Abstract
A series of 12 new thiazolidine-2,4-dione derivatives were obtained by microwave-assisted synthesis. All compounds were physicochemically characterized by quantitative elemental
Introduction
Heterocyclic compounds containing nitrogen, oxygen, and sulfur atoms are known for exhibiting a wide spectrum of pharmacological activities.1–5 Since the anticonvulsant, antimicrobial, and antihyperglycemic potential of several thiazolidine-2,4-dione-bearing compounds was reported, this moiety has been intensively studied in the last decades. 3 Thiazolidine-2,4-dione derivatives have been also identified as potential inhibitors of lanosterol 14α-demethylase. 4
Infections caused by invasive and pathogenic fungi, especially in high-risk and immunocompromised patients, represent some of the most life-threatening diseases worldwide.5–7 The opportunistic yeasts that belong to the
Lipophilicity is a physicochemical property that affects a drug’s pharmacokinetics and pharmacodynamics; 9 thus, computational prediction of lipophilicity is essential for the improvement of the absorption, distribution, metabolism, excretion, and toxicity (ADMET) properties from the first steps of drug discovery. Suboptimal pharmacokinetic properties and increased toxicity can lead to a drug’s failure in the development phases; therefore, in silico prediction of ADMET properties of new drug candidates could be useful for eliminating the molecules that will probably fail in the early stages of drug development.
Based on these facts and as a continuation of our research on heterocyclic compounds bearing sulfur and nitrogen atoms, we designed and synthesized under microwave irradiation new thiazolidine-2,4-dione derivatives as potential antifungal agents. The compounds’ antifungal properties were assessed in vitro against several fungal strains, and their affinity toward the fungal lanosterol 14α-demethylase and their ADMET properties were determined in silico in order to select potential lead compounds that could be further optimized into potential anti-
Materials and Methods
Chemistry
