Abstract
Cell-based assays have long been important within hit discovery paradigms; however, improving the disease relevance of the assay system can positively affect the translation of small-molecule drug discovery, especially if adopted in the initial hit identification assay. Consequently, there is an increasing need for disease-relevant assay systems capable of running at large scale, including the use of induced pluripotent stem cells and donor-derived primary cells. Major hurdles to adopting these assays for high-throughput screening are the cost, availability of cells, and complex protocols. Miniaturization of such assays to 1536-well format is an approach that can reduce costs and increase throughput. Adaptation of these complex cell assays to 1536-well format brings major challenges in liquid handling for high-content assays requiring washing steps and coating of plates. In addition, problematic edge effects and reduced assay quality are frequently encountered. In this study, we describe the novel application of a centrifugal plate washer to facilitate miniaturization of a range of 1536-well cell assays and techniques to reduce edge effects, all of which improved throughput and data quality. Cell assays currently limited in throughput because of cost and complex protocols may be enabled by the techniques presented in this study.
Introduction
Cell-based assays have been routinely used in high-throughput screening (HTS) to screen large libraries of small molecules to identify potential drug candidates. However, over the past few decades, this has largely focused on a reductionist target-based approach centering on a specific protein. In contrast, cell assays that adopt a phenotypic approach have the advantage of revealing a target or targets necessary to drive a desired cell phenotype and associated disease mechanisms in an endogenous system. A study of the success of different strategies undertaken in the discovery of 50 new drugs that were approved by the U.S. Food and Drug Administration between 1999 and 2008 revealed that the contribution of phenotypic screening exceeded that of target-based approaches, with 28 being phenotypic-based discoveries and 17 using a target-based approach. 1 Considering this analysis was based on an era in which the major focus was on target-based approaches, it is not surprising that the drug discovery field is once again embracing a phenotypic approach for small-molecule discovery.
Drug discovery programs that examine specific targets within an endogenous background are also being used more heavily. These include new screening techniques such as transcriptomics and proteomics or through specific antibody labels within a high-content screening paradigm. The initial hit identification assay is of critical importance in the ultimate success of drug discovery programs involving phenotypic, transcriptomic, or proteomic approaches. Recent studies suggest that the most predictive cell assays involve use of disease-relevant assay systems including use of induced pluripotent stem cells (iPSCs) and donor-derived primary cells. 2 Major barriers to adopting these assays for HTS are the complex techniques, along with the cost and availability of the relevant cells, with some iPSC-based assays costing up to £2,000 per 384-well microtiter plate. Miniaturization of such assays would inevitably reduce costs, increase throughput, and ultimately improve the translation of hit identification screens.
Despite the potential impact, there are only limited examples of effective adaptation of complex cell-based HTS screens to a 1536-well format. A 1536-well primary human organotypic assay containing primary human fibroblasts, mesothelial cells, and extracellular matrix showed promising results, achieving an average Z′ of 0.6. However, the limited scale of the screen (2420 compounds) does not address the complexities involved in adaptation of assays to the scale required for HTS, which often requires screening up to 2 million compounds. 3 Miniaturization to 1536-well format has also been reported for an assay using primary retinal neurons studying photoreceptor differentiation using a fluorescent reporter and cell survival using a CellTiter Glo viability assay. 4 Despite a limited number of protocol steps, the differentiation assay reported high variability (percentage coefficient of variance [CV] = 20%). Other studies have reported issues with edge effects during longer incubations, resulting in exclusion of the outer wells of the 1536-well plate to improve the precision of the assay. 5 Compromised assay quality has also been observed in miniaturization of an immunohistochemistry assay from 384-well format (Z′ = 0.51 ± 0.09; signal/background = 4.07 ± 0.39) to 1536-well plate format (Z′ = 0.36 ± 0.08; signal/background = 3.34 ± 0.42). 6 This study addresses some common issues observed for miniaturization to 1536-well format, such as liquid-handling challenges for immunohistochemistry assays requiring washing steps and coating of plates. Edge effects can also result in compromised assay quality and throughput. Our results demonstrate that novel applications of techniques such as centrifugal washing instead of tip-based aspirations can have significant impact on the quality and feasibility of miniaturization of cellular assays to 1536-well format.
To demonstrate the feasibility of 1536-well miniaturization for HTS, a variety of cellular assays were used in this study:
A phenotypic assay previously used for the identification of small molecules that inhibit the inducible degrader of low-density lipoprotein receptor (IDOL) by monitoring levels of low-density lipoprotein receptor–green fluorescence protein (LDLR-GFP). This reporter system was miniaturized to 1536-well format and demonstrated compatibility with high-content imaging systems.
A phenotypic assay previously used for the identification of small molecules that promote proliferation of human pancreatic β-cells was used in this study to demonstrate the application of centrifugal plate washing for complex phenotypic assays requiring multiple washing steps.
The Corning Epic system measures cell dynamic mass redistribution (DMR) by refraction of light from a biosensor integral to an Epic microtiter plate. A DMR assay had previously been used in 384-well format to identify tool compounds that interact with GPR83, a G-protein coupled receptor (GPCR). 7 However, the technology has previously been limited at large scale because of the high cost of the specialized plates. Development of a 1536-well format DMR assay was enabled by using centrifugal plate washing for both in-house plate coating and a media exchange step, allowing the assay to be screened in higher throughput.
A 1536-well CellTiter-Blue viability assay was previously used with the aim of identifying and understanding the levels of undesirable cellular toxicity within HTS outputs. Increases in plate batch sizes and compound numbers to an HTS scale resulted in significant loss of assay performance. Problematic edge effects observed were alleviated by using plate seals, which improved throughput and efficiency of the assay.
Materials and Methods
Cells and Media
All cell lines were grown under standard culture conditions at 37 °C with 95% humidity and 5% v/v CO2. Unless otherwise stated, cells were cryopreserved as described previously, 8 and all lab reagents were obtained from Sigma.
PathHunter eXpress GPR83 CHO-K1 β-Arrestin Orphan GPCR cells (DiscoverX, Fremont, CA) were screened in UltraCHO Serum Free CHO medium and supplement (Lonza, Basel, Switzerland) containing 100 U/mL penicillin and 100 µg/mL streptomycin.
