Abstract
This study aimed to develop and compare single and multiple 3D models such as multicellular tumor spheroids and to investigate the influence of Matrigel on their morphological and functional behavior. MDA-MB-231 3D models were generated in the presence and absence of Matrigel and their key biological properties within 6 days of culture were monitored. Our results revealed the formation of well-defined 3D models in the presence of Matrigel, with a uniform morphology, increased diameter, good circularity, and increased expression of a proliferation marker (PCNA). In comparison, 3D models generated without Matrigel were characterized by an irregular border, reduced dimensions and circularity, and a decrease of PCNA expression. Similarities between the single and multiple 3D cultures were found in their viability, Nrf2 expression, and glutathione (GSH) content. The influence of Matrigel on MDA-MB-231 spheroids metabolism under hypoxic conditions was highlighted by released lactate dehydrogenase and nitric oxide, GSH levels and expression of Nrf2 and Hsp70 proteins. Based on the increased expression of PCNA and the development of the hypoxia process in the presence of extracellular matrix, our study showed that the addition of Matrigel improves the growing environment of tumor spheroids, making it closer to that of in vivo tumor conditions.
Introduction
Selection of the experimental model represents the critical step of each in vitro experiment, in order to obtain a situation close to that existing in human tissues. Two-dimensional (2D) cell culture is the most used experimental model, but mechanical and chemical cues, tissue-specific architecture, and cell–extracellular matrix interactions, which are elements essential for the functioning of real tissues, are lost in most cases. 1
New molecules with excellent antitumor properties in vitro proved to be ineffective in in vivo preclinical tests because 2D cell cultures could not reproduce the complexity and heterogeneity of tumor tissues. 2 In order to compare the results obtained in vitro with the in vivo ones and to rely on the mechanisms identified in cell culture, it is important to develop and perform the experiments using a research model that is morphologically and functionally similar to in vivo tissues.
For the past three decades, three-dimensional (3D) experimental models were developed and explored. These models proved to be more adequate for studying cell biology and physiology because they mimic the in vivo environment better than 2D culture. 3 They are represented mainly by spherical models that are classified in four groups: multicellular tumor spheroids (MCTSs), tumor spheres, tissue-derived tumor spheres, and organotypic multicellular spheroids. 4 The MCTSs, which are the most used 3D models are cell aggregates with many cell–cell and cell–extracellular matrix interactions that restore the parameters of original tissues characterized by nutrient gradients, gases, waste, and hypoxia. 1 These models are organized in three layers of cells found in different stages of the cell cycle: the first one is characterized by proliferative cells, followed by cells in a quiescent state, and the last one in the spheroid’s center is represented by necrotic cells. A hypoxic core is formed due to the lack of oxygen in the spheroid’s center, which is a specific structure of this model. However, the development of the necrotic center and hypoxia condition were mostly found in spheroids larger than 500 µm, reflecting the environment of avascular tumors presented in many types of cancers. 5
Various techniques have been developed in order to generate uniform and reproducible MCTSs in a short time. 6 The most used method is the liquid-overlay technique that is characterized by the presence of a nonadherent surface such as agarose or 2-methacryloyloxyethyl phosphorylcholine. A low-attachment surface determines the appearance of many cell–cell interactions in favor of those established between cells and the surface, leading to aggregates’ formation. The liquid-overlay technique has better efficiency, generating spheroids of uniform size 6 compared with other approaches. This technique can also be used to generate 3D co-culture models. The 3D co-cultures are formed from two or more cellular types and differentiate from MCTSs through the presence of stromal cells, important components of the in vivo tumor environment. 7 As a consequence, the development of such a complex experimental model leads to different behavior, dynamics, and responses, closer to those found in real tissues.
The liquid-overlay technique can generate single and multiple spheroids. Frequently, individual spheroids are formed when the technique is performed in 96-well round-bottom plates, while flat culture dishes are more suitable for obtaining multiple spheroids. 8 By comparison, single spheroids are homogenous in size and shape, while multiple spheroids might have various dimensions, which could influence the uniformity of results. Usually, multiple spheroids merge in time, leading to large ones, in the advanced stages of culture. The greatest advantage of using multiple spheroids is the high number of cells obtained at the same time in a single dish. On the other hand, due to their uniform size, single spheroids represent suitable tools for drug screening 3 even if their use is still limited to a narrow range of methods for the development of therapeutic agents to treat cancer.
MCTSs could be successfully used for cancer research in order to understand cancer cell interactions or for drug screening, 9 but at present, 3D culture models are facing many problems, challenges, and limitations. Size and uniformity represent a critical parameter for the evaluation of drug penetration and transport in spheroids. 3 At the same time, the low penetration of the drug is one of the most significant limitations concerning the interaction between drugs and MCTSs. 9 Another important limitation is represented by the poor development of techniques and equipment for spheroid analysis; at this moment, only a few are optimized for 3D cultures. 10 For example, 3D models generated on scaffolds could be difficult to analyze, due to limited possibilities to extract the cells from the spheroids. 11 Other drawbacks are represented by the difficulty to control culture conditions when the 3D models are generated in gels and to develop analysis and data acquisition tools for more efficient screening. 11
Moreover, some of the 3D models are characterized only as aggregates and their transition to spheroids requires the presence of additional additives 12 (e.g., Matrigel) that are based on extracellular matrix proteins and provide a high efficiency in the generation of tight cell–cell interactions and cell–extracellular matrix interactions that are implicated in the regulation of signaling pathways and the induction of cellular responses. 13
In this context, we aimed to generate and compare single and multiple spheroids for breast cancer research and to study the influence of Matrigel on the biology of 3D models. Thus, single and multiple 3D cultures were grown from the MDA-MB-231 cell line in the presence or absence of Matrigel and compared by analyzing similarities and differences between their biological properties, such as morphology, viability, degree of necrosis, and hypoxia.
Materials and Methods
Cell Culture and Generation of MCTSs
Human breast adenocarcinoma cell line MDA-MB-231 (ATCC HTB-26, Manassas, VA) was cultured in Dulbecco’s Modified Eagle Medium (DMEM; cat. no. 31600-083, Gibco, UK) supplemented with 3.5 g/L glucose, 1.5 g/L NaHCO3, 1% antibiotics-antimycotics solution (cat. no. A5955, Sigma-Aldrich, St. Louis, MO), and 10% fetal bovine serum (cat. no. 10270-106, origin South America, Gibco, by Life Technologies, Carlsbad, CA).
Cells were grown in 75 cm2 culture flasks until 80%–90% confluency at 37 °C in a 5% CO2 air atmosphere. The cells were trypsinized using a 0.25% trypsin/0.53 mM EDTA solution and the culture media was refreshed every 2 days.
Harvested cells were used to generate MCTSs through the liquid-overlay technique, using low-cell-attachment Nunclon Sphera Microplates (Thermo Fisher Scientific, Waltham, MA), which are coated with a hydrophilic polymer. Thereby the Nunclon Sphera surface supports the formation of consistent cancer spheroids in suspension in both round- and flat-bottom culture plates. Nunclon Sphera 96-well round-bottom plates (cat. no. 12-566-430, 174925) were used to generate a single spheroid in a 200 µL volume of culture media, while multiple-spheroid cultures were generated from MDA-MB-231 cells in Nunclon Sphera six-well plates (cat. no. 12-566-435, 174932) in a 3 mL volume of culture media ( Fig. 1 ). Cells number was measured using a Bürker-Türk counting chamber before cell seeding.
