Abstract
Neurotrophic assays are phenotypic methods to identify molecules that stimulate differentiation of neuronal cells. Bioactive small molecules with neurotrophic actions hold great promise as therapeutic agents for the treatment of neurodegenerative diseases and neuronal injuries by virtue of their ability to stimulate neuritic outgrowth. A combined in vitro method, which measures neurotrophic activity and cytotoxicity in a single assay, has been described. This assay, performed in 96-well microplates with PC12 and Neuroscreen-1 (NS-1; a subclone of PC12) cells, is a simple tool for identification of new neurotrophic agents. Stimulation of neurite outgrowth was measured with NIS software by analysis of digital cell images as multiple parameters, namely, mean neurite length, neurite length/cell, nodes/cell, and number of neurites/cell. The assay has been standardized and validated with dose-response analysis for nerve growth factor (NGF) and mechanism-based inhibitors of NGF-induced neurite outgrowth, namely, SU6656 (an Src family kinase inhibitor) and PD98059 (a MEK inhibitor). The assay has been successfully applied for screening natural and synthetic compound libraries for cytotoxicity and neurotrophic activity. Screening of a set of harmala alkaloids identified harmine as a potential neurotrophic molecule that significantly stimulated NGF-induced neurite outgrowth in the NS-1 cells. Important advantages of this method are its simplicity and determination of cytotoxicity and neurotrophic activity in a single assay. This assay may be suitable for primary and cultured neuronal cells.
Keywords
Introduction
Neurite outgrowth is an intricate process that involves three stages, namely, sprouting (the initiation of neurite formation), elongation of axon, and branching of dendrites followed by formation of synapses. 1 Neuritogenesis is a fundamental cellular differentiation process for formation of new neurons. Understanding this process is important, because it is required for proper wiring of the brain and nerve regeneration and has been associated with various neurodegenerative diseases. 2 Reports indicate significance of neurite outgrowth in the process of neuroregeneration and repair of damaged neurons. Neurotrophic substances that promote neuritic outgrowth can repair damaged neurons and may play a significant role in restoration and readjustment of normal neuronal functions of the damaged neurons. 3 Neurite outgrowth in cultured neurons is a good indicator of the neuroregenerative potential of a particular compound tested. In the field of drug development and toxicology, in vitro assays are used to evaluate the effects of compounds on neurite outgrowth and cell survival in primary and transformed neurons.4,5 There are various cell models to study neurite outgrowth. These includes primary neuron culture such as cortical neurons, cerebellar granule neurons and cell lines such as PC12 cells, and neuroblastoma cells such as NG108-15 as SH-SY5Y.6,7 Assays measuring neurite outgrowth have critical application for screening of new compounds that stimulate, complement, and inhibit neuritogenesis or neurite outgrowth. These assays also have important utility for understanding the cellular and molecular mechanisms for repair of axons and also assist to build up knowledge to overcome repair limitations. 4 There are many traditional manual, semiautomated, and automated assays/methods to measure the neurite outgrowth in various models. Traditional quantitative methods involve manual analysis of individual images to measure the neurite outgrowth. The utility of traditional methods for screening of chemical libraries is reduced as the methods are labor intensive, time-consuming, and error prone. However, manual methods are still being used in research laboratories owing to the relatively low cost of reagents and analytical instruments and ease of implementation.8,9 Improved or advanced automated neurite outgrowth assays using an algorithm particularly intended for measuring various aspects of neurite outgrowth have been developed.4,10 These methods have high throughput as compared with manual methods. To meet the increasing demand for robust high-throughput systems, several commercial instruments have also been developed for high-resolution imaging and fast processing. Despite their availability, these instruments are highly expensive owing to the cost of operation, acquisition, and maintenance. 11 To address the use of simple inexpensive methods that are easy to implement and that could be affordable in any lab, we have developed a 96-well plate format assay for measuring cell viability (cytotoxicity) and neuritogenesis in the same plate with a single assay protocol. This method uses an Alamar Blue (resazurin) reduction assay for cytotoxicity. Resazurin is soluble in physiologically buffered formulations. It is a simple method due to single-step direct homogenous addition of the Alamar Blue to growing cultures. The cell viability is measured by reduction of resazurin to resorufin. 12 The resazurin-based cell viability methods have advantages such as rapidity, reliability, sensitivity, cost, and safety. 13 The same culture after cell viability assay may be processed further for neurite growth analysis. The digital microscopic images are captured under bright field, followed by tracing of the neurites with a computer mouse using the NIS element software. From the tracing of neurites, different parameters for neurite outgrowth can be measured. The data obtained may be directly exported to the MS Excel format and added to the macro-based MS Excel template designed to measure neurite outgrowth multiparameters. Here, we show a comparison of nerve growth factor (NGF) dose-response analysis in PC12 and Neuroscreen-1 (NS-1; a subclone of PC12) cells to optimize the assay for screening of more compounds with a wide range of concentrations. This assay can easily be used for analyzing various concentrations of compounds for the potential to cause cytotoxicity, the potential to induce or inhibit neurite outgrowth, and for the mechanism of neurotrophic action. This method has been successfully compared and measured with NGF, an indigenous neurotrophic agonist, for dose-response in both the PC12 and NS-1 cell line. This assay, which can also be performed with other adherent primary as well as cultured neuronal cell lines, has been applied from screening different sets of natural and synthetic low-molecular compounds for potential neurotrophic activity.