Human LDLR (NM_000527, ORF 188-2767 with Kozak sequence introduced before the start codon) was fused to GFP (U55762.1, ORF 679-1398) cloned into the Nhel and BsiWl restriction sites of pIREShyg3 (Clontech, Mountain View, CA). Full-length wild-type IDOL (inducible degrader of LDLR; NM 013262, ORF 238-1569) was subcloned into pIRESneo3 mammalian expression vector (Clontech). In HEK293 cells, a single clone expressing LDLR-GFP was generated that was then transfected with the wild-type IDOL construct to create an LDLR-driven GFP reporter cell line (HEK293 LDLR-GFP/IDOL), which was cultured and expanded in DMEM containing 10% v/v fetal bovine serum (FBS) and selection antibiotics hygromycin and geneticin. Cells were screened in OptiMEM (Invitrogen, Carlsbad, CA) containing 1 % v/v fetal calf serum (FCS), 100 U/mL penicillin, and 100 µg/mL streptomycin.
EndoC-BH1 cells 9 were obtained from EndoCells and either cultured in low-glucose DMEM (Gibco, Waltham, MA) or screened in glucose-free DMEM (Gibco) supplemented with 2.8 mM glucose. Both culture and screening media contained 2% w/v albumin from bovine serum fraction V (Roche, Basel, Switzerland), 50 µM 2-mercaptoethanol, 10 mM nicotinamide, 5.5 µg/mL transferrin, 6.7 ng/mL sodium selenite, 100 U/mL penicillin, and 100 µg/mL streptomycin.
THP-1 cells were cultured in suspension in roller bottles rotated at 115 rpm in RPMI containing 10% v/v FCS (PAA) and 2 mM L-glutamine (Invitrogen). THP-1 cells were screened in phenol-red free RPMI containing 10% v/v FCS, 2 mM L-glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin.
Screening Assays
Unless otherwise stated, all reagents were added to microtiter plates using a Multidrop Combi (Thermo Fisher Scientific, Waltham, MA), and centrifugal removal of media from microtiter plates was carried out using a BlueWasher (Blue Cat Bio) at 5 g for 5 s. All test compounds used were solubilized in 100% v/v DMSO.
GPR83 Assay
Fibronectin (Invitrogen) was solubilized at 50 µg/mL in sterile phosphate-buffered saline (PBS) and added to 1536-well Epic cell microtiter plates (Corning, NY), which were centrifuged for 1 min at 300 g. After 1 h equilibration at ambient temperature, excess fibronectin was removed from the wells by either (1) manually flicking out well contents, rinsing with PBS, and flicking again or (2) by emptying centrifugally. GPR83-CHO K1 cells in GPR83 screening media were dispensed into the coated plates at a density of 4000 cells/well. Plates were centrifuged as before, prior to incubation overnight under standard culture conditions. Cell media were removed using the centrifugal washer and Hank’s balanced salt solution (HBSS) containing 2 mM HEPES pH 7.4 and 1.5 % v/v DMSO (Epic assay buffer) added using a vertical pipetting station (V-Prep; Agilent Technologies, Santa Clara, CA). Plates were equilibrated within the Corning Epic System at 26 °C for 60 min, prior to a baseline DMR measurement being taken. To generate compound transfer plates (CTPs), compounds were acoustically dispensed using an Echo555 (Labcyte, Sunnyvale, CA) into 1536-well v-bottom polypropylene microtiter plates (Greiner Bio-One, Kremsmünster, Austria), and HBSS containing 2 mM HEPES pH 7.4 was added to create 3X compound stocks. All single concentration screening was at a final concentration of 1.5% v/v DMSO and a compound concentration of 10 µM. Compound was transferred from the CTP to the Epic cell plate using a V-Prep. The Epic cell plate was placed into the Corning Epic System, and the DMR response was measured.
IDOL Assay
HEK293S LDLR-GFP/IDOL cells in IDOL screening media were dispensed at a density of 3000 cells/well into 1536-well microtiter plates coated with poly-D-lysine (Greiner Bio-One 792091) and incubated under standard culture conditions for 20 h. Test compounds were added using acoustic dispensing to give a final concentration of 10 µM and 0.1% v/v DMSO, and plates were then incubated under standard culture conditions for a further 6 h. Bafilomycin A1 (Invitrogen) was added to a final concentration of 0.75 µM and 0.1 % v/v DMSO. Media were removed using centrifugation, and 3.7% v/v formaldehyde in PBS containing 2 µg/mL Hoechst 33342 (Invitrogen) was added to cell plates. Plates were centrifuged and incubated at ambient temperature for 20 min protected from light. Formaldehyde/Hoechst solution was removed by centrifugation, and wells were washed twice with PBS. Plates were sealed and stored at 4 °C until image acquisition on the Operetta CLS high-content analysis system (PerkinElmer, Waltham, MA) or CellInsight (Thermo Fisher Scientific). Image-based quantification of GFP was carried out using the Compartmental Analysis BioApplication on CellInsight. Whole-well fluorescence intensity (FI) using 420 nm excitation (ex)/520 nm emission (em) was also measured using an EnVision multilabel plate reader (PerkinElmer).
EndoC βH1 Assay
Coating media (DMEM high-glucose [Gibco], penicillin-streptomycin [Invitrogen], 2 µg/mL fibronectin, and 1% v/v extracellular matrix) was added to CellBIND 384-well microtiter plates (Corning). After a minimum of 24 h incubation at 37 °C, the coating solution was removed using centrifugation. EndoC βH1 cells in EndoC culture media were dispensed at a density of 8000 cells/well into coated plates and incubated under standard culture conditions for 24 h. Media were removed using either centrifugation or tip-based aspiration (ELx405 Microplate Washer; Biotek, Winooski, VT) and replaced with EndoC screening media. To generate CTPs, compounds were acoustically dispensed into 384-well v-bottom polypropylene microtiter plates (Greiner Bio-One), and EndoC screening media were added to create compound stocks. Single concentration screening was at a final compound concentration of 10 µM and 0.3% v/v DMSO. A V-Prep was used to transfer compound from CTP to cell plate, and cells were incubated for 68 h under standard culture conditions. Using the EdU Click-iT kit (Invitrogen), cells were treated with 10 µM EdU for 4 h, before fixation, permeabilization, and labeling as per the manufacturer’s instructions. Plates were sealed and stored at 4 °C until image acquisition on the CellInsight using target activation measurement (channel 1: cells identified and segmented by nuclear marker; channel 2: percentage EdU-positive cells).
