Abstract
A crude extract from earthworms was used as an environmentally friendly and easily accessible biocatalyst for asymmetric direct aldol reactions. The β-hydroxy carbonyl compounds could be prepared in yields of up to 94%, with ee values of up to 98% and dr of up to >99:1. A wide range of substrates could participate in the reaction. This earthworm catalyst was also efficient in the large-scale reaction, providing product in excellent yield and good selectivity. The protocol as an example gives inspiration for the development of sustainable catalysts from nature, which also provides a potential possibility for application of earthworm catalyst in industrial production.
Introduction
In view of the importance of safety, health, and environmental issues, the use of nonpolluting, environmentally friendly, and easy-to-handle catalysts for reactions has long been a pursuit of chemists. 1 Nature is an important source of catalysts, but the use of natural catalysts in organic synthesis is very limited. Therefore, to find easy methods for use of environmentally benign natural catalysts is highly desirable. Some crude extracts of organisms have been employed as biocatalysts because of their simple and convenient preparation, environmental friendliness, and cost-effectiveness.2–5 Earthworms, just like humans, rely on bacteria in their intestines to oxidize and convert plants and minerals into nutrients. 6 Among the enzymes that are secreted by the alimentary tract of earthworms, a large number of serine proteases are most studied. 7 These enzymes are stable and strongly resistant to organic solvents and detergents, and they retain full activity for years at room temperature. 8 To use earthworm enzymes expediently, we developed a practical way to get crude extract of earthworms as an all-natural catalyst that avoids expensive and time-consuming enzyme purification. Our group recently reported that crude extract of earthworms, as a versatile, environmentally friendly, and sustainable biocatalyst, could catalyze various organic reactions including Mannich, aldol, Biginelli, Henry, aza-Diels-Alder reactions, and domino reactions for the synthesis of coumarin derivatives.9,10
