Abstract
Objective
There is a lack of established biological, psychological, social, and digital markers for the prediction, identification, and stratification of patients with major depressive disorder (MDD). We therefore aimed to evaluate serum nerve growth factor (NGF) in MDD patients.
Methods
In this case–control study, we recruited MDD patients and age- and sex-matched healthy controls (HCs). A qualified psychiatrist evaluated study participants according to the Diagnostic and Statistical Manual of Mental Disorders, fifth edition. Serum NGF levels were analyzed using a commercially available enzyme-linked immunosorbent assay kit.
Results
We analyzed data from 106 MDD patients and 88 HCs. Mean serum NGF concentrations were significantly higher in MDD patients (104.70 ± 6.43 pg/mL) than in HCs (72.09 ± 7.69 pg/mL). Receiver operating characteristic curve analysis revealed the good diagnostic performance of serum NGF in MDD.
Conclusions
Higher serum NGF levels might be involved in MDD pathophysiology, and altered NGF levels may be an early warning sign of depression. The present findings will aid in the development of new and improved therapies for depressive patients. Further interventional studies are recommended to explore the underlying mechanisms, risk factors, disease course, and treatment responses of NGF in MDD.
Introduction
Depression is a clinically important and rapidly expanding public health problem. Major depressive disorder (MDD) is a debilitating disease associated with sadness and irritability that disrupts the day-to-day lives of those affected. MDD is predicted to be the major cause of global health problems by 2030 because of its high prevalence. 1 Some common symptoms of MDD are a persistent depressed mood, anhedonia, feelings of guilt, frequent changes in appetite, low energy levels, a short attention span, agitation, difficulty sleeping, and suicidal thoughts. Major depression is a mental illness that is currently ranked fourth among global disabilities; it is estimated to become the second most common disease by 2030. 2 A population study revealed that women are twice as susceptible to depression as men. 3 Furthermore, MDD is associated with medical comorbidities as well as financial burdens. 4 Neurological diseases, such as Parkinson's disease or stroke, and the initial months following childbirth can both increase the likelihood of serious depression. 5 MDD is a complex issue that affects approximately 324 million people worldwide, and around one in every six adults during their lifetime. 6 The number of individuals experiencing depression has expanded by 18.4% between 2005 and 2015. 7 MDD is more common in older adults, especially in women (7.5%) and men (5.5%) between 55 and 74 years old. 8 A comprehensive study of mental health conditions in Bangladesh revealed a prevalence of mental health issues of 6.5% to 31% in adults and 13.4% to 22.9% in adolescents. 9 Moreover, depressive symptoms are reportedly present in 29.7% of the rural population in Bangladesh. Over the last 25 years, the prevalence of MDD has risen in adolescents and adults in the United States. 10 In 2012 and 2013, more than 10% of US individuals had experienced MDD in the previous 12 months, and more than 20% had experienced MDD during their lifetime. 10 The current prevalence estimates of MDD in China are reported to range from 1% to 6.5%, but 8.3% of those surveyed were diagnosed with the disorder. 11 Healthcare professionals thus encounter depression as a common mental issue in their routine clinical practice; it is currently a prevailing cause of disability worldwide. 12
The etiology of MDD is very complex and includes physiological, hereditary, ecological, and psychological components. Growth hormones and thyroid imbalances are also involved. Past mental trauma can also trigger depression later in life.13,14 The pathophysiology of MDD includes alterations in neurotrophic factors, which play an important role in the disease; 15 neurotrophic factors such as growth factors can also influence MDD. A growth factor is a physiological substance that influences cell growth. Growth factors send signals to intracellular components to alter gene expression by acting on specific cell surface receptors. 16 Nerve growth factor (NGF) is a neurotrophic factor that regulates the growth, production, maintenance, and survival of specific neurons. Preclinical and clinical depression models have indicated that antidepressants may control depression through NGF. 17 Changes in growth factor levels have also been linked to the clinical signs and pathogenesis of many mental conditions, including MDD, bipolar disorder, schizophrenia, and anxiety. 18 Together, these findings suggest that NGF might act as marker for mental health problems such as MDD or mood disorders.
