Abstract
The present study examined the involvement of purinergic signaling components in the rat model of hippocampal degeneration induced by trimethyltin (TMT) intoxication (8 mg/kg, single intraperitoneal injection), which results in behavioral and neurological dysfunction similar to neurodegenerative disorders. We investigated spatial and temporal patterns of ecto-nucleoside triphosphate diphosphohydrolase 1 (NTPDase1/CD39) and ecto-5′ nucleotidase (eN/CD73) activity, their cell-specific localization, and analyzed gene expression pattern and/or cellular localization of purinoreceptors and proinflammatory mediators associated with reactive glial cells. Our study demonstrated that all Iba1+ cells at the injured area, irrespective of their morphology, upregulated NTPDase1/CD39, while induction of eN/CD73 has been observed at amoeboid Iba1+ cells localized within the hippocampal neuronal layers with pronounced cell death. Marked induction of P2Y12R, P2Y6R, and P2X4-messenger RNA at the early stage of TMT-induced neurodegeneration might reflect the functional properties, migration, and chemotaxis of microglia, while induction of P2X7R at amoeboid cells probably modulates their phagocytic role. Reactive astrocytes expressed adenosine A1, A2A, and P2Y1 receptors, revealed induction of complement component C3, inducible nitric oxide synthase, nuclear factor-kB, and proinflammatory cytokines at the late stage of TMT-induced neurodegeneration. An increased set of purinergic system components on activated microglia (NTPDase1/CD39, eN/CD73, and P2X7) and astrocytes (A1R, A2AR, and P2Y1), and loss of homeostatic glial and neuronal purinergic pathways (P2Y12 and A1R) may shift purinergic signaling balance toward excitotoxicity and inflammation, thus favoring progression of pathological events. These findings may contribute to a better understanding of the involvement of purinergic signaling components in the progression of neurodegenerative disorders that could be target molecules for the development of novel therapies.
Keywords
Introduction
Neurotoxicants, such as trimethyltin (TMT)-chloride, have been reported as risk factors for the development of neurodegenerative disorders (Kotake, 2012; Pompili et al., 2020; Yegambaram et al., 2015). In rats, TMT selectively targets the limbic region, particularly the hippocampus, with a similar pattern as observed in humans and with comparable behavioral alterations (Corvino et al., 2013, 2015; Ferraz da Silva et al., 2017; Geloso et al., 2011; Haga et al., 2002; Lattanzi et al., 2013; Lee et al., 2016; Trabucco et al., 2009). TMT-induced neurodegeneration in rats is characterized by early astrocyte activation followed by sustained astrogliosis, the response of resident microglial to hippocampal neuronal loss that progressively worsens over 3 weeks (Dragic et al., 2019b, 2021; Haga et al., 2002; Little et al., 2002, 2012), as well as a cognitive deficit in various tasks similar to human neurodegenerative disorders such as Alzheimer's disease and temporal lobe epilepsy (Chvojkova et al., 2021; Corvino et al., 2013; Geloso et al., 2011; Lattanzi et al., 2013; Lee et al., 2016; Pompili et al., 2020; Trabucco et al., 2009; Ye et al., 2020). Thus, TMT neurotoxicity is a valuable tool for studying changes in molecular signatures of glial cells during the progression of neurodegeneration that accompanies hippocampal dysfunction.
In general, glial cells, microglia, and astrocytes are crucial in monitoring, maintaining, and preserving the metabolic and structural integrity of the central nervous system (CNS), and respond to noxious stimuli and insults to the brain. Alterations in CNS homeostasis immediately lead to changes in microglial cells morphology and functional polarization toward one of the two complex phenotypes, detrimental that release proinflammatory cytokines and reactive oxygen species (ROS) and reactive nitrogen species or prorepair, an antiinflammatory phenotype that express molecular markers such as arginase-1 (Arg1) (Illes et al., 2020; Zabel & Kirsch, 2013), with a full repertoire of transitional states between them. In response to brain injury, astrocytes assume reactive states that may be discriminated based on the proliferation and induction of proinflammatory mediators and ROS (Verkhratsky et al., 2014). Furthermore, different polarized states of reactive astrocytes are characterized, determined as dominantly harmful, a proinflammatory type that might releases the neurotoxic complement C3 directly leading to neuron death, and the dominantly neuroprotective type (Liddelow & Barres, 2017), but also with the repertoire of transitional microenvironment-dependent states.
Communication between astrocytes, microglia, and degenerating neurons is mediated via different signaling molecules, and one of the strongest is adenosine triphosphate (ATP) (Sperlagh & Illes, 2007). A large amount of extracellular ATP, released from injured neurons and activated glial cells, acts as a “danger signal” and activates specific ligand-gated P2X channels and G-protein-coupled P2Y receptors (Burnstock, 2017; Di Virgilio et al., 2009; Sperlagh & Illes, 2007), promoting microglial chemotaxis and phagocytosis as well as the release of proinflammatory cytokines (Bernier et al., 2013; Franke et al., 2012; Haynes et al., 2006; Illes et al., 2020). Enzymes responsible for calibrating the duration, and degree of P2 receptor activation are functionally coupled membrane-bound ectonucleotidases named ecto-nucleoside triphosphate diphosphohydrolase 1 (NTPDase1/CD39) and ecto-5′ nucleotidase (eN/CD73) that rapidly hydrolyze ATP to adenosine (Grkovic et al., 2019a; Matyash et al., 2017; Zimmermann et al., 2012). NTPDase1/CD39 is dominantly expressed at microglia and endothelial cells and hydrolyzes ATP and adenosine diphosphate (ADP) to adenosine monophosphate (AMP) (Braun et al., 2000; Grkovic et al., 2019b; Matyash et al., 2017; Robson et al., 2006; Zimmermann et al., 2012). The resulting AMP is hydrolyzed to adenosine by eN/CD73, widely expressed in the hippocampus (Grkovic et al., 2019a, 2019b; Zimmermann et al., 2012). Adenosine G-protein-coupled receptors (A1R, A2AR, A2BR, and A3R) mediate modulatory effects of adenosine in an inflammatory environment (Hasko & Cronstein, 2013; Nedeljkovic, 2019). The two ectonucleotidases act together as an immune checkpoint since they determine the ATP/adenosine ratio and the inflammatory status of the tissue. Therefore, an altered function of NTPDase1/CD39 and eN/CD73 and dysregulation of the purinergic signaling are largely implicated in the pathophysiology of several neurological diseases, including Alzheimer's and Parkinson disease, multiple sclerosis, and astroglioma (Burnstock, 2017), but their cell-specific localization during neurodegeneration is rarely explored. Furthermore, NTPDase1/CD39 and eN/CD73 represent promising pharmacological targets in the treatment of neuroinflammatory processes (Antonioli et al., 2013).