Reagents and solvents used for synthesis were of analytical-grade purity and purchased from Alfa Aesar (Karlsruhe, Germany) and Merck (Darmstadt, Germany). Microwave-assisted synthesis was performed using a CEM Discover BenchMate reactor, in open-vessel mode, under a condenser and with magnetic stirring. The synthetic route (
Fig. 1
), followed to obtain the series of compounds
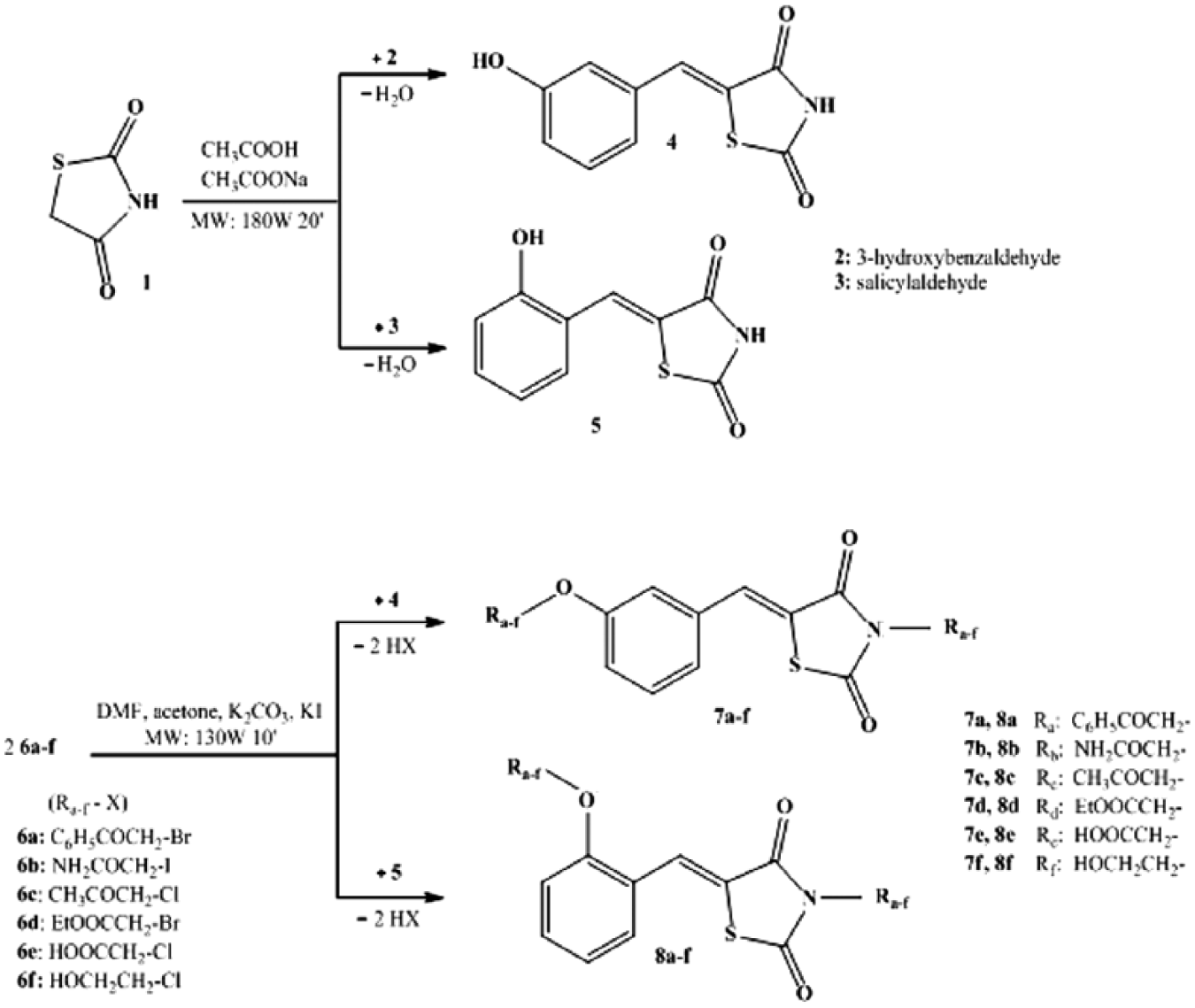
Route followed in order to obtain the thiazolidine-2,4-dione derivatives
The protocols for the synthesis of 5-(hydroxybenzylidene)thiazolidine-2,4-diones (
Antifungal Assay
Stock solutions (1 mg/mL) were prepared by dissolving the test compounds
Determination of Inhibition Zone Diameters
The antifungal screening was based on the agar disk diffusion method against
Determination of Minimum Inhibitory Concentration and Minimum Fungicidal Concentration Values
For the compounds that exhibited the best in vitro anti-
The microorganisms used for this assay were obtained from the University of Agricultural Sciences and Veterinary Medicine, Cluj-Napoca, Romania. The MIC and MFC values were determined against cultures of
Antifungal activity was tested by using the broth microdilution method according to the Clinical and Laboratory Standards Institute (CLSI) guidelines, 12 following a previously reported protocol. 13 The growth control, sterility control, and control of antifungal compounds were used. The MIC was defined as the lowest concentration required for arresting the growth of fungi. The MFC was defined as the lowest concentration of the agent at which no colonies are observed. All MIC and MFC experiments were repeated three times.
Statistical Analysis
The results of the inhibition zone diameter determination assay were expressed as mean ± standard deviation (SD) of three independent experiments. Statistical comparisons between groups were made using one-way analysis of variance (ANOVA). A value of
Molecular Docking Study
The molecular docking study was carried out using AutoDock 4.214 against a fungal lanosterol 14α-demethylase. A validated experimental structure of lanosterol 14α-demethylase isolated from
ADMET and Molecular Property Predictions
The new compounds were subjected to a theoretical in silico ADMET prediction study using the web tool SwissADME (http://www.swissadme.ch/), taking into account the Lipinski rule of 5 (RO5).
