich (St. Louis, MO, catalog No. A2383, A5285). The LOPAC 1280 library was obtained from Sigma-Aldrich (St. Louis, MO, catalog No. LO4100). A diversity library was obtained from ChemBridge (San Diego, CA, DIVERSET, EXP), and a subset of 1600 compounds was used for screening.
Instrumentation and Analysis
PHERAstar Plus, a microplate reader from BMG Labtech, was used to measure both FP and TR-FRET readouts. For the Transcreener FP assays, a black nonbinding low-volume 384-well plate from Corning was used (Corning, NY, catalog No. 4514), and for the TR-FRET assays, a low-volume nonbinding white 384-well plate was used (Corning, catalog No. 4513). After adding all the enzyme components, the reaction was mixed in an orbital shaker for 30 s. The FP assays used an excitation wavelength of 620 nm and emission wavelength of 670 nm. The TR-FRET settings were excitation 320/emission 620/emission 670 nm. EC50 and EC85 values, Hill slopes, and curves were generated by GraphPad Prism. The raw data from the readouts were analyzed using a four-parameter fit (minimal mP, highest enzyme concentration; maximal mP, no enzyme control; inflection point, EC50; and Hill coefficient, slope of the curve) using GraphPad Prism. The raw data were converted to AMP or adenosine (for CD39 and CD73, respectively) using a standard curve run under similar conditions.
Analysis of CD39 Activity Using the Transcreener AMP2 Assay
The composition of the assay buffer was 50 mM Tris, 10 mM CaCl2, 5 mM MgCl2, 0.01% Brij-35, pH 7.5. ATP or ADP was used as substrate at a concentration of 100 µM. Optimum Transcreener antibody concentration was determined by serially titrating AMP2 antibody in the presence of 100 µM ATP or ADP in the reaction buffer containing 2 nM AMP2 Alexa 633 tracer in a final 20-µL volume. AMP antibody was used at a concentration of 85% of the amount required for saturated binding to tracer (i.e., the EC85). The assay buffer used for TR-FRET assays is similar to the one for FP. The optimal tracer concentration (EC85) for the TR-FRET assays was determined by titrating the AMP2 HiLyte 647 tracer in the presence of 100 µM ADP or 100 µM ATP. CD39 was titrated in the presence of 100 µM ATP or ADP to determine optimal enzyme concentration, and raw data (millipolarization units for FP or ratios for TR-FRET) were converted to AMP using a standard curve run under similar conditions to show a linear correlation of the enzyme with the product formed. Z′ measurements were calculated using these optimized CD39 reaction conditions, and dose-response curves for CD39 probe compounds POM1 and PSB069 were conducted to determine IC50 values and correlate the experimental values with those reported in the literature.
Analysis of CD73 Activity Using the Transcreener ADP2 Assay
The Transcreener ADP2 Assay was used to develop and optimize an assay for CD73 activity for measurement of adenosine using FP readout. To optimize assay conditions, ADP2 antibody was titrated in the presence of 10 µM ATP. Adenosine kinase-1 (10 ng/µL) and tracer (2 nM) were included in buffer containing Tris-50 mM, 5 mM MgCl2, DMSO-1%, Brij-0.01%, at pH 7.5. The plate was mixed well, incubated for 1 h at room temperature, and read in PHERAstar Plus. The optimal antibody concentration (EC85) was calculated as approximately 20 µg/mL for monoclonal ADP2 antibody. To determine the extent of cross-reactivity of ADP2 antibody, competition curves were performed using AMP, ATP, and adenosine. A standard curve (10 µM adenosine/ADP) was generated using the Transcreener ADP2 assay by including the coupling enzyme, adenosine kinase (AK). Recombinant CD73 was titrated, and raw data (mP units) were converted into adenosine to show a linear correlation of the enzyme with the product formed. The CD73 reactions were not quenched by EDTA; thus, a kinetic readout was used for this assay. Z′ measurements were calculated using these optimized CD73 reaction conditions, and dose response for CD73 inhibitor PSB12379 was conducted to determine IC50 values and to correlate the experimental value with those reported in the literature.