It has been previously described an early change in astrocyte morphology that precedes neuronal loss, particular reactive astrocyte phenotypes, and their dynamic remodeling after TMT intoxication (Dragic et al., 2019b). It was also found that TMT-induced mitochondrial depolarization is independent of extracellular Ca2+ and disturbed antioxidative defense, but also upregulated main proinflammatory factors and components of signaling pathways responsible for astrocyte reactivity, and markers of proinflammatory subtype of astrocytes in vitro (Dragic et al., 2021). Induction of P2X2R in glial cells has been reported after TMT intoxication (Latini et al., 2010), but the involvement of other purinergic signaling components has not been explored. The main goal of the present study was to explore the cell-specific localization of NTPDase1/CD39 and eN/CD73 and the expression of purinergic receptors specific for microglia in the early and the late stage of hippocampal neurodegeneration induced by TMT. Furthermore, there is no information on whether TMT-induced inflammation in rats is caused by reactive microglia and/or astrocytes. Thus, in the present study, we analyzed NTPDase1/CD39 and eN/CD73, and purinergic receptors expression patterns in the context of activation of glial cells, inflammation, and its potential resolution after TMT intoxication. We also hypothesized that components of purinergic signaling may assign functional states of glial cells.
Material and Methods
Animals, Surgical Procedure, and Treatment
Two-month-old female rats of the Wistar strain (200–220 g) maintained in the local animal facility were used in the study. Appropriate actions were taken to alleviate the pain and discomfort of the animals following the compliance with the European Communities Council Directive (2010/63/EU) for animal experiments, and the research procedures were approved by the Ethical Committee for the Use of Laboratory Animals. Animals were housed 3–4/cage, in a 12 h light/dark regime, constant humidity and temperature, and free access to food and water.
It was previously shown that TMT-induced hippocampal neurodegeneration and gliosis, the pattern of which was comparable in adult rats of both sexes (Corvino et al., 2015; Dragic et al., 2019b; Geloso et al., 2011; Haga et al., 2002; Little et al., 2012; Trabucco et al., 2009). However, given that the expression of ectonucleotidases in the brain is modulated/regulated by gonadal steroids and differs in two sexes (Grkovic et al., 2019b; Mitrovic et al., 2016, 2017), the study was performed in female rats, bilaterally ovariectomized 3 weeks before TMT injection as we described previously (Dragic et al., 2019b).
On day zero, animals of the TMT group received TMT (8 mg/kg dissolved in 1 mL 0.9% w/v saline) (in the form of a single intraperitoneal [i.p.] injection), whereas the control (Ctrl) group received an adequate volume of 0.9% saline solution. The animals were returned to their cages, and monitored for unusual signs of behavior until sacrifice, as reported previously (Dragic et al., 2019b). At 7 and 21 days post intoxication (dpi), animals of TMT and age-matched Ctrl groups (10 animals/group) were sacrificed by decapitation (Harvard apparatus, Holliston, MA, USA).
Histochemistry, Immunohistochemistry, and Immunofluorescence Microscopy
Brains (
Nissl Staining
Alterations in hippocampal cytoarchitecture induced by TMT injection were evaluated by Nissl staining. Sections were kept in 0.5% thionine solution for 20 min, washed in tap water, dehydrated in graded ethanol (70%–100%), cleared in xylene for 2 × 5 min, and covered with DPX-mounting medium (Sigma Aldrich, USA).
Immunohistochemistry and Immunofluorescence
Slides were kept at room temperature (RT) for 30 min before staining. After washing in phosphate-buffered saline (PBS), slides were put in 0.3% H2O2 in methanol for 20 min, to block endogenous peroxidase, and then immersed in 5% donkey normal serum at RT for 1 h to block nonspecific binding. Sections were probed with primary antibodies, overnight at 4 °C in a humid chamber. After washing in PBS (3 × 5 min), sections were incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies (2 h, RT in a humid chamber). The list of antibodies used for immunohistochemistry (IHC) and immunofluorescence (IF) is presented in Table 1. The immunoreaction was visualized with 3,3′S-diaminobenzidine-tetrahydrochloride (Abcam, UK), which is converted to the insoluble brown precipitate by HRP. Sections were washed in distilled water, dehydrated in graded ethanol solutions (70%–100%), cleared in xylene, and mounted with the use of DPX-mounting medium (Sigma Aldrich, USA). Sections were analyzed under a LEITZ DM RB light microscope (Leica Mikroskopie & Systems GmbH, Wetzlar, Germany), equipped with a LEICA DFC320 CCD camera (Leica Microsystems Ltd, Heerbrugg, Switzerland), and LEICA DFC Twain Software (Leica, Germany). All images were captured at 40× magnification.
List of Antibodies.
The identical protocol has been applied for double and triple IF staining, with the omission of the methanol/H2O2 step. After incubation with primary antibodies (Table 1), sections were probed with fluorescence dye-labeled secondary antibodies and mounted with Mowiol (Calbiochem, La Jolla, CA). For double and triple IF staining, primary and secondary antibodies were separately applied for each labeling. Sections incubated without primary antibodies or with rat preimmune sera were used as negative Ctrls. Sections were analyzed by a confocal laser-scanning microscope (LSM 510, Carl Zeiss GmbH, Jena, Germany), using Ar multiline (457, 478, 488, and 514 nm), HeNe (543 nm), and HeNe (643 nm) lasers using 63× (2× digital zoom) DIC oil, 40× and monochrome camera AxioCam ICm1 camera (Carl Zeiss GmbH, Germany).
Enzyme Histochemistry
Ectonucleotidase enzyme histochemistry based on the ATP/ADP- and AMP-hydrolyzing activities of NTPDase1/CD39 and eN/CD73, respectively, have been applied (Dragic et al., 2019a; Grkovic et al., 2019b). Briefly, cryosections were preincubated for 30 min at RT in Tris-maleate sucrose (TMS) buffer, containing 0.25 M sucrose, 50 mM Tris-maleate, 2 mM MgCl2 (pH 7.4), and 2 mM levamisole, to inhibit tissue nonspecific alkaline phosphatase. The enzyme reaction was carried out at 37 °C/60 min, in TMS buffer, containing 2 mM Pb(NO3)2, 5 mM MnCl2, 3% dextran T250, and 1 mM substrate (ATP, ADP, or AMP). After thorough washing, slides were immersed in 1% (v/v) (NH4)2S, and the product of enzyme reaction was visualized as an insoluble brown precipitate at a site of the enzyme activity. After dehydration in graded ethanol solutions (70%–100% EtOH, and 100% xylol), slides were mounted with a DPX-mounting medium (Sigma Aldrich, USA). The sections were examined under a LEITZ DM RB light microscope (Leica Mikroskopie & Systems GmbH, Wetzlar, Germany), equipped with a LEICA DFC320 CCD camera (Leica Microsystems Ltd, Heerbrugg, Switzerland) and analyzed using LEICA DFC Twain Software (Leica, Germany).
IF Quantification
Raw multiimage IF micrographs were used to measure integrated fluorescence density expressed as arbitrary units (AUs) and the density confined within five predefined regions of interest (ROIs), with background fluorescence subtraction for at least three images per ROI and
Gene Expression Analysis by Quantitative Reverse Transcriptase-Polymerase Chain Reaction
Total RNA was extracted from the hippocampal formation (7 and 21 dpi and appropriate age-matched Ctrls,
Primer Sequences Used for RT-qPCR.