We have assessed the topological polar surface area (TPSA) 15 and the logarithm of the compound partition coefficient between n-octanol and water (LogP),16,17 important descriptors in the prediction of bioavailability and the passive transport of an active compound by the blood–brain barrier. 18 Bioavailability is highly multifactorial but is primarily driven by gastrointestinal absorption. 13 Solubility is an important aspect in designing new molecules and was computed. 19 Being a substrate to P-glycoprotein or an inhibitor of various CYP isoenzymes was computed by built-in functions of SwissADME.
Results and Discussion
Chemistry
The structures of the synthesized compounds were confirmed by elemental analysis and spectral data (
1
H-NMR, 13C-NMR, Fourier transform [FT]–IR, and MS). All
Microwave-assisted Knoevenagel condensation in position 5 of 2,4-thiazolidinedione (
In the 1H-NMR spectra of the final compounds
Antifungal Activity
Determination of Inhibition Zone Diameters
The results of the antifungal activity screening of compounds
Zone of Inhibition (mm) of Tested Compounds

The value obtained for each compound represents the mean of three independent measurements ± SD. The values obtained for the most active compounds are marked in bold.
Both intermediate compounds
Minimum Inhibitory Concentration and Minimum Fungicidal Concentration
In the initial in vitro anti-
Minimum Inhibitory Concentration (μg/mL) and Minimum Fungicidal Concentration (μg/mL) of Compounds
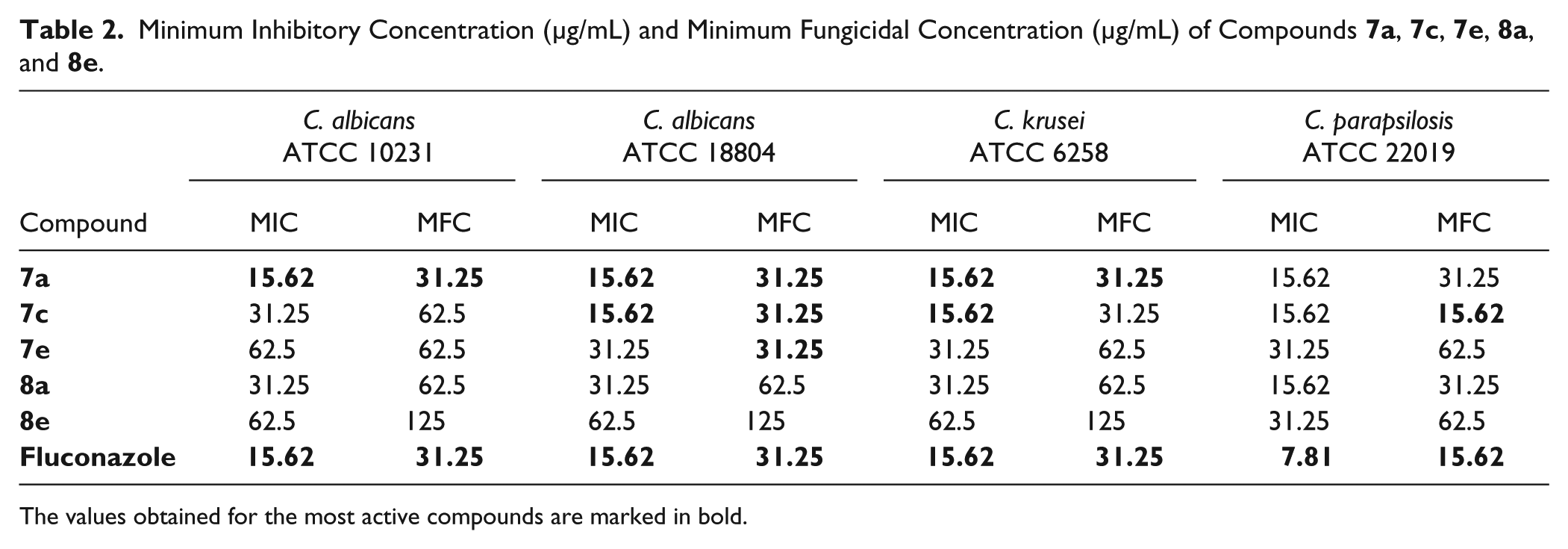
The values obtained for the most active compounds are marked in bold.