The asymmetric direct aldol reaction as one of the most powerful carbon-carbon bond formation methods has become one of the central study issues in organic synthesis. 11 In recent years, a lot of successful metal catalysts and organocatalysts for direct asymmetric aldol reactions have been reported with high efficiency and enantioselectivity, 12 including some methods under solvent-free mechanochemical conditions,13–15 yet the development of eco-friendly and sustainable catalysts for the asymmetric aldol reaction is still a significant challenge. There are some reports about enzyme-catalyzed direct asymmetric aldol reactions using aldolases or some promiscuous enzymes.16–21 However, it is still difficult to find a cost-efficient and simple procedure for biocatalytic direct asymmetric aldol reaction for scale-up. 22 Thus, we think it is still necessary to further explore the application of earthworm extract for the asymmetric direct aldol reaction and especially for the large-scale reaction, although we have concisely reported the use of earthworm extract in asymmetric direct aldol reaction in which eight examples were investigated. 9 Herein, we report detailed studies of earthworm extract-catalyzed asymmetric direct aldol reaction with largely expanded substrates, in which 18 new examples were included and a scale-up reaction was tested. This study further demonstrates the excellent utility of earthworm extract for asymmetric direct aldol reaction.
Materials and Methods
Materials
Live earthworms (
General Procedure for the Synthesis of Compounds 3
A round-bottom flask was charged with the crude earthworm extract (75 mg), aldehyde
Results and Discussion
First, a cheap and readily obtainable crude earthworm extract was prepared from
Control Experiments and Solvent Screening. a
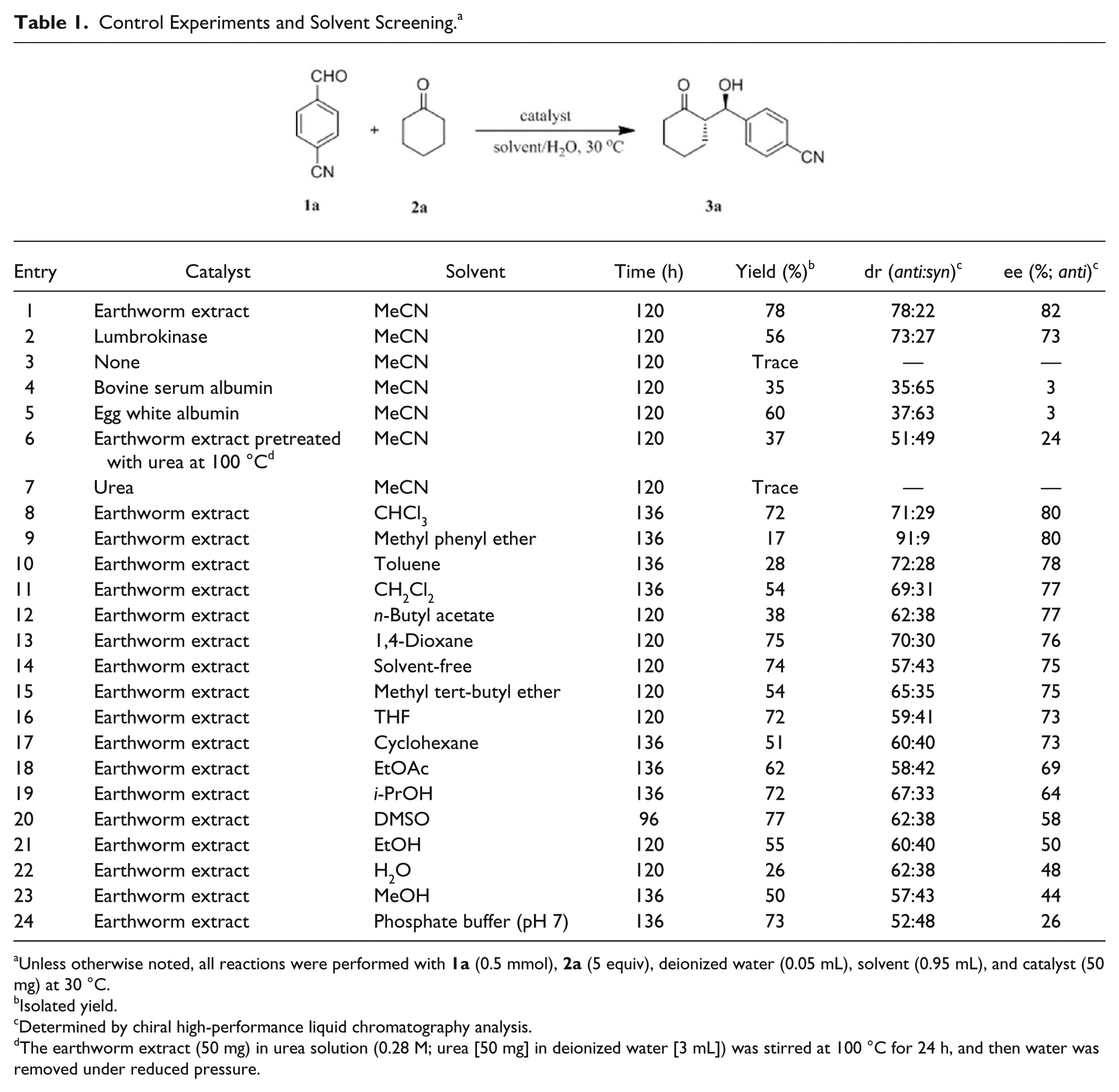
Unless otherwise noted, all reactions were performed with
Isolated yield.
Determined by chiral high-performance liquid chromatography analysis.
The earthworm extract (50 mg) in urea solution (0.28 M; urea [50 mg] in deionized water [3 mL]) was stirred at 100 °C for 24 h, and then water was removed under reduced pressure.