CellTiter-Blue Cell Viability Assay
Assay-ready compound plates (ARPs) were prepared by acoustically dispensing compounds into 1536-well black microtiter plates (Greiner Bio-One). Single concentration screening was at a final compound concentration of 50 µM and 0.5% v/v DMSO. For concentration response (CR) screening, a range of volumes was dispensed to create 10-point curves with a final compound concentration range between 100 µM and 2 nM. Wells were backfilled with the appropriate volume of DMSO to ensure a consistent final screening concentration of 0.5% v/v. THP-1 cells in THP-1 screening media were dispensed at a density of 1000 cells/well into ARPs and incubated under standard culture conditions for 48 h. CellTiter-Blue Cell Viability Reagent (Promega, Madison, WI) diluted 1:16 in THP-1 growth media was then added before incubating under standard culture conditions for a further 2 h. Plates were briefly centrifuged at 300 g prior to measuring FI (ex 540 nm/em 590 nm) using an EnVision multilabel plate reader.
Data Analysis
All assays included control wells. Neutral wells (0% compound activity) contained a matched concentration of DMSO to compound wells. Inhibitor/stimulator wells (100% compound activity) contained an assay-specific control compound. All screening data were analyzed using Genedata Screener. The activity of each test compound was normalized to the median values of neutral and inhibitor/stimulator control wells, apart from the GPR83 assay, in which only neutral control wells were used.
For the GPR83 assay, compound activity was determined by the DMR response at the final frame of the read after compound addition (20 min unless otherwise stated).
IC50 values reported for the CellTiter-Blue viability assay were calculated by nonlinear regression fitting to a four-parameter logistic model using the automated curve-fitting technology in Genedata Screener.
TIBCO Spotfire 6.5.2 was used to generate various scatter plots, box plots, and comparison circles.
Results and Discussion
As the pharmaceutical and biotechnology industries strive to become more successful, they must face the challenge of using more sophisticated and expensive cellular assay systems in HTS. Miniaturization of assays and use of robotic automation can significantly decrease both screening times and the amount of cells and media needed to optimize, validate, and screen complex cell assays. Miniaturization may, in fact, be the only way certain screens can be run as an HTS. Our results indicate that by adopting several new techniques and technologies, it is possible to overcome some of the challenges faced when miniaturizing cellular assays to 1536-well format.
Centrifugal Plate Washing Maintains the Cell Monolayer and Is an Enabling Alternative to Traditional Tip-Based Aspiration, Particularly for 1536-Well Format Assays
