Abstract
Patient-specific induced pluripotent stem cells (iPSCs) have tremendous potential for development of regenerative medicine, disease modeling, and drug discovery. However, the processes of reprogramming, maintenance, and differentiation are labor intensive and subject to intertechnician variability. To address these issues, we established and optimized protocols to allow for the automated maintenance of reprogrammed somatic cells into iPSCs to enable the large-scale culture and passaging of human pluripotent stem cells (PSCs) using a customized TECAN Freedom EVO. Generation of iPSCs was performed offline by nucleofection followed by selection of TRA-1-60–positive cells using a Miltenyi MultiMACS24 Separator. Pluripotency markers were assessed to confirm pluripotency of the generated iPSCs. Passaging was performed using an enzyme-free dissociation method. Proof of concept of differentiation was obtained by differentiating human PSCs into cells of the retinal lineage. Key advantages of this automated approach are the ability to increase sample size, reduce variability during reprogramming or differentiation, and enable medium- to high-throughput analysis of human PSCs and derivatives. These techniques will become increasingly important with the emergence of clinical trials using stem cells.
Keywords
Introduction
Advances in technology have enabled the reprogramming of adult somatic cells into human induced pluripotent stem cells (iPSCs),1–3 which can be subsequently differentiated into cells of interest, providing a potentially inexhaustible supply of cells with disease-specific genotypes and phenotypes. A main bottleneck in using iPSCs for disease modeling lies in the vast amount of time and manpower necessary to maintain cells in culture, making large-scale population studies almost unfeasible within the constraints of an average-sized laboratory. The use of robotics could enable this type of research. A key advantage of an automated approach is the ability to increase sample size and reduce variability during the reprogramming, maintenance, and differentiation of iPSCs. Current applications of automated technology for stem cell research are still limited. Among the very few systems reported for automation of stem cell culture and differentiation are the Automation Partnership Biosystems (TAP Biosystems, Royston, UK), which has already been used to maintain human mesenchymal stem cells 4 as well as human bone marrow–derived cells, 5 and the AutoCulture system (Kawasaki Heavy Industries), which has been used to maintain human cardiac stem cells. 6 In addition, prototypes have been developed to change medium for cell culture of embryonic stem cells. 7 The automated platform TECAN Freedom EVO (TECAN, Männedorf, Switzerland) has been adapted to maintain mouse embryonic stem cells and differentiate them toward a neuronal lineage. 8 Similarly, a few groups have reported on the reprogramming, maintenance, and differentiation of human PSCs on automated platforms. The New York Stem Cell Foundation uses a self-designed robotic platform composed of three platforms using STAR liquid-handling systems (Hamilton Robotics, Reno, NV) to carry out the maintenance and passaging of human iPSCs. 9 Another automated platform was described for the maintenance of human iPSCs, using a robotic arm (MELFA, RV-4FC-D; Mitsubishi, Tokyo, Japan) for liquid handling. 10 A smaller liquid handler was recently described for the maintenance and passaging of human PSCs, based on a self-contained Gilson’s pipette Max liquid handler, that can process up to 96-well plates. 11 However, this system requires additional offline steps as there is no associated incubator and many steps require human contribution.
Our initial requirement for an automated platform was the ability to maintain large numbers of iPSC lines or progeny, with minimal variation, to model complex diseases, such as age-related macular degeneration or primary open angle glaucoma. Indeed, those conditions involve various genetic and environmental factors, and thus large numbers of samples are necessary to robustly confirm biological effects. Herein we describe a customized TECAN Freedom EVO platform for the maintenance of human fibroblasts undergoing reprogramming to iPSCs, as well as maintenance and passaging of undifferentiated colonies of PSCs. We also report the feasibility of using this platform for long-term differentiation of cells, demonstrated with guided differentiation of human PSCs to retinal cells, including retinal ganglion cells (RGCs) and retinal pigment epithelium (RPE) cells. We describe the maintenance protocols used and the adaptations required for using this automated platform. This system allows for large sample–sized research, reduced variability, and future high-throughput analysis of the transcriptome and metabolome of progeny cells derived from patient iPSCs. The performance of our system could be deemed to lie between that of simple liquid handlers 11 and a more complex fully integrated platform. 9 Our system enables the maintenance of human fibroblasts undergoing reprogramming; allows for the long-term maintenance, passaging, and differentiation of iPSCs, which are not available with smaller liquid handlers; and offers an economical alternative to more complete equipment.
Materials and Methods
Ethics
All experimental work performed in this study was approved by the Human Research Ethics committees of the Royal Victorian Eye and Ear Hospital (11/1031H, 13/1151H-004) and University of Melbourne (0605017, 0829937) with the requirements of the National Health & Medical Research Council of Australia and conformed with the Declarations of Helsinki. 12
Platform Material
The TECAN system is a liquid-handling platform that requires a tissue culture plate format. All cells were cultured and handled in a six-well plate format. Tips used were 5 mL disposable conductive sterile tips with filter for the liquid handling (LiHa) arm (TECAN). Cell culture medium was aliquoted into 50 mL Falcon tubes and placed into specific carriers in the TECAN platform.