Materials and Methods
Reagents and Chemicals
Culture medium (RPMI 1640), horse serum, fetal bovine serum, L-glutamine, penicillin 100 U/mL, and streptomycin 100 µg/mL were obtained from Hyclone (Logan, UT). NGF, collagen IV–coated 96-well plates were purchased from BD Biosciences (San Jose, CA). DMSO, Geimsa staining solution, and phosphate-buffered saline (PBS) obtained from Sigma Aldrich (St Louis, MO). Alamar Blue was purchased from Thermo Scientific (Waltham, MA).
Cell Cultures
NS-1 cells were procured from Cellomics (Pittsburg, PA), and PC12 (CRL-1721) cells were purchased from the ATCC (Manassas, VA). PC12 cells were cultured in F12 medium or RPMI 1640 medium containing 10% equine serum, 5% fetal bovine serum, 100 U/mL penicillin, and 100 µg/mL streptomycin and incubated at 37 °C in a 5% CO2 and 95% humidified incubator. PC12 cells were subcultured every 2 to 3 d at 70% to 80% confluence of cells. NS-1 is a neuronal-like cell line optimized for screening. It is a subclone of PC12, a widely used cell line derived from rat pheochromocytoma cells that serves as a standard model system for neurons. NS-1 cells display several substantial advantages over PC12 cells, including faster growth, no aggregation, and better responsiveness to NGF. NS-1 cells were cultured in RPMI 1640 medium containing 10% horse serum, 5% fetal bovine serum, 2 mM L-glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin and incubated at 37 °C in a 5% CO2 and 95% humidified incubator. The NS-1 cells were also subcultured after 2 to 3 d or twice a week for maintaining culture.
Cell Treatments and Differentiation
NS-1 cells and PC12 cells were seeded, 2000 cells/well and 3000 cells/well, respectively, onto a clear flat-bottom 96-well collagen IV–coated plate. Cells were allowed to adhere for 24 h in an incubator having 37 °C, 5% CO2, and 95% humidity. After 24 h, medium was replaced with fresh culture medium with and without different concentrations of NGF. For evaluation of test compounds for neurotrophic activity and cytotoxicity, the test compounds were added alone or with NGF (the concentrations as stated in the Results section). Each concentration of the test compound and NGF was tested in triplicate. NS-1 cells and PC12 cells, after treatment with NGF and/or the test compounds, were incubated further for 96 h and 6 d, respectively.
Cell Viability/Cytotoxicity Assay
Cell viability for determination of cytotoxicity was determined using the Alamar Blue colorimetric quantitative method. This method uses a resazurin reduction assay.12,14 In physiologically active cells, resazurin is reduced to resorufin, giving a bright red fluorescence. After 72 h for the NS-1 cell cultures and after 5 d of incubation for the PC12 cells, 10 µL of the Alamar Blue reagent was added directly to the media into each well. Plates were further incubated overnight. The fluorescence was measured at 544 nm excitation and 590 nm emission, and raw data were exported to a MS Excel–based macro and analyzed to determine viability/cytotoxicity (IC50 values) employing XLFit.
Image Capture and Image Analysis for Neurite Outgrowth
After determination of viability for cytotoxicity, the same assay plates were processed further for analysis of neuritic outgrowth. The media were discarded. The adherent cells were washed with PBS and fixed with methanol. The plates were allowed to dry, and cells were stained with Geimsa stain (1:40 dilution) for 45 min. After staining, the plates were washed twice with distilled water, air dried, and then observed under a 10× objective lens of an inverted microscope under bright light. The digital images of the cells treated with compounds with/without NGF were captured with a Nikon digital camera using NIS element software and stored as JPEG files. Multiple images covering at least five different fields of view in each well were captured. These images were further analyzed with NIS element software for neurite outgrowth. The publically available NIH Image-J software (Neuron J plugin) 15 can also be used. For each image captured, 50 cells per/field were analyzed by counting the total number of neurites. Neurite lengths were measured by tracing along the full length of individual neurites, starting from the cell body ( Fig. 1 ). The neurotrophic action was assessed according to the following parameters: 1) mean neurite length (sum of the length of total neurites measured/number of neurites measured), 2) neurite length/cell (sum of the length of total neurites measured/number of cells counted), 3) neurites/cell (total number of neurites/number of cells counted), and 4) neurite nodes/cell, indicative of neurite branching (total numbers of neurite nodes or branches/total number of neurites).

