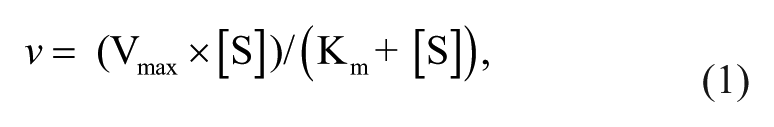Abstract
Amoxapine has been demonstrated to be a potent inhibitor of Escherichia coli β-glucuronidase. This study aims to explore the factors causing unsatisfactory efficacy of amoxapine in alleviating CPT-11–induced gastrointestinal toxicity in mice and to predict the outcomes in humans. Amoxapine (100 µM) exhibited poor and varied inhibition on β-glucuronidase activity in gut microbiota from 10 healthy individuals and their pool (pool, 11.9%; individuals, 3.6%−54.4%) with IC50 >100 µM and potent inhibition toward E. coli β-glucuronidase (IC50 = 0.34 µM). p-Nitrophenol formation from p-nitrophenyl-β-D-glucuronide by pooled and individual gut microbiota fitted classical Michaelis-Menten kinetics, showing similar affinity (Km = 113–189 µM) but varied catalytic capability (Vmax = 53–556 nmol/h/mg). Interestingly, amoxapine showed distinct inhibitory effects (8.7%–100%) toward β-glucuronidases of 13 bacterial isolates (including four Enterococcus, three Streptococcus, two Escherichia, and two Staphylococcus strains; gus genes belonging to OTU1, 2 or 21) regardless of their genetic similarity or bacterial origin. In addition, amoxapine inhibited the growth of pooled and individual gut microbiota at a high concentration (6.3%–30.8%, 200 µM). Taken together, these findings partly explain the unsatisfactory efficacy of amoxapine in alleviating CPT-11–induced toxicity and predict a poor outcome of β-glucuronidase inhibition in humans, highlighting the necessity of using a human gut microbiota community for drug screening.
Keywords
Introduction
β-Glucuronidases (EC 3.2.1.31) belong to glycoside hydrolase family 2 and could hydrolyze β-linked glucuronides. 1 Bacterial β-glucuronidases in human gut play an essential role in cleaving glucuronide conjugates of dietary compounds, endobiotics, and xenobiotics, 2 which could result in enhanced systemic exposure of many compounds through facilitating enterohepatic recirculation to release more parent compound for absorption or cause toxicity due to production of active metabolites in gut lumen. Recently, there has been increasing research interest in applying bacterial β-glucuronidase to enhance drug exposures 3 and developing bacterial β-glucuronidase inhibitors to relieve drug toxicity.4–7 For example, intraduodenal administration of an anticancer compound (JNJ-10198409) together with Escherichia coli (E. coli) β-glucuronidase to catheterized rats resulted in elevated systemic exposures of both the parent compound and its glucuronide. 3 On the other hand, inhibition of bacterial β-glucuronidases alleviated gastrointestinal toxicity of irinotecan (CPT-11) by inhibiting the release of the active metabolite SN-38 from the glucuronide SN-38G in gut lumen. 4
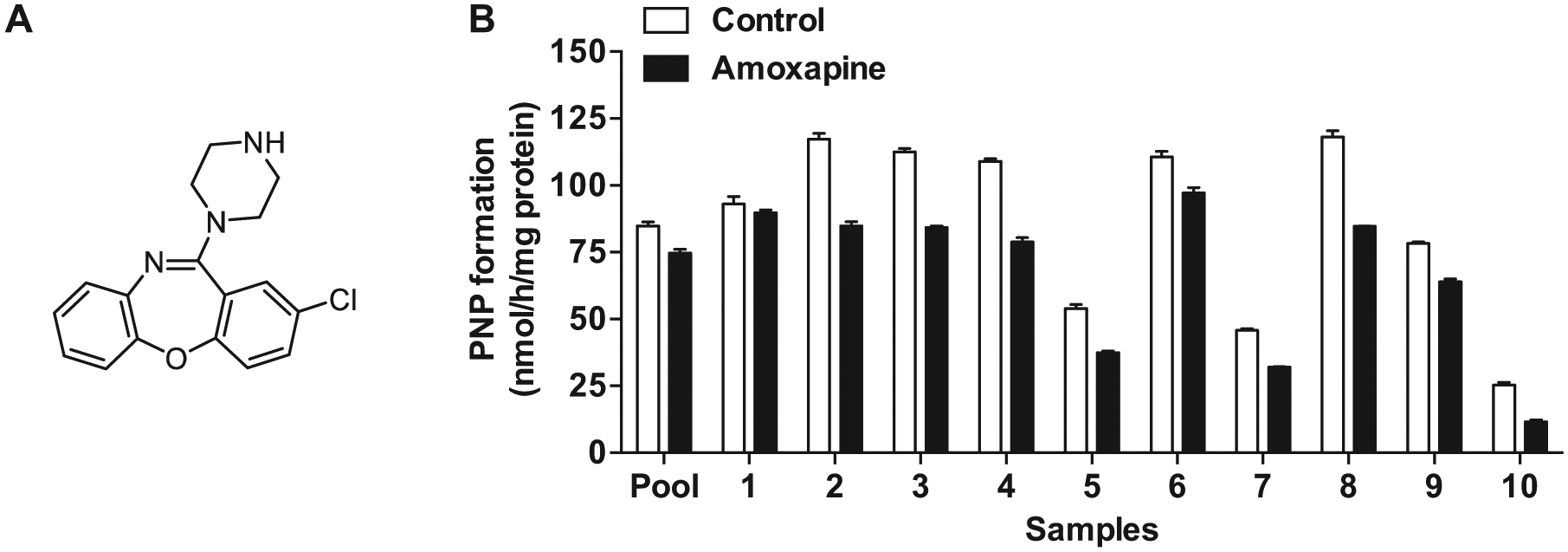
CPT-11 is one of the most commonly used anticancer agents for colon cancer therapy. 8 CPT-11 is converted to SN-38 by carboxylesterases in liver. The active metabolite is further conjugated to form SN-38G by hepatic UDP-glucuronosyltransferases (UGTs). The glucuronide could then be exported into the gut lumen and subjected to hydrolysis by gut bacterial β-glucuronidases to generate SN-38, which causes damage of intestinal epithelial cells and severe diarrhea in up to 40% of treated patients, limiting dose intensification of CPT-11.4,9 Recent studies reported the applications of β-glucuronidase inhibitors in the alleviation of CPT-11 gastrointestinal toxicity.4,6,10 Amoxapine ( Fig. 1A ), a tricyclic antidepressant, showed a potent inhibitory effects toward E. coli β-glucuronidase (IC50 = 0.39 µM) but had no effect on the orthologous mammalian β-glucuronidase (Bos taurus) in a cell-based assay. 11 Concomitant use of amoxapine significantly suppressed severe diarrhea-induced by CPT-11 on days 7 to 9 in mice, but in a later stage of treatment, 70% of the mice still progressed to severe diarrhea. 6 The authors speculated that the complicated manipulation of CPT-11 metabolism by hepatic carboxylesterases and UGTs may cause biliary excretion of the toxic SN-38 into gut lumen, which might account for the diarrhea in a later stage, while further study is warranted to confirm the explanation.