The diagnosis of depression is challenging because its neurobiological causes are not yet well understood. Several past studies have indicated that monoamine insufficiency, neurotrophic abnormalities, dysfunctional hypothalamic–pituitary–adrenal axis activity, and inflammatory changes are linked to MDD;19–23 however, a complete understanding of the pathophysiology of MDD is yet to be established.24,25 Furthermore, the current diagnostic criteria for depression are flawed because they mainly rely on subjective measures. Importantly, the heterogeneity of individuals with MDD impacts the development of effective interventions. This study therefore aimed to support the validation of a biological marker of depression by evaluating serum NGF concentrations in MDD patients and healthy controls (HCs) in Bangladesh. It is hoped that the findings of this study will improve the analysis, diagnosis, and management of MDD patients.
Materials and methods
Ethics approval
The study protocol was approved by the Research Ethics Committee, University of Asia Pacific (Ref: UAP/REC/2022/101). The study was performed in accordance with the Declaration of Helsinki. We briefed each participant about the objective and purpose of this study and obtained their written informed consent before participation. In cases where an individual’s intellectual capacity was suspected to be impaired, consent was obtained from their legal guardians. Moreover, all patient details were de-identified so that they are unable to be identified in any way.
Study population
This case–control study involved MDD patients and sex- and age-matched HCs. Patients were from a tertiary care teaching hospital in Dhaka, Bangladesh, and HCs were selected from various parts of Dhaka city. Before beginning the trial, each participant was examined by a qualified psychiatrist. The screening steps included a psychiatric interview; a standardized diagnostic session based on the Diagnostic and Statistical Manual of Mental Disorders, fifth edition (DSM-5); a depression intensity evaluation using the Hamilton Rating Scale for Depression (HAM-D); an assessment of past psychiatric problems; and the collection of social demographic information using a pre-configured inquiry form. In the present study, all MDD patients had been experiencing depressive symptoms for at least 2 weeks and scored more than seven on the HAM-D. Exclusion criteria were as follows: alcohol or illegal drug abuse or substance misuse in the previous 6 months, kidney or inflammatory diseases, heart disease, or acute or chronic infection. Because antidepressant and antipsychotic medications can affect the blood levels of many substances, all participants were asked to stop using them for at least 1 week before the test.
Blood sample collection, serum separation, and storage
A syringe was used to draw 5 mL of blood from the cephalic vein of each MDD patient and HC in the study. Blood samples were collected between 8.00 a.m. and 9.00 a.m. after overnight fasting. The blood was then dispensed into a Falcon tube and left to clot for an hour at 25°C. The clotted blood was then centrifuged at 1000 × g for 15 minutes to separate the blood cells and serum. Next, the serum was isolated from the cells and collected in an Eppendorf tube before being frozen at −80°C until further analysis.
Serum NGF quantification
Serum NGF concentrations were determined using a commercially available enzyme-linked immunosorbent assay kit (catalog number: EZ0469; Boster Bio, Pleasanton, CA, USA). All reagents and materials were equilibrated at 18°C to 25°C before the experiment. Samples, plates, working standards, and reagents were prepared according to the manufacturer’s instructions. First, 100 µL of sample, standard, or control was added to each appropriate well and the plate was carefully covered and incubated for 120 minutes at 25°C. The liquid was then discarded before 100 µL of biotinylated goat anti-human NGF polyclonal antibody was added to each well. The plate was covered and incubated for 90 minutes at 25°C before being washed three times with phosphate-buffered saline. Next, 100 µL of avidin–biotin–peroxidase complex was added to each well and incubated for 40 minutes at room temperature. After five washes with phosphate-buffered saline, each well was incubated in the dark at room temperature for 30 minutes with 90 µL of color-developing reagent. Finally, 100 µL of stop solution was added to each well; absorbance was measured at 450 nm using a microplate reader within 30 minutes of stopping the reaction.