Washing steps are a frequent requirement in cell-based assay protocols, particularly for imaging assays requiring fixation, permeabilization, and staining steps. Traditionally, washing protocols have employed tip-based aspiration or manual flicking of plates for removal of liquid from wells. An alternative technology was needed to facilitate miniaturization of many phenotypic assays as most commercial tip-based plate washers are either not compatible with 1536-well plates or in the case of unfixed cells wash too vigorously, leading to removal of some or all of the cells in the wells of repeatedly washed plates. The centrifugal plate washer offers an alternative to tip-based aspiration by removing media via centrifugation. A number of features of the centrifugal plate washer used in this study also facilitated integration onto Agilent and HighRes Biosolutions robotic systems used for automated HTS in our laboratory. These included a small device footprint, accessibility of the plate nest by robot grippers, less than 30 s for complete plate emptying, and no requirement for either compressed air or vacuum lines to run.
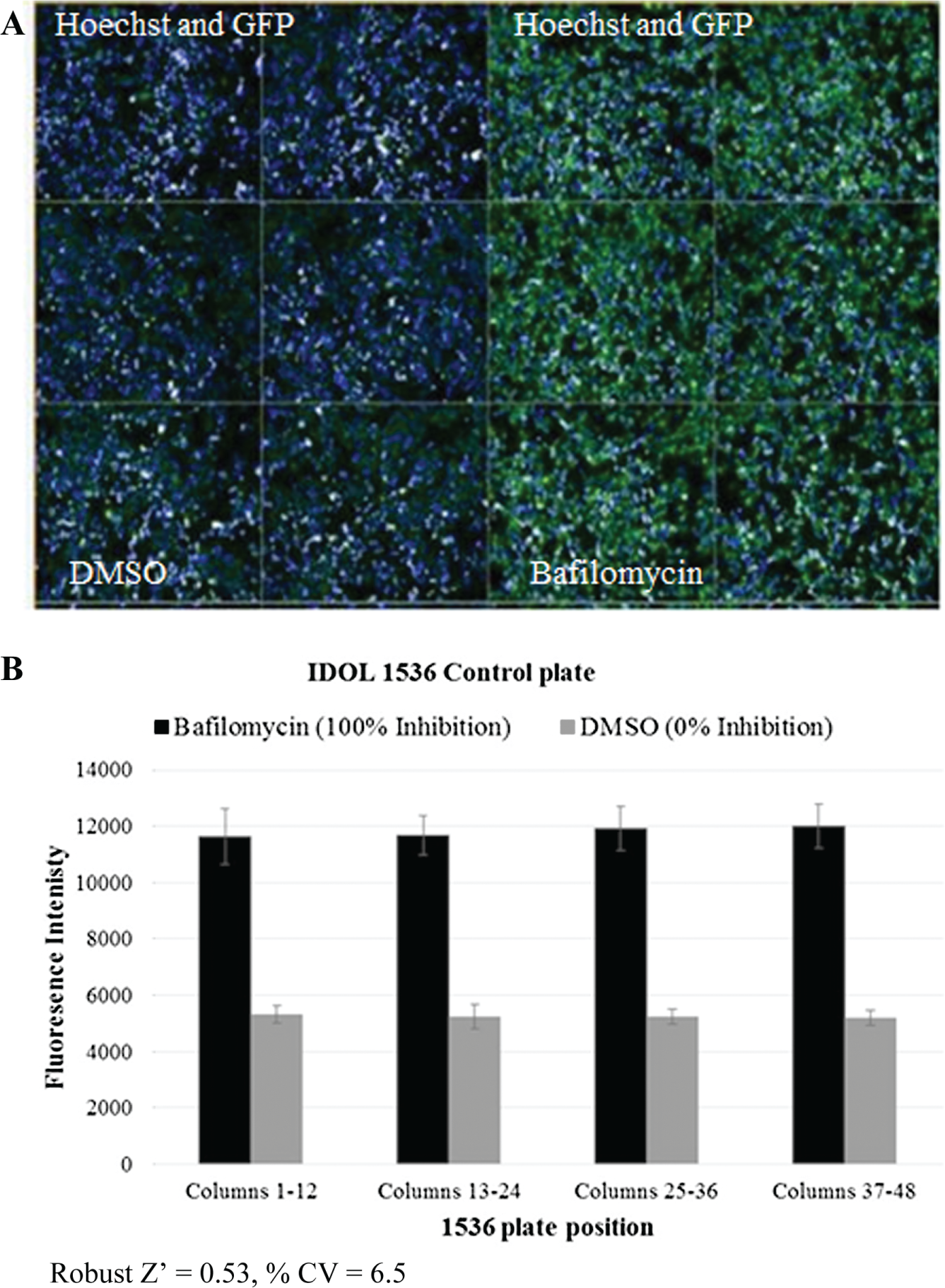
A disrupted monolayer is a frequent obstacle in many cell-based assays when using tip-based washing steps, often leading to cell loss and variability. An even monolayer and minimum cell loss is demonstrated for HEK293 LDLR-GFP/IDOL cells after fixation with two centrifugal washing cycles of 1536-well plates ( Fig. 1A ). This shows that the centrifugal plate washer offers an improved washing method by reducing the risk of monolayer disruption often associated with tip-based aspiration. Employing centrifugation for washing steps requires optimization of both time and speed of centrifugation but is more adaptable to changes in plate dimensions than tip-based aspiration, which requires optimization of the correct height of the aspiration and dispense tips relative to the plate dimensions.
(
Reduced Residual Volume with Centrifugal Plate Washing Reduces Liquid-Handling Steps in Several Cell Assays while Achieving Comparable Assay Quality in 384- and 1536-Well Plates
Reducing liquid-handling steps is advantageous in an HTS setting, particularly when using 1536-well plates, as it reduces robotic handling steps and minimizes potential variation introduced during washing steps. Residual liquid associated with tip-based aspiration in 384-well plates frequently leads to a requirement for numerous washing cycles (typically three to five cycles of aspiration and dispense steps) after media change, fixation, permeabilization, and fluorescent staining. Centrifugal plate washers (we use the BlueCat Bluewasher) can reduce this residual volume from 10 to 15 µL for typical 384-well cell assays using tip-based aspiration to <0.2 µL, reducing the need for so many wash steps. When reagents are added back into the plate, the reduced residual volume leads to reduced dilution of these components. We exemplify this with a reduction in washing cycles while maintaining assay quality (Z’, % CV and background) when an IDOL assay was miniaturized from 384- to 1536-well format. Z′ is a measure of statistical effect size and uses the means and standard deviations of controls to quantify the suitability of a particular assay for use in screening. 10 Control plates with neutral (DMSO) and inhibitor (bafilomycin A1, prevents lysosomal degradation of LDLR 11 ) controls, and plates with neutral controls only, were used to assess the quality of the assay in 1536-well format. After fixation of HEK293 LDLR-GFP/IDOL cells, 1536-well plates were washed twice by centrifugation. The IDOL 1536-well assay achieved Z′ = 0.53, % CV = 6.5%, and signal/background ~2 ( Fig. 1B ) using whole-well FI. This was similar to the historic IDOL 384-well data, which employed three tip-based wash cycles achieving Z′ = 0.6, % CV = 5%, and signal/background ~2 (data not shown) using whole-well FI. The IDOL phenotypic assay was previously used to screen ~1.8 million compounds over a period of 10 weeks. Miniaturization of the assay to 1536-well high-content format enabled by the Bluewasher would reduce screening time by fourfold and reduce the number of cells required to complete the screen by 50%.