Fibroblast Culture
Human fibroblasts were cultured in DMEM with high glucose, 10% fetal bovine serum (FBS), L-glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin (all from Life Technologies, Carlsbad, CA). All cell lines were confirmed to be mycoplasma free using the MycoAlert mycoplasma detection kit (Lonza, Basel, Switzerland) per the manufacturer’s instructions. A total of 77 fibroblast lines were used for reprogramming.
Generation of iPSCs
iPSCs were generated using human skin fibroblasts obtained from subjects over the age of 18 years by episomal method, as described previously. 13 Briefly, reprogramming was performed on passage of 8 to 10 fibroblasts by nucleofection (Lonza Amaxa Nucleofector) with episomal vectors expressing OCT4, SOX2, KLF4, L-MYC, LIN28, and shRNA against p53 14 in feeder- and serum-free conditions using TeSR-E7 medium (STEMCELL Technologies, Vancouver, Canada). The reprogrammed cells were maintained on the automated platform using TeSR-E7 medium, with medium change every day (2 mL/well).
Selection of iPSCs
Pluripotent cells were selected using a MultiMACS Cell24 separator (Miltenyi Biotec, Bergisch Gladbach, Germany) by TRA-1-60 sorting using anti-human TRA-1-60 microbeads in combination with Multi-24 column blocks (Miltenyi). Briefly, reprogrammed cells from one well of a six-well multiwell plate were washed in phosphate-buffered saline (PBS) and incubated with TrypLE Select (5–10 min, 37 °C; Life Technologies), and cells were collected and gently triturated in TeSR-E8 medium supplemented with Y27632 (10 µM; Selleck Chemicals, Houston, TX). The cell suspension was filtered through a preseparation filter (30 µm, Miltenyi) into a 15 mL tube, and cell number was determined. Cells were then centrifuged (5 min, 300g) and resuspended in 80 µL ice-cold TeSR-E8 medium containing Y27632 (10 µM) and incubated with 20 µL anti-TRA-1-60 beads (5 min, 4 °C). Volume was adjusted to 1 mL in TeSR-E8 medium containing Y27632 (10 µM), and each suspension containing magnetically labeled cells was loaded onto a MultiMACS column. Columns were washed twice with TeSR-E8 medium containing Y27632, then eluted with 1 mL TeSR-E8 medium containing Y27632. Cell number was determined and cells were then plated into one well of a six-well plate coated with vitronectin XF (40 µL/well in 2 mL cell adhere dilution buffer; STEMCELL Technologies), then placed back into the online incubator. Quantification of successful reprogramming was performed post TRA-1-60 selection by immunomagnetic beads (MACS), measuring the frequency of lines able to form undifferentiated colonies, no colony, or colonies of differentiated cells. The frequency of reprogrammed fibroblasts that formed small colonies that did not grow was also reported.
Maintenance and Passaging of PSCs
Subsequent culturing was performed on the automated platform using TeSR-E8 (STEMCELL Technologies), changing medium every 2 days (2 mL/well). Passaging of newly generated iPSC lines and of the iPSC line CERA007 15 was performed on the automated platform using ReLeSR (STEMCELL Technologies) onto vitronectin XF plated wells. In parallel, iPSCs and the human embryonic stem cell line H9 (WiCell, Madison, WI) were maintained in the same conditions but manually passaged as a comparison to automation.
Automated Passaging
Passaging was performed weekly. The new vitronectin-coated plates were accessed from the incubator and placed onto the carrier, four at a time. Vitronectin XF was removed, 2 mL of PBS was added per well, the plate carrier was angled and tilted four times to ±40°, PBS was removed, 2 mL of medium was added, and then the carrier was angled and tilted four times to ±40° and plates were returned to the incubator. In parallel, the plates to be passaged were retrieved from the incubator and placed onto the carrier (four at a time), medium was removed, 1 mL of PBS was added, and the plate carrier was angled and tilted 20 times to ±10°. PBS was removed, replaced by 800 µL of ReLeSR, and the plate carrier was angled and tilted 20 times to ±10°. The plate carrier was angled and tilted as follows for change of liquid: four times to ±40° for washes with PBS and 20 times to ±10° when shaking was needed. Seven hundred microliters of ReLeSR was removed, and the cells were left to incubate for 10 min. Medium (1 mL) was added for 3 min, followed by shakes of the carrier (angled and tilted 100 times ±5°, 20 times ±40°, 1000 times ±0.5°), twice. Visual assessment of cell detachment was then performed. Medium was then aspirated with a 52° angled carrier and transferred to 15 mL tubes (six wells into one tube: final volume of 4.8 mL). Cell suspension was mixed by pipetting up and down two times with 4 mL and seeded into the vitronectin XF–coated plates retrieved from the hotel at chosen concentration and returned to the incubator.