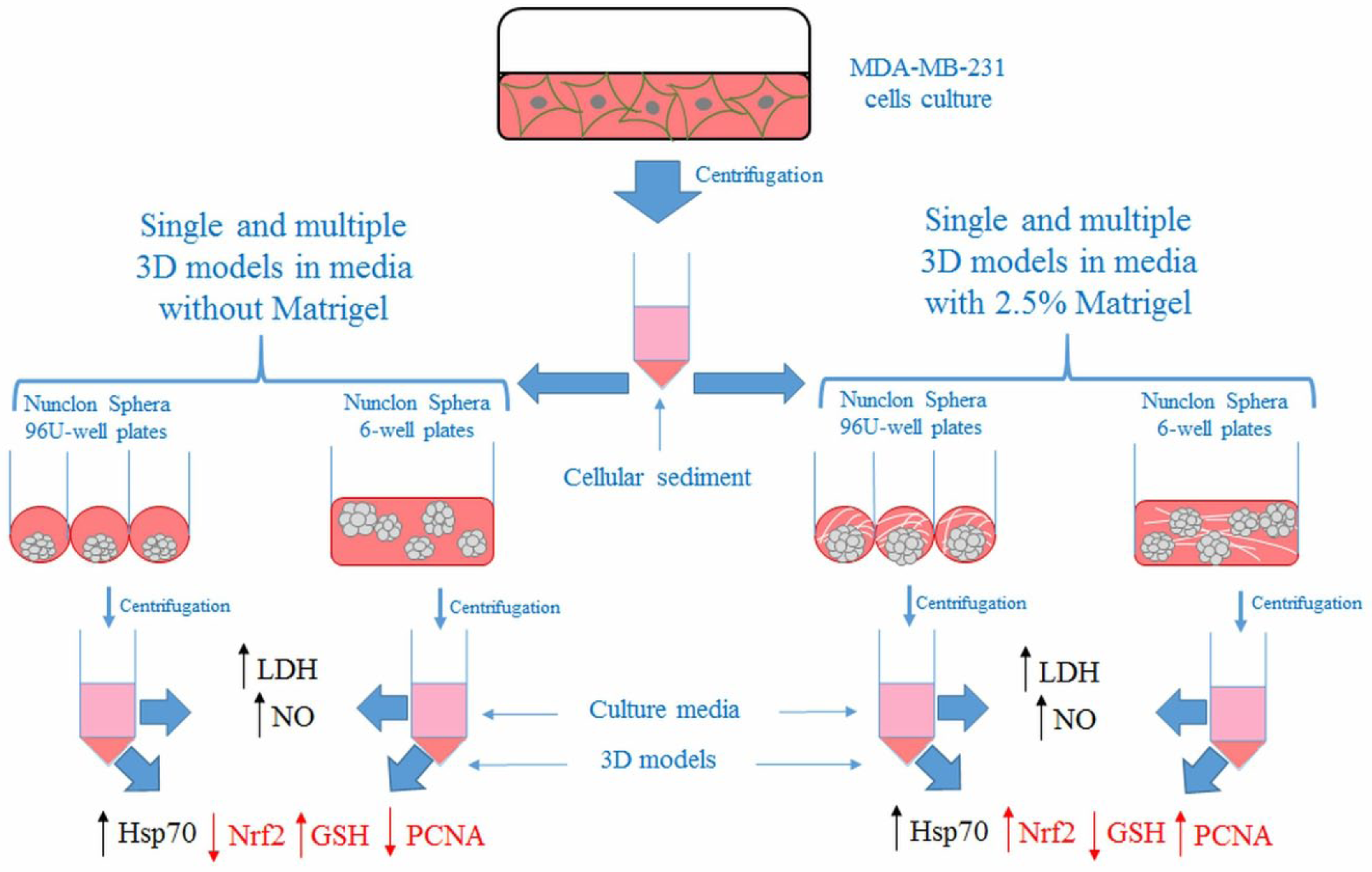
Schematic illustration of the generation and characterization of single and multiple 3D models grown in media without and with 2.5% Matrigel from MDA-MB-231 cells. The cellular sediment resulted after the centrifugation of MDA-MB-231 cells was used to produce single and multiple 3D cellular models in Nunclon Sphera plates in the presence or absence of Matrigel. Collected supernatants and cell pellets were used for further determinations. High levels of LDH and NO in culture supernatants and elevated levels of Hsp70 expression in cell lysate were found in both conditions of single and multiple 3D model cultures (black). Differences were highlighted in the expression of PCNA, Nrf2, and GSH content (red).
Single and multiple 3D cultures were generated in the absence and presence of Matrigel Basement Membrane Matrix (356237, Corning, NY). Matrigel Basement Membrane Matrix was added in the culture media at a percentage of 2.5%, according to Ivascu and Kubbies. 14 In order to generate a single spheroid, cells were seeded at various densities of 2500, 5000, and 8000 cells/well, while multiple spheroids were generated using 5 × 105, 12.5 × 105, and 25 × 105 cells/well.
The culture media was refreshed every 2 days, starting with the second day of culture, by replacing half of the culture media with a fresh one. When multiple spheroids were generated in the presence of Matrigel, half of the culture media was replaced with a fresh one containing 2.5% Matrigel.
All the biological experiments were performed using spheroids generated with and without Matrigel.
Morphology of MCTSs
All generated 3D models were monitored until the sixth day of culture using an Olympus IX73 (Olympus, Tokyo, Japan) microscope equipped with a Hamamatsu ORCA-03G camera (A3472-06, Hamamatsu, Japan). The images were acquired using the CellSens Dimension software (v1.11, Olympus).
The variation in time of diameter and circularity was also analyzed. The diameter of 3D models was measured from the second day until the sixth day of culture using ImageJ software (v1.51k, NIH, Bethesda, MD) for 3D models generated from 5000 cells and 12.5 × 105 cells/well. For this purpose, the diameter of each 3D model was measured using the “Straight line” function from the ImageJ software, with the value of the “length” being associated with that of the 3D model diameter. For the same time intervals, circularity was measured using the “Freehand selection” tool from the ImageJ software and circularity parameter (a value of 1 was associated with a perfect circle) according to Gong et al. 15
Fifty single 3D models and 10 microscopic fields of multiple 3D models were analyzed for each time interval.
Cellular Viability Assessment
Cellular viability was evaluated using the LIVE/DEAD Viability/cytotoxicity kit (L3224, Invitrogen, Carlsbad, CA) assay according to the manufacturer’s instructions. Live cells were labeled with calcein AM, and dead ones were stained with ethidium bromide (EthD-1). For the assessment of viability, 5000 cells/well and 12.5 × 105 cells/well were cultured in medium with and without Matrigel in a 96-well plate and a 6-well plate, respectively. Each day, between the third and sixth days of culture, 3D model viability was measured in every three wells. Thus, the medium was removed and 2 µM calcein AM and 4 µM EthD-1 serum-free media solutions were added directly to cellular models that were incubated for 20 min at 37 °C. Finally, the labeled 3D models were analyzed under an Olympus IX73 fluorescence microscope (Olympus) and the images were acquired using fluorescein isothiocyanate (FITC) and tri-rhodamine-isothiocyanate (TRITC) filters and CellSens Dimension software. The fluorescence intensity of live and dead cells was quantified using ImageJ software (v1.51k, NIH). For each time interval and experimental condition, three images were analyzed. From the ImageJ menu (Analyze—Set measurements), the parameters “area,” “integrated density,” and “mean gray value” were selected. Then, the spheroid was selected using the “Freehand selection” tool and a region close to the spheroid that had no fluorescence (background) was also selected. The corrected total cell fluorescence (CTCF) for live cells (green fluorescence) and dead cells (red fluorescence) was calculated, using the formula CTCF = Integrated density of selected spheroid – (Area of selected spheroid × Mean fluorescence of background readings). According to the ImageJ guide, the parameter “integrated density” represents the sum of the values of the pixels in the selection, while “mean gray selection” is the average gray value within the selection (the sum of the gray values of all the pixels in the selection divided by the number of pixels). Using CTCF values, the results were expressed as a percentage of live cells and dead cells, respectively, relating to the total CTCF value (live and dead cells), which was considered 100%.
Lactate Dehydrogenase Assay
The lactate dehydrogenase (LDH) activity in culture media was assessed using the Cytotoxicity Detection Kit (LDH) (v10, cat. no. 14115700, Roche, Basel, Switzerland) based on the reaction between lactate and NAD+ with the generation of NADH. Then, NADH participated in the generation of formazan, which is detected spectrophotometrically. The level of LDH, considered a marker of necrosis, was determined up to the sixth day of culture. After the generation of 3D cellular models, a 50 µL volume of Matrigel-free medium and medium with 2.5% Matrigel was transferred into an optically clear 96-well flat-bottom microplate and treated with a 50 µL reaction mixture consisting of two reagents: catalyst represented by a diaphorase/NAD+ mixture and a dye solution made from iodotetrazolium chloride and sodium lactate. The absorbance of samples was measured after 15 min of incubation at 490 nm using a Flex Station 3 microplate reader (Molecular Devices, San Jose, CA). The values of LDH activity were first divided by cell number, and the LDH level in the sample media was calculated in relation to one of the controls (seeding moment).
Preparation of Cellular Lysate
To obtain the cellular lysate, at each tested time point, 3D models were collected from culture plates and centrifuged for 5 min at 1500 rpm. The cell pellets were then washed and resuspended in phosphate-buffered saline (PBS) solution. Further, the cellular models were sonicated for 30 s, three times on ice using a UP50H ultrasonicator (Hielscher Ultrasound Technology, Teltow, Germany) at 80% amplitude. The samples were then centrifuged for 10 min, 3000 rpm, 4 °C, and the supernatants were collected and used for further determinations. The protein concentration was measured using the Bradford method and bovine serum albumin (BSA) as standard protein.