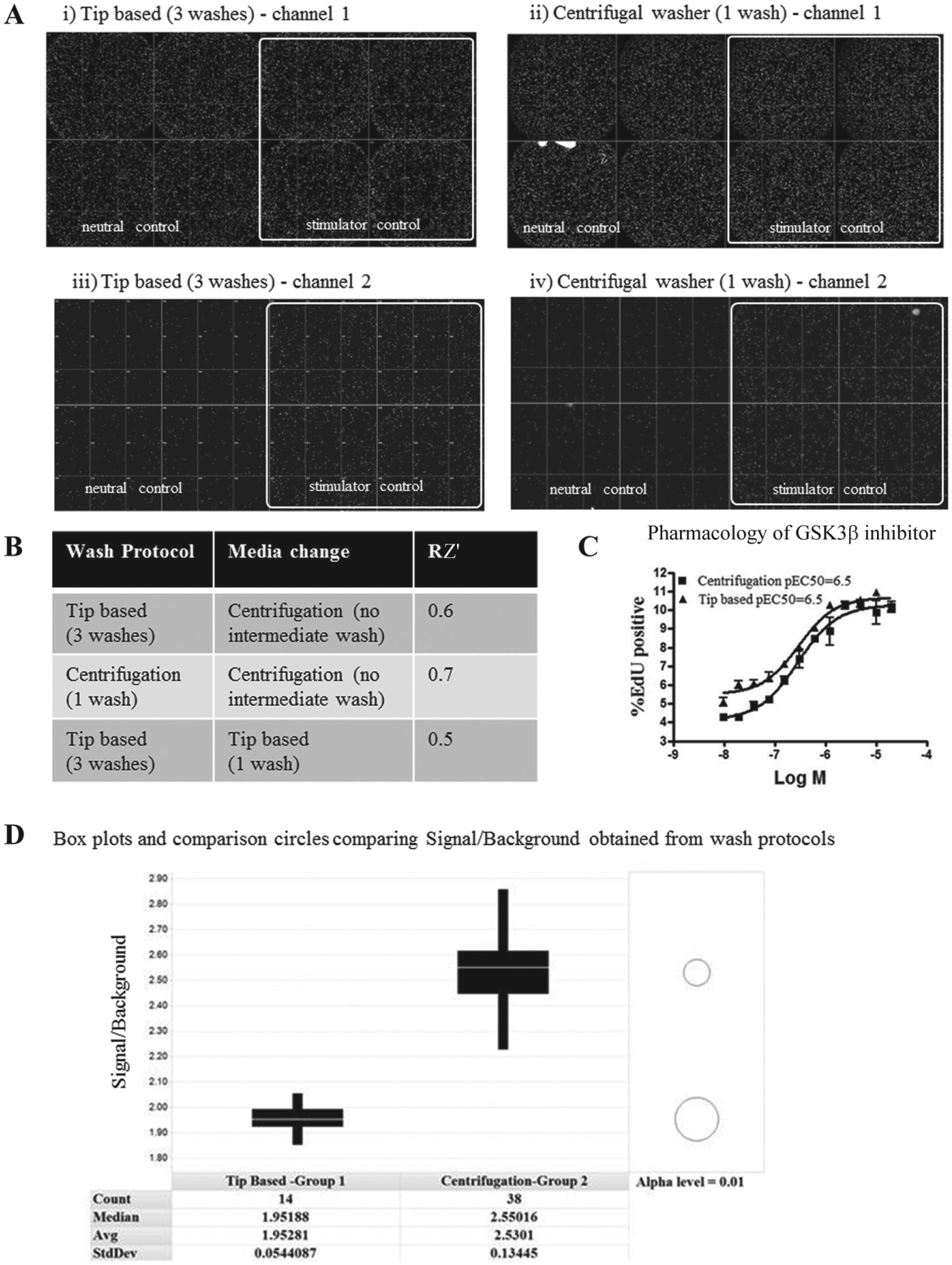
We performed a direct comparison of assay quality using either tip-based aspiration or centrifugation in the EndoC βH1 proliferation assay after fixation, permeabilization, and fluorescent staining. This demonstrated that a reduction in number of washing cycles (from three with tip-based aspiration to one with centrifugation) resulted in identification of similar cell numbers in channel 1 and EdU-positive cells in channel 2 ( Fig. 2A ). Control plates with neutral (DMSO) and stimulator (glycogen synthase kinase 3 [GSK3β] inhibitor—GSK3β has previously been shown to drive proliferation of pancreatic β-cells 12 ) controls were used to evaluate the quality of the assay. Employing three washing cycles with tip-based aspiration yielded Z′ = 0.6, whereas one wash cycle with centrifugation resulted in Z′ = 0.7 ( Fig. 2B ). Glucose starvation and serum starvation steps are frequently required in cell-based assays and require media change and washing steps on live cells, which can lead to cells detaching from microtiter plates. The EndoC βH1 cell proliferation assay required a glucose starvation step, and when the centrifugal plate washer was used to remove media, the low residual volume meant that no additional washing step was required prior to adding glucose starvation media and showed improved results (Z′ = 0.6–0.7) over tip-based aspiration (Z′ = 0.5; Fig. 2B ). A pEC50 of 6.5 was measured in both protocols using a GSK3β stimulator control ( Fig. 2C ), highlighting the suitability of deploying the centrifugal plate washer for the EndoC βH1 proliferation assay.
(
The advantages of using centrifugation in a phenotypic HTS were further exemplified in the EndoC βH1 cell proliferation assay, in which an additional wash step during media change was found to be unnecessary, and the signal/background was also improved. A 14-plate 384-well screen employing tip-based aspirations and one intermediate wash resulted in a signal/background of 1.9, whereas a similar 38-plate 384-well screen employing centrifugal washing resulted in a significantly improved signal/background of 2.5 ( Fig. 2D ). Minimizing washing of live cells without compromising assay quality will benefit 1536-well formats by limiting cell loss and reducing assay variability. Even though the EndoC βH1 proliferation data were generated in 384-well format, the reduction in number of wash cycles further demonstrates the potential of centrifugal washing to enable 1536-well miniaturization by reducing the liquid-handling steps and also the number of robotic steps required to process the plates.
Reduced Residual Volume with Centrifugal Plate Washing Enables Standardized Protocols for Coating 1536-Well Plates and Improves Assay Performance
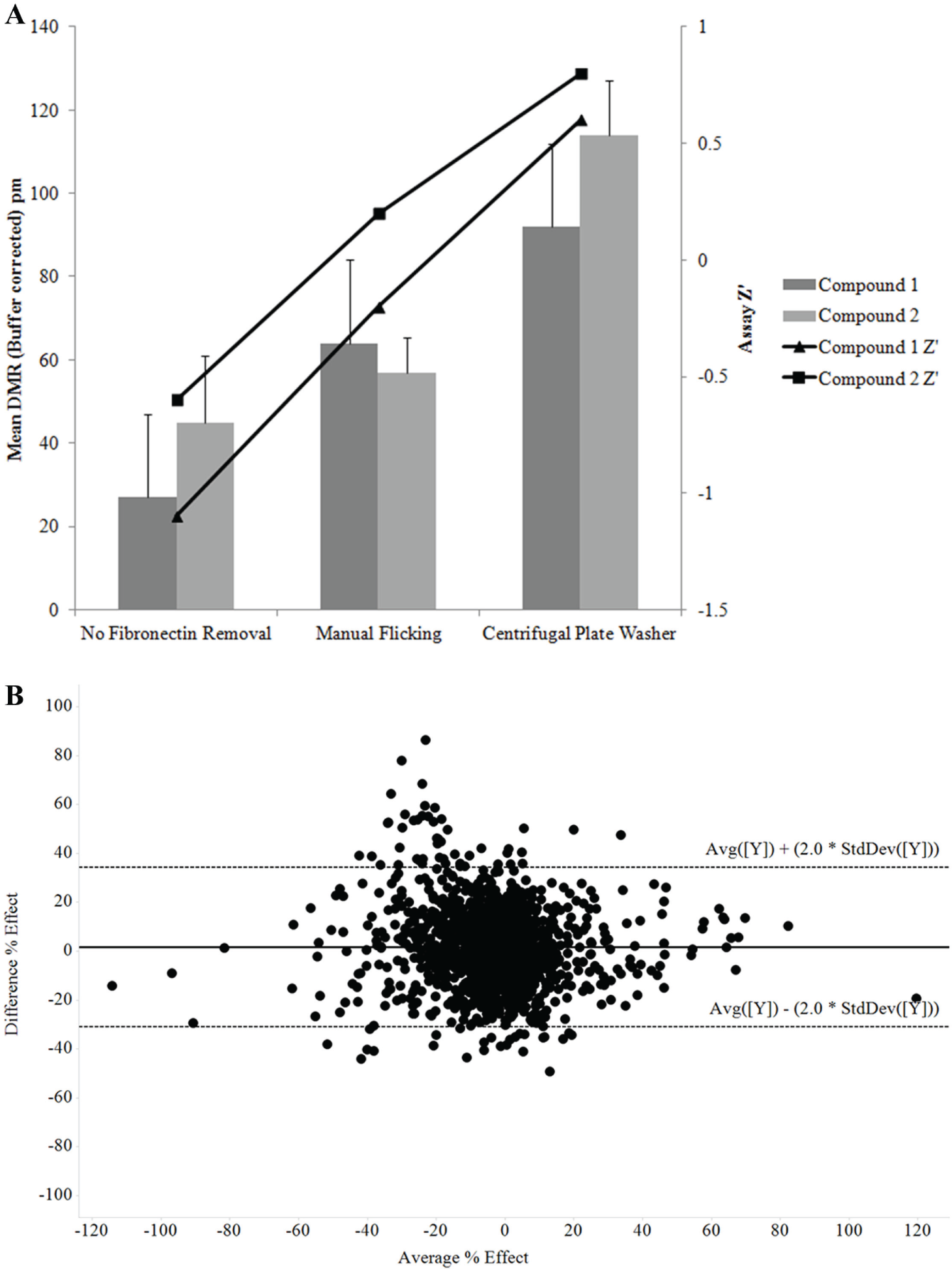