Measurement of neurite outgrowth by analysis of digital cell images with NIS element software. (
Analysis of Data for Cytotoxicity and Neurite Outgrowth
Results for each set of assays were obtained at least in triplicate with three independent experiments. The data obtained were exported into MS Excel and added to the macro-based MS Excel template designed to measure neurite outgrowth multiparameters. The final results are expressed as mean ± SD of different experiments. The results of the cytotoxicity assay were analyzed by XLFit for determination of IC50 values.
Results
Comparison of Dose Response of NGF in Terms of Cell Viability and Neurite Outgrowth in PC12 and NS-1 Cells
Figure 1
shows representative digital images of control (
Fig. 1A
) and NGF-treated (
Fig. 1B
) NS-1 cells.
Figure 1B
also shows the approach for tracing the neurites and measurement of length and number of neurites. An area blown to show neurites and neurite nodes is presented as
Figure 1A
. PC12 cells and NS-1 cells were incubated with different concentrations of NGF for 96 h to determine the viability of cells using the Alamar Blue assay. The cytotoxicity results are expressed in viability percentage (
Suppl. Fig. S1A
[PC12 cells] and
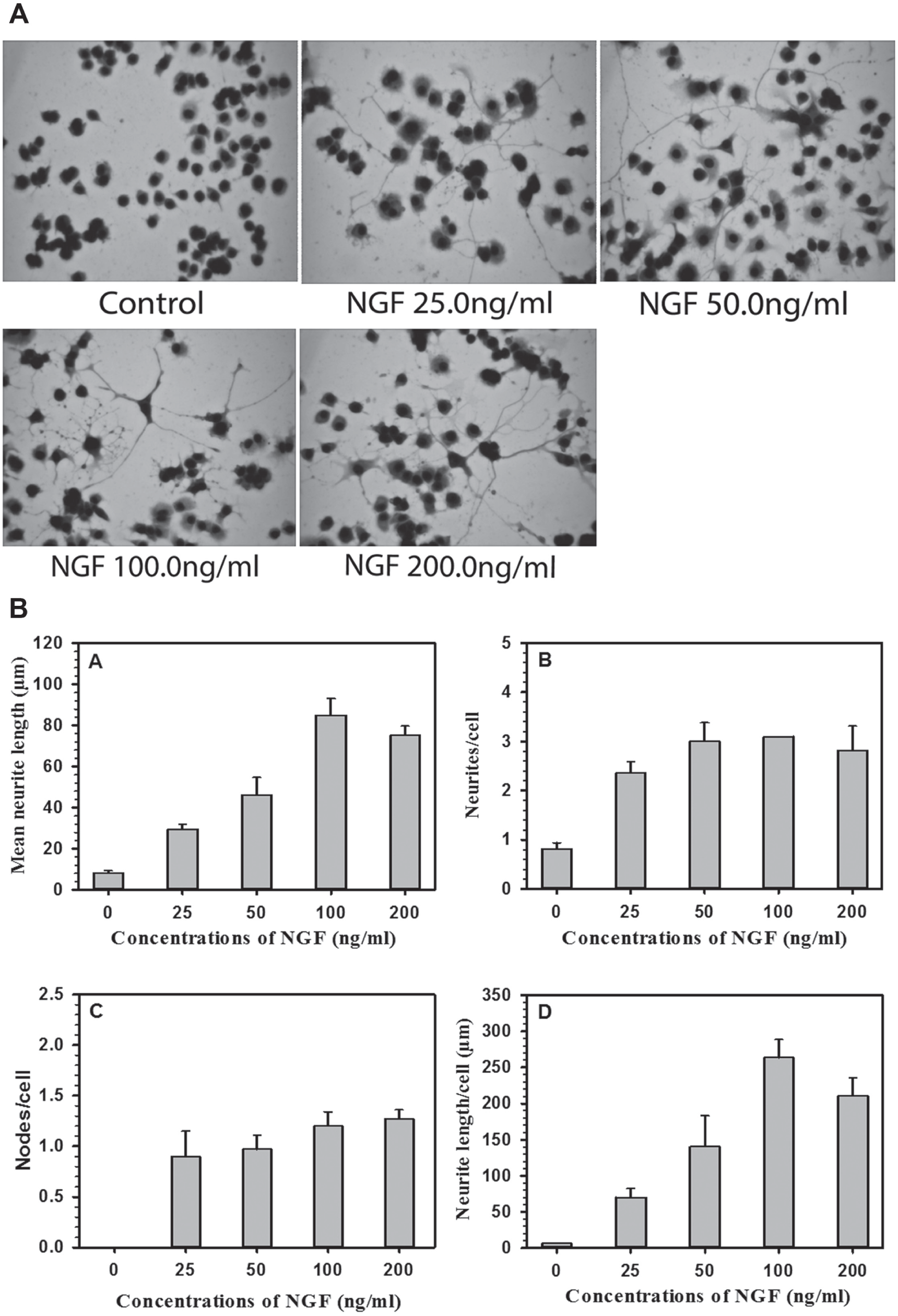
(
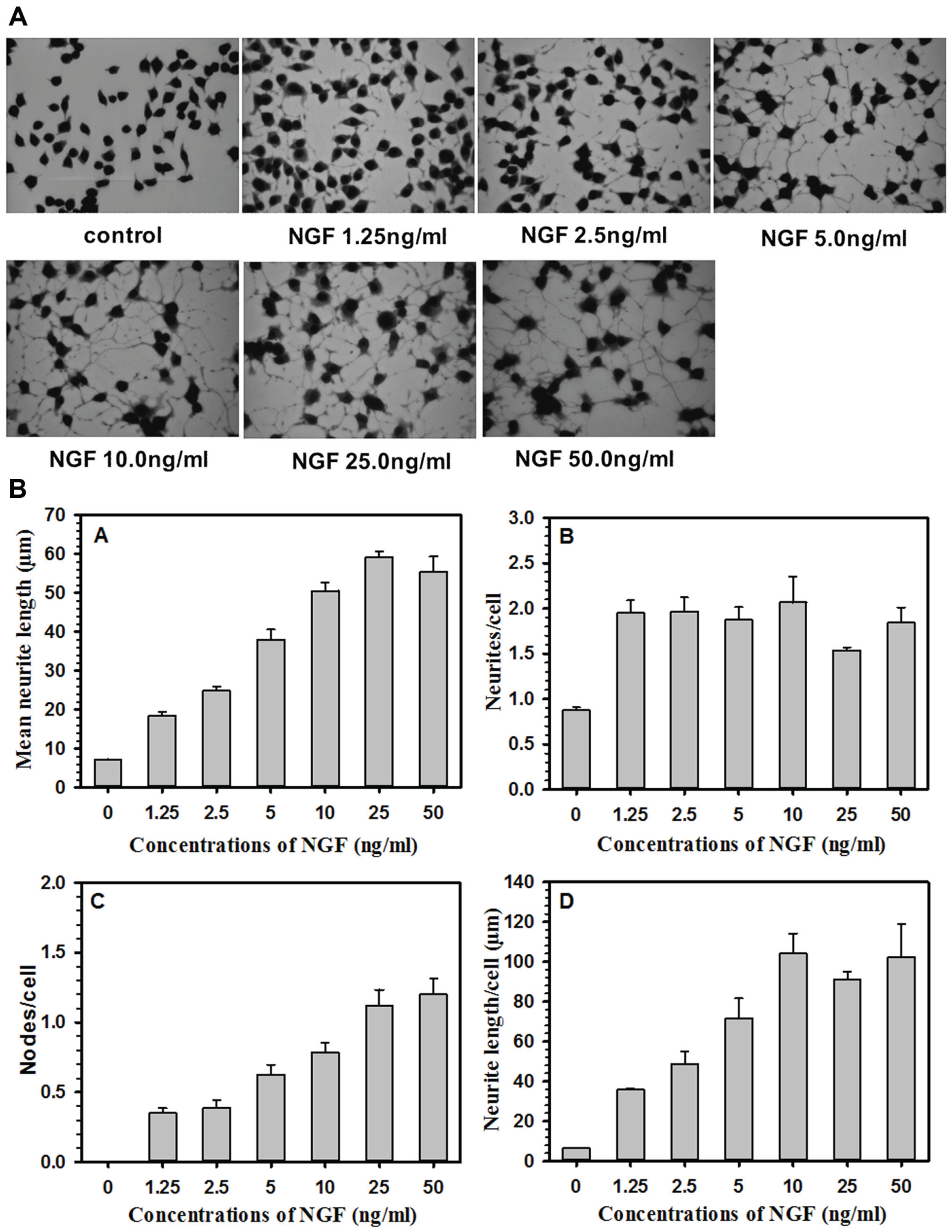
(
Validation of the Assay with Mechanism-Based Inhibitors