Western Blot Analysis
Western blot technique was performed to analyze the expression of some proteins of interest: proliferating cell nuclear antigen (PCNA), nuclear factor erythroid 2-related factor 2 (Nrf2), and 70 heat shock protein (Hsp70) from the 3D culture up to the sixth day. First, protein samples (30 µg) were denatured in the presence of β-mercaptoethanol at 95 °C for 5 min and ran on 10% and 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) gels, respectively, in TRIS-glycine-SDS buffer at 90 V for 2 h. After separation in SDS-PAGE gels, the proteins were transferred to a polyvinylidene fluoride (PVDF) membrane (cat. no. IPVH00010, Merck, Darmstadt, Germany) in a wet transfer system, using a TRIS-glycine-methanol buffer (90 min).
Membrane development was achieved using Western Breeze Chromogenic Anti-Mouse and Anti-Rabbit kits (WB7103 and WB7105, Invitrogen); polyclonal anti-PCNA (sc-56, Santa Cruz, Biotechnology, Dallas, TX), anti-Nrf2 (sc-722, Santa Cruz), and anti-Hsp70 (sc-24, Santa Cruz) specific antibodies; and monoclonal anti-β-actin antibody (A1978, Sigma-Aldrich), following the manufacturers’ instructions. Alkaline phosphatase-conjugated anti-mouse and anti-rabbit secondary antibodies were added for 30 min. In the end, BCIP/NBT substrate was used to detect the protein bands, which were visualized with the ChemiDoc Imaging System (BioRad, Hercules, CA) and quantified using Image Lab software (v5.1, Bio-Rad, Hercules, CA). The β-actin was used as reference protein to normalize the results.
Histology
Single 3D models and multiple 3D models cultured in media with and without Matrigel were histologically analyzed on the third and sixth days of culture. The 10 µm thick sections were cut at MNT cryostat (SLEE, Mainz, Germany) and mounted on Superfrost Plus slides. Cryosections were fixed in 4% paraformaldehyde for 5 min and stained with hematoxylin and eosin, according to the protocol of the Rapid Frozen Sections staining kits (Bio-Optica, Milano, Italy). The slides were analyzed under light microscope (Olympus BX43 microscope, Olympus, Hamburg, Germany; XC30 software).
Immunohistochemistry
Immunochemical detection of cleaved-PARP (cPARP) was performed on the third and sixth days of culture for single and multiple 3D models generated without and with 2.5% Matrigel. The cryosections were fixed in 4% paraformaldehyde for 5 min and incubated with blocking solution (Novocastra Leica Biosystems, Wetzlar, Germany) for 10 min. Exposure to primary antibody cPARP (Cell Signaling, Danvers, MA) at 1:50 dilution was done for 2 h. Detection was performed using a polymer detection system (Novolink Max Polymer, Novocastra Leica Biosystems) and 3,3′-diaminobenzidine (DAB; Novocastra Leica Biosystems) as chromogenic substrate. Hematoxylin staining was applied before dehydration and mounting. Images were acquired by light microscopy (Olympus BX43).
Quantification of Intracellular Reduced Glutathione
The glutathione (GSH) content in 3D cultures within a 6-day interval was measured using the Glutathione Assay Kit (CS0260, Sigma-Aldrich). Briefly, a volume of cell lysate was deproteinized with an equal volume of 5% sulfosalicylic acid (SSA) solution (S2130, Sigma-Aldrich) and centrifuged (10 min, 10,000 rpm, 4 °C). Then, a 10 µL volume of the supernatant was mixed with 150 µL of 5,5′-dithio-bis(2-nitrobenzoic) acid (DTNB; D8130, Sigma-Aldrich) solution, and GSH oxidation and formation of 5′-thio-2-nitrobenzoic acid (TNB), measurable at 405 nm, occurred. A solution of 200 µM GSH prepared in 5% SSA (G6529, Sigma-Aldrich) was used to prepare a calibration curve. The GSH concentration of samples was determined by extrapolation on the calibration curve and was calculated in relation to the GSH level of control.
Nitric Oxide Production
The amount of nitric oxide (NO) released by cellular models in culture media was determined up to the sixth day of culture using a colorimetric Griess reaction for the oxidative status evaluation. Thus, 3D models were cultured in media with and without Matrigel at a density of 5000 cells/well and 12.5 × 105 cells/well, respectively. An 80 µL volume of sample culture media was mixed with 80 µL of Griess reagent made of equal volumes of 10% N-(1-naphthyl) ethylenediamine dihydrochloride (cat. no. 222488, Sigma-Aldrich) and sulfanilamide (S9251, Sigma-Aldrich) prepared in 85% phosphoric acid (H3PO4). Finally, the absorbance was read at 540 nm and the NO concentration was calculated using a 0–200 µM NaNO2 standard curve. The values of the NO concentration were first divided by cell number and all samples were reported to the control values.
Statistical Analysis
All experimental data were represented as an average of three biological replicates. Statistical significance was determined by Student’s t test using Microsoft Excel software. A P value <0.05 was considered significant. All sample values were reported to the control ones.
Results
Morphology of MCTSs
The morphology of single and multiple 3D models was evaluated within six days of culture and the optical microscopy images of spheroids revealed different morphological characteristics between those cultured in medium with and without Matrigel.
Figure 2 shows single 3D models obtained from various cellular densities (2500, 5000, and 8000 cells/well), most of them characterized by a spherical morphology (about 90%). Starting with the first day of culture, higher compaction of cells was observed in the spheroids cultured in the presence of 2.5% Matrigel ( Fig. 2B ) compared with those generated without Matrigel ( Fig. 2A ). In the absence of Matrigel, the generated 3D models were represented by compact aggregates and not spheroids; their borders became irregular in time and they decreased in size. Those generated in media with Matrigel were represented by spheroids with a uniform morphology, a well-defined border until the last day of the experiment, and a size increase in a time- and cellular density-dependent manner. Moreover, a dependency between the cellular density and the 3D culture dimensions for both conditions was observed. Analyzing Figure 2C , it can be seen that after the second day, the 3D models generated from 2500 cells in culture medium had the same diameter in both conditions. In the case of those generated from 5000 and 8000 cells, this parameter also presented comparable values. But starting with the third day, a downward trend of diameters of cellular aggregates generated in media without Matrigel and an increased tendency of the diameters of those grown in the presence of Matrigel were noticed. Also, it was observed that the diameter of 3D models generated from 5000 cells in medium without Matrigel reached 350 μm in size on the sixth day of culture, while in the presence of Matrigel they became about 700 μm in size.
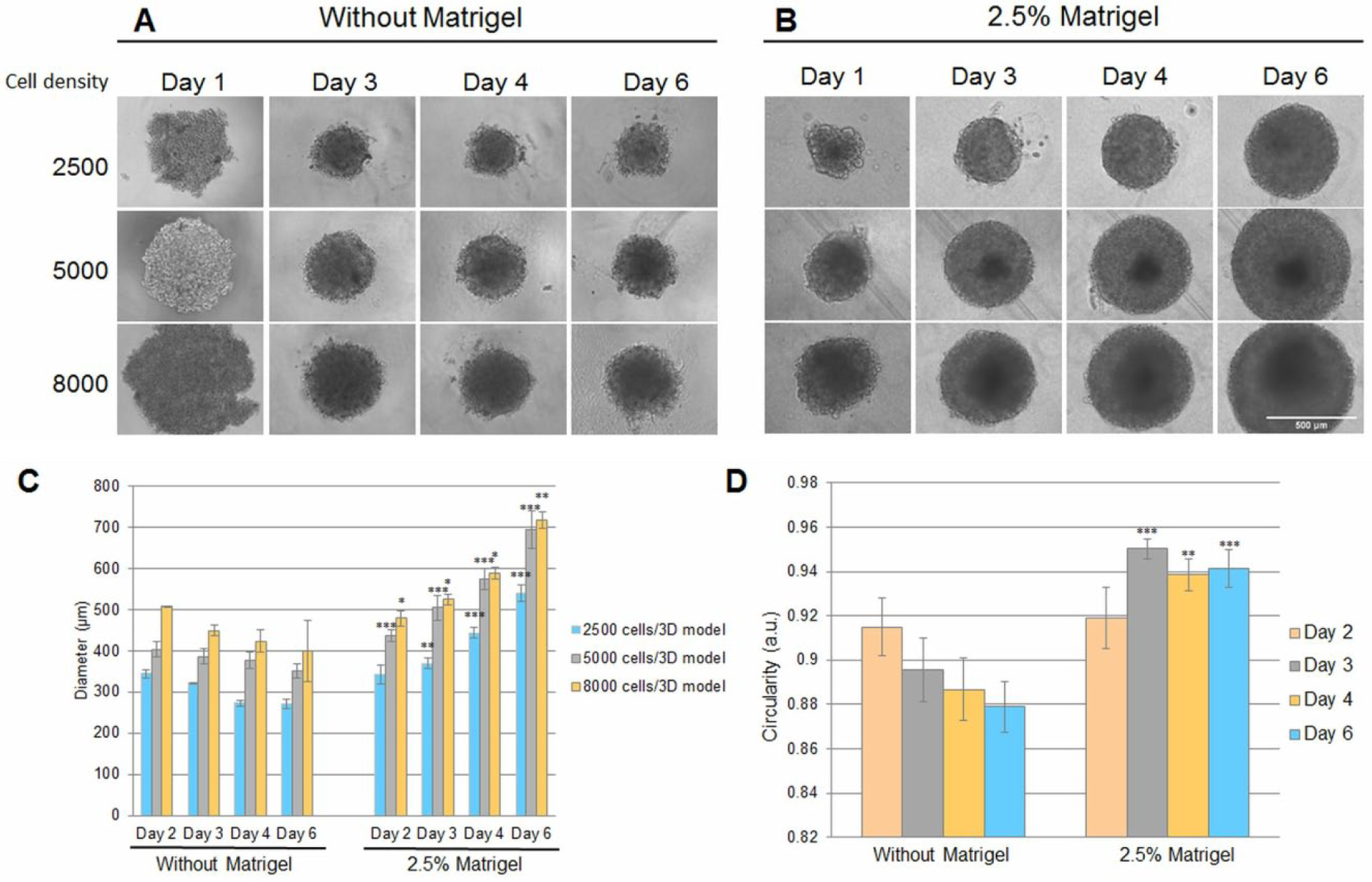
Morphology of single MDA-MB-231 breast cancer 3D models after 1, 3, 4, and 6 days of culture seeded at various cellular densities (2500, 5000, and 8000 cells/well). The figure presents bright-field images of 3D models cultured in (
The spherical morphology of generated 3D models was estimated by analyzing their circularity, with a value of 1 being assigned to a perfect circle and its decrease indicating an elongated polygon. The circularity of single spheroids varied between those generated in the presence of Matrigel and the ones obtained in its absence ( Fig. 2D ). After 2 days, the circularity was 0.915 ± 0.006 and 0.919 ± 0.01 for 3D cultures grown in the absence and presence, respectively, of Matrigel. But after the third day, its variation tendency was opposite depending on medium composition. The circularity of 3D models decreased in the absence of Matrigel up to 0.895 ± 0.01 and increased to 0.950 ± 0.004 in its presence.