High-Throughput Screen for CD39/CD73 Inhibitors
High-throughput screens for inhibitors of CD39 were performed using a 1280-compound LOPAC using the FP format and a 1600-compound subset of a ChemBridge diversity library using the TR-FRET format. Without running analytical tests for each compound, we cannot be certain about their solubility in the screening reactions. However, both the LOPAC and the ChemBridge libraries are widely used commercial collections of compounds that, from our experience, are generally soluble under aqueous conditions at concentrations of at least 50 µM. In any event, if some wells contained lower concentrations of compound because of limited solubility, it would not have an impact on the conclusions drawn in this study. Both libraries were screened at 10 µM final concentration using 100 µM ATP as the substrate. The assay conditions were similar to what is described above. The compounds were plated using an acoustic dispenser, to which 5 µL of 100 pM CD39 was added. The plates were preincubated with the enzyme for 30 min at room temperature. This was followed by the addition of 5 µL of ATP substrate to initiate the reaction. After 1 h of incubation, 10 µL of detection reagents was added to the plate. The plates were mixed well and read after an hour in PHERAstar Plus. The pilot screen for CD73 was performed using a subset of the ChemBridge diversity library, 1600 compounds. The compounds were plated at 20 µM final concentration using the acoustic dispenser. Five microliters of 5 pM CD73 was preincubated with the compounds for 30 min. AMP (5 µL) was added to initiate the reaction, followed by the addition of detection reagents (10 µL). The plates were read in PHERAstar Plus.
Results and Discussion
To determine the feasibility of the Transcreener AMP2 Kit for identifying inhibitors of CD39 activity, assay conditions were optimized for screening using FP detection. An antibody/tracer binding analysis was performed (