The most active compound was
The MFC/MIC ratio for all tested compounds ranged from 1 to 2, suggesting that all the synthesized thiazolidine-2,4-one derivatives could act as fungicidal agents. 20
Molecular Docking Study
The results of the molecular docking study show the best binding affinities of the compounds in the active site of lanosterol 14α-demethylase and their consequent predicted inhibition constants ( Table 3 ).
Predicted Binding Affinity of Tested Compounds to Lanosterol 14α-Demethylase (ΔG) and the Predicted Inhibition Constant (Ki) and Its Cluster Analysis.
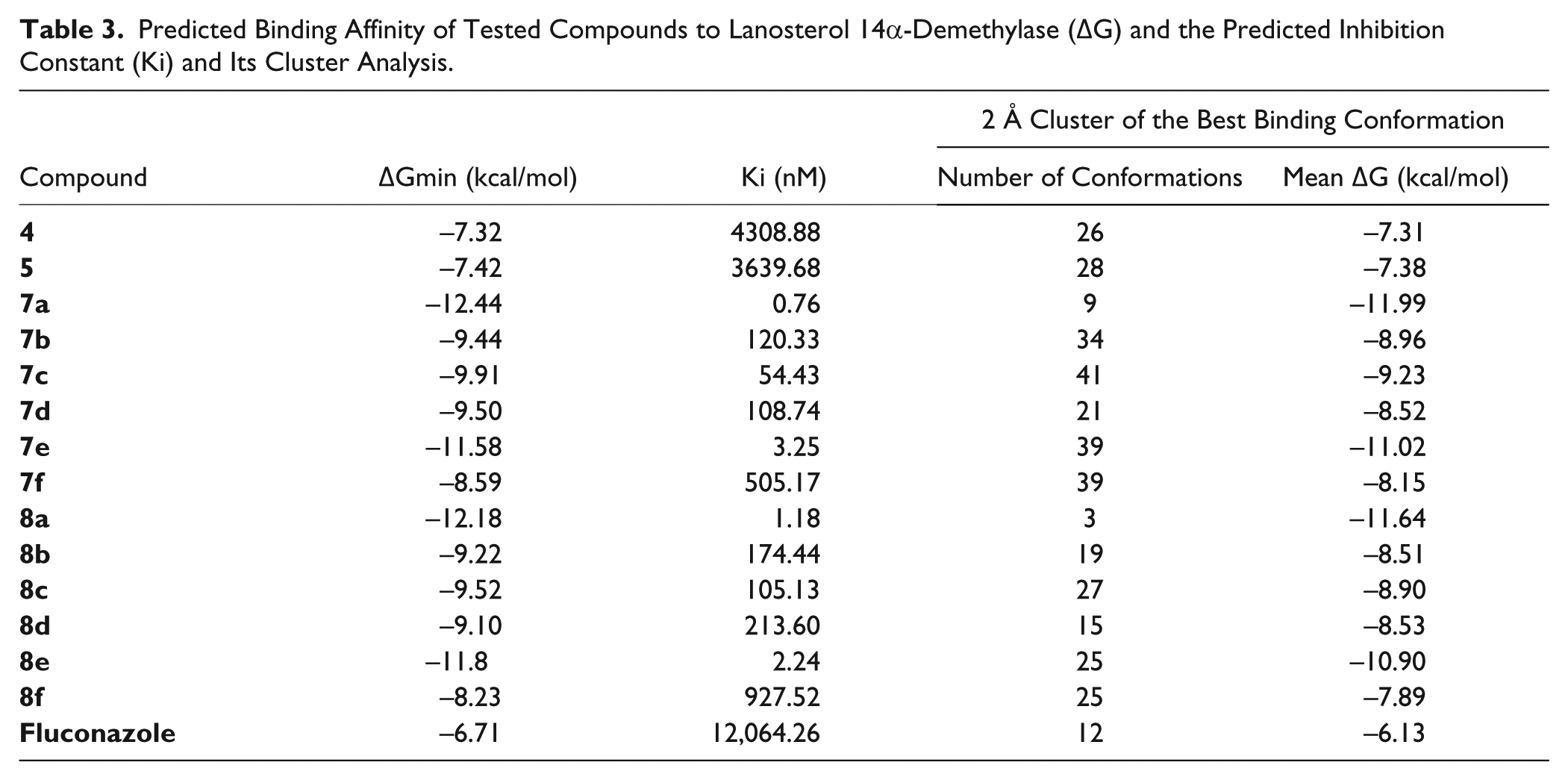
The best binding compound is considered
An interesting aspect is that ligands substituted with aromatic rings have fewer conformations in the 2 Å cluster (their proposed binding modes are less homogeneous than those substituted with smaller alkyl moieties); thus, they have many clusters with few conformations.