When MDA-MB-231 cells were grown in six-well plates in media without 2.5% Matrigel, multiple tight cellular aggregates with different shapes were formed only at densities below 12.5 × 105 cells/well. Above this cellular density, no aggregation of cells was observed ( Fig. 3A ). In the presence of Matrigel, well-defined and round-shaped spheroids were obtained at densities below 12.5 × 105 cells/well ( Fig. 3B ). A higher density of cells induced the formation of large cell aggregates in the media with Matrigel. However, the formed spheroids had different sizes (between 150 and 300 μm) and have grown over time (to 200–650 μm) either by cellular multiplication or by spheroid fusion.
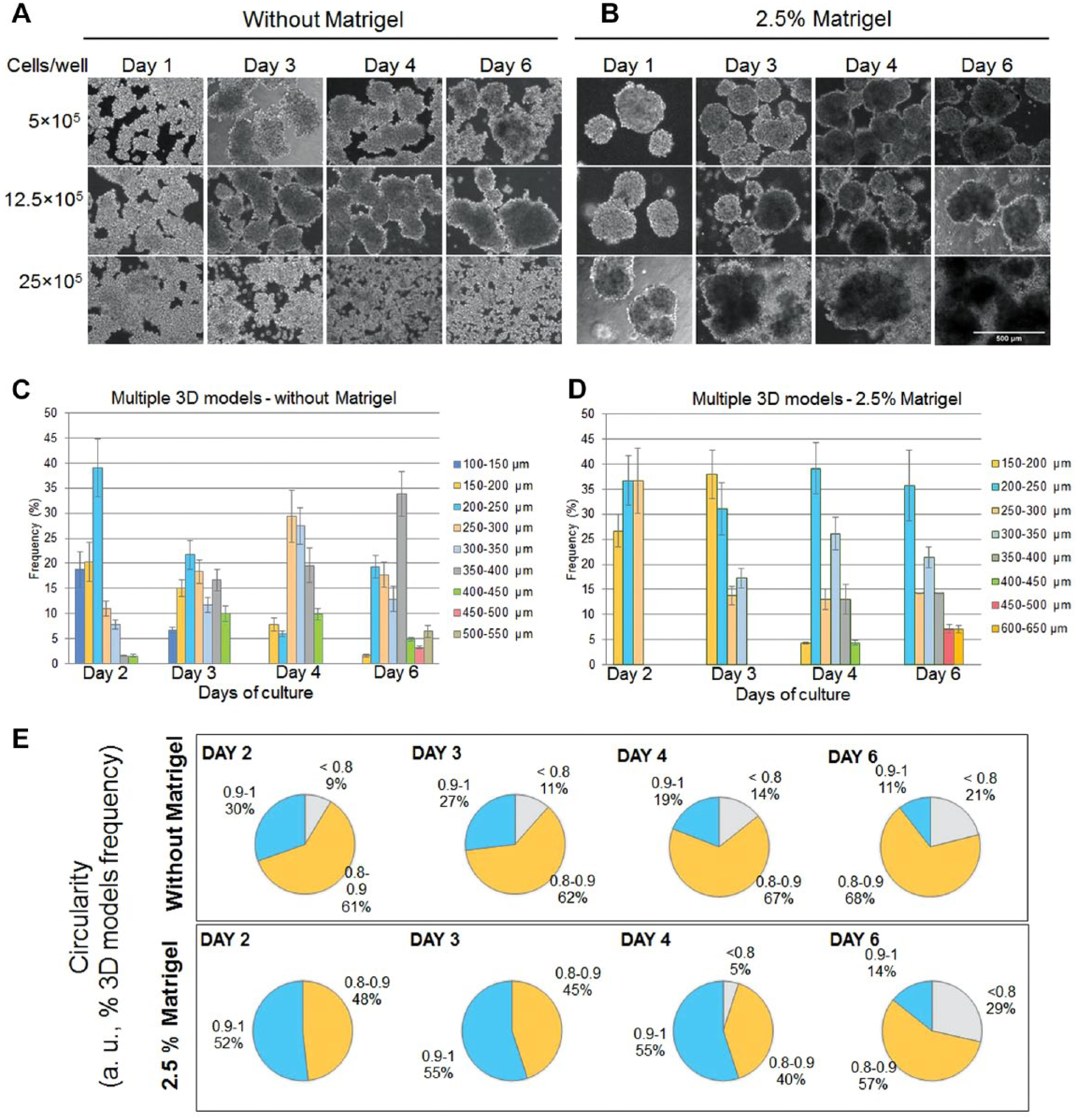
Morphology of multiple MDA-MB-231 breast cancer 3D models after 1, 3, 4, and 6 days of culture seeded at various cellular densities (5 × 105, 12.5 × 105, and 25 × 105 cells/well). The figure presents bright-field images of multiple 3D models cultured in (
The diameter size was measured for multiple 3D models generated from a density of 12.5 × 105 cells/well. The variation in a time-dependent manner of 3D model diameters in the two conditions is presented in
By comparison, the frequent size of multiple spheroids generated in the presence of Matrigel was between 200 and 300 μm starting with the second day of culture and remained so until the sixth day. Larger spheroids with a diameter size over 350 μm were observed starting with the fourth day of culture.
Between the second and fourth days, the presence of Matrigel in the culture media generated a higher circularity of spheroids compared with normal growth media, but on the sixth day, a decrease of circularity between 0.8 and 0.9 in the presence and absence of Matrigel ( Fig. 3E ) was noticed. Also within 6 days, the use of Matrigel favored the formation of better individualized spheroids ( Fig. 3B ).
Viability and Proliferation Capacity of MCTSs
In both single and multiple 3D models, dead cells were observed in their centers starting with the third day, up to the sixth day ( Fig. 4A ). For single compact aggregates generated without Matrigel, no significant change in their size occurred within 6 days of culture, whereas the presence of Matrigel determined an increase in their dimensions of about 30%, which correlated with the level of dead cells from the inner core. The rate of live cells to dead ones was decreased on the sixth day of culture compared with the third one in both conditions ( Fig. 4B ), but in the presence of Matrigel this was lower (67.8% ± 4.72) than that in its absence (73.24% ± 2.58).