Statistical analysis
Data were processed and examined using IBM SPSS Statistics for Windows, version 25.0 (IBM Corp., Armonk, NY, USA). Independent sample t-tests were applied for descriptive statistical analyses. All data are presented as the mean ± standard error of the mean, or as the frequency and percentage. Box and scatter plots were used to visualize the data. Additionally, receiver operating characteristic (ROC) curve analysis was performed to evaluate the predictive performance of serum NGF in depression. A value of p < 0.05 was considered significant. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 26
Results
Socio-demographic profiles of the study population
In the present study, 106 patients with MDD and 88 HCs were recruited. The socio-demographic profiles of participants are shown in Table 1. The different parameters considered in this study were level of education, occupation, economic impression, marital status, area of residence, and smoking history. These features were included to observe any associations between these factors and MDD severity. Of the MDD patients, 64.15% were women and 35.84% were men; 59.43% were married and 40.57% were unmarried. Additionally, 61.32% of MDD patients were in the normal body mass index (BMI) range. Furthermore, economic status was considered high in 4.71% of patients, medium in 19.81% of patients, and low in 75.47% of patients. The presented data indicated that people in the age range of 18 to 25 years were more prone to depression. Among the MDD patients, 55.66% had a previous history of MDD and 32.07% had a family history of MDD. The MDD patients and HCs were relatively similar with respect to age, sex, level of education, BMI, income, occupation, and smoking habits.
Socio-demographic profiles of study participants.
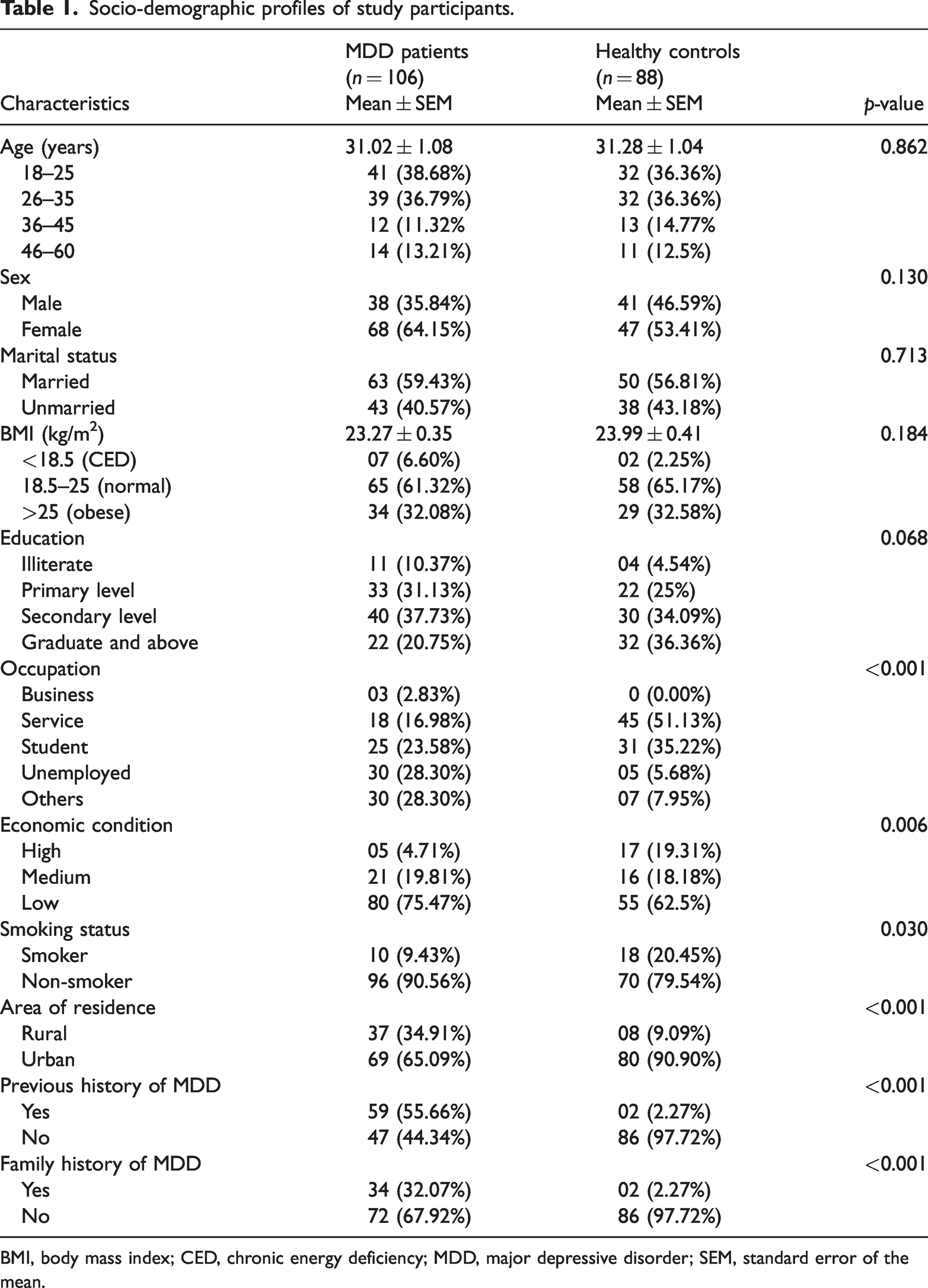
BMI, body mass index; CED, chronic energy deficiency; MDD, major depressive disorder; SEM, standard error of the mean.