The cytotoxicity and neurite outgrowth assay was further validated with the mechanism-based inhibitors of NGF-induced neurite outgrowth, namely, SU6656 (an Src family kinase inhibitor) and PD98059 (a MEK inhibitor). These inhibitors antagonize neurotropic action of NGF through attenuation of the activation of neurotrophic signal transduction functions. Both inhibitors produced concentration-dependent inhibition of neurotrophic action of NGF on NS-1 cells ( Suppl. Figs. S3, S4, and S7 ). Low concentrations (>10 µM) of SU6656 and PD98059 inhibited neurite outgrowth without any cytotoxic action. However, higher concentrations (100 µM) of these inhibitors were cytotoxic against NS-1 cells ( Suppl. Fig. S2 ). Further, the cytotoxicity assay was also validated with doxorubicin, a potent cytotoxic drug. Doxorubicin showed potent cytotoxic action against NS-1 cells ( Suppl. Fig. S2C )
Application of the Assay for Screening a Set of Harmala Alkaloids for Cytotoxicity and Neurotrophic Activity
The results presented in the previous section show that NS-1 cells respond better than parent PC12 cells to the neurotrophic action of NGF. This allows the use of low and suboptimal concentrations of NGF to determine potentiation or synergism of the neurotrophic action of NGF with the test compounds. The method has been successfully applied for screening different libraries of natural and synthetic compounds. To demonstrate the utility of this