Quantification of Expression of Pluripotency Markers and of Cell Growth
TRA-1-60 quantifications were performed using a MACSQuant (Miltenyi) on iPSCs just prior to passaging to fresh plates and at different passage numbers. Cell counts of live cells when plated down and at passaging were performed to determine cell growth.
Retinal Cell Differentiation
Retinal differentiation of BRN3B-mCherry A81-H7 human embryonic stem cells (hESCs) 16 was performed via an adapted protocol originally described by Lamba et al. 17 using DMEM F12 with glutaMAX (Life Technologies), 10% Knockout Serum Replacement (Life Technologies), IGF1 (10 ng/mL; Peprotech, Rocky Hill, NJ), Dkk1 (10 ng/mL; Peprotech), Noggin (10 ng/mL; R&D Systems, Minneapolis, MN), basic fibroblast growth factor (bFGF; 5 ng/mL), B27 and N2 (both 1×; Life Technologies) as described in Gill et al., 18 changing medium every second day. We adapted the protocol to automation by starting with a monolayer of PSCs plated on vitronectin XF in place of embryoid body formation. Cells were assessed at day 24, and no further enrichment was performed. 18 Successful differentiation into RGCs was determined by appearance of mCherry-positive cells, which is indicative of BRN3B expression. Differentiation of H9 hESCs (WiCell) into RPE cells was performed in feeder-free conditions as described in Lidgerwood et al. 19 using vitronectin XF and RPEM medium (α-MEM, 0.1 mM nonessential amino acids, 0.1 mM N2, 1% L-glutamine–penicillin–streptomycin solution, 250 µg/mL taurine, 20 ng/mL hydrocortisone, 13 pg/mL triiodothyronine, all from Sigma-Aldrich, St. Louis, MO; 25 mM HEPES), supplemented with 5% FBS, IGF1 (10 ng/mL), Dkk1 (10 ng/mL), Noggin (10 ng/mL), bFGF (5 ng/mL), B27 and N2 (both 1×), changing medium every 2 days. Cells were assessed at day 35. Successful differentiation into RPE cells was determined by cobblestone morphology and pigmentation, as well as PMEL expression by immunocytochemistry.
Immunocytochemistry
Immunocytochemistry was performed using OCT3/4 (C-10, Santa Cruz Biotechnology, Santa Cruz, CA), TRA-1-60 (Abcam, Cambridge, UK), and PMEL (Abcam). Cells were then immunostained with isotype-specific secondary antibodies (Alexa-Fluor, Life Technologies). Nuclei were counterstained using DAPI (Sigma-Aldrich).
Statistical Analysis
Data are expressed as mean ± standard error of the mean (SEM). All statistical analyses and graphical data were generated using Graphpad Prism software (v6, www.graphpad.com). TRA-1-60 quantifications were performed on 10 individual lines maintained on the automated platform (n = 5 at passage 1 and n = 5 at passage 4), as well as 4 lines maintained manually. Cell counts were performed on 10 individual iPSCs lines over three passages each. Statistical methods used were one-way analysis of variance followed by Tukey’s multiple comparisons test. Statistical significance was established from p < 0.05.
Results
Description of the Automated Platform
Our modular platform is composed of a TECAN Freedom EVO 200, which includes a class 2 biosafety cabinet, a robotic liquid-handling arm with eight independent channels (eight channels LiHa), and a robotic manipulator arm (RoMa with eccentric fingers), in conjunction with a Liconic STX110 automated incubator mounted behind the Freedom EVO and a carousel LPX220 for dispenser tips (DiTis) on the right side of the working platform ( Fig. 1 ). A 5 L glass bottle and peristaltic pumps are also present; however, these were not used for the routine maintenance described here. We opted not to use the tubing and troughs that are necessary in that option and replaced them with sterile Falcon tubes as a mean to ensure sterility and volume of reagents required ( Fig. 1 ). The overall system dimensions are 3.4 m (length) × 1.8 m (width) × 2.7 m (height). To the left side, some space was reserved for a computer, keyboard, monitor, and operator ( Fig. 1 ). A MultiMACS Cell24 Separator and a MACSquant flow cytometer were accessed offline. As illustrated in Figure 1 , on the workbench, from left to right, are a cabinet with medium bottle and pump (1) which can deliver medium to a LiHa medium refill trough contained within a Torrey Pines heater; three carriers for up to 24 × 50 mL Falcon tubes (2), a LiHa wash station (3), a carrier for 5 mL DiTis (4), a large waste for DiTi boxes and used tips (5), a carrier for tip boxes (6), a tilting carrier (7), a microplate carrier (8), the transfer station (9) to the Liconic incubator (12), a hotel for tissue culture lids (10), and a carousel (11). Autoclaved water bottles for the liquid-handling system were stored under the bench (13). The LiHa arm was used solely with 5 mL syringes: 8 × 5 mL syringes for DiTis, although they are capable of handling smaller 1 mL tips (which were not used for our protocols). The Liconic LPX220 carousel was used for the storage of tip boxes. It contains one rotary plate with 10 interchangeable cassettes and an internal robotic handler, as well as a transfer station to place tip boxes onto the worktable. The Liconic STX110 incubator comprises an internal robotic handler to access five independent stackers that can store up to 85 culture plates (17 per stacker), an internal barcode scanner, as well as a transfer station to bring culture plates onto the worktable. The incubator has a controlled environment, which was set to 37 °C and 5% CO2. The TECAN EVO runs with two independent software packages: the freedom EVOware Plus pipetting software (EVOware) and the Workflow Planning Tool. These are used to direct pipetting and device commands (EVOware) and to plan and execute each line of the workstation’s workflow (the Workflow Planning Tool). Each protocol used on the platform was entered as an independent template.