Clinical outcomes and laboratory findings
Table 2 presents the clinical outcomes and laboratory findings of this study in terms of age, BMI, DSM-5 score, HAM-D score, and serum NGF concentration. There was no significant difference in the mean ages of MDD patients (31.02 ± 1.08 years) and HCs (31.28 ± 1.04 years). Among the MDD patients, 38 were men (mean age 29.68 ± 1.49 years) and 68 were women (mean age 31.76 ± 1.46 years). There was no significant difference in BMI between MDD patients (23.27 ± 0.35 kg/m2) and HCs (23.99 ± 0.41 kg/m2). Mean DSM-5 scores were 7.49 ± 0.11 in patients and 2.05 ± 0.15 in controls. Mean serum NGF concentrations were significantly higher in MDD patients (104.70 ± 6.43 pg/mL) than in HCs (72.09 ± 7.69 pg/mL; p = 0.001). There was a significant positive relationship between depression and serum NGF concentrations. In addition, in MDD patients, mean serum NGF concentrations were 108.77 ± 10.00 pg/mL in men and 102.43 ± 8.36 pg/mL in women. Figure 1 presents a box plot showing the distribution of serum NGF levels in MDD patients and HCs. A sex-specific scatter plot is presented in Figure 2. These graphs demonstrate the mean differences and associations of HAM-D scores with NGF levels in the study population.
Clinical features and laboratory outcomes of study participants.

BMI, body mass index; DSM-5, Diagnostic and Statistical Manual for Mental Disorders, fifth edition; HAM-D, 17-item Hamilton Rating Scale for Depression; MDD, major depressive disorder; NGF, nerve growth factor; P/C, patients/control; SEM, standard error of the mean.

Distribution of serum NGF levels in MDD patients and healthy controls. Box plot showing the median, maximum, and minimum values.

Sex-specific scatter plots showing the associations between and the mean differences in serum NGF and HAM-D scores of study participants.
ROC curve analysis
The ROC curve analysis of serum NGF is shown in Table 3. One important NGF parameter was the cut-off value, which was 71.32 pg/mL in this study. The sensitivity and specificity, which are two important parameters of ROC curve analysis, were also determined. The sensitivity was 68.4% and the specificity was 67.1%. Moreover, the positive and negative predictive values in this analysis were 82.5% and 81.4%, respectively. The area under the curve was 0.774, which is considered a good diagnostic value in statistical analysis, and the 95% confidence interval was 0.707 to 0.842 (p < 0.001). In Figure 3, the ROC curve for NGF is shown. The ROC curve illustrates the relationship between sensitivity and specificity at possible cut-offs. This figure indicates that the ROC curve has a good diagnostic value.
Receiver operating characteristic curve analysis of serum NGF.