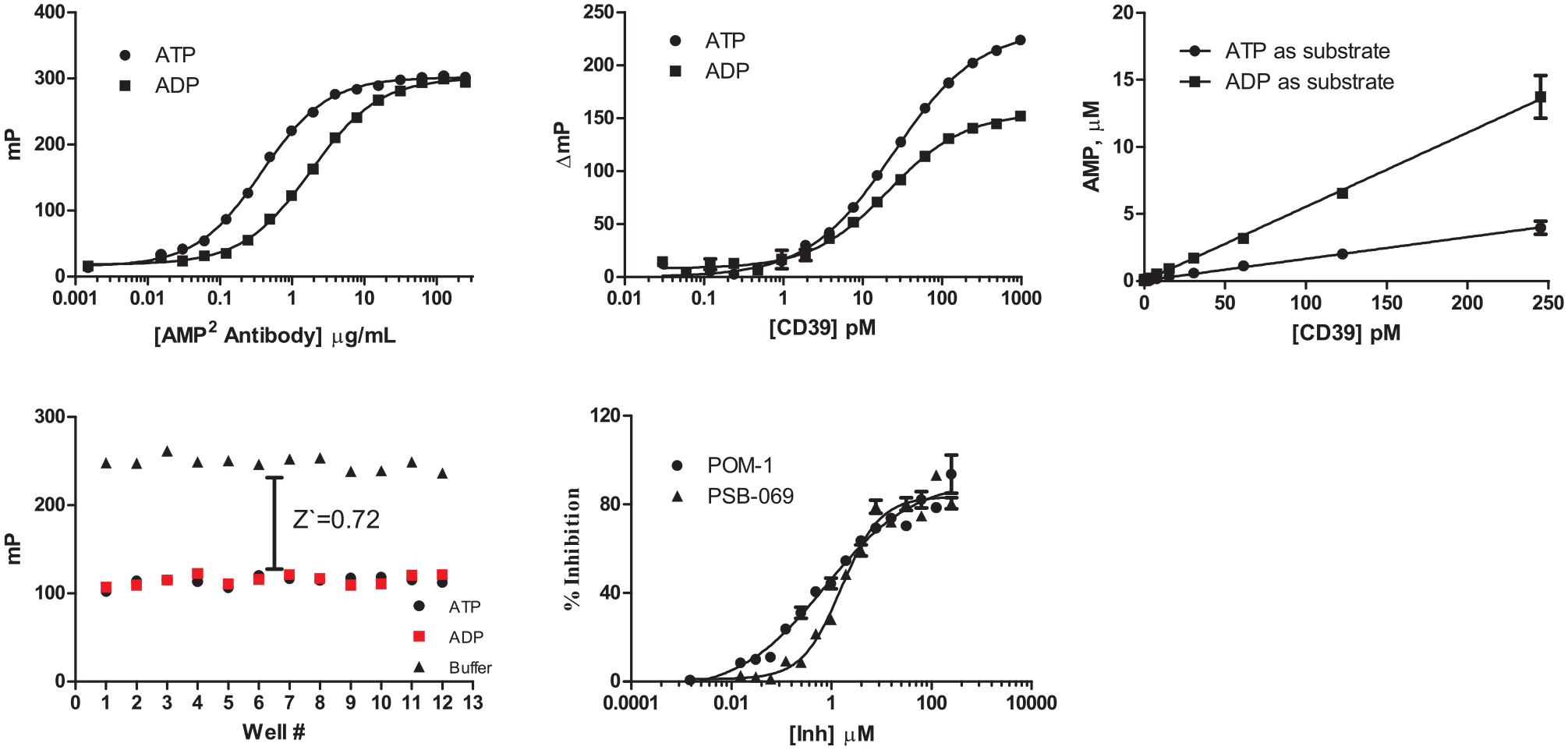
CD39 activity optimization and screening in fluorescence polarization. (
Two previously characterized CD39 inhibitors were used to generate dose-response curves for experimental determination of IC50 using the Transcreener AMP2 assays system. The inhibitors are members of the polyoxometalate family of compounds, which are inorganic cluster metal complexes. 6 POM1 inhibits ecto-nucleoside-triphosphate-diphosphohydrolases, and PSB069 is a nonselective CD39 inhibitor.7,8 Each inhibitor yielded IC50 values that correlated with literature values: an experimental determination of 0.8 µM for POM1 and 1.6 µM for PSB069 compared with previously reported IC50 values of 2.58 µM and 2.22 µM, respectively.9–11
To test the CD39 assay in an HTS setting, we performed a pilot screen of 1280 bioactive compounds from a LOPAC library with 100 µM ATP as the substrate (
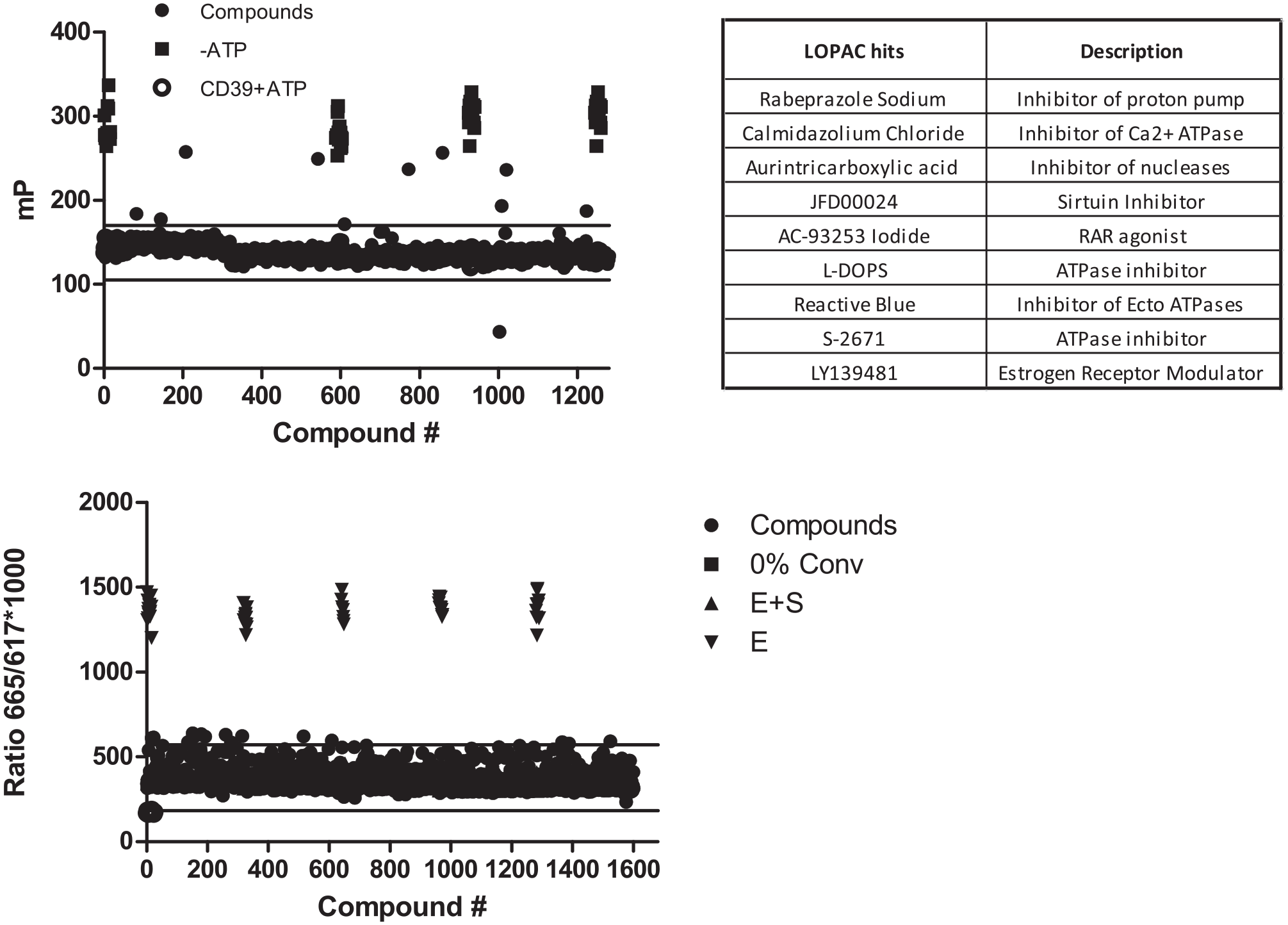
Screening CD39 using the Library of Pharmacologically Active Compounds (LOPAC) and a subset of the diversity library from ChemBridge. (
The CD39 assay is a direct detection method; that is, the antibody recognizes the AMP formed in the enzyme reaction. Ideally, we would have liked to have developed a direct assay for CD73 by developing an antibody for adenosine, but all of our initial attempts proved unsuccessful. We therefore developed a coupled assay method using AK to convert adenosine to AMP. We used an excess of AK to minimize compound interference with the coupling enzyme. Nevertheless, as with any coupled assay, stringent hit confirmation would require counterscreening against the coupling enzyme to triage AK inhibitors. In addition, whereas the CD39 assay was designed as an endpoint assay with EDTA as a quenching reagent, we were unable to identify a reagent that would quench CD73 without an effect on AK. Therefore, the CD73 assay was used in continuous mode, which required coordination of reaction start and read times.