Statistical Analysis
Data were analyzed for normality and appropriate parametric tests were used. All values are presented as mean ± SD or SEM. Between-group comparisons for 7 and 21 dpi were analyzed using an unpaired
Results
Spatiotemporal Patterns of Neurodegeneration and Gliosis After TMT Exposure
TMT-induced hippocampal degeneration was confirmed by Nissl staining (Figure 1a). As we have shown previously (Dragic et al., 2019b), cell injury was observed in the hilar/proximal CA3 (hilus/pCA3) at 7 dpi. This is followed by the almost complete disappearance of staining in neuronal somata in CA1 and the proximal and medial CA3 (p/mCA3) regions at 21 dpi (Figure 1a), as already reported (Geloso et al., 2011; Haga et al., 2002; Latini et al., 2010; Little et al., 2012). As reported previously (Dragic et al., 2019b), immunostaining of astrocyte marker glial fibrillary acidic protein (GFAP) showed the presence of pronounced astrogliosis at 7 dpi as well as 21 dpi (Figure 1b).

Spatiotemporal pattern of hippocampal neurodegeneration and gliosis after TMT exposure (a) thionine staining of coronal sections obtained from control animals and at 7 and 21 dpi. Arrowheads indicated injured neuronal cell layers in the hippocampus. Scale bar = 500 μm. (b) Immunohistochemical staining of GFAP in control animals and at 7 and 21 dpi. Scale bar = 500 μm. (c) Immunohistochemical staining of Iba1 in the whole hippocampal area and corresponding enlarged CA1 and mCA3 at 7 and 21 dpi. Scale bar = 500 μm (under 5× magnification), and 100 μm (under 20× magnification). (d) Representative images of different Iba1-
A great morphological diversity of reactive microglia was observed (Figure 1c and d). Specifically, highly ramified Iba1-immunoreactive (
Expression of NTPDase1/CD39, eN/CD73, and Purinoreceptors Involved in Microglial Reactivity