(
A recent study carried out by Wallace and coworkers 12 also revealed that different bacterial β-glucuronidases showed varied propensities for inhibition. For instances, one inhibitor (inhibitor 1) showed potent inhibition on E. coli β-glucuronidase (IC50 = 0.16 µM) but no inhibition on Bacteroides fragilis β-glucuronidase. This finding offers another explanation for the unsatisfactory efficacy of amoxapine in alleviating CPT-11–induced toxicity: amoxapine may selectively inhibit β-glucuronidases of some bacterial origins, including E. coli β-glucuronidase tested in the study by Wallace et al., 12 while the alleviation of CPT-11 toxicity should rely on a potent inhibition of multiple bacterial β-glucuronidase activities instead of E. coli β-glucuronidase alone, which was used for screening inhibitors. In addition, the catalytic activity and propensity for inhibition of gut bacterial β-glucuronidases may have great species difference. Thus, it is necessary to assess the inhibitory effects of amoxapine on human gut bacterial β-glucuronidases, which will provide more reliable information to predict the potential of amoxapine on alleviating CPT-11–induced toxicity in humans.
Therefore, in this study, the effects of amoxapine on β-glucuronidase activity of pooled and individual human gut microbiota and human gut bacterial isolates were investigated in vitro with intentions to (1) explore the factors causing unsatisfactory efficacy of amoxapine in alleviating CPT-11–induced toxicity in mice and (2) predict the outcomes of β-glucuronidase inhibition by amoxapine on the gastrointestinal toxicity of CPT-11 in humans. The effects of amoxapine on the growth of human gut microbiota were also determined to evaluate whether amoxapine disrupts gut microbial homeostasis.
Materials and Methods
Materials
DMSO, bovine hemin, vitamin K1, amoxapine, p-nitrophenol (PNP), p-nitrophenyl-β-D-glucuronide (PNPG), and E. coli β-glucuronidase (G8401) were supplied by Sigma-Aldrich (St. Louis, MO). L-cystine was purchased from Research Organics (Cleveland, OH). Purities were all >98%. Agar was purchased from Biosharp (Hirono, Japan). The BBL Brain Heart Infusion (BHI) medium was purchased from Becton Dickinson (Franklin Lakes, NJ). Dulbecco’s phosphate-buffered saline (PBS) was provided by Life Technologies (Carlsbad, CA). Deionized water was purified by a Milli-Q purification system (Millipore, Bedford, MA).
Preparation of Human Gut Microbiota
Ten fresh fecal samples were obtained from healthy volunteers (5 males and 5 females, Nos. 1–10, 24–41 years old) who had not taken any medicine and avoided alcohol 3 months before fecal collection. The experiment was approved by the internal Research Ethics Committee of the Institute of Chinese Medical Sciences, University of Macau. Fresh fecal samples were immediately stored at –80 °C until use. Gut microbiota were prepared at 4 °C according to our previous report 13 with minor modifications. Briefly, fecal samples were pooled at 0.5 g each or individually processed at 5 g each. The individual or pooled fecal samples were mixed well with 20 mL 100 mM potassium phosphate (PBS) buffer (pH 7.4). The resultant fecal suspension was centrifuged at 200 g for 5 min and the supernatant decanted and centrifuged at 5000 g for 30 min. The resultant precipitate was resuspended with 5 mL PBS buffer (pH 7.4) to produce gut bacteria suspension.