GPR83-CHO K1 cells require fibronectin-coated plates for optimal adherence and growth, and although Epic 1536-well plates precoated with fibronectin are commercially available (Corning), it would reduce costs to develop an in-house protocol for coating plates. Removal of excess fibronectin post coating was essential to maintain assay robustness as the excess fibronectin reduced the DMR response and Z′ generated using two tool stimulator compounds ( Fig. 3A ). The low residual volume achieved using centrifugation to remove excess fibronectin eliminated the requirement for a PBS wash step when using manual flicking of the plates and improved DMR responses and Z′ values ( Fig. 3A ). To compare commercial and in-house fibronectin-coated 1536-well Epic plates, a set of 1408 compounds was tested at 10 µM in the DMR GPR83 assay. A Bland Altman plot of these data ( Fig. 3B ) shows that there is no bias between the two methods of plate coating and that 95% of data points lie within the 95% limit of agreement, which is as we would expect with cell-based assay replicate data. It was therefore concluded that coating plates in house with fibronectin was an acceptable method to enable screening of larger compound libraries at lower cost. Use of centrifugal plate washing for both in-house plate coating and media exchange steps is enabling for HTS. The single-concentration screening capacity of the GPR83 DMR assay using a single Epic device increases from ~3000 compounds per day in 384-well format to ~14,000 compounds per day in 1536-well format, allowing libraries of greater than 100,000 compounds to be tested in HTS. Using two benchtop Epic devices would enable testing of more than 1 million wells in less than 2 months.
(
Compatibility of 1536-Well Imaging Plates with High-Content Imaging Systems and Automation Equipment
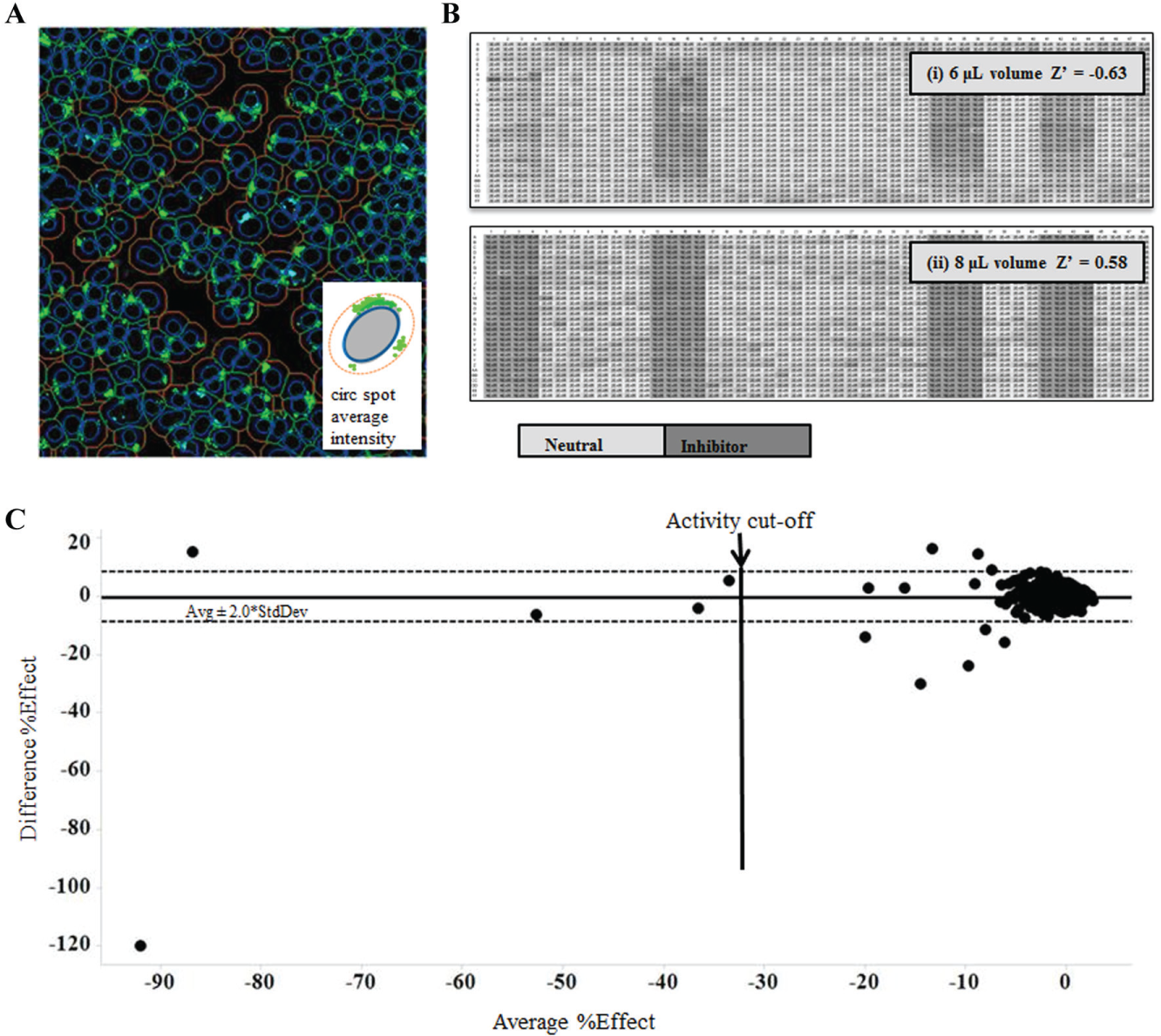
Several 1536-well plates previously investigated for microscopy-based assays were found to be incompatible with imagers and/or automation platforms used in HTS because of their raised or lowered well position, leading to issues with image focus and access for optics and dispensers or reduced plate height, leading to issues with robot gripper compatibility with lids, well content evaporation, and imager stage depth. The Greiner Bio-One 1536-well plate used for the 1536-well format of the IDOL assay combines a full-depth well with a plate height of 11.2 mm, compliant with the Society for Biomolecular Sciences standards. 13 This plate type is compatible with our automation with a variety of lids while maintaining the crucial low well base for imaging applications. To assess the compatibility of the 1536-well IDOL assay with the CellInsight imager, an algorithm was developed using the Compartmental Analysis BioApplication, which identified cells (primary objects) using Hoechst nuclear marker in channel 1 and quantified the level of GFP in channel 2 using circ spot intensity (average intensity of punctate objects) within the circ compartment (a cellular region derived from the area covered by the primary object; Fig. 4A ). The 1536-well IDOL assay was then used to screen a set of 1408 compounds at 10 µM with compounds dispensed into two plates into different well positions on each plate. Control plates (neutral (DMSO) and inhibitor (bafilomycin A1 controls) demonstrated the robustness of the assay (Z′ = Z′ = 0.58; Fig. 4B ). Reproducibly active compounds were identified based on a cutoff of ≥30% inhibition, resulting in a 0.3% hit rate, similar to that obtained with a previous FI 384-well format HTS screen (0.5%; Fig. 4C ). The 1536-well IDOL assay demonstrated the compatibility of miniaturized image-based assays with HTS imaging systems and automation equipment.