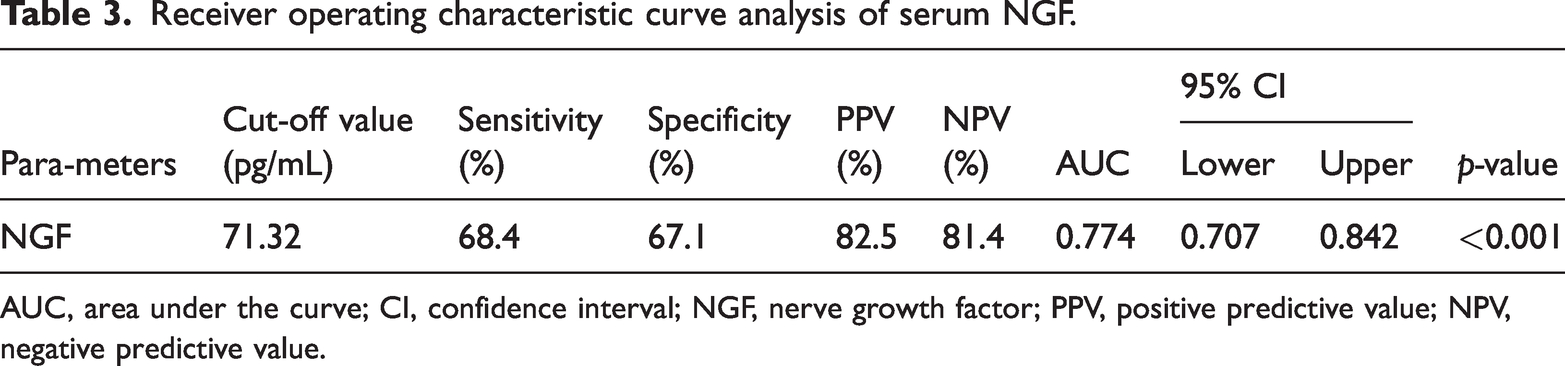
AUC, area under the curve; CI, confidence interval; NGF, nerve growth factor; PPV, positive predictive value; NPV, negative predictive value.
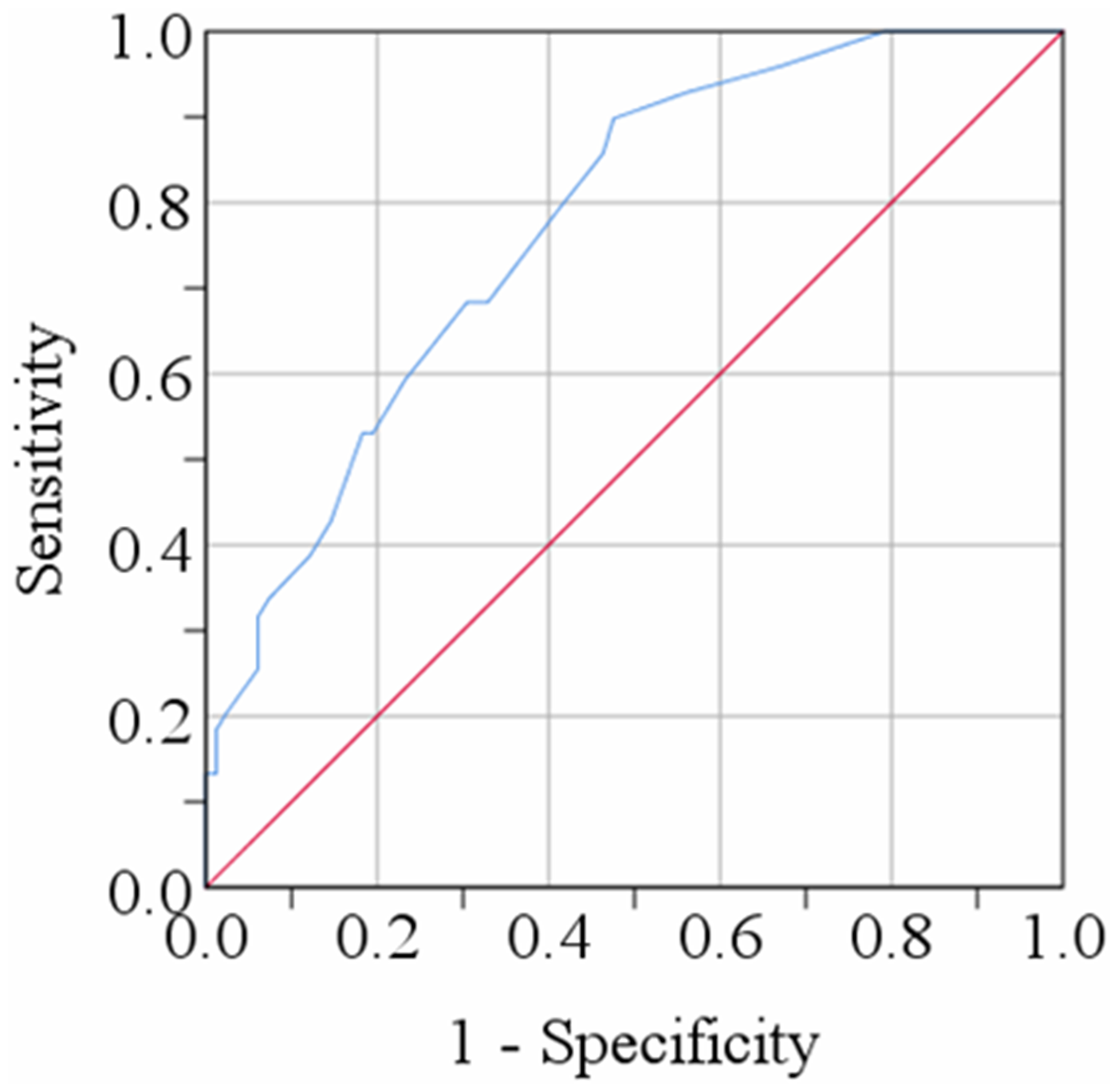
ROC curve for serum nerve growth factor. The cut-off point was detected as 71.32 pg/mL.