Isolation and Identification of Human Microbial Isolates
Individual human gut bacteria suspensions were diluted 10-fold serially in sterile water, inoculated on BHI agar plates (3.7 g BHI, 0.05 mg vitamin K1, 0.5 mg bovine hemin, 50 mg L-cystine, 1 g agar) and incubated at 37 °C for 48 h under anaerobic conditions (N2/H2/CO2 90:5:5; MACS-VA500 workstation; Meintrup DWS, Lähden, Germany). Following incubation, different colony types were counted, isolated in pure culture, and cultured in BHI broth at 37 °C for another 24 h. Cells were collected by centrifugation at 13,000 g for 10 min. Genomic DNA from gut bacterial isolates were extracted as described above. Bacterial universal primers 27F (AGAGTTTGATCMTG GCTCAG) and 1492R (TACGGYTACCTTGTTACGA CTT) were used to amplify the 16S ribosomal RNA (rRNA) from genomic DNA. Amplification was performed with Premix Ex taq DNA Polymerase (Takara, Tokyo, Japan) according to the manufacturer’s instructions. The amplification was performed as follows: initial denaturation for 5 min at 94 °C, 35 cycles each of denaturation for 30 s at 94 °C, annealing for 30 s at 55 °C, primer extension for 1.5 min at 72 °C, and a final extension for 10 min at 72 °C. 14 Sequencing with primers 27F and 1492R was performed on an Applied Biosystems (Foster City, CA) 3730 DNA Analyzer. The isolates were identified according to the databank of 16S rRNA sequences of the EzBioCloud server (http://www.ezbiocloud.net/). 15
Microbial β-Glucuronidase Gus Gene Identification by Degenerate PCR
Degenerate PCR was performed with genomic DNA of bacterial isolates as templates. Degenerate primers GNfor (TATTTAAAAGGITTYGGIMRICAYGARGA) and GNP-2mod (CCTTCTGTTGTIKBRAARTCIGCRAARTTCCA) were synthesized according to Dabek et al. 16 Amplification was performed with Premix Ex taq DNA Polymerase (Takara) according to the manufacturer’s instructions using a ramped annealing approach with conditions as follows: initial denaturation (3 min at 94 °C) and then 35 cycles of denaturation (30 s at 94 °C), ramped annealing (20 s at 55 °C, 5 s at 50 °C, 5 s at 45 °C, and 5 s at 40 °C), elongation (1 min at 72 °C), and a final extension (10 min at 72 °C). PCR products obtained were purified using the Quick Gel Extraction Kit (CWBIO, Beijing, China) and cloned into pGEM-T-Easy vector systems (Promega, Madison, WI) according to the manufacturer’s instructions. Sequencing with vector primer M13F was performed on an Applied Biosystems 3730 DNA Analyzer. Gus gene type was identified by denaturing-gradient gel electrophoresis and phylogenetic analysis using the program MEGA4.0 (our research group’s unpublished data).
In Vitro Assay for β-Glucuronidase Activity and Inhibitor Efficacy
β-Glucuronidase activity of human gut microbiota and bacterial isolates was determined by measuring PNP generated from PNPG by crude microbial proteins. Gut microbiota or microbial isolate suspension (0.3 mL) were dispersed in 3 mL PBS buffer (pH 7.4) and ultrasonicated for 10 min in cycles of 3 s of ultrasonication and 4 s of resting time using a BILON-250Y (Shanghai Bilon Instrument Co., China) (60% power, Φ6 probe) ultrasonic processor to prepare crude proteins. Protein concentrations of these samples were determined with a bicinchoninic acid kit (Pierce, Rockford, IL). The PNPG hydrolysis assays were conducted in 96-well flat-bottomed tissue culture plates (Nunc, Roskilde, Denmark). Reactions consisted of 10 µL PNPG (250 µM), 10 µL bacterial protein (0.05–0.3 mg/mL), and 80 µL PBS buffer (pH 7.4). After a 1-h incubation at 37 °C in a plate reader (Tecan, Japan Co., Kanagawa, Japan), the formation of the hydrolytic product PNP was monitored by recording the absorbance at 405 nm, and concentrations were determined from a standard curve (0–200 µM PNP in PBS buffer, pH 7.4).
PNPG hydrolysis activity by crude enzymes of human gut microbiota and bacterial isolates in the presence of amoxapine (100 µM) were also determined and conducted in parallel to controls without amoxapine. All reactions were performed in triplicate.
Kinetics of PNPG Hydrolysis by Human Gut Bacteria
The kinetics of PNPG hydrolysis by crude enzymes of human gut bacteria were determined under the initial rate conditions. The assays were carried out as described earlier, except that the concentrations of PNPG ranged from 50 to 1000 µM. All assays were performed in triplicate.
Determination of IC50 Values
IC50s of amoxapine toward crude enzymes of pooled and three individual (Nos. 1, 7, and 10) human gut bacteria and E. coli β-glucuronidase were determined in the aforementioned in vitro assays with minor modifications. Briefly, the reaction system consisted of 10 µL inhibitors at various concentrations (0.001–100 µM), 10 µL PNPG (250 µM), 10 µL protein samples from human gut microbiota (0.1 mg/mL) or E. coli β-glucuronidase (2 µg/mL), and 70 µL PBS buffer (pH 7.4). Relative activity was defined as percent of PNP formation in the presence of amoxapine compared with untreated controls. The IC50 values were calculated as the concentration of inhibitor that produced 50% in vitro inhibition and evaluated from the concentration-response curve using Prism 5.0 (GraphPad Software, La Jolla, CA). 10
Assays for Growth Inhibition of Human Gut Bacteria
The effects of amoxapine on the growth of pooled and three individual (Nos. 1, 7, and 10) human gut bacteria were determined at a concentration range between 25 and 200 µM (DMSO, final concentration 2%). Gut bacteria were cultured in LB broth to an OD600 of 0.3 to 0.4, and 5 µL fresh gut bacteria solution was added into 95 µL LB broth with different concentrations of amoxapine and incubated at 37 °C for 12 h. Bacterial growth was recorded by measuring absorbance at 600 nm. Controls (2% DMSO) and blank (deactivated bacteria) were made, and all assays were performed in triplicate. The percentage of bacterial growth inhibition was computed as described by La Fuente et al. 17 with minor modifications: percentage inhibition = 100 × (ODcontrol − ODamoxapine)/(ODcontrol − ODblank).