In addition, we explored effect of different solvents on the earthworm extract catalyzed model aldol reaction (
Table 1
, entries 1 and 8–24). The reaction medium had an obvious influence on this reaction. It seems likely that the reaction in low- to moderate-polarity solvents displayed better stereoselectivity than in high-polarity solvents. For instance, the reaction in MeCN, CHCl3, methyl phenyl ether, toluene, CH2Cl2,
As water contents are essential for the stereoselectivity and activity of enzymes in organic solvents,23,24 the influence of water contents on the earthworm extract catalyzed model aldol reaction was investigated (
Table 2
). The model reaction in MeCN (analytical purity) without addition of water gave the product in a yield of 73% with a low ee of 64% (
Table 2
, entry 1). The best yield of 85% was obtained with 74% ee when the water content was 20% (
Table 2
, entry 5). When the water content exceeded 25%, the yield and ee declined sharply (
Table 2
, entries 7 and 8). Taking into consideration both yield and ee, we chose 15% as the optimum water content, which gave a good yield of 83% with 78% ee and 76:24 (
Screening of Water Content. a
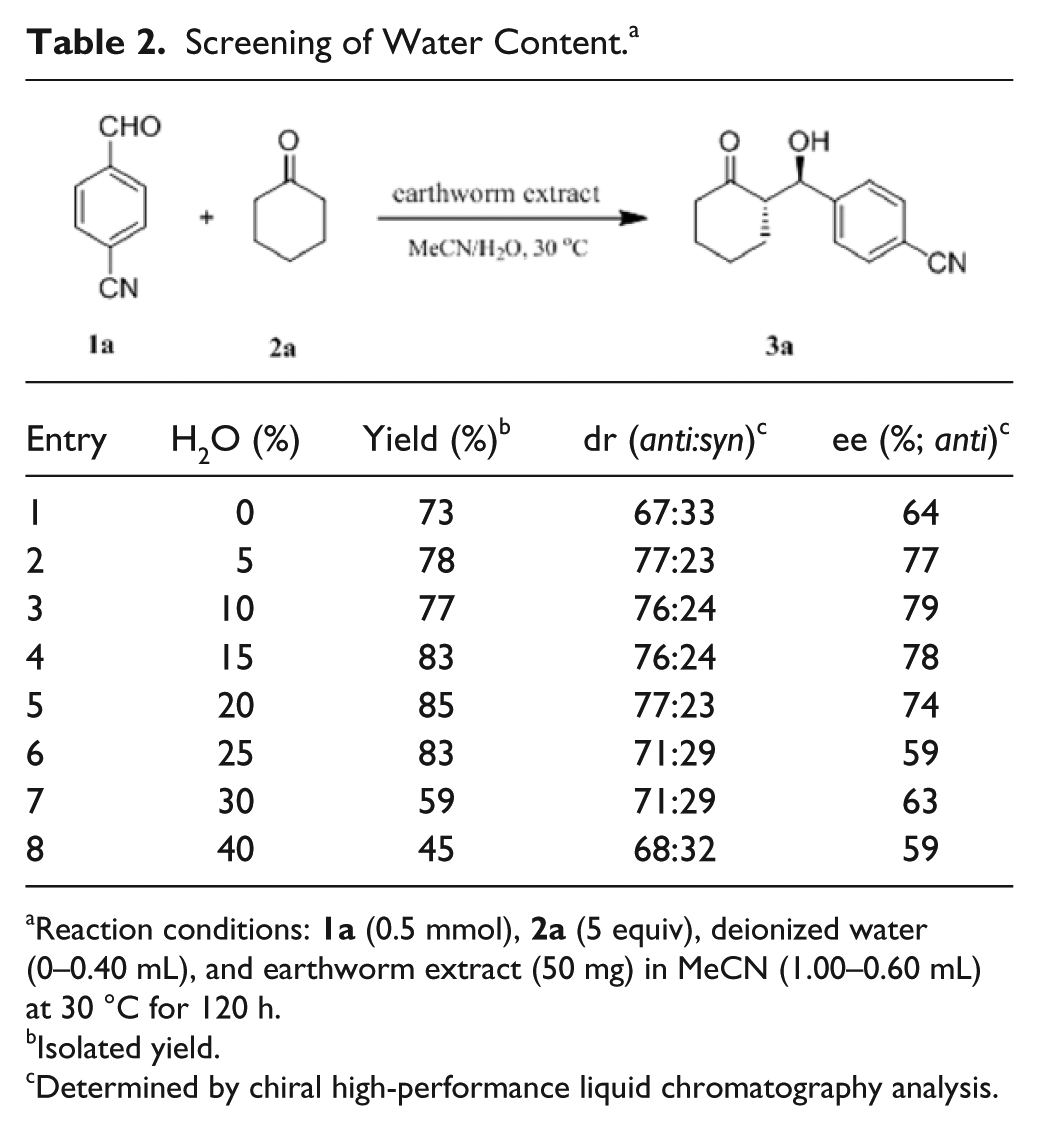
Reaction conditions:
Isolated yield.
Determined by chiral high-performance liquid chromatography analysis.