method, the results obtained with a set of harmala alkaloids are presented. The harmala alkaloids were selected for the evaluation of neutrophic action in view of their origin from the natural resources and spectrum of neuropharmacological actions. Figure 4 presents the structures of harmala alkaloids tested for neurotropic actions. The compounds were tested alone and also in combination with NGF. None of the harmala alkaloids, when tested alone and in combination with NGF, showed significant cytotoxicity up to 30 µM concentration, except harmine, which showed cytotoxicity only at 30 µM ( Fig. 5 ). Harmine was not toxic at 10 µM concentration. The neurotrophic action of the harmala alkaloids was monitored at 10 µM concentration alone and in combination with NGF (1.25 ng/mL; Fig. 6 ). None of the harmala alkaloids tested showed significant neurotrophic action alone. However, harmine produced significant potentiation of neurotrophic response of NGF on NS-1 cells. The harmine NGF co-treatment produced a significant increase in mean neurite length ( Fig. 6A ) as well as neurite length/cell ( Fig. 6D ) as compared with NGF treatment alone. No significant increase was noted in the numbers of neurites/cell ( Fig. 6B ) or neurite nodes/neurite ( Fig. 6C ).
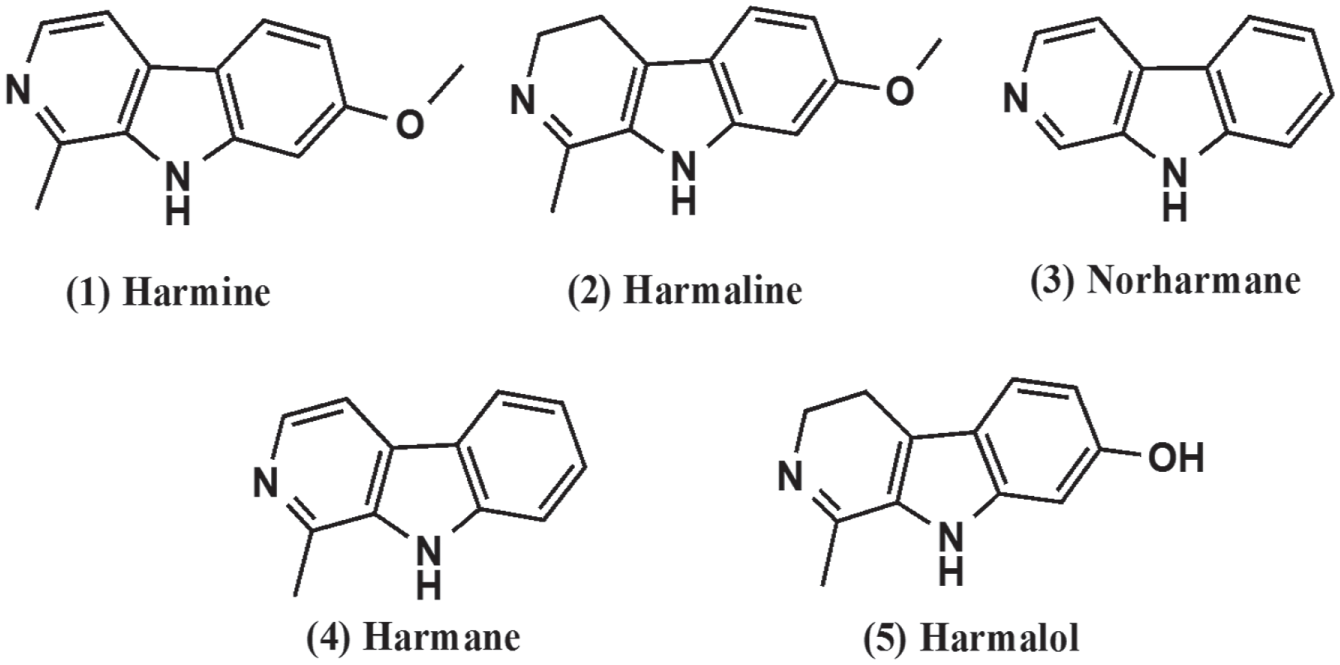
A set of harmala alkaloids tested for neutrophic activity and cytotoxicity on Neuroscreen-1 cells.