Data Analysis
All data are expressed as mean ± standard deviation (SD). Data are representatives of three independent experiments. The differences between groups were compared using the Student t test. P < 0.05 was considered statistically significant.
The apparent kinetics parameters of PNPG hydrolysis by crude enzymes of human gut bacteria were calculated by fitting the experimental data with a Michaelis-Menten (equation (1)) model with nonlinear regression analysis using Prism 5.0 (GraphPad Software). The in vitro intrinsic clearance (CLint) was then calculated with equation (2).
where v is the velocity of metabolite formation, Vmax is the maximum velocity, Km is the Michaelis constant defined as the substrate concentration at half of Vmax, [S] is the substrate concentration, and CLint is the intrinsic clearance.
Results and Discussion
Effects of Amoxapine on β-Glucuronidase Activity in Pooled and Individual Human Gut Microbiota
β-Glucuronidase activity in crude enzymes of pooled and individual gut microbiota samples from 10 healthy volunteers was determined with PNPG (250 µM) as the substrate. The activity of individual gut microbiota ranged within 25.3 to 118.1 nmol/h/mg protein ( Fig. 1B ). Sample of No. 8 showed the highest activity (117.5 nmol/h/mg protein), about 2.6 times that of activity of the sample from No. 7 (45.8 nmol/h/mg protein). These data indicate varied PNPG hydrolysis capability in healthy volunteers, which agrees with the data (PNP formation 138–294 nmol/h/mg) obtained from five healthy individuals reported previously. 18 Surprisingly, amoxapine showed poor inhibitory effects on β-glucuronidase activity of the crude enzymes of pooled human gut microbiota (7.7%), even at 100 µM. Moreover, the inhibitory effects of amoxapine (100 µM) on β-glucuronidase activity in the gut microbiota community from different individuals exhibited large variation, with percent inhibition ranging from 3.6% on sample No. 1 to 54.4% on No. 10. The poor and varied inhibition of amoxapine on the overall β-glucuronidase activity in gut microbiota from different individuals should be attributed to the individual differences in gut microbiota composition 19 and the varied propensities for inhibition of different bacterial β-glucuronidases. 12 Since the reactions were carried out with total bacterial proteins that contain complex proteins in addition to β-glucuronidases, we could not exclude that nonspecific protein binding may also account for the incomplete inhibitory effects of amoxapine.
To our knowledge, this is the first report on the inhibitory effects of amoxapine on β-glucuronidase activity of human gut microbiota. The results indicate poor outcomes of amoxapine in alleviating CPT-11–caused gastrointestinal toxicity in humans. Since the relative abundance of most of the dominant genera in mouse and human gut microbiota is quite different, 20 amoxapine might exhibit better efficacy in inhibiting β-glucuronidase activity of mouse gut microbiota, which could explain its efficacy in alleviating CPT-11–induced toxicity in earlier stages reported recently. 6
Kinetics of PNPG Hydrolysis by Human Gut Microbiota
Three representative gut microbiota samples, which showed low PNPG hydrolysis activity (No. 7) and lowest (No. 1) or highest (No. 10) inhibition on PNPG hydrolysis by amoxapine, were selected for an enzyme kinetics study. The effects of the concentration of PNPG on its hydrolysis activity by crude enzymes of human gut microbiota are depicted by the direct plots and the Eadie-Hofstee plots of the velocity and concentration data obtained from the pooled sample and three representative samples ( Fig. 2 ). The Vmax and Km as well as the in vitro intrinsic clearance (CLint), which were calculated from the formations of PNP, are summarized in Table 1 . PNPG hydrolysis by crude enzymes of pooled and three individual (Nos. 1, 7, and 10) gut microbiota followed typical Michaelis-Menten kinetics. The apparent Km ranged from 113 to 189 µM, indicating comparable affinity. However, the velocity of PNP formation by β-glucuronidases from three individuals ranged from 53 to 556 nmol/h/mg protein, with that of the No. 1 sample 7 to 10 times that of No. 7 and No. 10, indicating large individual variation in gut bacterial PNPG hydrolyzing capability. As a result, the in vitro intrinsic clearance of PNPG (CLint = 3.0 mL/h/mg protein) by gut microbiota from sample No. 1 was about six times those from Nos. 7 and 10 (CLint ~0.5 mL/h/mg protein) ( Table 1 ). These results indicate compounds undergoing enterohepatic recirculation may exhibit distinct systemic exposure or local exposure to toxic metabolite (e.g., CPT-11 metabolite SN38) in different individuals.