Also, compounds substituted in position 2 (the salicyl derivatives) bind less homogeneously and have lower binding energies in comparison with the 3-hydroxy derivatives.
The C=O groups from the substituents are important for the formation of hydrogen bonds with Ser378. The absence of ketone groups from the side chains of compounds
ADMET and Molecular Property Predictions
The results of virtual screening, carried out with SwissADME, for the following descriptors are presented in Table 4 : molecular weight (MW), number of rotatable bonds (RoB; the number of any single nonring bond, linked to nonterminal heavy atom-amide C–N bonds, is not considered because of their high rotational energy barrier 21 ), hydrogen bond acceptors (HBAs; the sum of all oxygen and nitrogen atoms, according to the RO5 definition 22 ), hydrogen bond donors (HBDs; the sum of all –OH and –NH, according to the RO5 definition 22 ), TPSA, and the LogP characterizing lipophilicity. The penetration of the blood–brain barrier was evaluated only based on the values predicted for MW, LogP, HBA, HBD, and TPSA, as specified in the literature. 23
Results of Virtual Screening Carried Out for ADMET Descriptors.
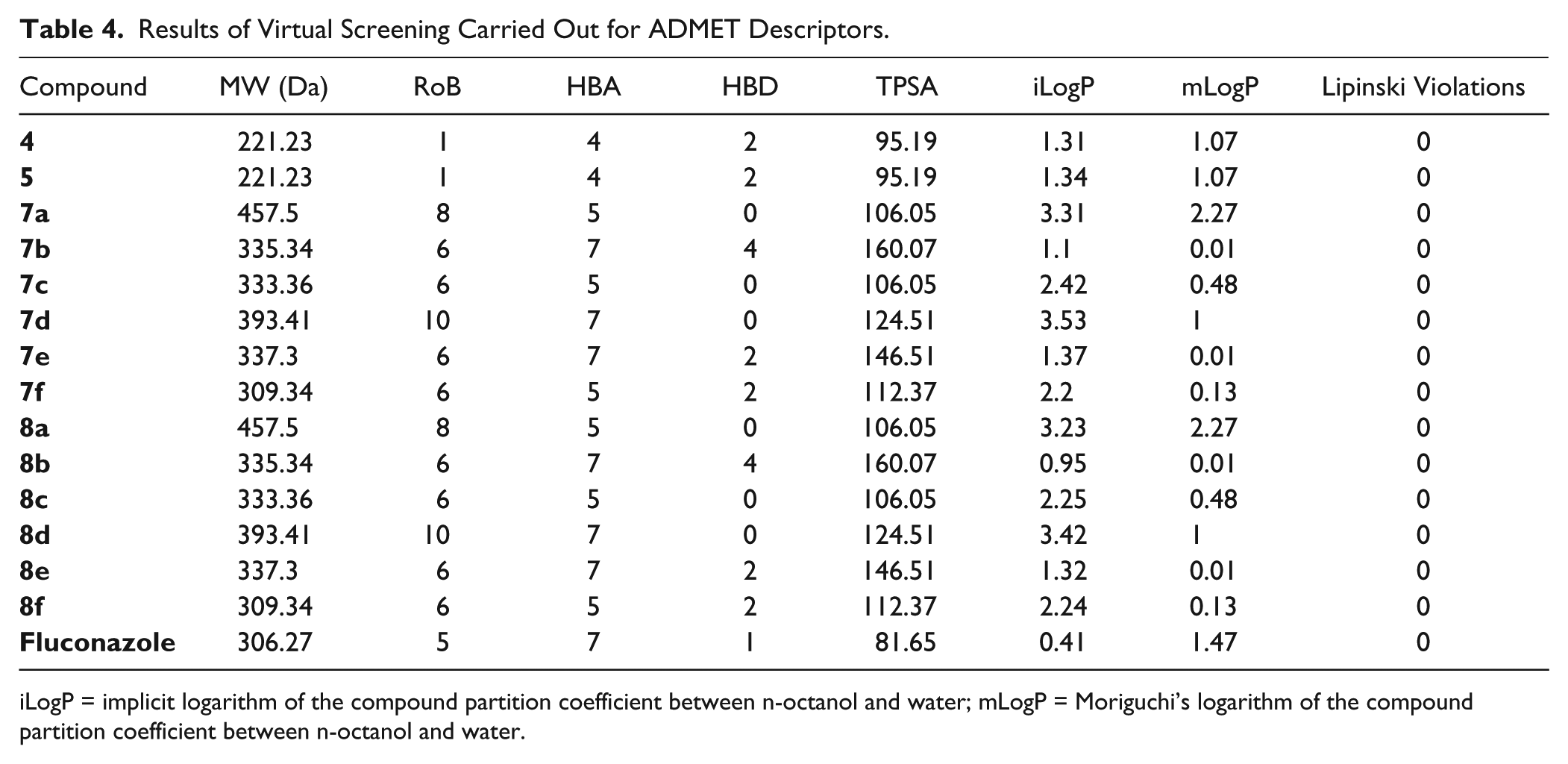
iLogP = implicit logarithm of the compound partition coefficient between n-octanol and water; mLogP = Moriguchi’s logarithm of the compound partition coefficient between n-octanol and water.
All compounds passed the RO5, having MW < 500 Da, mLogP < 4.15, less than 10 HBAs, and less than 5 HBDs. Also, all compounds passed the TPSA limit to 140 Å2, except
Regarding compounds’ lipophilicity, it was observed that the presence of voluminous substituents (such as phenylcarbonylmethyl in compounds
The predicted solubility, gastrointestinal absorption, blood–brain barrier penetrability, P-glycoprotein affinity, and CYP inhibition of the synthesized compounds are summarized in
Table 5
. Compounds
Predicted Solubility, Gastrointestinal Absorption, Blood–Brain Barrier Penetrability, P-Glycoprotein Affinity, and CYP Inhibition of the Synthesized Compounds.