To further improve the stereoselectivity and yield of the model reaction, the molar ratio of the substrates was screened (
Table 3
, entries 1–6). When the molar ratio of 4-cyanobenzaldehyde (
Screening of Substrate Molar Ratio and Catalyst Loading. a
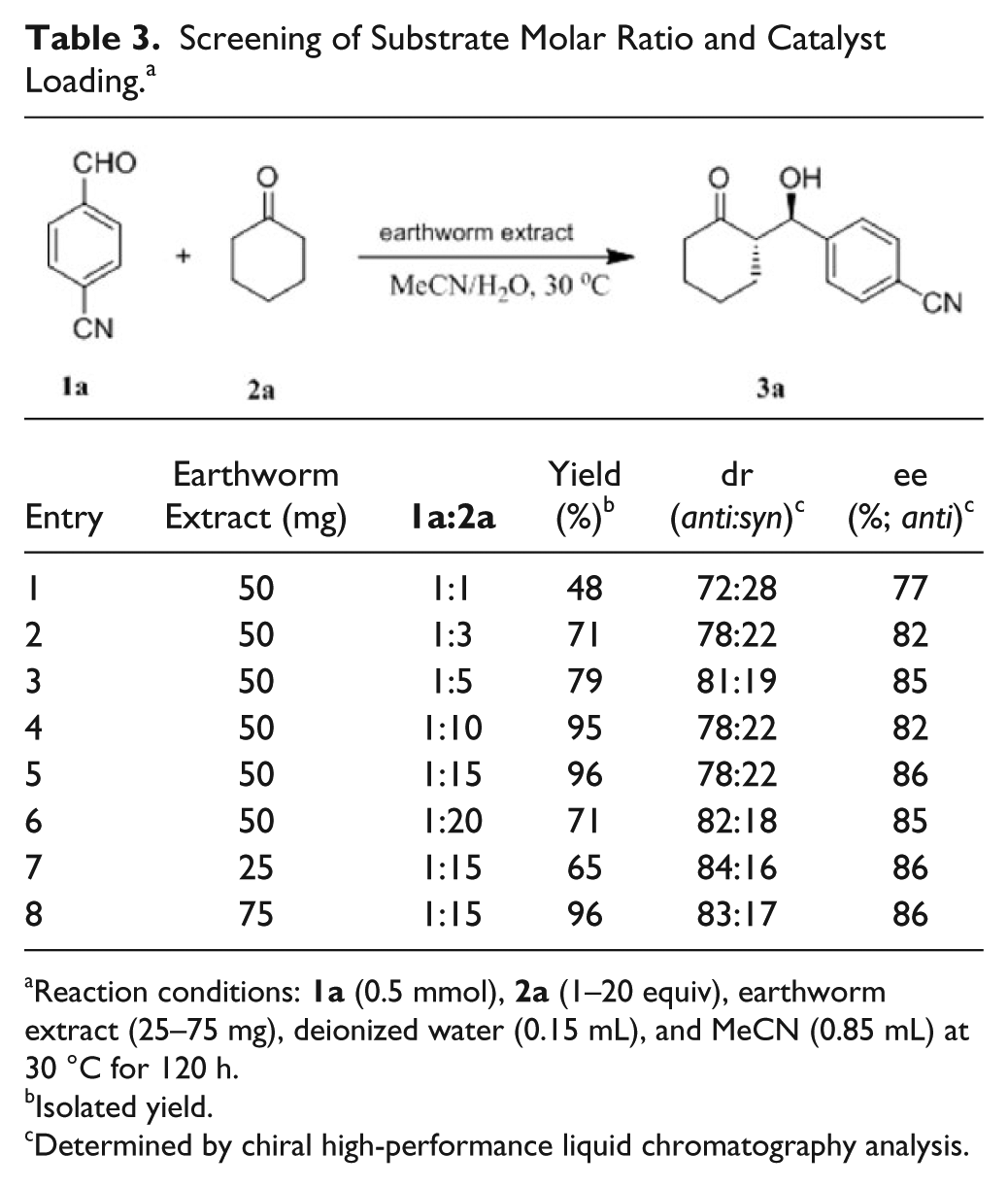
Reaction conditions:
Isolated yield.
Determined by chiral high-performance liquid chromatography analysis.