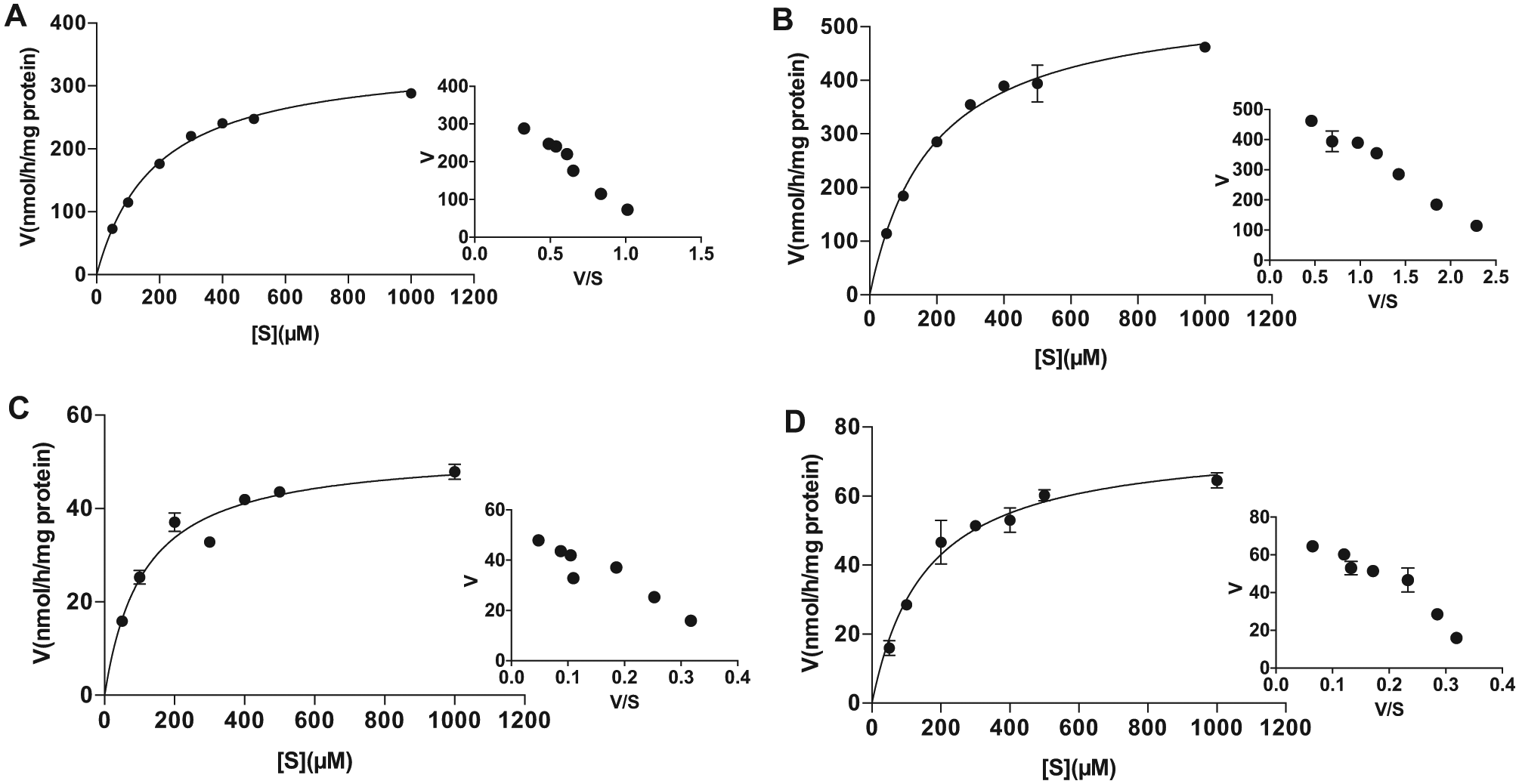
Effects of the concentration of p-nitrophenyl-β-D-glucuronide (PNPG) on formation rates of p-nitrophenol (PNP) in crude enzymes of pooled (
Apparent Kinetic Parameters of p-Nitrophenyl-β-D-Glucuronide Hydrolysis in Crude Enzymes of Pooled and Three Individual Human Gut Microbiota Samples Obtained by Fitting the Experimental Data with the Michaelis-Menten Equation.
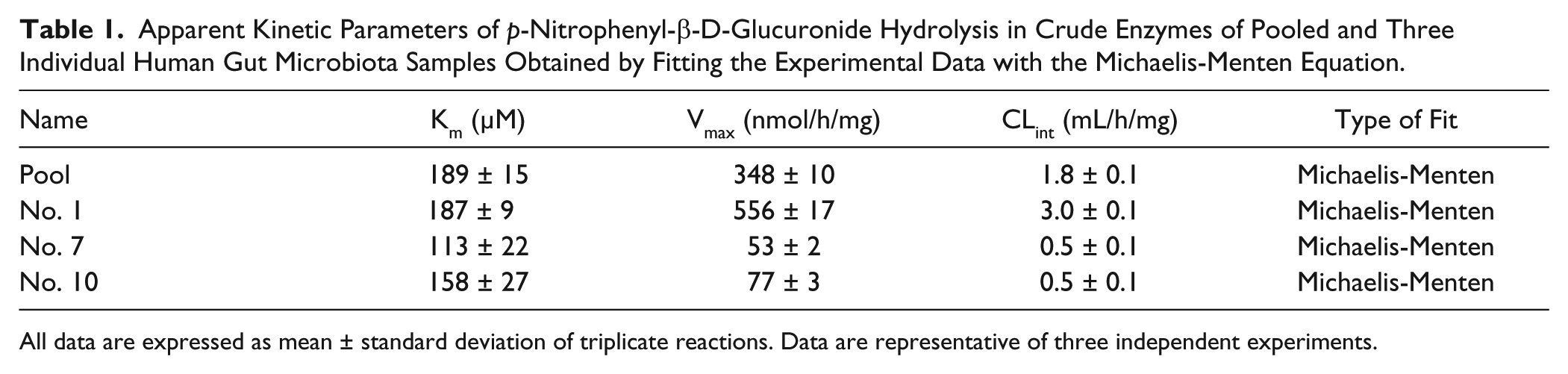
All data are expressed as mean ± standard deviation of triplicate reactions. Data are representative of three independent experiments.