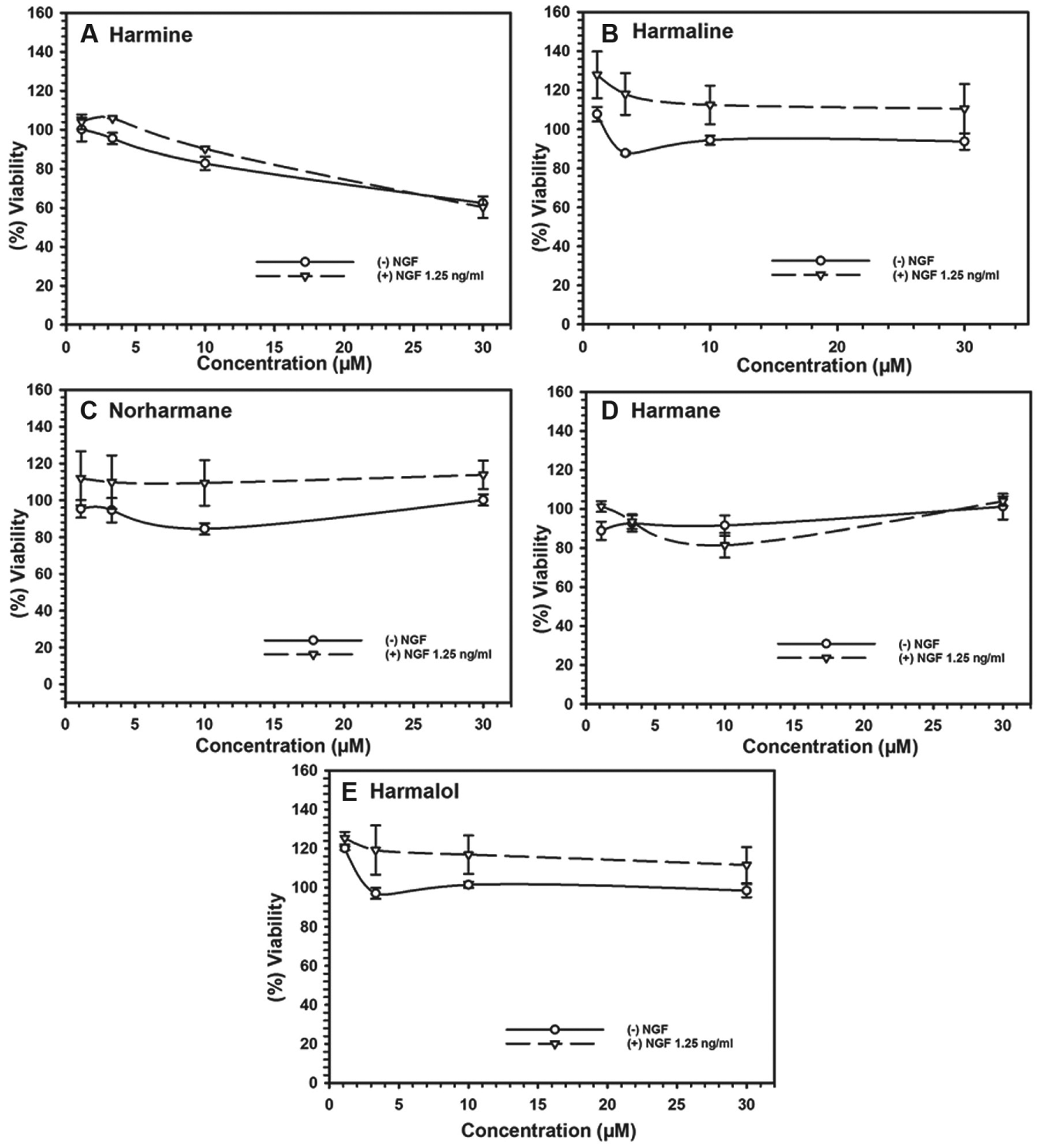
Cytotoxicity of harmala alkaloids against Neuroscreen-1 cells alone and in combination with nerve growth factor. Each point represents the mean ± SD of three observations.