Discussion
Various genetic, social, and biological factors are associated with depression. 27 In the present study, we examined serum NGF levels in individuals who had been depressed over both the short and long term, and identified significantly higher serum NGF levels in MDD patients than in HCs. A large proportion of our study population had lower socio-economic backgrounds; their living conditions were characterized by high levels of stress caused by financial struggles.
Our results showed consistency with other studies in which serum NGF levels of MDD patients were significantly higher than those of HCs.28,29 These findings are supported by a range of previous studies regarding this subject. In one study, for example, both acute stress and lactation and labor caused a rise in NGF serum levels; however, a repeated stress paradigm may cause long-term NGF downregulation. 30 In an earlier study by Pallavi et al., NGF concentrations were observed to be lower in depressed individuals than in controls. 31 Conversely, other studies have reported higher NGF in individuals with high stress levels. 29 When adults with extreme stress and chronic depression—such as caregivers of impaired spouses—were compared with controls, Hadjiconstantinou et al. discovered that these stressed adults had increased NGF levels. 28 Similarly, a study by Fontenelle et al. identified heightened NGF concentrations in individuals with obsessive-compulsive disorder, 32 and Barbosa et al. reported a correlation between NGF and depression in bipolar disorder patients. 33 Another study found that, although there were lower levels of neurotrophic factors in the developing phase of depression, these factors were increased after antidepressant therapy. 34 Our findings thus support previous data that NGF concentrations change in response to stressful situations, indicating that NGF might operate as a connection between neuroendocrine and immunological components. Increased NGF levels in depressive individuals might function as a coping mechanism to shield the body against stress.
NGF is a neuropeptide that controls the production and survival of specific neurons in the peripheral and central nervous systems. 35 NGF and related molecules (generally called neurotrophins) mediate many biological events in our physiology,36–38 with effects ranging from neurotrophic39,40 to immunotropic41,42 and metabotropic.36,43 NGF is a modulator of neurological system development; this was first discovered by Cohen and Levi-Montalcini, earning them the Nobel Prize in Physiology or Medicine in 1986.44–46 NGF has been linked to the development of a wide range of neuronal and non-neuronal disorders such as Alzheimer's disease, obesity, atherosclerosis, type 2 diabetes, cardiometabolic disorders, and other neurodegenerative diseases.42,47,48 NGF is a modulatory factor in the hypothalamic–pituitary–adrenal axis and helps to maintain the immunological process. 39 An association between NGF and MDD has been reported in some recent investigations, although results have been inconsistent. A study of suicide revealed that the hippocampal NGF levels of individuals who died from suicide differed from those of HCs. 49 Decreased NGF levels have also been reported in certain parts of the brain in multiple mouse models, including those associated with stress-induced disease, anxiety vulnerability, threatening treatment, and helplessness.50–53 Several investigations of peripheral NGF levels between depressed patients and HCs have also indicated that NGF levels in individuals with MDD are altered.31,33,54–57 In another study, NGF levels were assessed in a younger population that included depressed patients with and without suicidal tendencies; serum NGF was considerably lower in MDD patients (both with and without suicidal tendencies) than in controls. 58 In contrast, another report has indicated that serum NGF levels are higher in MDD patients than in HCs. 29 An association between NGF and various emotional states has also been reported; Emanuele et al. revealed elevated NGF levels in people who were actively focused on a favored partner, had an emotional dependency, and sought emotional attachment with that person. 59 Conversely, some studies have reported no significant differences in serum NGF levels between MDD patients and control groups.60–62