The main goal of the present study was to explore the involvement of the purinergic signaling system in TMT-induced hippocampal neurodegeneration and gliosis. We first analyzed the expression of genes encoding NTPDase1/CD39 and eN/CD73. There was a significant increase in the relative expression of NTPDase1/CD39-mRNA in the hippocampal tissue at 7 and 21 dpi (
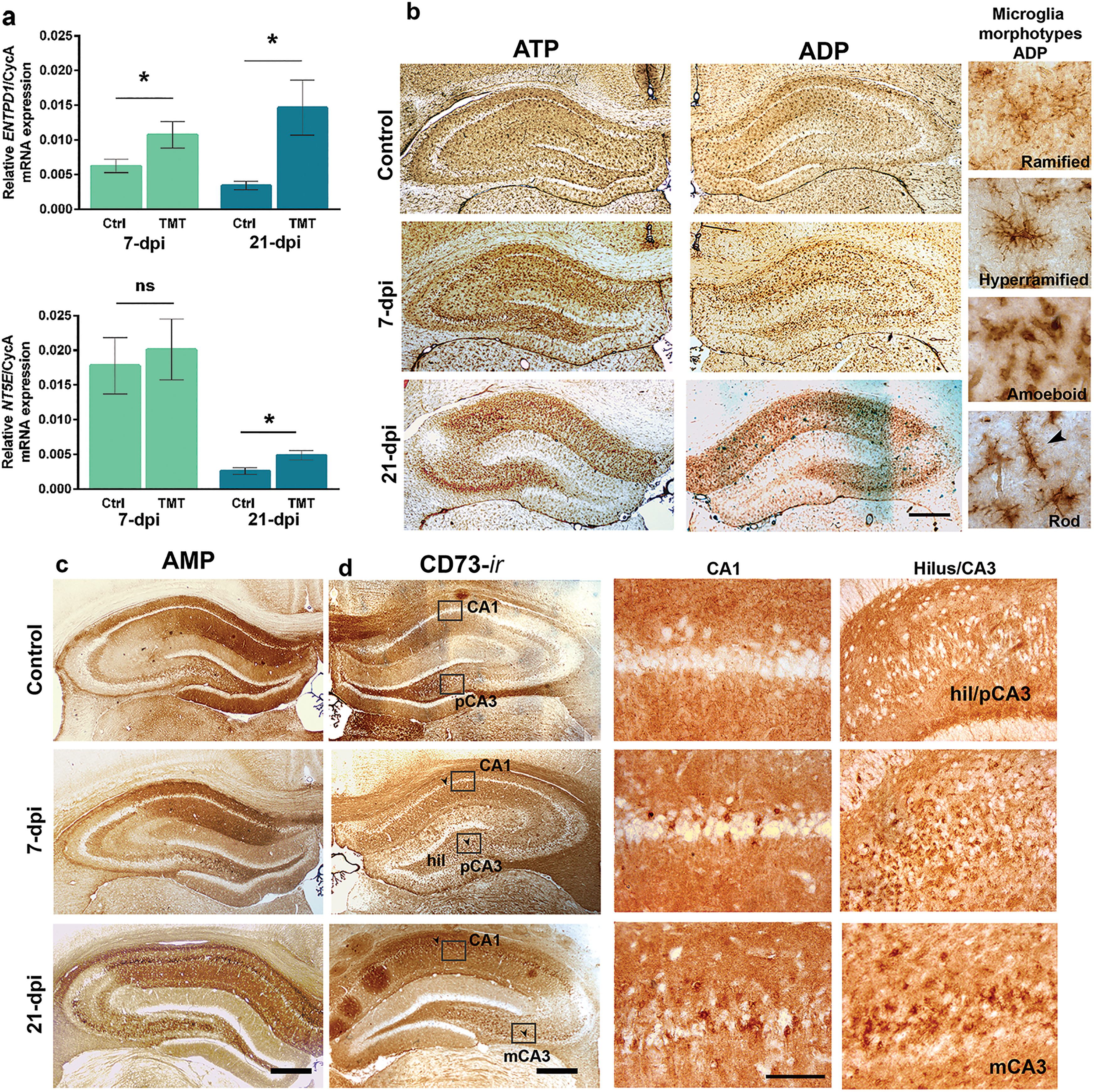
Expression and activity of NTPDase1/CD39 and eN/CD73 in the hippocampal region after TMT exposure (a) RT-qPCR analysis of genes encoding NTPDase1/CD39 and eN/CD73 in the Ctrl hippocampal tissue, 7 and 21 dpi, respectively. Bars represent mean mRNA expression of target gene relative to CycA ± SD. Significance shown inside the graphs: *
The pattern of the enzyme activity in the hippocampus and the localization of upregulated NTPDase1/CD39 in response to TMT were determined by enzyme histochemistry using ATP and ADP as a substrate
eN/CD73 activity and localization in response to TMT were determined using AMP-based enzyme histochemistry (Figure 2c) and eN/CD73-directed immunocytochemistry (Figure 2d). In intact hippocampal tissue, diffuse histochemical reaction and eN/CD73-
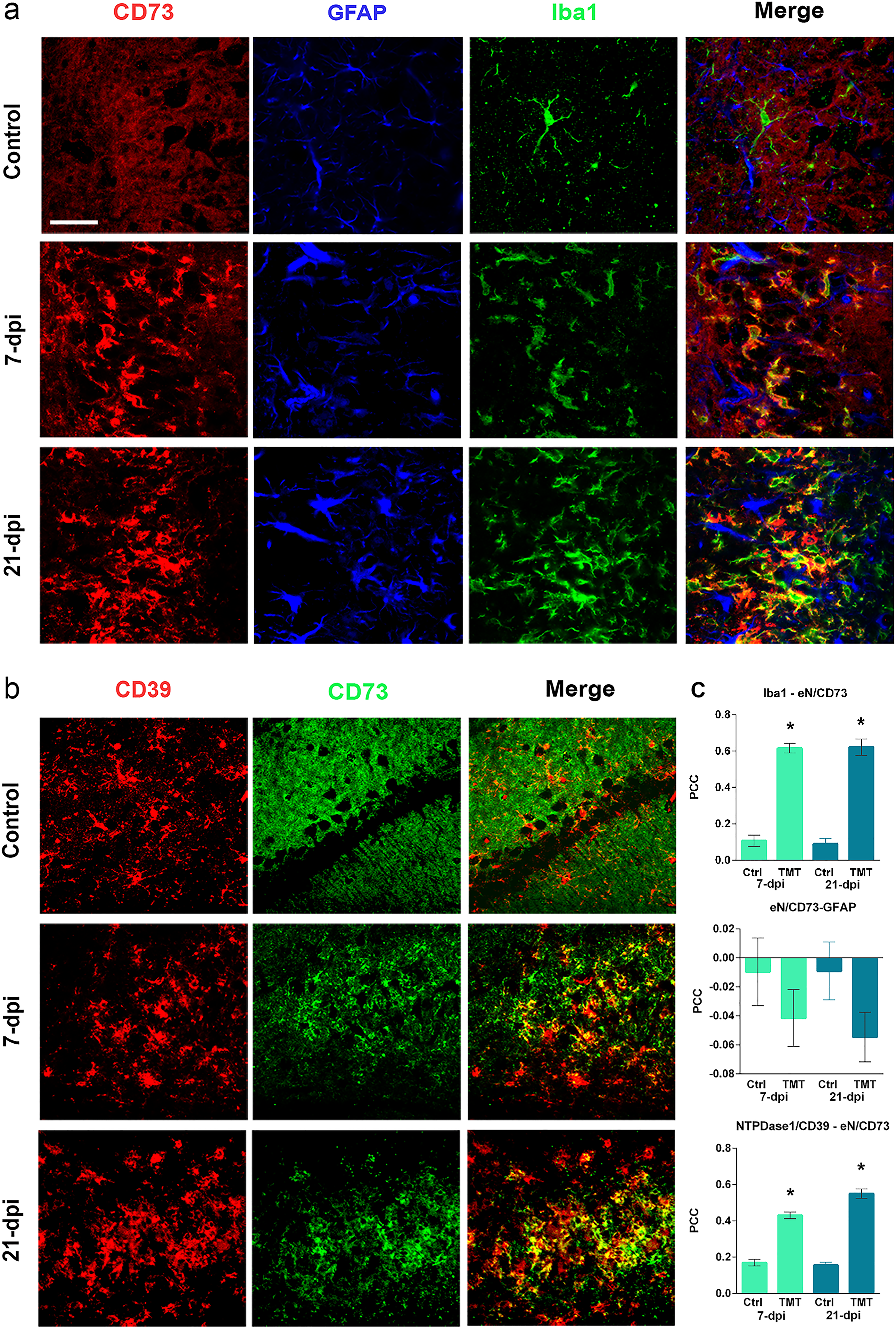
Identification of cells that upregulate eN/CD73 in the hippocampal region after TMT exposure (a) triple IF labeling directed to eN/CD73 (red), astrocyte marker GFAP (blue), and microglial marker Iba1 (green) in the Ctrl, 7 and 21 dpi hippocampi. Overlaid images (merge) reveal the overlapping signal corresponding to Iba1-
Since the role of extracellular ATP is closely related to its breakdown products, changes in mRNA expression of ATP/ADP-sensitive P2 and adenosine P1 receptors in the hippocampus during TMT-induced neurodegeneration were explored (Figure 4). Regarding ATP/ADP-sensitive P2 receptors mainly expressed by microglia (Illes et al., 2020), a significant increase in the relative abundance of P2X4R-, P2Y2R-, and P2Y6R-mRNA levels were observed, at both 7 dpi (
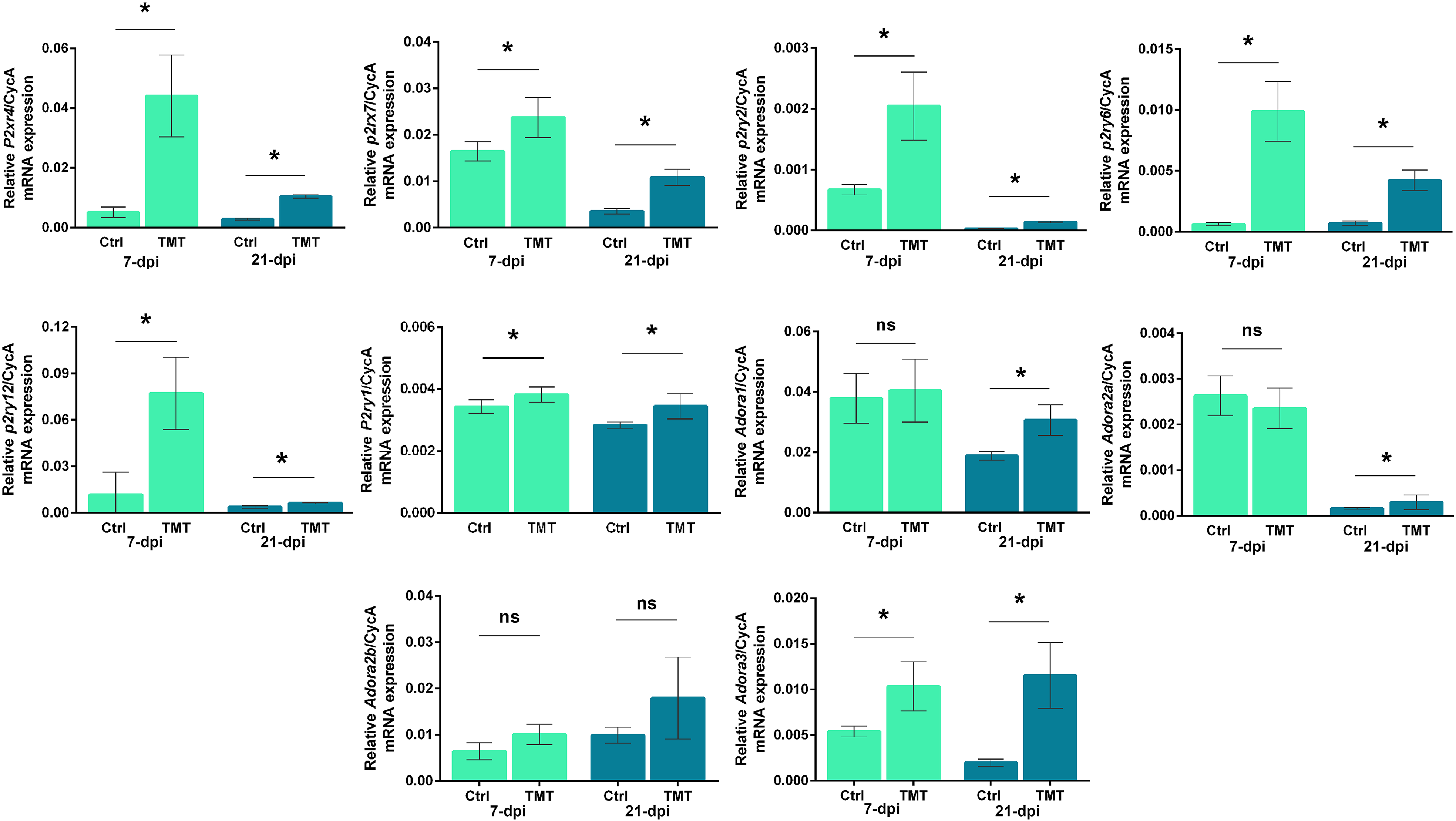