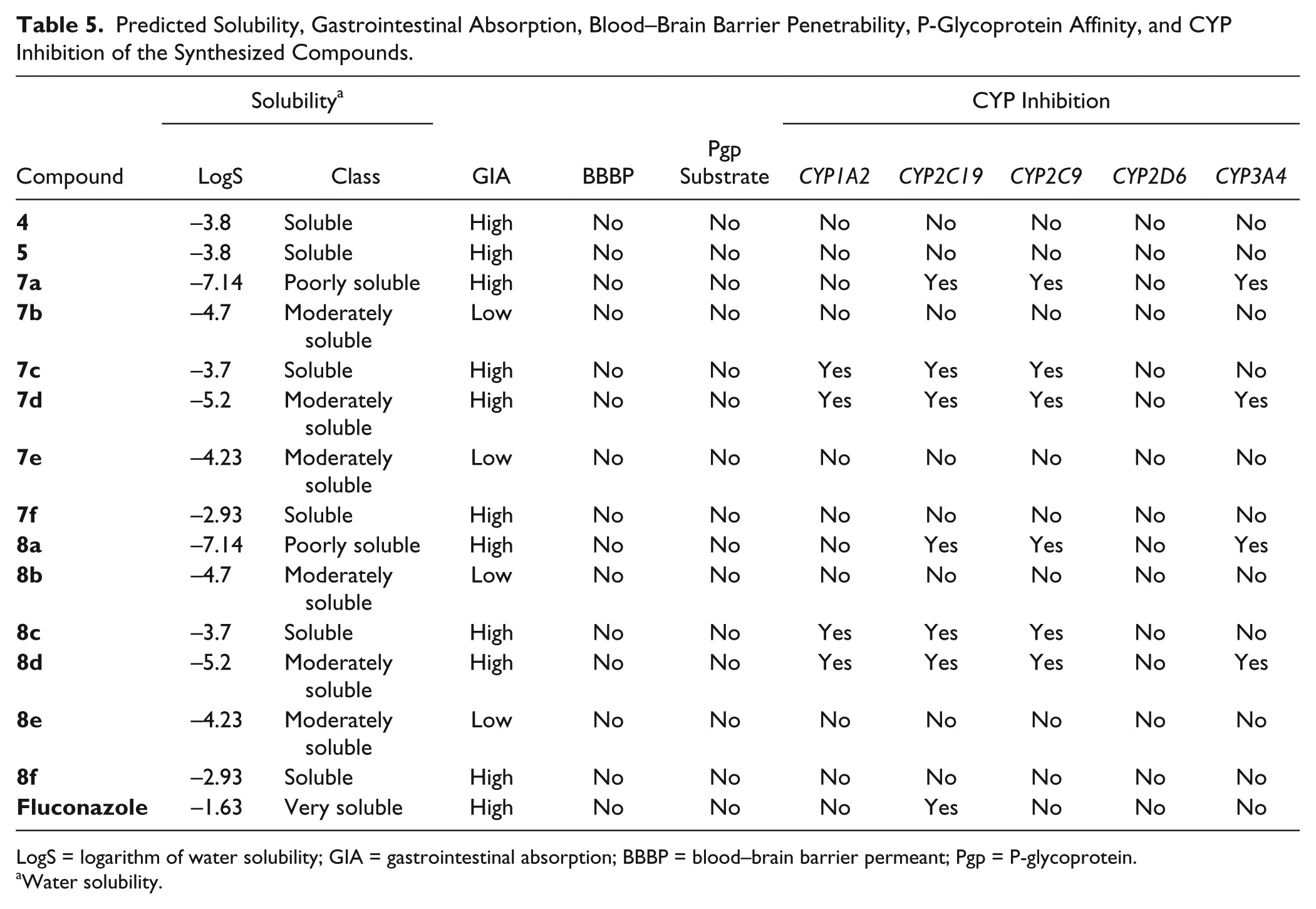
LogS = logarithm of water solubility; GIA = gastrointestinal absorption; BBBP = blood–brain barrier permeant; Pgp = P-glycoprotein.
Water solubility.
Conclusions
In the search for new antifungal agents, 12 novel thiazolidine-2,4-dione derivatives were synthesized by microwave-assisted synthesis and tested in vitro for their antifungal properties, against several strains of
For all the studied compounds, a lipophilicity augmentation was observed by introducing ester groups or phenylcarbonyloxomethyl rests. The thorough virtual screening carried out for ADMET profiling of the thiazolidine-2,4-dione derivatives led to the conclusion that based on the predicted values of physicochemical descriptors, all derivatives are small molecules with good predictions for oral bioavailability according to RO5, thus making them suitable for systemic use. An absence of blood–brain barrier penetrability was also forecasted for all the synthesized compounds, thus making them less likely to possess central nervous system adverse effects.
Footnotes
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work reported in this paper was financially supported by the “Iuliu Hat̨ieganu” University of Medicine and Pharmacy, Cluj-Napoca, PCD 7690/68/15.04.2016 and 5200/59/01.03.2017.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