Subsequently, the influence of temperature on the earthworm extract catalyzed model aldol reaction was investigated (
Table 4
, entries 1–7). It was shown that the yield increased from 56% to 96% as the temperature rose from 15 °C to 30 °C (
Table 4
, entries 1–4). Meanwhile, increasing the temperature from 15 °C to 45 °C led to a decrease in both the diastereoselectivity (from 89:11 to 60:40 [
Influence of Temperature on the Model Aldol Reaction. a
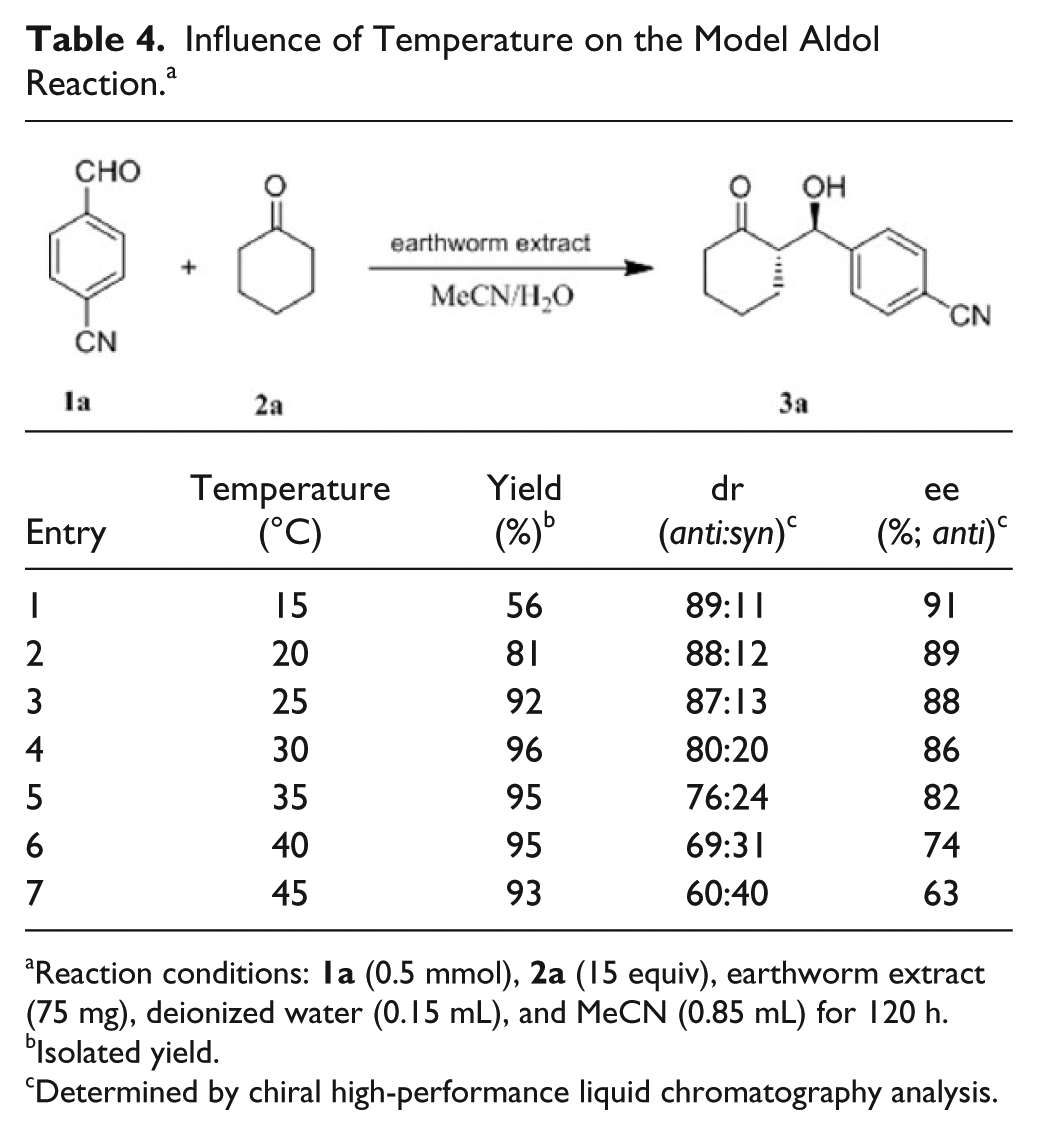
Reaction conditions:
Isolated yield.
Determined by chiral high-performance liquid chromatography analysis.