Purinoceptors gene expression in the hippocampal region after TMT exposure The abundances of transcripts coding for P2X4, P2X7, P2Y1R, P2Y2R, P2Y6R, P2Y12R, A1R, A2AR, A2BR, and A3R were assessed by RT-qPCR at 7 and 21 dpi. Bars represent mean mRNA expression of target gene relative to CycA ± SD. Significance shown inside the graphs: *
The Inflammatory Status of the Hippocampal Tissue After TMT Exposure
It is known that activated glial cells develop functional phenotypes, which may be roughly categorized as proinflammatory or antiinflammatory. Therefore, we first assessed the inflammatory status of the hippocampal tissue at the early (7 dpi) and the late (21 dpi) stage of TMT-induced neurodegeneration by determining the expression of several inflammatory markers. As shown in Figure 5, only tumor necrosis factor-α (TNF-α)-mRNA level was significantly increased at 7 dpi when compared to Ctrl (
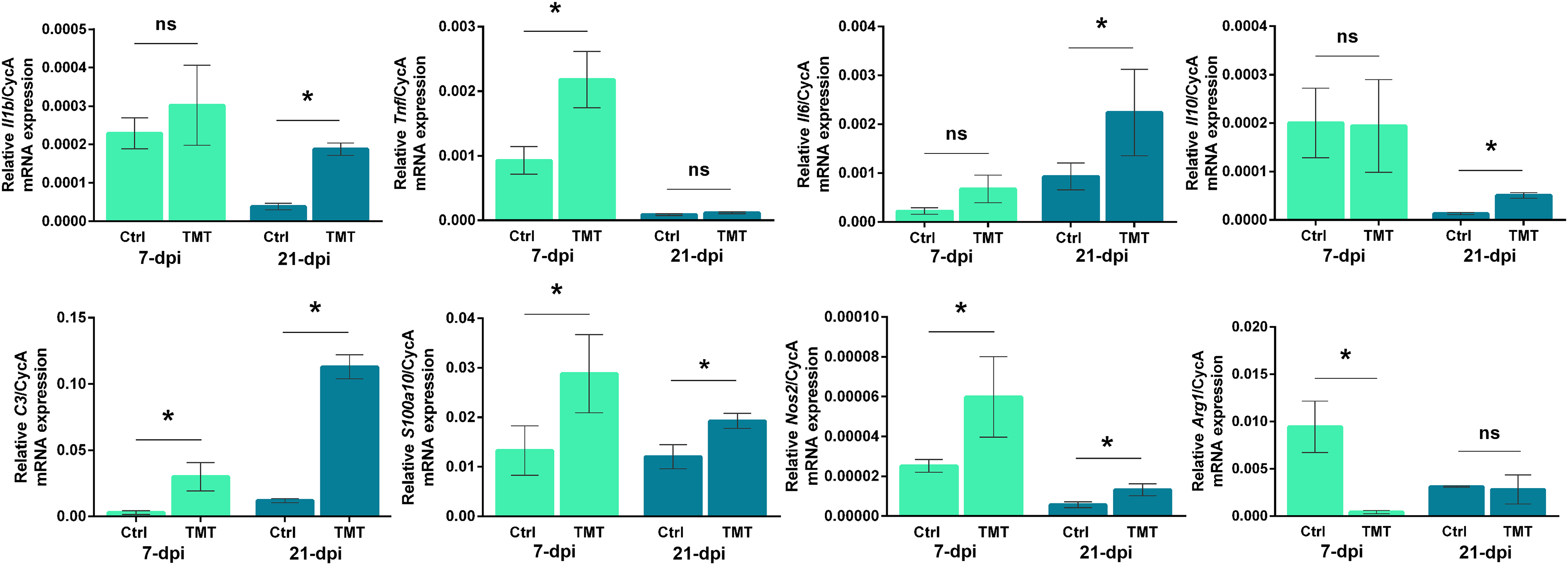
Proinflammatory status of the rat hippocampal region after TMT exposure The abundance of transcripts coding IL-1β, TNF-α, IL-6, IL-10, C3, S100a10, iNOS, and Arg1. Bars represent mean mRNA expression of target gene relative to CycA ± SD. Significance shown inside the graphs: *
Functional State of Reactive Microglia and Astrocytes
Next, we sought to determine the cellular source of inflammation and performed colocalization of Iba1 or GFAP against inflammatory markers. Neither of the tested proinflammatory cytokines (IL-1β, TNF-α, and IL-10) and C3 (data not shown) nor polarization marker iNOS (Figure 6) was found in association with Iba1-
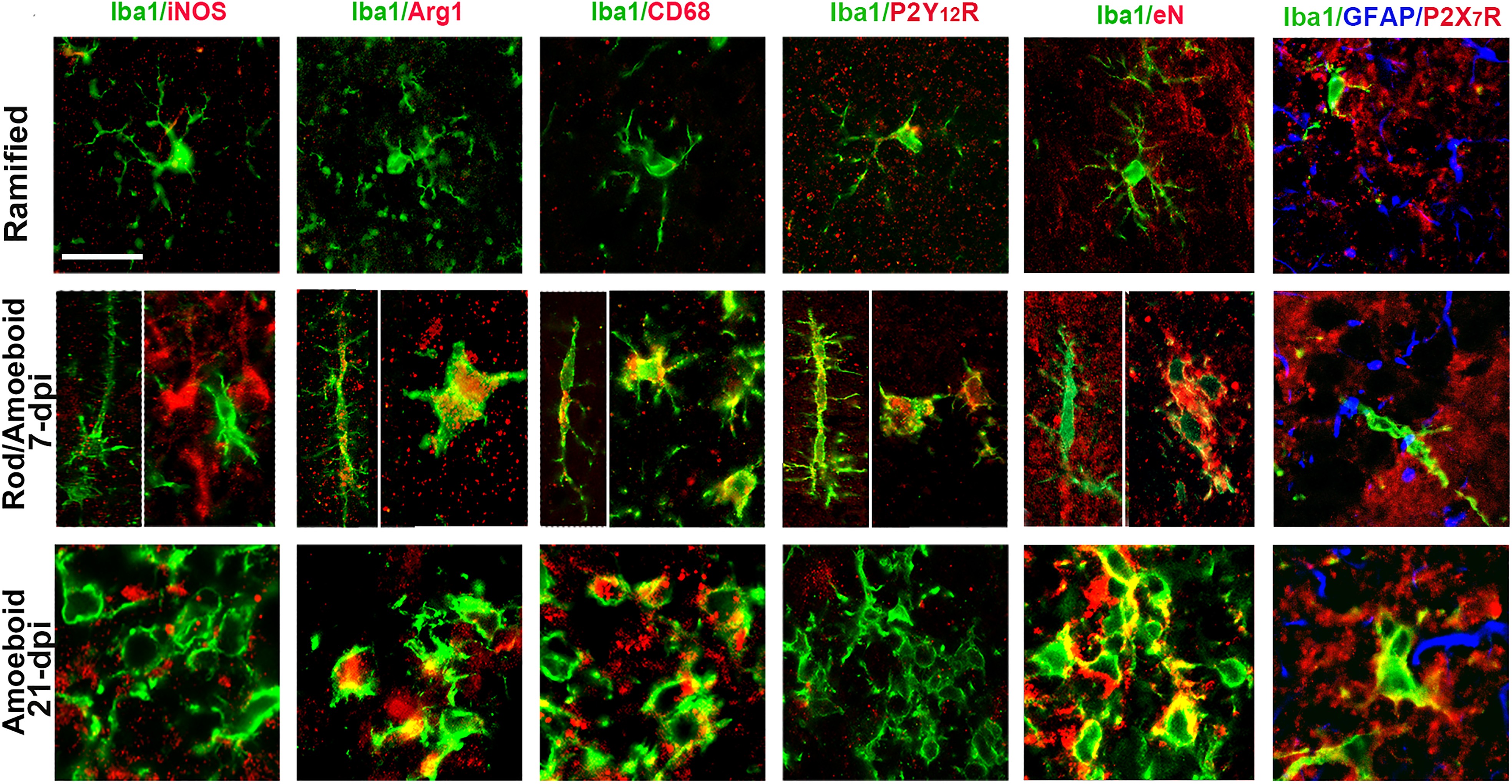
Assessment of the functional state of reactive microglia after TMT exposure. Ramified morphology of Iba1+cells corresponds to control microglia but also to ramified Iba1+ cells in the hippocampal areas distant from the site of neurodegeneration at both 7 and 21 dpi. Double immunofluorescent staining of Iba1 and iNOS, Arg1, CD68, P2Y12 receptor (R), and eN/CD73, and triple immunofluorescent staining of Iba1, GFAP and P2X7R in the injured area 7 and 21 dpi, reveal Iba1-
The lack of expression of proinflammatory cytokines and markers by Iba1-