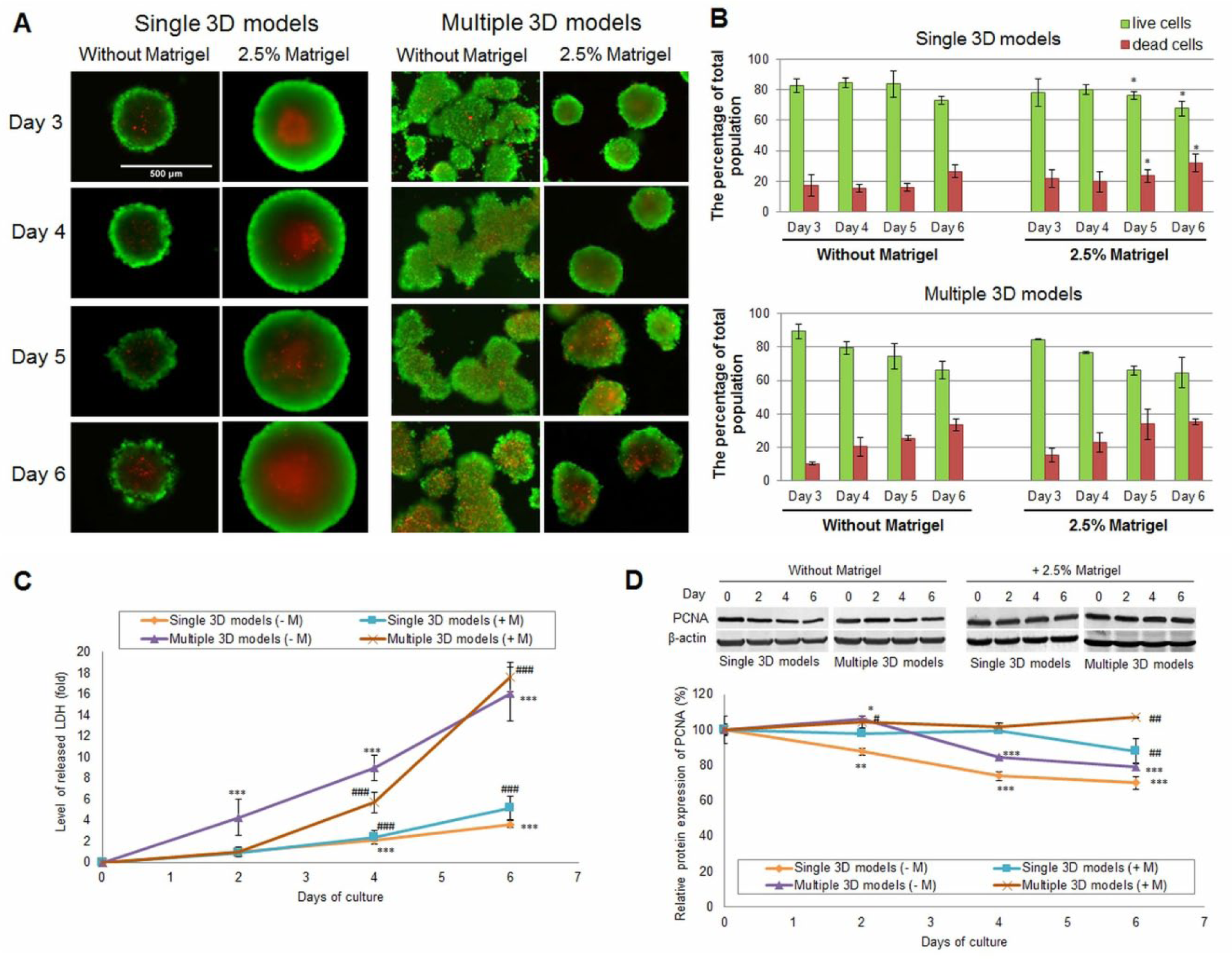
Cell viability and proliferation capacity of MDA-MB-231 breast cancer 3D models generated with and without Matrigel (days 3–6 of culture). (
Generation of multiple 3D models in a single well in the absence of Matrigel resulted in cell aggregates with no defined necrotic center. However, as shown in Figure 4A , many dead cells dispersed in the cellular mass and an increase in their number dependent on time were observed. In the presence of 2.5% Matrigel, multiple spheroids were smaller in size compared with single ones, and therefore the percentage of dead cells was lower after the first 3 days. From the fourth day, the viability of multiple spheroids started to decrease in a time- and size-dependent manner, leading to the formation of a necrotic center. By comparison, the percentage of viability was almost similar for the single and multiple 3D cultures in both the presence and absence of Matrigel ( Fig. 4B ).
The results obtained by fluorescence microscopy were also confirmed by the LDH assay and indicated the development of a necrotic center in the 3D cultures. As shown in Figure 4C , the level of LDH increased in time for both single and multiple cellular models. Also, it was higher in culture media of multiple breast cancer 3D cultures compared with single ones. This result is in correlation with the increase in percentage of dead cells by 3%–7% in multiple 3D models generated with and without Matrigel compared with the percentage of dead cells found in single 3D culture. In addition, on the second day of culture, the LDH level of multiple aggregates generated without Matrigel started to increase significantly.
At the same time, no significant influence of Matrigel on the media LDH level was observed in single-spheroid culture up to the second day. On the other hand, after 4 days of culture adding Matrigel into the multiple 3D culture decreased the LDH release by about 3 ± 0.19-fold in comparison with the Matrigel-free culture. However, on the sixth day of culture the release of LDH slightly increased in the medium of 3D models cultivated in the presence of Matrigel.
To evaluate the proliferation capacity of breast cancer 3D models over time, PCNA protein expression was investigated. This marker showed a clear difference between the 3D cultures generated with Matrigel and those generated without Matrigel. When 3D models were grown without Matrigel, PCNA expression decreased significantly in time ( Fig. 4D ). On the last day of culture, in single and multiple 3D cultures, the level of PCNA expression decreased by 30% ± 3.75 and 20% ± 2.25, respectively, compared with that registered at the seeding moment. In the presence of Matrigel, in single 3D cultures the expression of PCNA remained constant up to the fourth day of culture when it started to decrease significantly. A different situation was observed in the multiple 3D cultures with Matrigel, when the PCNA expression significantly increased up to the sixth day in comparison with the control ( Fig. 4D ).
Hematoxylin and eosin stain ( Fig. 5A ) revealed that after 3 days of culture, MDA-MB-231 single 3D models generated without Matrigel were compact and became apparently larger with a homogenous inner structure on day 6. In the presence of 2.5% Matrigel, larger MDA-MB-231 cells with pale cytoplasm, characteristic of necrotic cells (arrows), were observed in the core and at the interface to the outer spheroidal edge. On the contrary, in multiple spheroids, necrotic cells were not concentrated in the spheroid core but dispersed in the entire area, and their number increased with time, in correlation with live/dead assay results ( Fig. 4B ).