To investigate the generality and scope of this earthworm extract catalyzed direct asymmetric aldol reaction, several other substrates were tested (
Table 5
). The earthworm extract exhibited excellent catalytic activity and stereoselectivity toward aldol reaction, and satisfactory results were obtained with yields of up to 94%, dr of up to >99:1 (
Substrate Scope and Large-Scale Reaction. a
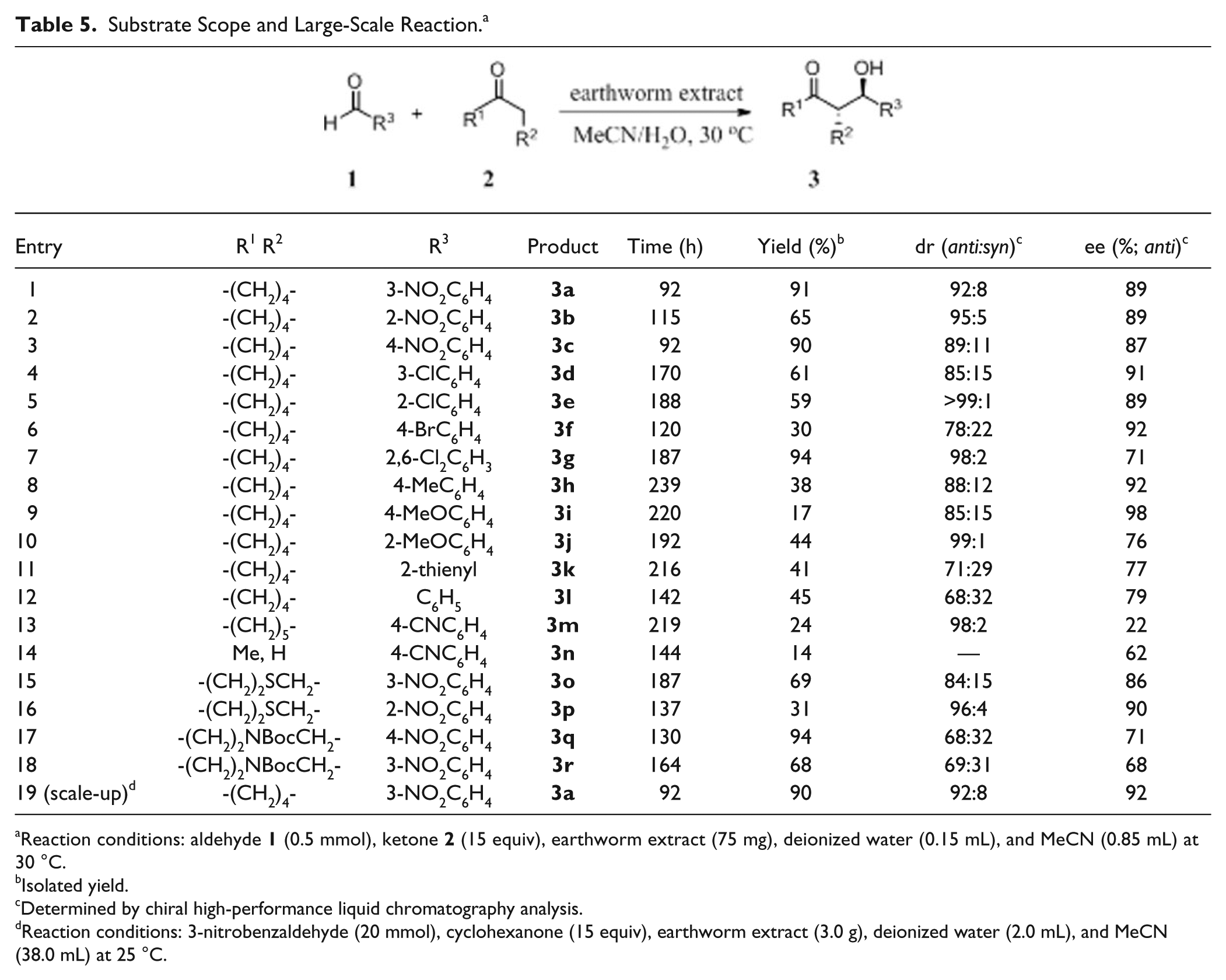
Reaction conditions: aldehyde
Isolated yield.
Determined by chiral high-performance liquid chromatography analysis.
Reaction conditions: 3-nitrobenzaldehyde (20 mmol), cyclohexanone (15 equiv), earthworm extract (3.0 g), deionized water (2.0 mL), and MeCN (38.0 mL) at 25 °C.
Finally, it is notable that this earthworm catalyst is also efficient in large-scale aldol reaction. For example, the reaction between 3-nitrobenzaldehyde and cyclohexanone could be easily performed on a large scale of 20 mmol, which gave an excellent yield of 90% with 92% ee and 92:8 dr (
In summary, a crude extract from the earthworm species
Footnotes
Acknowledgements
We acknowledge members of the Guan lab for their contribution to the extraction of earthworm enzymes.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (No. 21472152 and No. 21672174) and the Basic and Frontier Research Project of Chongqing (cstc2015jcyjBX0106).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