AK converts adenosine produced by CD73 to ADP and AMP; the ADP can be detected using the ADP2 antibody. The ADP2 antibody was titrated in the presence of 10 µM ATP, 10 µM adenosine, and 2 nM tracer in the assay buffer. The optimal ADP2 antibody concentration (EC85) was determined to be 20 µg/mL (
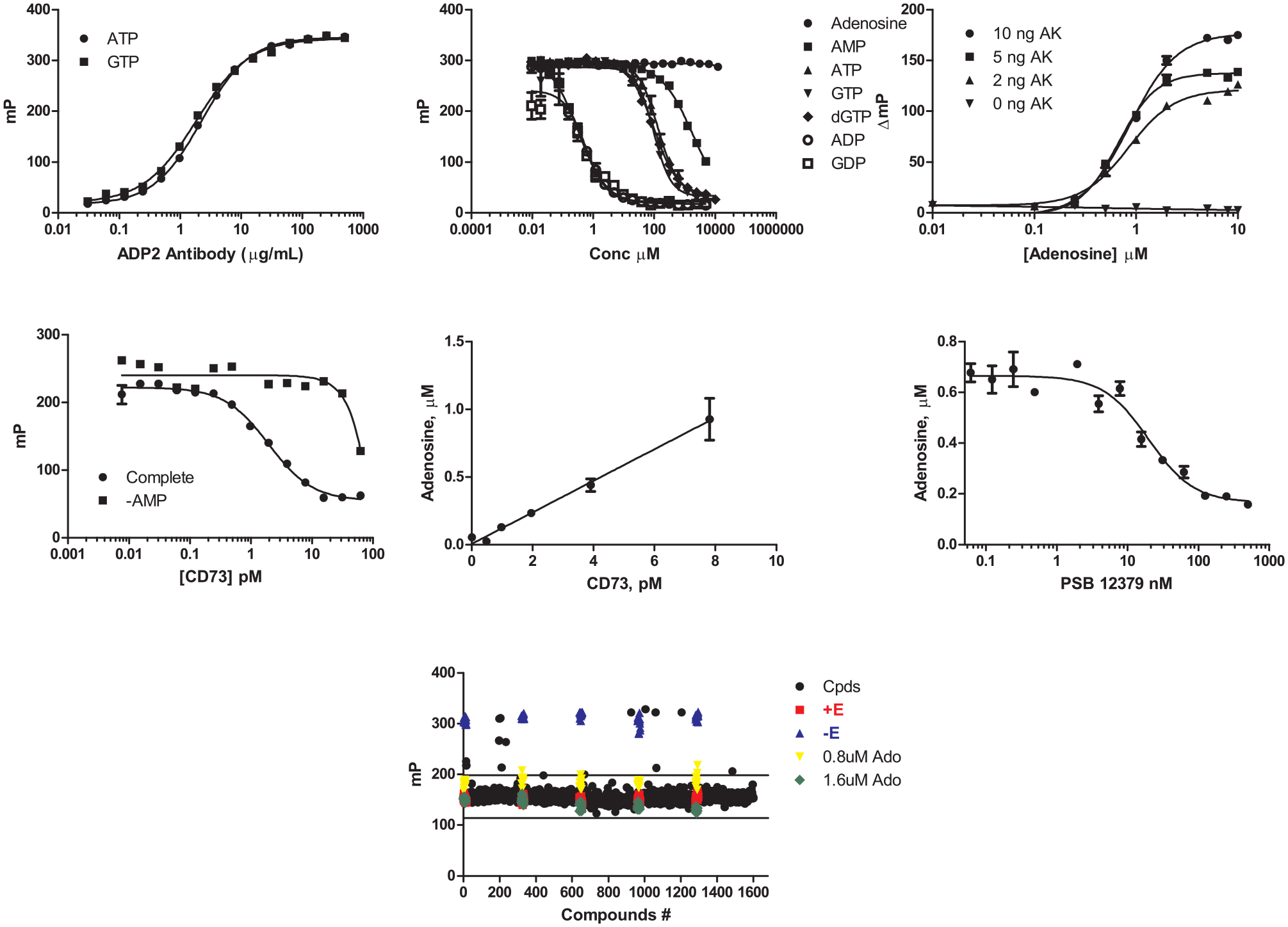
Development and optimization of CD73 activity assay for measurement of adenosine production in fluorescence polarization. (
PSB12379 is an α,β-methylene-ADP (AOPCP) derivative that inhibits ecto-5′-nucleotidase/CD73 with a reported IC50 of 2.2 nM.
14
The dose-response curve run using the AK-Transcreener ADP-coupled assay gave an IC50 value of 19 nM, slightly higher than what is reported (
Considering the pathophysiological roles of CD39 and CD73, details of which are only beginning to emerge, there is considerable untapped potential for drug discovery strategies that identify ectonucleotidase inhibitors. Biochemical inhibitors of CD39 include ATP analogs such as ARL67156, and nonnucleotide-derived NTPDase inhibitors such as dyes bearing sulfonate groups (e.g., suramin and related compounds), and POMs, which are inorganic, negatively charged metal complexes. 11 Fewer inhibitors of CD73 are known; they include a nucleotide analog called AOPCP and some naturally isolated flavonoids, such as quercetin and myricetin. 11 In general, such compounds are intrinsically useful for biochemical studies but lack utility as pharmacological agents because of limitations such as nonspecificity, high molecular weight, low potency, and unfavorable pharmacokinetic profiles.