(
Plate Seals and Increased Assay Volumes Reduce Edge Effect Patterns
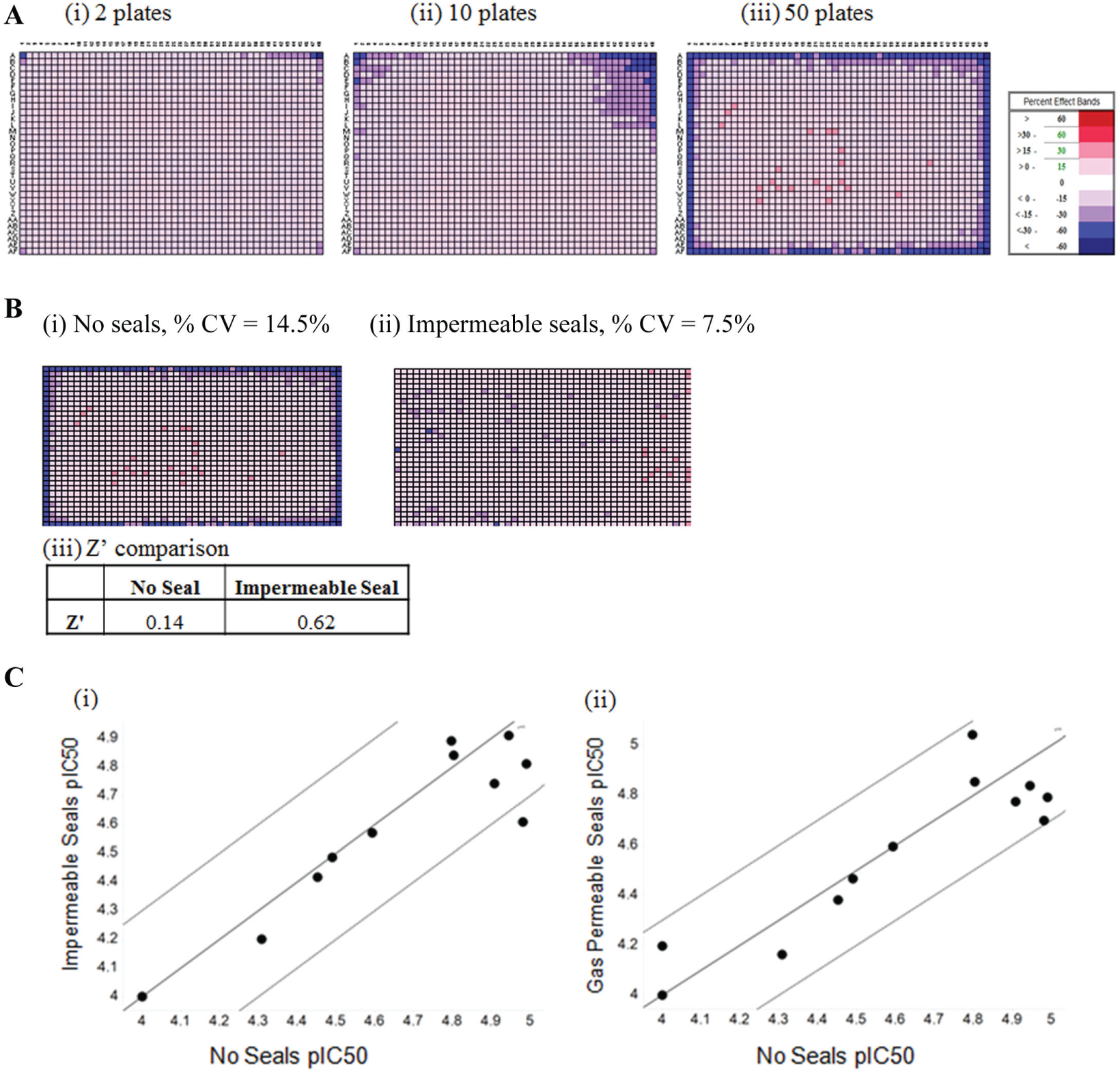
An edge effect pattern occurs in which liquid evaporates from the outer wells of microtiter plates during incubation steps. Edge effects are particularly problematic in 1536-well cell assays with their smaller volumes, especially when longer incubation periods are required. In an attempt to alleviate edge effects, researchers often decide not to culture cells in the outermost wells but to fill these with sterile water and use only the inner wells of microtiter plates. However, not using the outer wells reduces throughput and increases costs. A CellTiter-Blue cell viability assay was miniaturized to 1536-well format to increase throughput for an automated HTS to identify cytotoxic compounds. A similar cell viability assay was previously used to identify cytotoxic agents against a small library of 2000 compounds. 14 However, this study did not address the edge effects encountered at an HTS scale of screening. To determine if the 1536-well assay was suitable for screening at large scale, larger plate batch sizes were evaluated. Problematic edge effects were observed for 10 and 50 plate batches, compromising the scalability of the assay ( Fig. 5A ). To determine if plate seals could improve the observed edge effects, commercially available impermeable and gas-permeable seals were assessed. The variability of cell growth in 1536-well plates containing neutral controls (DMSO) in all wells was assessed using CellTiter-Blue with clear impermeable seals or no seals at positions 1 to 10 and positions 40 to 50 of a typical 50-plate batch test. Employing impermeable seals reduced the % CV from 14.1% to 7.5% and eliminated the edge effect observed with no seals and removed the requirement to exclude the outer wells of the 1536-well plate ( Fig. 5B ). Previous in-house data generated using the 1536-well CellTiter-Blue viability assay demonstrated that clear impermeable plate seals were as effective at reducing edge effects as gas-permeable plate seals designed to allow gas exchange. In addition, the Z′ of 1536-well control plates with maximal neutral control (DMSO) and minimal inhibitor control (Puromycin) dispersed across the plate (including outer wells) was increased from 0.14 with no seals to 0.62 with impermeable seals, again reflecting the improvement in assay performance by the use of plate seals ( Fig. 5B ). To determine if plate seals were affecting the overall viability of the cells or pharmacology over prolonged incubations, 12 AZ compounds from the AZ library were tested in CRs. The majority of pIC50 values generated using impermeable or gas-permeable seals showed good agreement (y = x ± 0.3 log) with those generated using no seals, demonstrating that any possible reduction in gas exchange due to plate seals did not affect the pharmacology of key compounds in the CellTiter-Blue cell viability assay ( Fig. 5C ). The CellTiter Blue assay was used to screen 500,000 compounds at a single concentration prior to the findings of using plate seals to improve assay performance, resulting in a 20% loss of compounds screened, as the outer wells of the plates had to be excluded.