The automated platform. (
Generation of iPSC Lines
We manually reprogrammed 77 skin fibroblast lines from individual patients to iPSCs using episomal vectors in feeder- and serum-free conditions in TeSR-E7 medium. Nucleofection of fibroblasts was performed in a six-well plate format. Following nucleofection, cells were placed into the online incubator. Medium was changed every day using the automated platform. To identify and isolate iPSCs, we used the marker TRA-1-60, which was previously shown to be a marker of fully reprogrammed iPSCs. 20 Instead of picking clonal-derived iPSCs, we performed bulk selection of polyclonal iPSCs as these were shown to be indistinguishable from clonal-derived iPSCs. Notably, the bulk generation of polyclonal iPSCs has been shown to be as effective in the generation of fully reprogrammed lines as manual selections of clones. 21 At approximately day 30, iPSCs were purified by MACS labeled with TRA-1-60, using a MultiMACS Cell24 Separator and maintained in feeder-free culture on vitronectin in TeSR-E8 medium. When post-reprogrammed cells were subjected to TRA-1-60 selection by MACS for selection of iPSCs, 52.31% ± 5.71% of whole cells sorted for TRA-1-60 were positive for the pluripotency marker and were plated for expansion ( Table 1 ). Quantification of reprogramming performed post TRA-1-60-MACS enrichment indicates that 92.2% of fibroblast cultures were successfully reprogrammed to iPSCs (presence of TRA-1-60–positive colonies that had grown and retained their characteristic undifferentiated morphology), whereas 2.6% of fibroblast cultures formed small colonies that did not grow, and 5.2% of reprogrammed fibroblast cultures formed no colony or colonies of differentiated cells (n = 77; Table 1 ).
Quantification of Successful Reprogramming.

n = 51 samples were sorted and satisfied internal specifications for cell counts and cell sizes on countess.
Maintenance of PSCs
iPSC lines were then used for maintenance and passaging on the automated platform. Pluripotency of all derived iPSC lines was further evidenced by immunocytochemistry for OCT-4 and TRA-1-60 expression ( Fig. 2A–F ). The maintenance and passaging templates allow for changing medium and passaging of iPSCs. Maintenance was optimized for automation in serum-free and feeder-free conditions using vitronectin-coated plates in TeSR-E8 medium. All lines were successfully passaged on the platform. iPSCs were maintained for multiple passages on vitronectin-coated plates using E8 culture medium and passaged using ReleSR. Representative bright field images of colonies following successive passaging are shown in Figure 2G–I . Immunocytochemistry confirmed that iPSCs remain pluripotent, as indicated by OCT-4 and TRA-1-60 expression, following successive passaging using this automated platform ( Fig. 2J–L ). Cells were passaged at a split of 1:6 without affecting maintenance and morphology. MACS quantification of TRA-1-60 immediately prior to passaging demonstrates that the cells were successfully maintained pluripotent on the platform. Lower splits would be achievable using 1 mL tips in place of 5 mL tips. Indeed, as shown in Figure 2M , the quantification of TRA-1-60, across lines and across passaging, clearly indicates reproducibility and little variability in TRA-1-60 expression, with 79.45% ± 7.51% (passage 1, n = 5 lines quantified) and 91.49% ± 4.81% (passage 4, n = 5 lines quantified) of TRA-1-60–positive cells in the culture. In parallel to the automated maintenance of iPSCs, some iPSC lines and H9 were also manually cultured, using the same feeder-free and serum-free conditions. Quantification of TRA-1-60 just prior to passaging indicates that 65.91% ± 4.56% cells were TRA-1-60 positive ( Fig. 2M , n = 4 lines quantified), indicating that automation is at least equivalent to manual maintenance in generating high-quality pluripotent iPSC colonies and is in range similar to that observed with other automated systems. 9 Similarly, cell counts performed at plating and before passaging on three successive passages of 10 iPSC lines maintained on the automated platform indicate that cell growth was similar between passages of the same lines and in the same order of magnitude across lines ( Fig. 2N ). These data thus demonstrate further the reliability of the platform to maintain iPSCs.