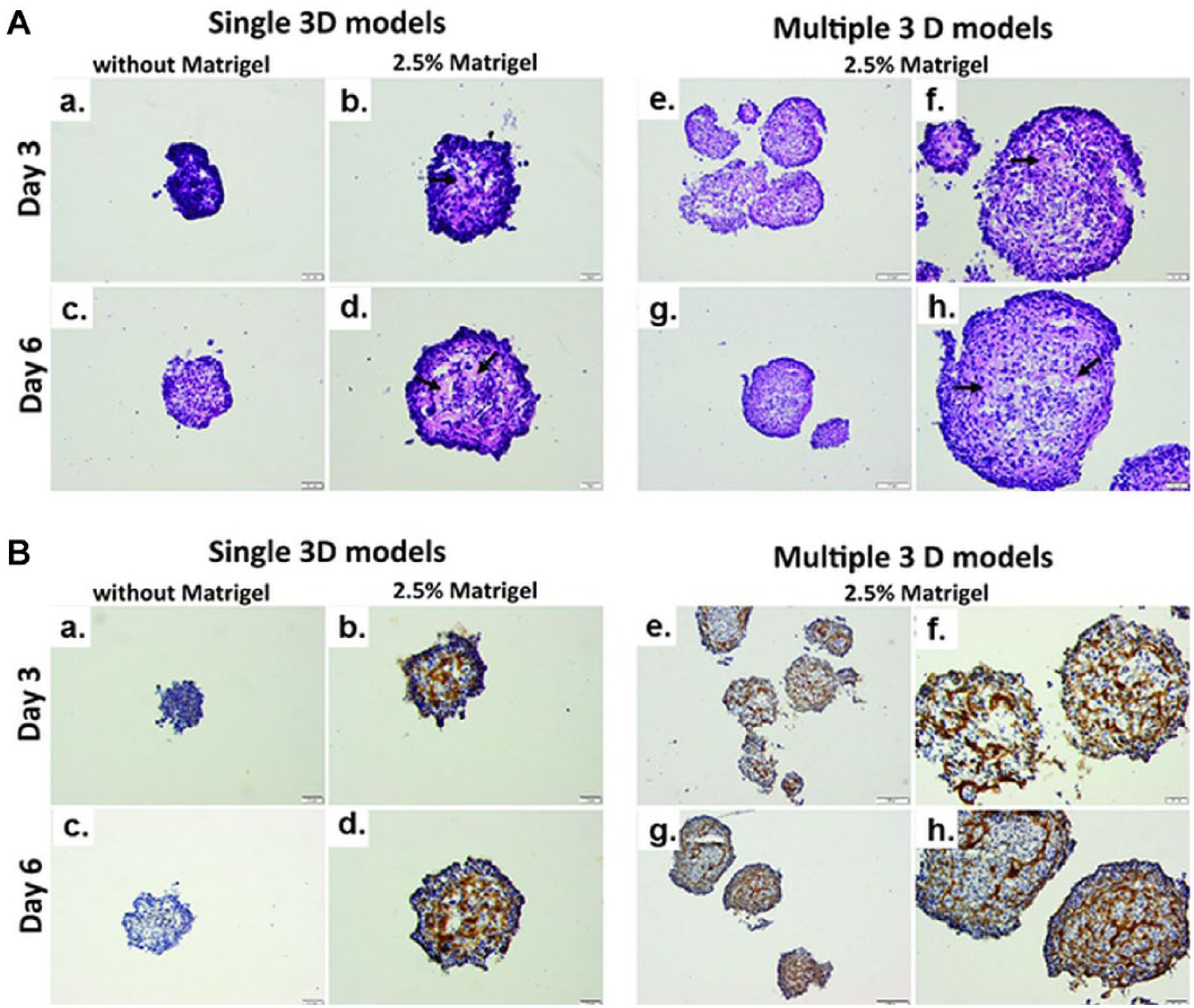
(
At the same time, immunohistochemistry images have shown that cPARP, a marker of apoptosis, was expressed in only a few cells on single 3D models without Matrigel for both analyzed time points. In the spheroid core of single 3D models obtained with 2.5% Matrigel, almost all cells were cPARP positive, but in the organized spheroidal edge they were cPARP negative. On the contrary, cPARP-positive cells were randomly spread in multiple 3D models and their number increased in time ( Fig. 5B ).
Hypoxia-Related Marker Expression
Several markers related to the hypoxia condition, such as the expression of Hsp70 and Nrf2 proteins, the concentration of GSH, and NO production, were analyzed in the 3D cultures for a period of 6 days.
In the presence of Matrigel, the expression of poly-ubiquitinated Nrf2 (100 kDa) increased in a time-dependent manner for both single and multiple spheroids ( Fig. 6B ), while in its absence a decrease was noticed. In this last condition, only in the single 3D culture, on the fourth day, was an increase of the Nrf2 protein level, by 22% ± 6.62, noticed compared with control.
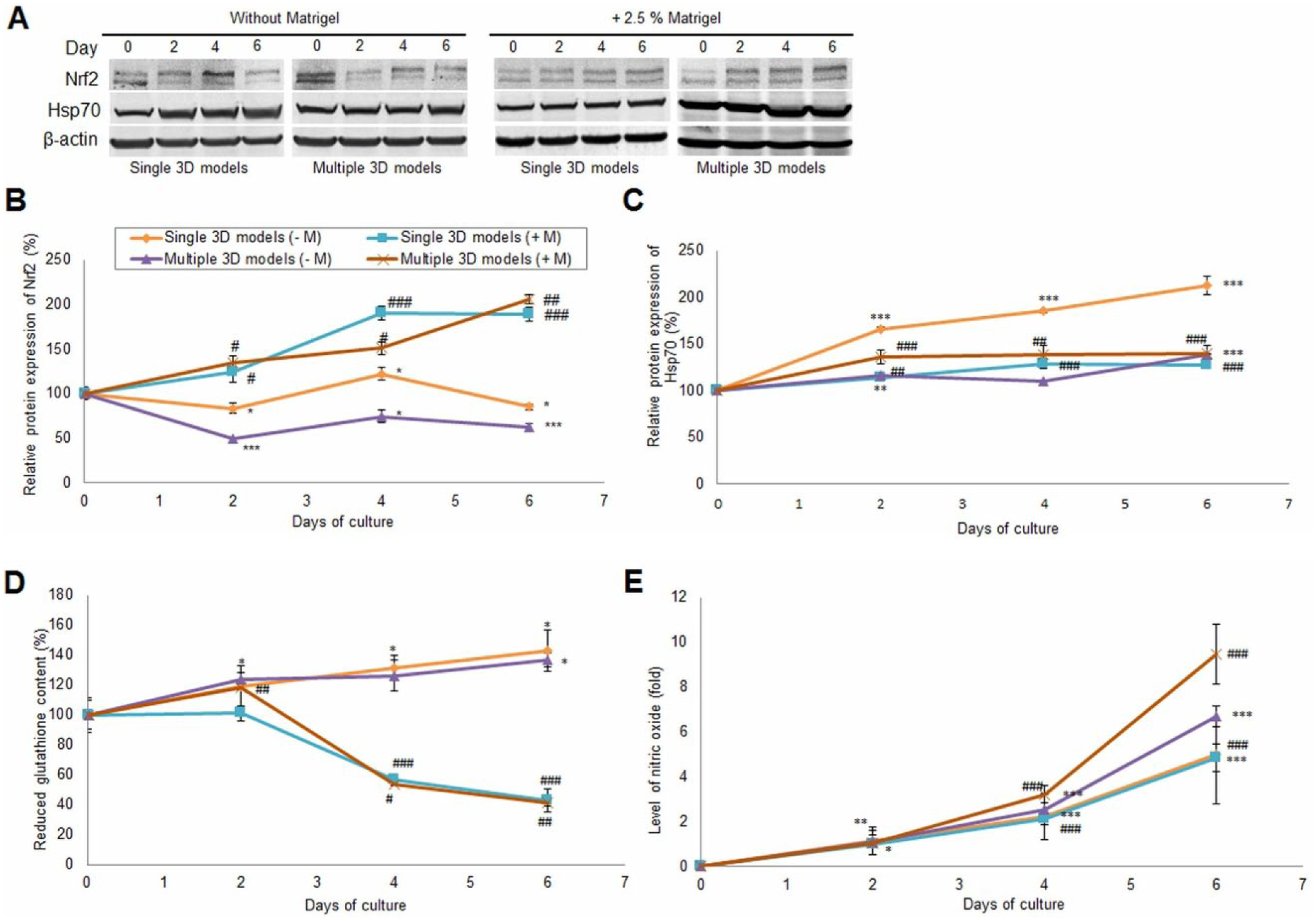
Hypoxia markers. (
In our experiment, a significant upregulation of Hsp70 protein expression for both types of 3D models and conditions was detected ( Fig. 6C ). However, on the sixth day, the highest increase of Hsp70 expression, by 112% ± 9.91 compared with the control, was registered for single compact aggregates cultivated in the absence of Matrigel. At the same time, single 3D models grown in Matrigel presented a lower expression of Hsp70 compared with the previous ones by about 85.22% ± 0.88. In multiple 3D models, the level of Hsp70 protein expression was insignificantly higher in those cultured in the presence of Matrigel (by only 2% ± 7.66 on day 6) compared with those without Matrigel).
The evaluation of intracellular GSH content showed a significant difference between 3D models cultured in the absence of Matrigel and in its presence. A similarity between single and multiple 3D cultures for each condition was also noticed ( Fig. 6D ). The level of GSH in single 3D models cultured in the absence of Matrigel increased in a time-dependent manner by approximately 40% ± 13.73 on the sixth day compared with the control. The addition of Matrigel in culture medium kept the level of GSH close to the initial one for the first 2 days, but after 4 days it started to decrease significantly (by approximately 60% ± 1.78 on day 6 in multiple spheroids) compared with the control.
The production of NO in culture media of 3D cultures generated in medium without and with 2.5% Matrigel increased over time ( Fig. 6E ). The level of NO was higher in the culture medium of multiple 3D models compared with single ones. An increase of NO level by 9.5 ± 0.26-fold was recorded on the sixth day of culture in the medium of multiple 3D models generated with 2.5% Matrigel compared with the control. By comparison, in medium without Matrigel the NO level increased by only 6.7 ± 0.09-fold on the sixth day of culture. In single 3D cultures, a similar variation of NO level was found in both conditions. An increase of NO level by approximately 5 ± 0.43-fold was observed on the last day of the experiment relative to the control in the culture medium of 3D models generated without Matrigel.
Discussion
Due to their remarkable characteristics, especially highlighted by the similarity with the real tissues, MCTSs have potential to be used in many research areas.