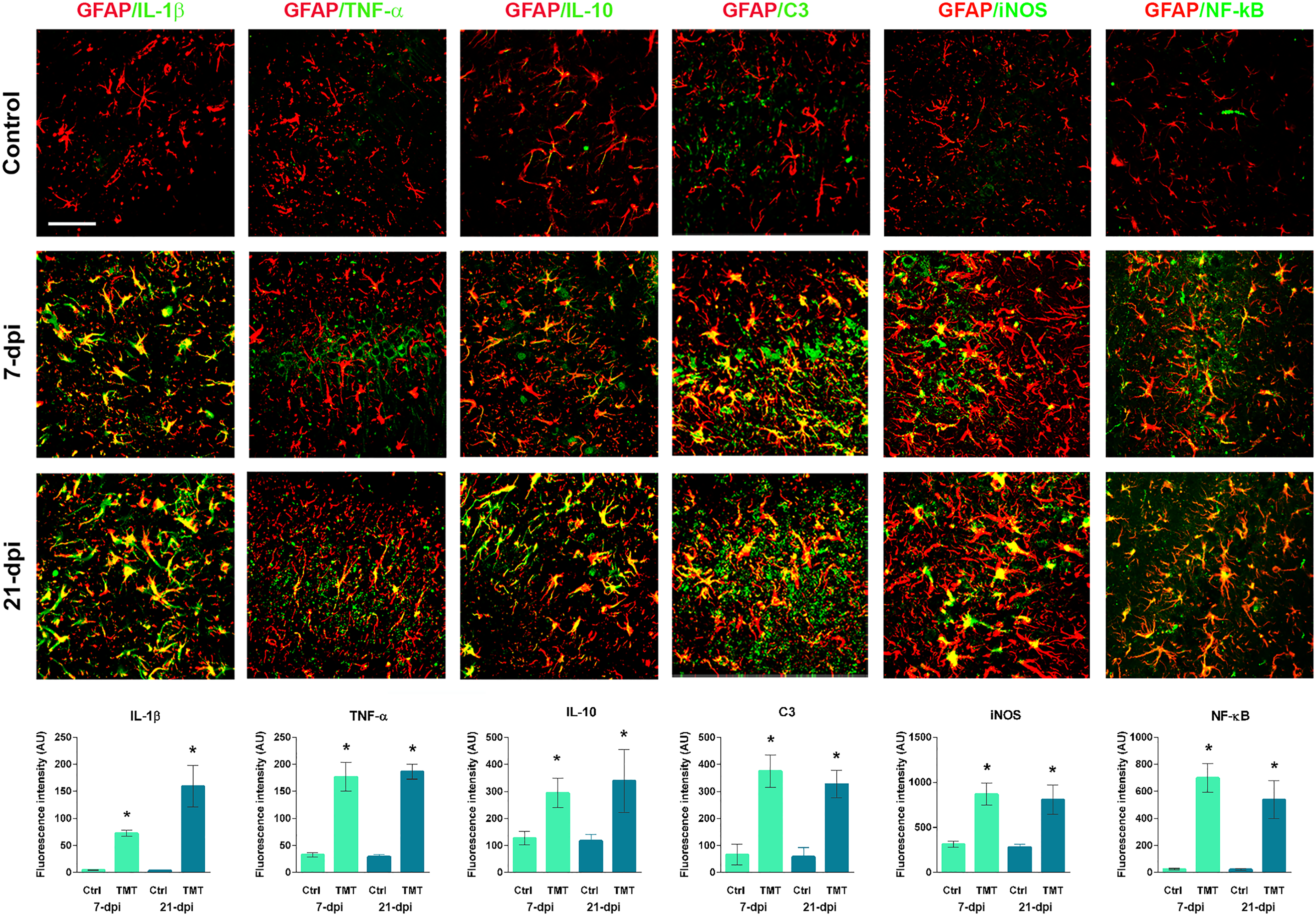
Assessment of the functional state of reactive astrocytes after TMT exposure. Double immunofluorescent staining of GFAP and IL-1β, TNF-α, IL-10, C3, iNOS and NF-kB and corresponding integrated fluorescence density expressed as AUs ± SEM in the injured CA area at 7 and 21 dpi. Significance shown inside the graphs: *
Since astrocytic P2Y1R is involved in the regulation of several cytokines/chemokines expression (e.g., IL-6, and TNF-α) (Kuboyama et al., 2011), and A2AR upregulation in activated glial cells facilitates the release of cytokines (Paiva et al., 2019), we explore their localization. In addition, prolonged adenosine A1R signaling and its cross-talk with A2AR might enhance A2AR-mediated neurotoxicity in neurodegenerative disorders (Stockwell et al., 2017). Thus, massive induction of P2Y1R and A2AR was found on GFAP-
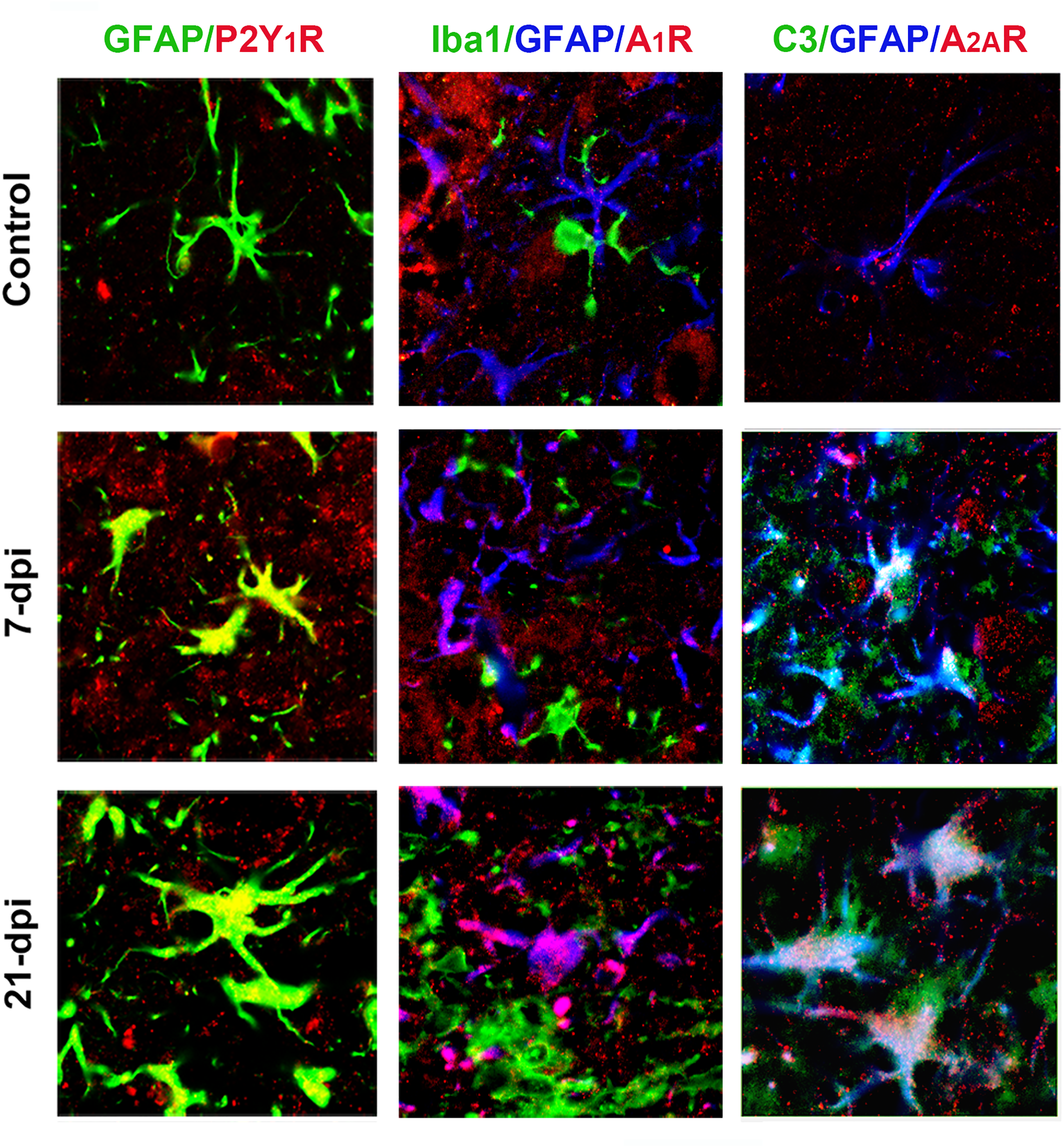
Association of P2Y1 and adenosine receptors with GFAP+ astrocytes in the hippocampus after TMT exposure. Double IF reveals that GFAP-
Discussion
The results of the present study corroborate the existing data on the spatiotemporal pattern of neurodegeneration and gliosis in the rat TMT model (Corvino et al., 2013, 2015; Haga et al., 2002; Latini et al., 2010; Little et al., 2012; Trabucco et al., 2009). As described and analyzed previously (Dragic et al., 2019b), reactive astrocytes (from day 2 post-TMT) were polarized toward the jeopardized regions, enclosing it and probably creating a protective glial barrier, keeping other regions from damage at the early stage of TMT-induced neurotoxicity (Dragic et al., 2019b). Microglial activation induced by TMT slightly lagged behind astrocyte reactivation, as observed earlier (Haga et al., 2002), and is manifested as a robust increase in the number of Iba1+ cells due to migration or proliferation of resident microglia (Little et al., 2002). However, we observed that synaptic layers in the affected sectors became largely populated with