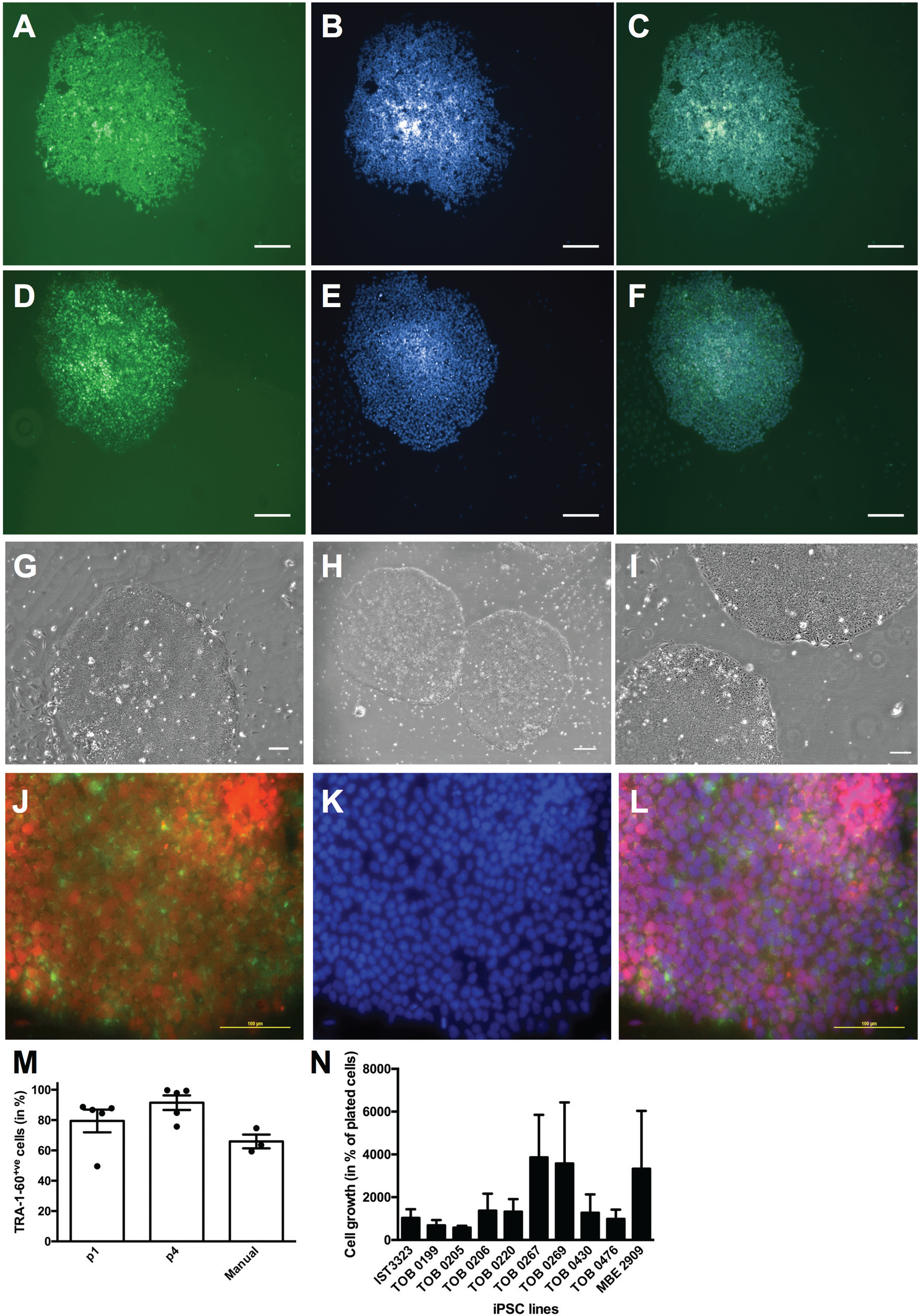
Stem cell maintenance using the automated platform. (A–F) Maintenance of cells post-TRA-1-60 selection. Representative images of colonies post-TRA-1-60 sorting, immunostained for OCT-4 (
Differentiation of PSCs into Retinal Cells on the Automated Platform
To assess the potential of this automated platform for long-term differentiation culture, we directed the differentiation of human PSCs toward two retinal lineages, RPE cells and RGCs, using protocols established within our group.18,19 Given the proof-of-concept nature and aim to ensure feasibility of long-term differentiation, no quantification was undertaken. We used the reporter line BRN3B-mCherry H7 16 for the RGC differentiation assay as this line fluoresces with expression of the RGC marker BRN3B, allowing for the screening of successful RGC differentiation. To make the RGC differentiation protocol described by Gill et al. 18 suitable for automation, it was slightly modified by replacing the initial embryoid body step with a monolayer differentiating culture. We then followed the protocol as previously described. As shown in Figure 3A , B , we could clearly observe expression of mCherry in the differentiated culture, indicating successful RGC differentiation using our automated platform.