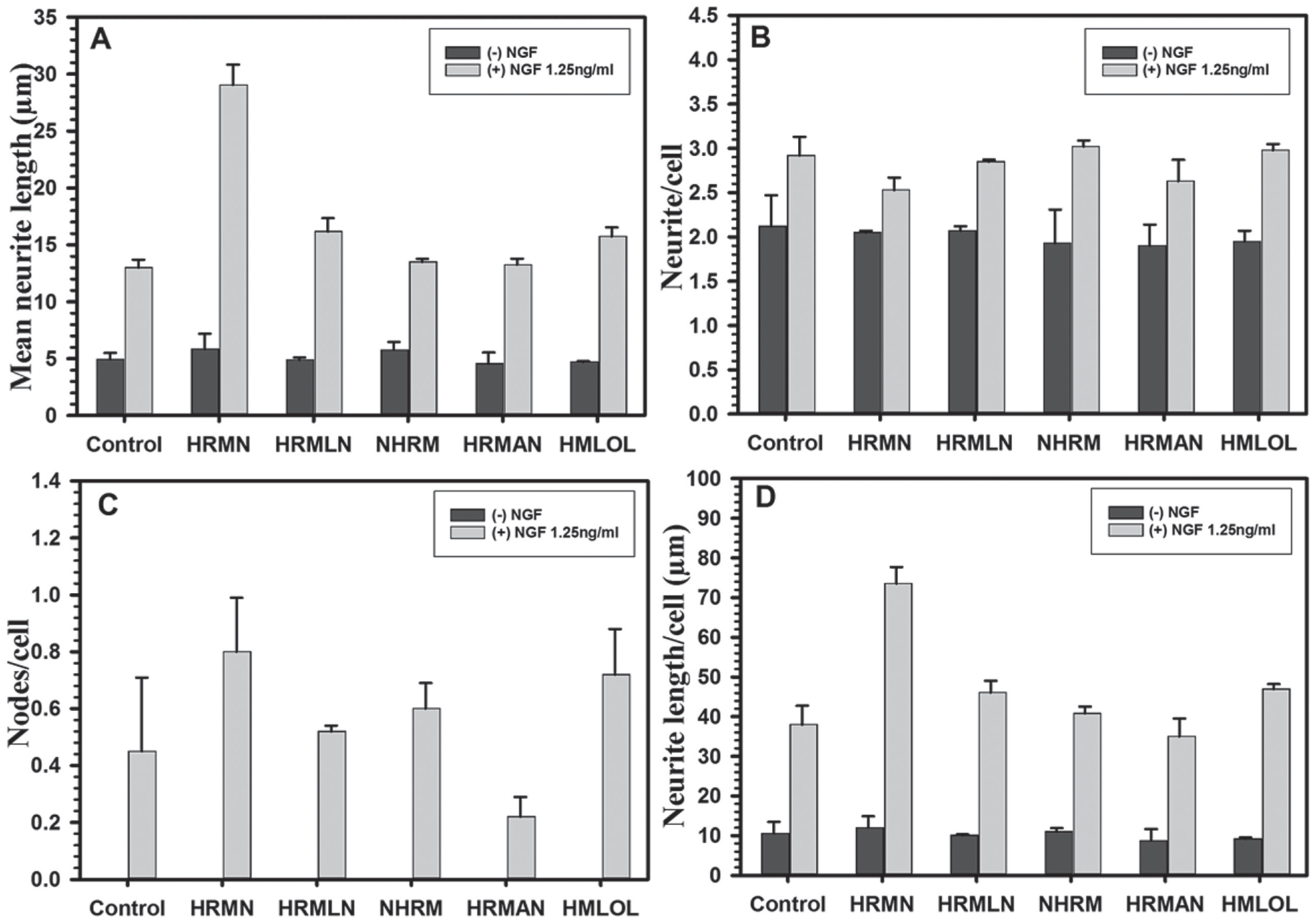
Neurotrophic activity of harmala alkaloids (10 µM) on Neuroscreen-1 cells alone and in combination with nerve growth factor (1.25 ng/mL). Each bar shows the mean ± SD of three observations.
Discussion
Neurotrophins such as NGF, brain-derived neurotrophic factor (BDNF), neurotrophin-3 (NT-3), and Neurotrophin-4/5 (NT-4) are intrinsic neurotrophic factors produced by the neuronal tissues as a part of normal neuronal development and induce neuronal growth, neuroregeneration, and repair of nerve injuries. 16 Recent studies have demonstrated roles of neurotrophins in regulation of neurogenesis in adult brain. 17
In this study, we evaluated a combined in vitro assay that measures and quantifies neurite outgrowth along with the cell viability (a measure for cytotoxicity) in a single assay protocol. This is a simple method, and it does not require costly reagents to measure the neurites. The assay carried out in a 96-well microplate format is suitable for screening of compounds over a wide concentration range. The main advantage of this method is simplicity and cost-effectiveness. This method does not require expensive automated systems, more expensive fluorescent dyes, or sophisticated software required for neurite outgrowth analysis. It can be done in any lab equipped with a simple light microscope attached with a digital image–capturing device. We have validated this method of neurite outgrowth analysis by comparing the dose response of NGF in PC12 and NS-1 cell lines and simultaneously performing the viability/cytotoxicity assay.