As a marker of microglia (Almolda et al., 2013; Braun et al., 2000), NTPDase1/CD39 activity and expression were markedly upregulated in all Iba1-
Microglial cell migration and chemotaxis depend on purinergic signaling via P2 receptors (Illes et al., 2020; Koizumi et al., 2007), which also triggers their shift to amoeboid phenotype (Illes et al., 2020). Thus, we found an increase in relative gene expression of P2Y12R specifically at the early stage (7 dpi) of TMT-induced neurodegeneration. This receptor is activated by extracellular ATP released from damaged cells that trigger microglial processes extension and migration to the site of injury (Illes et al., 2020). Further, we found an increase in P2X4 relative gene expression that may contribute to both migratory as well as secretory properties of microglia, and interacts with the P2Y12R in the regulation of chemotaxis (Illes et al., 2020). At this stage, an increase of P2Y6-mRNA level was observed, a receptor upregulated when neurons become damaged and send diffusible uridine 5′-diphosphate (UDP) signals to microglia (Illes et al., 2020). Adenosine also affects extension and chemotaxis to the site of active neurodegeneration via P2Y12R/A3R coactivation (Haynes et al., 2006; Ohsawa et al., 2012). The ADP-driven process extension was reversed to process retraction during proinflammatory condition coincident with P2Y12R protein downregulation (Orr et al., 2009), which we observed at the late stage of TMT-induced neurodegeneration. Additionally, P2Y6R stimulation blocks ATP-dependent migration of microglia, most likely by shifting its migratory phenotype to an amoeboid/phagocytic one (Bernier et al., 2013; Koizumi et al., 2007), and which upregulation persist at the late stage (21 dpi) of neurodegeneration. The mRNA levels of P2X7R, the ATP-sensitive receptor predominantly localized on microglial cells in the brain (Illes et al., 2020), were also increased at both the early and the late stage of neurodegeneration.