IC50s of Amoxapine on β-Glucuronidase Activity of Human Gut Microbiota and E. coli β-Glucuronidase
The effects of inhibitor concentration on β-glucuronidase activity in crude enzymes of pooled and three individual (Nos. 1, 7, and 10) human gut microbial proteins were further evaluated at different concentrations of amoxapine using E. coli β-glucuronidase as the positive control. As shown in Fig. 3 , amoxapine showed potent inhibition on E. coli β-glucuronidase with an IC50 of 0.34 µM, which was comparable to an IC50 of 0.39 µM reported by Ahmad et al. 11 However, amoxapine exhibited poor inhibition on β-glucuronidase activity in total bacterial proteins of both pooled (IC50: 337 ± 26 µM) and individual samples (IC50: No. 1, 240 ± 39 µM; No. 7, 176 ± 16 µM; No. 10, 123 ± 12 µM) with maximum inhibition less than 40% at 100 µM. The poor inhibition on β-glucuronidase activities in human gut microbiota might explain the unsatisfactory efficacy of amoxapine in alleviating CPT-11–induced toxicity, 6 highlighting the necessity of assessing the inhibitory effects on the β-glucuronidase activity of the gut microbiota community instead of single bacterial isolates. We can also speculate from this result that amoxapine might show high selectivity in inhibiting β-glucuronidases of different bacterial origins. Again, due to the existence of complex proteins other than β-glucuronidases, we could not rule out the impact of nonspecific protein binding on the inhibitory effects of amoxapine on β-glucuronidase activity in crude extracts of pooled and individual gut microbiota. Actually, using same amounts of crude enzymes, such as human liver microsomes, for enzyme kinetics or IC50 determination is commonly adopted when the content of target enzyme is unknown. 21 In this study, reactions were carried out with the same amounts of crude proteins, and the inhibitory effect was assessed based on the differences of PNP production in the gut microbiota of the same individual/pooled sample in the presence and absence of amoxapine, which minimized the impact of the individual differences in β-glucuronidases.
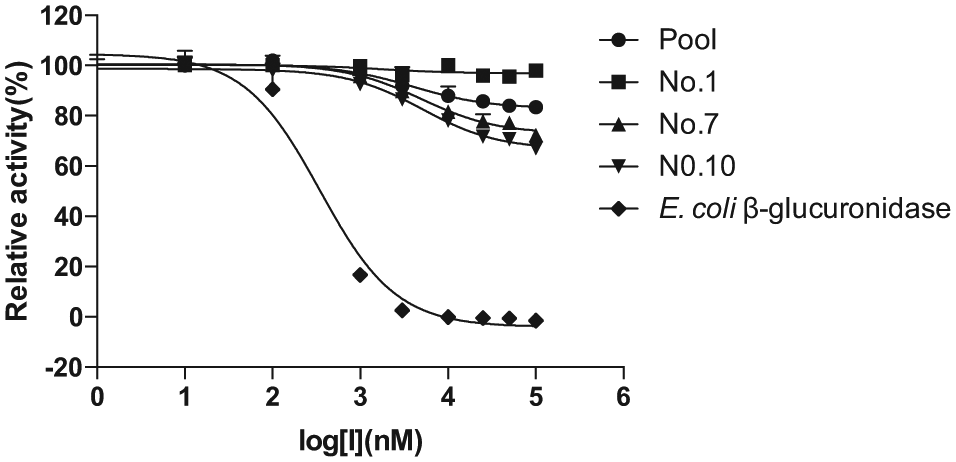
Effects of the concentration of amoxapine on p-nitrophenol (PNP) formation from p-nitrophenyl-β-D-glucuronide (PNPG) by crude enzymes of pooled and three individual human gut microbiota with Escherichia coli β-glucuronidase as control. The IC50 values (µM) of amoxapine toward β-glucuronidases from pooled, No. 1, No. 7, No. 10, and E. coli β-glucuronidase were 337 ± 26, 240 ± 39, 176 ± 16, 123 ± 12, and 0.34 ± 0.06, respectively. All data are expressed as mean ± standard deviation of triplicate reactions. Data are representative of three independent experiments.
Effects of Amoxapine on β-Glucuronidase Activity of Gut Bacterial Isolates
Among 23 human gut bacterial isolates tested, 13 showed detectable β-glucuronidase activity ( Fig. 4 ). The activity ranged within 6.3 to 320.2 nmol/h/mg protein, revealing large variation in PNPG hydrolyzing activity among different isolates. Moreover, the 13 isolates were screened for the β-glucuronidase gus gene by degenerate PCR and resulted in 10 isolates carrying gus genes that were further identified by cloning and sequencing ( Suppl. Table S1 ). Nine of the isolates (six carry gus gene OTU1 and three have no gus gene) could be fully inhibited by 100 µM amoxapine, while three (gus gene belongs to OTU1, OTU2, or OTU21) could only be partially inhibited and one (gus gene belongs to OTU2) could not be inhibited. For example, amoxapine only showed about 50% inhibition on β-glucuronidase activity from Klebsiella quasipneumoniae 3X26 (OTU1), Streptococcus agalactiae 3X23 (OTU2), and Staphylococcus pasteuri 3I10 (OTU21) and nearly no inhibition on β-glucuronidase from Staphylococcus warneri 3P7 (OTU2) ( Fig. 4 ). These results indicated that β-glucuronidase activity and the inhibitory effect of amoxapine vary with gut bacterial isolates regardless of their gus gene similarity or bacterial origin.