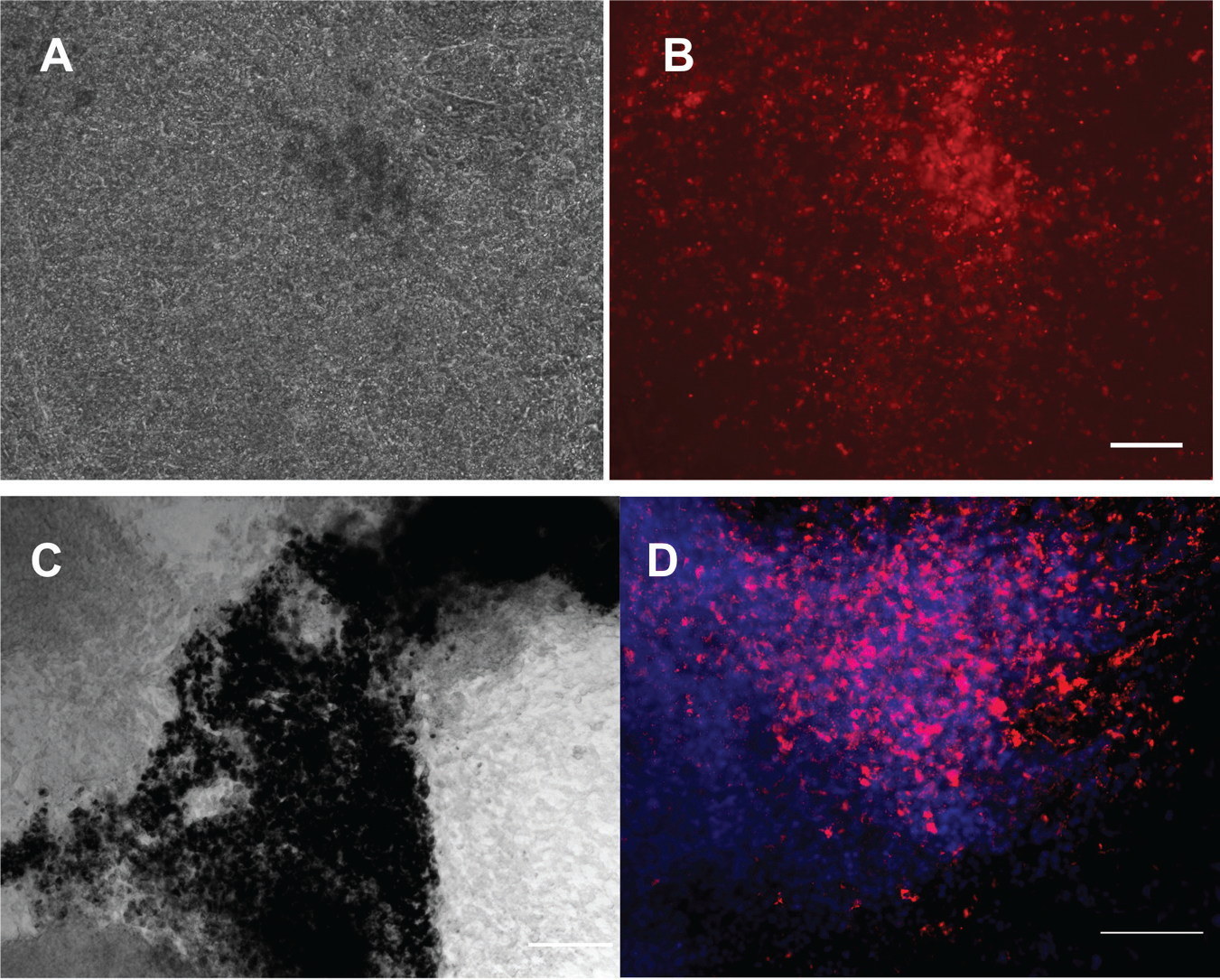
Retinal differentiation using the automated platform. (
Next, we performed RPE differentiation using the automated platform. The differentiation of PSCs into RPE cells is evident by the characteristic morphology and pigmentation of RPE cells. We directed differentiation of human PSCs plated in feeder-free conditions into RPE cells using IGF1, DKK-1, Noggin, and bFGF as described in Lidgerwood et al. 19 Medium was changed every other day. Pigmented cells started appearing approximately 4 weeks later. We confirmed the polygonal geometry of the RPE cells and expression of the RPE marker PMEL ( Fig. 3C , D ). Further enrichment would then be necessary to obtain purer population of cells of interest, by dissection or sorting of cells of interest as we previously described.18,19. Together, these results provide proof of concept that automation can be used to facilitate stem cell maintenance and retinal differentiation. Importantly, no contamination was observed during the reprogramming, maintenance, and differentiation procedures, demonstrating the robustness of the automated platform for long-term sterile cell culture. The workflow for all procedures and potential applications is presented in Figure 4 .