(
Problematic edge effects were also observed during optimization of the 1536-well IDOL assay. By increasing the volume of the assay from 6 µL to 8 µL, the Z′ of the quality control plates (neutral [DMSO] and inhibitor [bafilomycin A1] controls) improved from <0 to 0.58 ( Fig. 4B ). The higher volume likely reduced the effects of evaporation, which can change the concentration of salts and nutrients in the culture media in outer wells compared with the wells located in the center of the microplate.
Taken together, the use of plate seals and assay volume optimization can eliminate the need to exclude outer wells of 1536-well cell assays, thereby improving the throughput of such assays carried out at large scale.
Miniaturization of cell assays into 1536-well plates is estimated to reduce the cells required for a typical HTS by up to 50% and reduces the time taken to run a typical HTS campaign by up to fourfold. This article describes several techniques that, when employed, allow the minaturization of several phenotypic cell assays into 1536-well format, which have previously been limited to 384-well format. The novel application of a centrifugal plate washer (BlueCat Bluewasher) enabled the miniaturization of a range of 1536-well cell assays, reducing the number of washing cycles in phenotypic assays requiring fluorescent staining and facilitating in-house 1536-well plate coating. Techniques to reduce edge effects often observed with 1536-well cell assays also improved the throughput of cell assays and decreased the variability seen in such assays. Cell assays currently limited in throughput due to cost and complex washing protocols can benefit from the techniques presented in this study and enable the drug discovery industry to harness the improved translation to human disease associated with using primary and iPSC cells in the critical initial hit-finding stage.
This study has introduced technologies and techniques that enable miniaturization of cell assays to 1536-well format. However, liquid handling and cell dispensing remain a challenge. Application of emerging technologies such as bioprinting, which offers improved control of cell density and precision on spatial placement of cells and growth factors in a high-throughput manner, may also help to further deploy miniaturization of phenotypic assays in drug discovery.
Footnotes
Acknowledgements
The authors thank BlueCatBio for helpful technical support. We also thank Darren Plant and Brett Litten for extensive automation support, especially with the Agilent and HighRes Biosolutions robotic systems and Matthew Collier for his support in developing and screening the EndoC βH1 proliferation HTS. We acknowledge Johan Brengdahl and Tracy Gorman for development of the EndoC βH1 proliferation assay and Alleyn Plowright, David Smith, and Beverley Isherwood for guidance and support during HTS screening. We thank Paul Sharpe for generating the THP-1 cell bank and Eileen McCall for helping to generate the EndoC βH1 cell bank. We also thank Helen Boyd for supporting development of the 384-well format of the IDOL assay and guidance during IDOL HTS screening.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