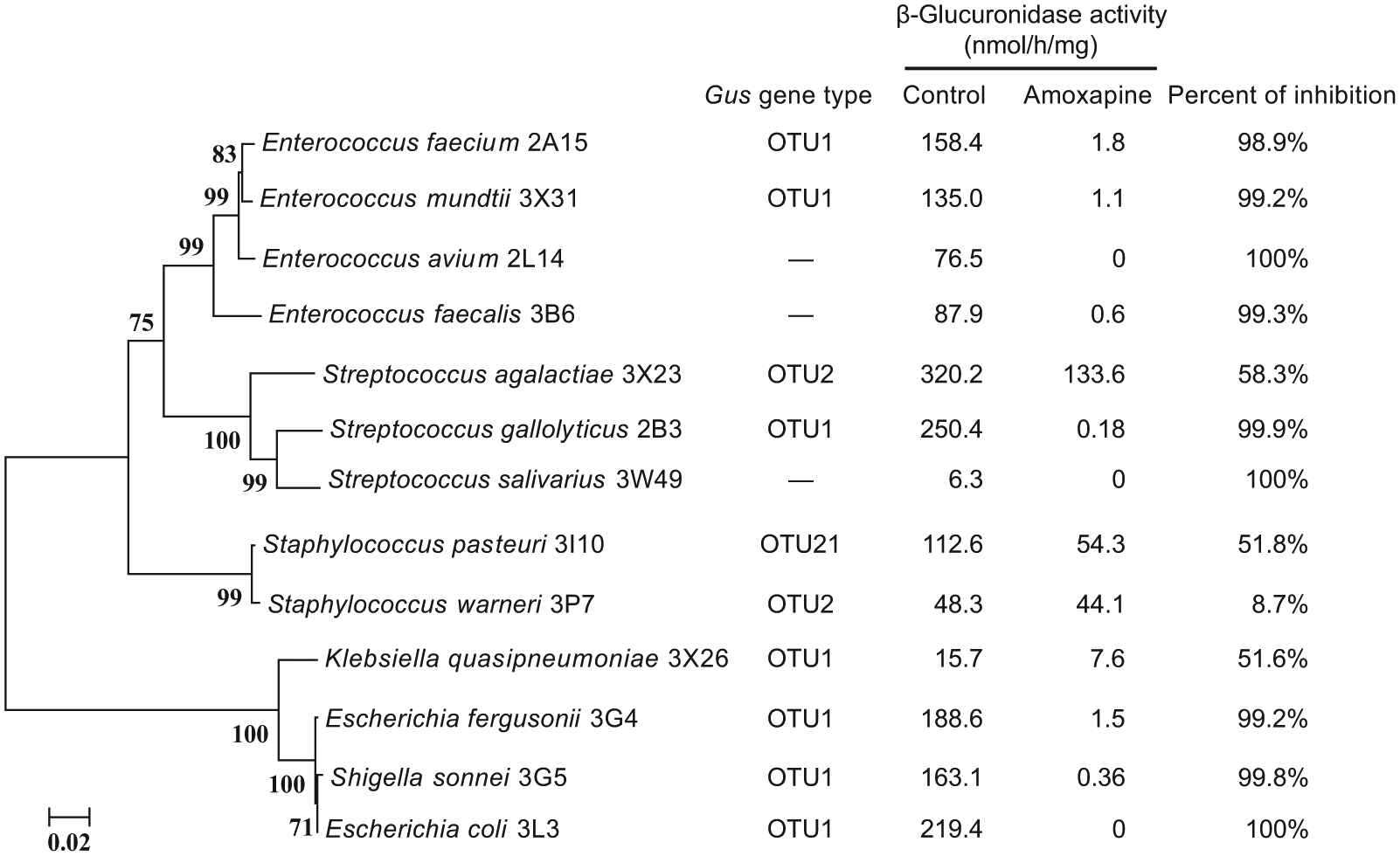
Phylogenetic tree (left) of bacterial isolates showing p-nitrophenyl-β-D-glucuronide (PNPG) hydrolysis activity and the inhibitory effects of amoxapine (right). The phylogenetic tree was constructed based on the 16S ribosomal RNA sequence with the program MEGA4.0 using the neighbor-joining method, 1000 times bootstrap, and distance model Kimura. Gus gene type was determined by degenerate PCR. —, gus gene not detectable. All reactions were performed in duplicate. Data are representative of three independent experiments.
In a recent study, amoxapine showed a potent inhibition on E. coli β-glucuronidase (IC50 = 0.39 µM) but an insignificant effect against mammalian β-glucuronidase (Bos taurus). 11 In this study, we confirmed the potent inhibition of amoxapine on E. coli β-glucuronidase (IC50 = 0.34 µM). However, as we have speculated, amoxapine could not completely inhibit β-glucuronidase activities of all 13 isolates tested, and the extent of the inhibition also showed great variation (8.7%–100%) regardless of their genetic similarity or bacterial origins. This finding is in concert with a recent study which reported that 10 compounds showed potent inhibition on E. coli β-glucuronidase but no inhibition on Bacteroides fragilis β-glucuronidase, 12 indicating that β-glucuronidases from different gut bacterial isolates may exhibit different propensities for inhibition. These findings further demonstrated that the effects on gut microbial community are more important than a single bacterial strain for screening β-glucuronidase inhibitors. Again, nonspecific binding to other proteins in the crude extracts may exist and prevent accurate comparison of the inhibitory effects of amoxapine among different bacterial isolates. Further study with pure β-glucuronidase of each isolate is necessary to determine the inhibition propensity of amoxapine on β-glucuronidases of different bacterial origins.