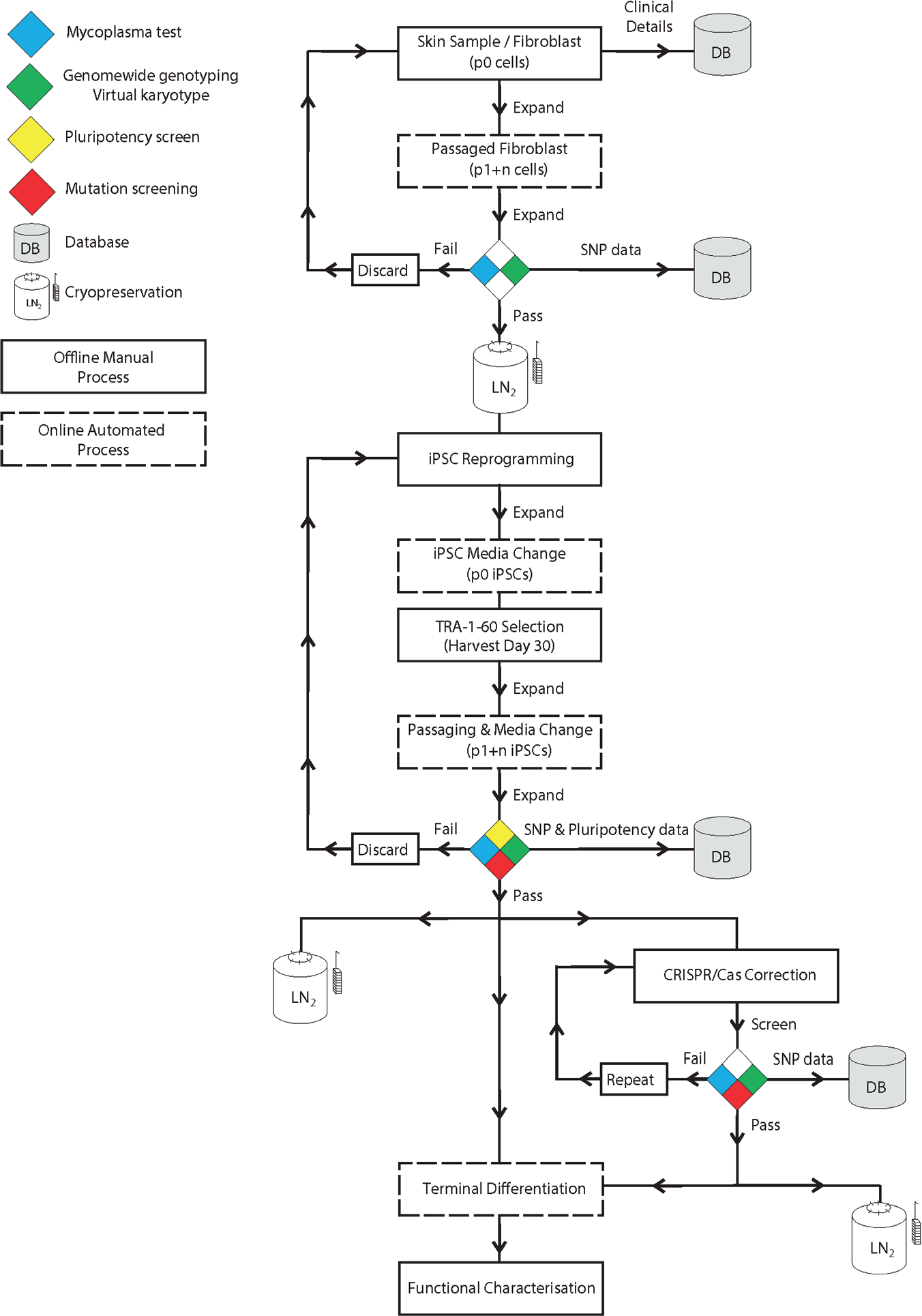
Workflow for the automation of fibroblast culture, reprogramming, maintenance, and differentiation of pluripotent stem cells. Quality controls (genome-wide single-nucleotide polymorphism genotyping and pluripotency analysis) and potential incorporation of gene editing are also indicated. Dotted lines indicate online automated steps, and solid lines indicate manual processes.