Concerning the polarization state of microglia, our data showed that Iba1-
According to the literature data (Little et al., 2002, 2012) early neurodegenerative response to TMT is not accompanied by increased gene expression of most proinflammatory cytokines. We found moderate induction of TNF-α in neurons at the early stage of TMT-induced neurodegeneration, while almost all GFAP+ cells around damaged areas (CA1 and mCA3) were the source of IL-1β, TNF-α, and IL-10 particularly at the late stage of TMT-induced neurodegeneration, supporting in vitro results (Dragic et al., 2021) and studies that showed upregulation of other proinflammatory mediators at the later stages of TMT-induced neurodegeneration (Lattanzi et al., 2013; Liu et al., 2005; Morita et al., 2008). Furthermore, P2X7R is considered as a major driver of inflammation (Di Virgilio et al., 2017; Erb et al., 2019; Franke et al., 2012; Peterson et al., 2010), and we observed increased neuronal staining with P2X7R at both the early and the late stage of TMT-induced neurodegeneration. A wave of neurodegeneration induced by TMT might provide conditions for a sustained ATP release and prolonged P2X7R activation with the resulting postponed induction of IL-1β, together with TNF-α, and IL-6. Reactive astrocytes might initiate upregulation of P2Y2R, as we observed after TMT intoxication, leading to a proinflammatory response (Peterson et al., 2010).
We have also observed that reactive astrocytes coexpressed iNOS, NF-kB, C3, and found increased expression of S100a10-mRNA. Taken together with the expression of main proinflammatory markers, it could be concluded that TMT-induced reactive astrogliosis exerts a complex molecular signature with the predominantly inflammatory phenotype (Escartin et al., 2021), mainly located around the sites of ongoing neurodegeneration. These proinflammatory astrocytes at the injured area also upregulate A1R and A2AR receptors. In the hippocampus, adenosine exerts inhibitory function under physiological conditions due to the high expression of neuronal A1R, modulating important processes such as learning and memory (Costenla et al., 2010; Stockwell et al., 2017). On the other hand, under pathological conditions, an increase of both mRNA and protein levels and aberrant signaling via hippocampal A2AR have been demonstrated to contribute to active neuroinflammation and cognitive deficits (Hu et al., 2016), both of which are seen in TMT-induced neurodegeneration (Geloso et al., 2011). Adenosine signaling via neuronal A1R supports survival, exerts neuroprotective effects, and has anticonvulsive properties (Glass et al., 1996). Thus, the shift of A1R immunoreactivity from neurons to astrocytes could render neurons vulnerable to secondary effects of TMT, such as seizures (Trabucco et al., 2009), while prolonged adenosine A1R signaling and its cross-talk with A2AR might enhance A2AR-mediated neurotoxicity in neurodegenerative disorders (Stockwell et al., 2017). Taken together with a concomitant increase of astrocytic A2AR, these results could be put in perspective of potential formation of A1R–A2AR heteromers, which are shown to contribute to dysregulation of glutamate homeostasis and favor excitotoxicity (Borroto-Escuela et al., 2018; Hou et al., 2020). A high amount of ATP released after TMT intoxication would be degraded by Iba1+/CD39+/CD73+ cells thus producing high levels of adenosine around the sites of injury. Adenosine in such a microenvironment may activate astrocytic A1R and A2AR supporting their proinflammatory phenotype (Nedeljkovic, 2019; Paiva et al., 2019; Popoli & Pepponi, 2012). Additional experiments are necessary to fully elucidate the role of adenosine in such a complex pathological environment. Induction of P2Y1R on reactive astrocytes further confirms their detrimental phenotype after TMT-induced neurotoxicity, since this receptor is also involved in the regulation of several cytokines/chemokines expression (Kuboyama et al., 2011), and causes astrocytic hyperactivity and dysfunction in an animal model of neurodegeneration (Delekate et al., 2014).
In summary, identification of expressional timeline of selected purinoreceptors and ectonucleotidases provides a framework for the reconstruction of their involvement in the initiation and progression of neurodegenerative events after TMT intoxication. This study suggests that proinflammatory astrocytes phenotype is possibly developed as a response to TMT intoxication. Increased availability of ligands such as ATP and adenosine coupled with a distinct set of activated glial purinergic repertoire (P2X7, A2AR, P2Y1, and A1R) and loss of homeostatic glial and neuronal purinergic pathways (P2Y12 and A1R) may shift purinergic signaling balance toward excitotoxicity and inflammation, thus ultimately favoring progression of pathological events. Targeting the upstream nucleotide metabolic pathway that controls adenosine production to modulate neural-immune interactions and neurodegeneration-related machinery represents a promising therapeutic strategy for intervening in disease progression.
Supplemental Material
sj-docx-1-asn-10.1177_17590914211044882 - Supplemental material for Microglial- and Astrocyte-Specific Expression of Purinergic Signaling Components and Inflammatory Mediators in the Rat Hippocampus During Trimethyltin-Induced Neurodegeneration
Supplemental material, sj-docx-1-asn-10.1177_17590914211044882 for Microglial- and Astrocyte-Specific Expression of Purinergic Signaling Components and Inflammatory Mediators in the Rat Hippocampus During Trimethyltin-Induced Neurodegeneration by Milorad Dragić, Nataša Mitrović, Marija Adžić, Nadežda Nedeljković and Ivana Grković in ASN Neuro
Footnotes
Acknowledgments
The research was funded by the Ministry of Education, Science, and Technological Development of the Republic of Serbia Nos. 451-03-1/2021-16/14 –0902102 and 451-03-68/2020-14/200178. The authors thank Professor Jean Sevigny from the Faculté de Médecine, Université Laval, Quebec City, QC, Canada for a kind gift of rabbit anti-rat NTPDase1/CD39 (mN1-2C) and eN/CD73 (rNu-9L) antibodies used in this study. We would also like to thank our Dr. Ivana Bjelobaba, Institute for Biological Research “Siniša Stanković,” National Institute of the Republic of Serbia, University of Belgrade, Belgrade, Serbia, for providing us with qPCR primers used in this study.
Author Contributions
All authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article. I.G. conceived and directed the projects. M.D. and I.G. designed experiments and performed all histology, analyzed the data, and wrote the manuscript. N.M. performed qPCR experiments. M.A. was involved in confocal microscopy and image acquisition. N.N. was involved in data interpretation and wrote the manuscript. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis. All authors read, revised, and approved the final manuscript.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethics Approval
The Ethical Committee approved all animal procedures for the Use of Laboratory Animals of “VINČA” Institute of Nuclear Sciences—National Institute of Republic of Serbia, University of Belgrade, Belgrade, Serbia, and animals were treated following the European Community Council Directive of 86/609/ EEC for animal experiment.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministarstvo Prosvete, Nauke i Tehnološkog Razvoja (grant numbers 451-03-68/2020-14/200178 and 451-03-1/2021-16/14 –0902102).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