Effects of Amoxapine on the Growth of Human Gut Microbiota
The effects of amoxapine on the growth of pooled and three individual (Nos. 1, 7, and 10) human gut microbiota were investigated by measuring absorbance at 600 nm after incubation for 12 h. As shown in Figure 5 , amoxapine showed no inhibition on the growth of pooled and three individual human gut microbiota at lower concentrations (25–100 µM), and significant inhibition was only observed at 200 µM, which was 11.6% for the pooled sample and 30.8%, 9.3%, and 6.3% for No. 1, No. 7, and No. 10, respectively, indicating that the in vitro inhibitory effect on bacterial β-glucuronidases observed at 100 µM amoxapine in this study is not a consequence of bacterial growth inhibition.
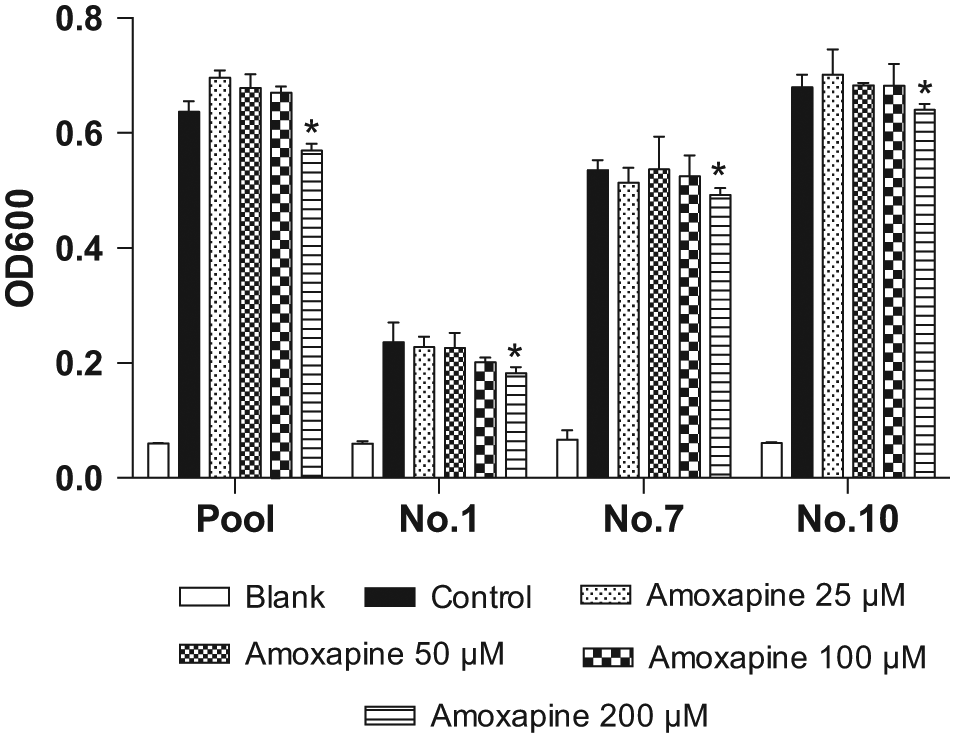
Effects of amoxapine on the growth of pooled and three individual human gut microbiota after 12-h incubation. All data are expressed as mean ± standard deviation of triplicate reactions. Data are representative of three independent experiments. Significant differences between samples were determined with the Student t test. *P < 0.05 versus controls.
In the recent study reported by Ahmad and coworkers, 11 the toxicity of amoxapine on E. coli cells was assessed with a metabolic viability assay that quantified the number of living cells in a culture through measuring the formation of formazan product. They demonstrated that amoxapine showed no signs of toxicity at up to 100 µM. In this study, we found that amoxapine only suppressed the growth of the human gut microbiota community at 200 µM. In fact, amoxapine showed antidiarrhea efficacy in mice at an oral dosage of 1 mg/kg, 6 which may result in high local exposure of amoxapine to gut bacteria. Considering the poor inhibition of amoxapine on β-glucuronidases in pooled and individual gut microbiota samples, we could not exclude that inhibition of amoxapine on the growth of gut microbiota could partially account for its efficacy in alleviating CPT-11–induced toxicity.
It is interesting to note that individual No. 1 showed much lower OD600 values, one-third to half of the pooled sample and the other two individuals, indicating a much slower growth of the gut microbiota from this individual. This might be due to large individual variation in microbial composition, and more bacteria in sample No. 1 might be unculturable, leading to its insensitivity to amoxapine inhibition.
In conclusion, the present study revealed poor and varied inhibitory effects of amoxapine on β-glucuronidase activities of the human gut microbiota community and gut bacterial isolates and inhibition on the growth of human gut microbiota at high concentrations. These findings offer an explanation for the reported unsatisfactory efficacy of amoxapine in alleviating CPT-11–induced toxicity in mice and predict a poor outcome in humans when used together with CPT-11 to alleviate β-glucuronidase-mediated toxicity. Considering the wide distribution of β-glucuronidases in gut microbiota, our study also highlights the necessity of using the human gut microbiota community for drug screening.
Footnotes
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation (Ref. 81473281), University of Macau (MYRG2015-00220-ICMS-QRCM), and the Science and Technology Development fund of Macao SAR (043/2011/A2, 029/2015/A1).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.