According to previous studies, MCTSs from MDA-MB-231, a triple-negative breast cancer cell line, are difficult to obtain. Some researchers characterized MDA-MB-231 3D models only as aggregates, while others mentioned that this cell line cannot generate spheroids without an extracellular matrix (such as Matrigel). 16 This cell line expresses low levels of E-cadherin, which is involved in intercellular junctions, thus limiting the ability of cells to organize themselves spontaneously into compact spheroids. 17
The efficiency of Matrigel in spheroid formation was indicated previously. 14 The rapid formation of spheroids in the presence of Matrigel is stimulated by its composition. A Matrigel matrix is extracted from the Engelbreth-Holm-Swarm (EHS) mouse sarcoma and contains a high percent of laminin, collagen IV, and enactin, proteins found in the extracellular matrix of cells but also proteoglycans and growth factors. Beside its role in spheroid formation and the improvement of their characteristics, Matrigel also has important applications in differentiation, tissue explants, and stem cell culturing, suggesting the importance of its composition and functional complexity. In stem cell culture, the main application of Matrigel is in maintaining the pluripotency and self-renewal capacity of stem cells, and in differentiation it is associated with a more differentiated phenotype of cells, dependent on the cell type. 18
Matrigel combined with differentiation factors can also be used in the generation of more advanced 3D models, like organoids, complex experimental models derived from primary tissues, embryonic stem cells, or induced pluripotent stem cells. 19
Along with Matrigel, other natural (collagen, chitosan, hyaluronic acid) or synthesized (polyethylene glycol) polymers are used as additives that support MCTS formation. 1 Despite their high efficiency in the generation of MCTSs, additional additives, or the main components of them, could influence the cancer cell metabolism and thus the experimental results. For example, hyaluronic acid, an important natural polymer found in the extracellular matrix of malignant tumors, was used as a scaffold for the generation of 3D cancer models, 1 but it can interfere in cell signaling and tumorigenesis. Previous studies have shown that in the presence of hyaluronic acid, important properties of cancer cells, such as metastasis or therapeutic resistance, are affected. 20 Also, the chitosan used for surface coating can induce modifications in the morphology of colon cancer cells, enhancing cell motility, drug resistance, and self-renewal capacity. 21 Moreover, cells cultured on the hyaluronic acid–chitosan scaffold showed a higher expression of stem cell-like markers. 20 Also, other studies revealed differences between natural and synthetic polymers used as matrices for spheroids. Thus, prostate cancer cells (DU145 cells) presented a different proliferation capacity and response to drugs in correlation with the nature of matrices. In the presence of natural matrices (e.g., Matrigel, Culture Basement Membrane Extract [BME]), the proliferation rate was lower than in the presence of synthetic matrices (such as Alvetex). These differences were associated with the variation in protein concentration and matrix stiffness between matrices. In addition, cells cultured in Matrigel and BME responded similarly by docetaxel compared with the cells in the presence of Alvetex that were more resistant. 22 In this regard, more experiments are needed to understand and eliminate any influence of additional components.
In this study, we aimed to generate single and multiple 3D culture models through the liquid-overlay technique and to compare their biological properties in the presence and absence of Matrigel. The biological properties of single and multiple 3D cultures and their similarity with the in vivo tumors were evaluated by the analysis of key markers involved in the viability and development of the necrotic center (LDH), proliferation (PCNA), and hypoxia (NO, Nrf2, Hsp70), as defining characteristics of MCTSs.
In the present paper, we show higher compactness of cells and a better circularity for the 3D models generated in media with 2.5% Matrigel. Improvement of spheroid morphology was also observed when different additives were used. In the presence of MethoCel, a cellulose-based additive, the circularity of MDA-MB-231 spheroids generated by the hanging drop technique was improved. 23 The compactness of cells after using MethoCel was also observed for other cell lines: MCF7, A549, and DU145. 23 However, the PC3 spheroids presented no improvement in circularity, suggesting that the efficiency of additives can be cell type dependent. 23 Moreover, the circularity of spheroids was also studied in relation to the technique used for their generation. In 2016, Raghavan et al. 24 reported no significant differences in the circularity of MCF7 and OVCAR8 spheroids generated by different techniques. The influence of additives was also observed in spheroid uniformity, especially when single spheroids were generated compared with those grown without Matrigel. A significant improvement of uniformity correlated with the higher compaction of cells was also detected for multiple 3D models generated with Matrigel.
Depending on the additive type, the spheroid diameter could vary. MDA-MB-231 spheroids generated with 2% Geltrex presented a larger diameter 18 than those generated in the presence of 2.5% Matrigel 14 in the same conditions. Our results were consistent with those of Ivascu and Kubbies. 14 In the presence of 2.5% Matrigel, we obtained a diameter of ~420 µm for a spheroid generated from 5000 cells after 24 h. At the same time, Dubois et al. (2017) 17 demonstrated that the percentage of additive (Geltrex) and the moment of its addition in culture media represent other important parameters in obtaining MDA-MB-231 compact spheroids or unique loose aggregates.
Furthermore, we observed a correlation between the diameter size of a single 3D culture generated with and without Matrigel and the expression of the PCNA protein. Some protein components of the extracellular matrix were correlated with expression of PCNA, a marker of the S phase. 25 The extracellular matrix has an important role in cell proliferation, probably due to its influence on the transition from the G1 phase to the S phase of the cell cycle. Thus, the decrease of diameter of 3D models obtained without Matrigel could be due to the time-dependent downregulation of PCNA expression and a higher rate of necrosis in the core of the 3D culture. This can also be associated with a larger area of quiescent cells compared with the proliferative one. The influence of Matrigel in spheroid size can be based on the high protein and growth factor content of Matrigel presented in culture medium, which can sustain a higher proliferation rate, similar to extracellular matrix presented in vivo. Our results revealed similar levels of PCNA expression in single- and multiple-spheroid culture, suggesting the similarity of these two models in both conditions, despite the differences in size, shape, and degree of cell agglomeration.
The importance of Matrigel in the spheroid generation was also illustrated when multiple spheroids were obtained in a single well of a Nunclon Sphera six-well plate. Besides Matrigel, the special-treated surface of the plates likely also contributed to the spheroid formation. When multiple spheroids were seeded without Matrigel, the 3D models formed were required to stay in the incubator without any movement for the first 3 days. This period was associated with the appearance of the cellular interactions necessary to allow cell aggregation. Instead, the spheroids cultured in media with Matrigel were characterized by a compact morphology even from the first day, indicating the presence of strong cell–cell and cell–extracellular matrix interactions. The formation of multiple 3D structures in a single well using the overlay technique is very difficult and rarely described in literature. Usually, the multiple-spheroid cultures are obtained when cells are either grown on matrix (e.g., agarose, collagen, hyaluronic acid, Matrigel) or embedded within matrix, but the prior preparation steps and subsequent extraction of spheroids are laborious and time-consuming. 26
An agitation-based approach using a bioreactor represents another alternative method to produce a large number of spheroids, but it is not always accessible. Scaffold-free cell spheroids in suspension can be obtained in ultra-low-attachment plates, but the technique still requires optimization regarding the cell density, number, and size of the spheroids. Nigjeh et al. (2018) 27 showed the formation of primary multiple-spheroid cultures derived from the MDA-MB-231 cell line in serum-free media after 5–7 days using ultra-low-attachment plates.
Using the six-well Nunclon Sphera microplates, we managed to obtain multiple compact MDA-MB-231 3D structures in 2–3 days in media with 10% fetal bovine serum in a single well using the liquid-overlay technique. We also observed that formation of these is dependent on cell density; at more than 12.5 × 105 cells/well, they were not produced. Moreover, in this situation an increase in time of multiple-spheroid diameters was highlighted. Besides the proliferation of spheroids, we associated the high size of multiple 3D models with spheroid aggregation. Thus, the evolution of spheroid diameter in time is changing dependent on cellular proliferation and aggregation, for both single and multiple spheroids.
Necrosis is a representative phenomenon for in vivo tumors. In MCTSs of large dimensions (>500 µm), the necrotic core was developed with the increase of the spheroids’ diameter. 5 In our study, the development of the necrotic center for both single and multiple 3D models generated with and without Matrigel was detected and correlated with the release of LDH in the culture media, which is considered a marker of necrosis. 28 In 3D cultures, gradients in oxygen and metabolites are developed, which generate necrosis as well as apoptosis. 29 As shown by immunohistochemistry analysis, areas with high expression of cPARP (an apoptosis marker) were highlighted in single and multiple 3D cultures with Matrigel and single 3D cultures without Matrigel.