PC12 cells are widely used as a model system for the study of neurite outgrowth and for understanding cellular and molecular mechanisms for neuronal differentiation.18–20 Previously reported data indicated that the NS-1 cell line, a subclone of PC12 cells, retains many of properties of the parent cell line that make it a valuable model system for the study of neurite outgrowth and differentiation.5,21 We found a dose-dependent increase in neurite outgrowth with NGF in NS-1 cells that is similar to that of PC12 cells. Our results also suggest that PC12 cells require >5 d (>120 h) of exposure to NGF treatment to induce optimum neurite outgrowth, whereas NS-1 cells require only 72 to 96 h for the maximum response. NS-1 cells showed greater response to NGF treatment as compared with parental PC12 cells. The NGF treatment was able to induce neurite outgrowth in NS-1 cells at doses as low as 1.25 ng/mL. To quantify neurite outgrowth in NS-1 and PC-12 cells, four parameters were measured using the NIS element software, namely, mean neurite length, neurite length/cell, neurites/cell, and nodes/neurites. The NS-1 cells show better threshold for determination of neurotrophic activity of the compounds because of their superior sensitivity for NGF-stimulated neurite growth compared with PC12 cells. For evaluation of the compounds in combination with NGF, the threshold of the assay can be adjusted with an appropriate concentration of NGF. For positive neurotrophic activity, suboptimal concentrations of NGF may be used. This allows for further potentiation of neurite outgrowth. For negative neurotrophic action (inhibition of neurite outgrowth), higher concentrations of NGF may be employed. This allows evaluation of inhibition or attenuation of NGF-stimulated neurite outgrowth.
There are several automated cell-based screening methods, which are known as high-content screening. High-content screening combines automated technology of image acquisition, image analysis, and data collection with fluorescent microscopy.22,23 This approach of analysis is mainly used by pharmaceutical industry labs for the high-throughput drug discovery. 7 These methods are fast and can analyze multiple parameters. However, owing to their cost, these are not affordable in low-cost-setting labs. Our method is cost-effective and eliminates the need for an additional assay for measuring cytotoxicity by combining both viability analysis and neurite outgrowth.
The assay has been successfully used for screening sets of synthetic as well as natural compounds. Screening a set of harmala alkaloids identified the potential neurotrophic property of harmine in combination with NGF. Earlier studies have reported significant neuropharmacological properties of harmine, namely, inhibition of human monoamine oxidase-A, 24 up-regulation of expression of glutamate transporter in astroglia, 25 enhancement of short-term memory in rats, 26 inhibition of dual-specificity tyrosine phosphorylation–regulated kinase 1A (DYRK1A), 27 in vivo improvement of functional impairment and neuronal death following traumatic brain injury in rats, 28 and neuroprotection and attenuation of cerebral infarct due to global cerebral ischemia in rats. 29 Potentiation of NGF-stimulated neurotrophic action of serotonin, as reported earlier, was also confirmed with the current assay. This assay showed that serotonin at high concentration (100 µM) shows some cytotoxicity against the NS-1 cells ( Suppl. Fig. S2B, S5, and S6 ) The assay was further validated with mechanism-based inhibitors of neurotrophin signal transduction pathways. SU6656, an inhibitor of the Src-family kinase, showed concentration-dependent inhibition of the neutrophic action of NGF, which was comparable with the observations reported earlier with a high-throughput screening assay employing cells expressing green fluorescent protein or native cells stained with HCS Cell Mask Red. 11 Similarly, PD98059, which attenuates NGF-linked Trk-A activation, also showed concentration-dependent inhibition of NGF-stimulated outgrowth. This assay, which may be performed with primary and cultured neuronal cells, will have important utility for in vitro screening and identification of small-molecule neurotropic agents.
Footnotes
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies are partly supported by the National Institute of General Medical Sciences (NIGMS), a component of the National Institutes of Health (NIH; grant P20GM104931) and U.S. Department of Agriculture–Agricultural Research Service (USDA-ARS) specific cooperative research agreement No. 58-6408-2-0009. The contents are solely the responsibility of the authors and do not necessarily represent the official view of the NIGMS, NIH, or the USDA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