Notably, Fiene et al. 9 reported the use of Transcreener assays for highly sensitive detection of NTPDases. The authors demonstrated direct detection of ADP when using ATP as a substrate (for NTPDase2, NTPDase3, and NTPDase8) or of AMP upon using ADP as a substrate (for CD39/NTPDase1). Because the FP and TR-FRET–based Transcreener assays are highly sensitive, low concentrations of substrate and enzyme can be used, which is advantageous when Km values are low and/or enzymes turn over slowly. This is especially helpful for the detection of poorly soluble competitive inhibitors.
In this study, we showed that the Transcreener AMP2 assay serves as a robust HTS method for CD39. The IC50 values obtained using the assay for two tool compounds for CD39 corresponded well with published values, and the assay was used successfully to identify hits from two pilot libraries. The ADP2 assay was similarly robust for detection and screening of CD73 when coupled with AK to convert adenosine to ADP. Although further investigation is needed to determine whether ectonucleotidase inhibitors could be viable drug candidates, drugs directed at purinergic pathway receptors suggest that the effort could prove fruitful. Success will ultimately require highly specific and sensitive HTS methods, such as the Transcreener AMP2 and ADP2 assays.
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are employed by BellBrook Labs, and their research and authorship of this article was completed within the scope of their employment with BellBrook Labs. BellBrook Labs manufactures and markets the Transcreener AMP2 and Transcreener ADP2 assay kits used in the research reported here. The authors declare no other potential conflicts of interest with respect to the research, authorship, and/or publication of this technical note.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