In the 3D environment, the expression of hypoxia-inducible factors (HIFs) is stable, which makes MCTSs suitable models for studying hypoxia in vitro. 29 In vivo, hypoxia plays multiple roles in the progression of cancer by increasing angiogenesis and chemoresistance. Also, it was proven that the invasive and migratory behavior of cells is enhanced under hypoxia, facilitating their survival. 30
It is well established that, under hypoxia, tumor cells produce high reactive oxygen species (ROS) levels as a consequence of the reduction of oxidative phosphorylation. In order to counteract these effects, cells upregulate metabolic pathways that generate NADPH and restore GSH levels. 29
An important role under hypoxia conditions is also played by NO. NO is a major signaling and effector molecule that mediates the body’s response under hypoxia. In breast cancer, the level of NO increased with the reduction of respiration rate, upregulating some hypoxia markers, including hypoxia-inducible factor-1α (HIF-1α). 31 Similarly, we found a time-dependent increase of NO production in all generated 3D models, suggesting not only the induction of hypoxia but also an increase of the oxidative status.
Nrf2 is a protein involved in the counteraction of oxidative modifications during hypoxia. In normal conditions, the Nrf2 protein is coupled with a homodimer protein, Kelch-like ECH associated protein (Keap1). This association enhances ubiquitination and proteasomal degradation of Nrf2 and prevents its activation. When Nrf2 is activated, it is translocated in the nucleus due to the inhibition of ubiquitination and saturation of Keap1 with Nrf2 occurs. 32
In our study, the expression of poly-ubiquitinated Nrf2 protein (~100 kDa) was quantified and we found important differences between the 3D models generated in the presence of Matrigel and those generated in its absence. The addition of Matrigel induced an increase of the poly-ubiquitinated Nrf2 protein expression, suggesting its accumulation in the cytoplasm. In contrast, the significant downregulation of Nrf2 in 3D cultures generated without Matrigel could indicate the translocation of Nrf2 in the nucleus and its activation. The increase of Nrf2 expression in time could also suggest that single and multiple spheroids show similar characteristics in the same culture conditions. According to recent studies, the activation of Nrf2 is a key step in the development of the hypoxia process. Nrf2 activation supports cell survival during hypoxia, but in several cancer types increased expression of Nrf2 was also correlated with a decreased survival and increased metastasis. 33 Syu et al. (2016) 34 found that Nrf2 protein is activated in breast cancer cells under hypoxic conditions. The Nrf2 activation could be induced by the production of intracellular ROS and was associated with chemoresistance. A significant activation of Nrf2 was also observed in cardiomyoblasts after the cells’ exposure to hypoxia or to reperfusion after the hypoxia condition. The upregulation of Nrf2 expression observed in spheroids generated in medium with Matrigel was probably a result of antioxidant defense activation.
The Nrf2 protein contributes to the GSH synthesis and maintenance by increasing the cysteine supply and regulating transcription of several antioxidant enzymes that protect against oxidative damage, including GSH peroxidase and several glutathione S-transferases. Under the hypoxic condition, activation of Nrf2 stimulates the induction of GSH levels. GSH is the main nonenzymatic cellular antioxidant. It is already known that many types of tumors are characterized by an elevation of GSH level. 35
By analyzing the GSH levels, we found a significant difference regarding the concentration between the 3D models generated without Matrigel and those cultured in medium with it.
As mentioned before, hypoxia is associated with changes in cellular metabolism, including high levels of intracellular GSH and increased activities of several antioxidant enzymes, thus explaining the high levels of GSH in the cellular aggregates generated without Matrigel. Instead, spheroids generated in the presence of Matrigel were characterized by a decrease of GSH content in a time-dependent manner. Similarly, Mansfield et al. (2004) 36 reported a decrease in a time-dependent manner of total cellular GSH in hypoxic cells. The mechanism was associated with the increase of mitochondrial ROS and inhibition of GSH synthesis. An important role in the inhibition of GSH synthesis was considered the restriction of cystine uptake. Similar to ROS activity, NO partially leads to an adaptive increase in GSH through a mechanism that requires the induction of cystine transport. So, it was shown that acute exposure to NO leads to GSH depletion, while a chronic exposure leads to elevation of cellular GSH. 37 As shown in our study, the high levels of NO were registered in all 3D cultures in both the presence and absence of Matrigel. Interestingly, the depletion of GSH content was found only in those with Matrigel, both single and multiple 3D cultures. These results suggest that the addition of Matrigel in culture media of spheroids could influence the cell metabolism by interfering with GSH synthesis, independent of the 3D culture system.
High levels of 70 kDa heat shock proteins (Hsp70) were also correlated with the hypoxia condition. The Hsp70 proteins are central components of folding process of proteins and are implicated in the growth and survival of cancer cells. It was shown that the induction of Hsp70 during hypoxia required the upregulation of HIF-1, which is formed from two subunits: HIF-1α and HIF-1β. The expression of the HIF-1α subunit was demonstrated to be regulated by the concentration of oxygen. 38 Thus, in normoxia HIF-1α is degraded by proteasome, while in hypoxic conditions HIF-1α accumulates in the cytoplasm and then is translocated into the nucleus, where it induces the transcription of target genes involved in the adaption of cells to hypoxia. Moreover, recent studies revealed that another heat shock protein associated with hypoxia, Hsp90, is secreted by MDA-MB-231 cells in order to survive in hypoxic conditions. 39 In our culture model, a high expression of Hsp70 in both single and multiple 3D cultures generated in Matrigel-free and Matrigel media was noticed, suggesting the presence of a hypoxic environment. The high expression of Hsp70 in parallel with high levels of NO, increased GSH content, and activation of Nrf2 protein indicates a more pronounced hypoxic environment in 3D cultures generated in the absence of Matrigel. This might rely on the lack of extracellular matrix proteins found in Matrigel that could sustain high viability and proliferation capacity of spheroids.
Through this study, we accomplished the generation of single and especially of multiple 3D models derived from MDA-MB-231 cells in Nunclon Sphera microplates from cell suspension using the liquid-overlay techniques ( Fig. 1 ). To validate the 3D models, the morphology, viability, proliferation, degree of necrosis, and expression of related-hypoxia markers in spheroids were evaluated. The four experimental conditions (single and multiple 3D cultures, with the addition of Matrigel or not) were analyzed and compared. The technique for multiple 3D structure formation and their similarities with single-spheroid cultures were highlighted in this paper and presented in detail. Also, some aspects regarding the contribution of Matrigel in spheroid formation were described and clarified.
Considering the results and the characteristics of the in vivo tumors and MCTSs, the spheroids generated in the presence of Matrigel represent the most adequate 3D experimental models to study the biology of breast cancer. Also, the high similarity between single and multiple 3D models can represent a step forward for the use of multiple spheroids in research studies.
In conclusion, in this study we showed the formation of breast tumor 3D models from the MDA-MB-231 cell line and provided a biological characterization by analyzing the key factors involved in their formation and development in the presence and absence of reconstituted basement membrane (Matrigel). Moreover, we accomplished the generation of multiple spheroids and their comparison with single-spheroid culture, which provided valuable information for the use of MCTSs in breast cancer research. Thus, the novelty of this study consists mainly of the successful generation of reproducible multiple-spheroid cultures. Moreover, their comparison with single 3D spheroids and the description of the main processes that are developing at the molecular level of the spheroids bring new information about the characterization of in vitro 3D models.
The results of our research led to the following main conclusions: (1) All generated 3D models in the presence of Matrigel showed the characteristics of MCTSs with a necrotic center surrounded by a layer of proliferative cells. (2) The addition of Matrigel in culture media decreased the time period needed for spheroid formation to 2–3 days and influenced the morphology of MCTSs that presented a higher compaction of cells, a good circularity, and proliferation capacity. (3) The hypoxia was restrained in spheroids generated in the presence of Matrigel as a result of its biological activity. (4) The single- and multiple-spheroid cultures expressed a high similarity, which was reflected by the PCNA levels, the GSH content, and the Nrf2 protein expression.
This study represents a step forward in the characterization of 3D models and understanding their functionality for breast cancer research as it presents the description of the key processes that characterize a solid tumor, with a starting point already being reached after the generation of multiple spheroids. The development of techniques for multiple breast cancer spheroids and their optimization could represent advantageous tools for future research studies. Moreover, MDA-MB-231 single and multiple spheroids can represent important requisites in future studies regarding drug evaluation or therapeutic strategies for the treatment of triple-negative breast cancer. At the same time, the optimization of the generation of spheroid cultures by evaluating various conditions, such as the addition of Matrigel to the medium, could ultimately facilitate proper drug candidate selection for drug discovery as well as for applications in translational research. As a future perspective, it might be interesting to further expand the findings to a different breast cancer cell line or other cancer cell types, mainly for the generation of multiple spheroids, as a validation of the methods used to obtain 3D cultures.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financed by a grant of the Romanian National Authority for Scientific Research and Innovation, UEFISCDI, project number PN-III-P2-2.1-PED-2016-0904, within the PNCDI III program.