Discussion
We describe the use of a modular platform to maintain, passage, and differentiate human iPSCs. All protocols were adapted to automation using a feeder-free system for maintenance and differentiation. Some aspects of the work were performed offline, notably the reprogramming of cells and selection of successfully generated iPSCs. Further optimization could allow these steps to be performed online, by integration within the modular platform, as done by others. 9 However, we did not to integrate those in anticipation of potential changes in methods of reprogramming (in which nucleofection might become obsolete) or TRA-1-60 selection. The automated system allows for substantial customization of both equipment and cell-handling parameters providing the flexibility needed for cell culture of various cell types. We did notice a few limitations of the automated platform. Despite a successful pipeline for the maintenance, passaging, and differentiation of human PSCs, as well as sterility, improvements can be made to facilitate, streamline, and reduce costs of the cell culture processes further. To reduce the risk of cross-contamination, each line was cultivated within its own six-well tissue culture plate. This format has the advantage of allowing for selections of cells for multiple applications, such as multiple long-term differentiation in various wells, or harvesting of samples for genomics, proteomics, or lipidomics. On the other hand, this format could also imply that some wells within plates might stay empty, hence increasing unnecessary cost and waste. It would be very useful to also work with single-well tissue culture plates in addition to multiwell plates. However, those are currently not commercially available with the required height to be handled by the robotic arm of this platform. Such single-well tissue culture plates would be very beneficial as they could allow increasing a plating format as observed in flasks. Second, the conductive sterile filter tips are expensive. Disposable filtered tips are required, as fixed tips can easily carry contaminations, and the sterility of the platform and in particular of the system liquid is paramount. There is also the risk of cross-contamination of samples by using fixed tips; thus, we elected to solely use disposable tips for all liquid-handling procedures. Also, maintenance of the platform and routine checks are essential but time-consuming. Importantly, we have not had any single contamination using this platform, demonstrating the sterility of the system. Further, current templates for cell handling using the automated platform are relatively slow. Hence, some adaptation had to be made to ensure cells were properly maintained, not left without medium for too long, and placed back in the incubator in a timely manner. Future modification to the templates, such as reducing redundant movement of robotic arms, would reduce the time required for cell handling and increase the efficiency for the automated platform to process samples.
Finally, given the rapid pace of advances in the stem cell field, many assays and techniques for stem cell reprogramming, maintenance, or differentiation can become rapidly obsolete and replaced with newer methods. Hence, a modular platform should allow for novel techniques to be included in the workflow to replace less useful methods. New protocols can be generated and adapted to the automated platform. However, a basic level of computing and training with the software are necessary. To fully realize the potential of the automated platform, the research group must be versatile and have a combined expertise in stem cell biology (reprogramming, cell maintenance and differentiation), as well as computing skills and engineering. To reduce these demands, workflow could be improved and made more generally useful by increasing the user friendliness of the interface between human and machine, thereby allowing modifications to the protocols to be made more easily.
The automated system we introduce here is novel and provides additional functions to the automated platform described by Konagaya et al. 10 ; it allows for the long-term maintenance and passaging of human iPSCs. In addition, we have also optimized maintenance of fibroblasts undergoing reprogramming as well as the directed differentiation to retinal lineages. This work can be placed in parallel to that of Paull et al., 9 who successfully used an automated platform for the generation, maintenance, and differentiation of a large cohort of human PSCs. In their study, the authors used a remarkable eight modular platform covering all aspects of cell handling. The system we describe here is not as complete but provides an economical alternative to the maintenance of PSCs and their differentiation.
In summary, we report the successful maintenance, passaging, and differentiation of human PSCs using an automated platform equipped with liquid handler and robotic arms, to handle cells in tissue culture plates. Despite some limitations, this platform shows excellent handling of sterility and dramatically expands the potential of human PSC research. By increasing sample size and reducing variability, it allows for more defined parameters for future high-throughput analysis of the transcriptome and metabolome of progeny cells derived from patient iPSCs. This will be particularly important for iPSC modeling of complex genetic diseases, which will require large sample sizes to provide sufficient power for statistical analyses.
Footnotes
Acknowledgements
We thank Marco Zalivani for the drawing of the computed-aided design images of the automated platform.
Author Contributions
D.E.C., M.D., H.H.L., T.K., R.C.B.W.: concept and design, collection and/or assembly of data, data analysis and interpretation, manuscript writing, final approval of manuscript. F.L., G.E.L., A.C., D.H., S.S.H., K.P.G., E.D.S., L.S.K., L.C..: collection and/or assembly of data, data analysis and interpretation, editing and final approval of manuscript. V.M.S., X.C., D.J.Z.: generation of cells, editing and final approval of manuscript. A.W.H., A.P.: concept and design, seeking of financial support, data analysis and interpretation, manuscript writing, final approval of manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the Ophthalmic Research Institute of Australia (10.13039/501100001108), the Joan and Peter Clemenger Foundation, the Philip Neal bequest, a National Health and Medical Research Council Practitioner Fellowship (10.13039/501100000925 to A.W.H.), an Australian Research Council Future Fellowship (10.13039/501100000923 FT140100047 to A.P., FT140100047), the University of Melbourne and Operational Infrastructure Support from the Victorian Government.