NGF is involved in the pathophysiology of other mental health issues. Moreover, increased NGF levels may serve as a warning sign for stress and anxiety levels, or be used to estimate stress levels. Numerous recent investigations have revealed that stressful events—such as circumstances that provoke anxiety or mental stress, enhanced environmental conditions, and parturition—induce a rise in certain growth factors. Research in both mice and humans has demonstrated that emotional stress raises the plasma levels of the growth factor NGF.63,64 For example, the plasma NGF concentration of troops who had their first experience with parachute jumping increased by more than 100% compared with soldiers who did not jump; the NGF levels of these parachuters were much higher the evening before the jump. 65 This finding indicates that an anxious state can initiate the production of NGF. This conclusion is in line with our earlier findings of higher NGF plasma levels in dominant fighting mice than in subordinate fighting mice. 64 Increased NGF levels in depressed individuals can thus be interpreted as a sign of stress, and may guide MDD patients to take the necessary steps to protect their body from such stress. In addition, the previous finding of a rise in NGF levels before cortisol and adrenocorticotropic hormone increases indicates that NGF might play a role in some kind of alert system related to homeostatic adaptation. 63 Notably, the present study is one of the few that reported elevated NGF levels in MDD patients compared with controls. Another significant finding is that the ROC curve analysis demonstrated a good diagnostic value of serum NGF concentrations. Although many previous studies have evaluated NGF levels in MDD patients and identified a link between them, to the best of our knowledge, none have assessed the accuracy of NGF levels for predictive or diagnostic purposes. In the current study, the ROC curve was plotted to evaluate the diagnostic value of serum NGF concentrations for MDD patients. The area under the curve was 0.774 and the cut-off point was 71.32 pg/mL.
Neurotrophic factors are important mediators that control synaptic neurotransmitter release as well as responsiveness in the adult nervous system. 66 Although various biological factors are responsible for the pathophysiology of MDD, there are no consistent markers of this disorder. 67 Several recent studies have determined a connection between biological markers and depression.68–71 To our knowledge, however, the present study is the first NGF research of its kind to be conducted in Bangladesh with a relatively large sample size. By collecting blood samples from a hospital that serves patients from all over the country, we were able to create a research population that was both uniform and diverse. However, there were some drawbacks to our research. For example, the single measurement of a marker may not provide information about the status of depressive symptoms. Furthermore, a range of nutritional factors are involved in the pathophysiology of depression,72,73 and some of these factors increase NGF levels in the body. The lack of any analysis of dietary intakes and supplementations in the present study is thus a major limitation. Additionally, a case–control observation of a single marker might restrict the broader scope of this research. To clarify the role of NGF in depression, further interventional studies in a large, homogeneous sample are required. Further investigations may provide a greater understanding of neurotrophic factors and their role in depression, thereby allowing the development of more selective diagnostic measurements and treatment interventions.
Conclusions
The current study evaluated serum NGF in MDD patients and evaluated the connection between serum NGF concentrations and the pathophysiology of depression. Our findings indicate that the development of depression might be associated with altered serum NGF levels. These results will aid our understanding of the underlying mechanisms, risk factors, disease course, and treatment outcomes of depression. Furthermore, serum NGF may be an option for early risk assessment or as a diagnostic marker of MDD. However, additional large-scale research on NGF is needed to gain a more comprehensive understanding of its role in the pathophysiology of MDD.
Footnotes
Acknowledgements
We thank the study participants and their relatives for their participation and cooperation in this study. We also thank all physicians and administrative staff in the Department of Psychiatry, Bangabandhu Sheikh Mujib Medical University, for their cooperation and administrative support during this study.
Author contributions
Conceptualization: LS and MRI. Formal analysis: LS and MS. Writing – original draft: LS. Writing – review and editing: SMAI, MAB, and MMASQ. Supervision: MRI. All authors have read and approved the final version of the manuscript. The corresponding author had full access to all data in this study and takes complete responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
