Abstract
Polyethylene terephthalate (PET) fibers have been widely used in various industrial fields due to their excellent chemical and physical performance. However, there is still a crucial problem of the massive precipitation of oligomers from the interior to the surface of product, which results in decreased production efficiency and harm to human health. A quantitative characterization method for surface oligomers of PET fibers based on Soxhlet extraction was developed, with lower-temperature extraction, high accuracy, excellent extraction effect and wider operability. The extraction temperature (Toe) decreased by 30°C compared to the conventional extractant of tetrachloroethylene (Te-Ex), benefiting from the trichloroethylene (Tr-Ex) extractant. The pre-wash process with trichloroethylene could ideally remove oil impurities of about 0.09% but retain the surface oligomers on the fibers, endowing the method of with promising accuracy. PET staple fibers with various contents of surface oligomers were extracted with Tr-Ex, and the achieved contents (i.e. 0.320%/0.527%/0.876%) were similar to those with Te-Ex (i.e. 0.307%/0.516%/0.828%), and the contents with Tr-Ex were all slightly higher than those with Te-Ex, indicating that the extraction efficiency of Tr-Ex improved by ∼4.10% compared with that of Te-Ex. It was confirmed that Tr-Ex method has excellent accuracy compared with as the Te-Ex method and better extraction effect than the Te-Ex method; maybe possibly dipole–dipole interaction between trichloroethylene and oligomer molecules was responsible for the better effect. This quantitative characterization method has potential application in industrial online measurement of surface oligomers on PET fibers, films and related materials.
Introduction
As a typical polyester material, polyethylene terephthalate (PET) polymers have been widely used in fibers,1,2 packaging bottles 3 and films 4 fields due to their excellent chemical and physical performance and multiple functions. However, there is still a crucial problem in the complex- and post-processing process5–7 with adverse effects on production efficiency, product quality 8 and human health,9–11 which is caused by certain equilibrium content of cyclic oligomers (PET oligomers) formed during polymerization12,13 and precipitated from the interior of the PET to the surface, forming a powder or granules (PET surface oligomers). With the expansion of application fields and the increasing social requirements for the environment and product quality, investigating and regulating the precipitation of oligomers on the surface of PET products has become a research hotspot in the industry.14–16
Extensive research, focusing on the formation process, mechanism and characterization methods of PET total oligomer, including surface and interior oligomers, shows that the total oligomer content is in the range of 1–3%,5,17 and cyclic oligomers are the main component with cyclic trimers the highest.18,19 Forming via cyclization reactions between intramolecular terminal hydroxyl and carbonyl groups, the oligomer content is affected by multiple factors, including the mass ratio of ethylene glycol/terephthalic acid during polymerization, polymerizing temperature and catalyst categories.20,21 Resulting from the complicated manufacturing and processing, some interior oligomers could migrate to the PET surface, mainly reliant on the changes in processing temperature and polyester crystallinity.22–24 These research works provide effective technical support for reducing the generation and precipitation of PET oligomers.
Even though significant progress has been achieved in exploring the properties and structure of oligomers on PET fibers, quantitative characterization research on surface oligomers of PET fiber is insufficient to adapt to the rapid development of industrial applications and environment protection. Owing to the content of surface oligomers greatly varying owing to subsequent processing conditions,12,22 developing an accurate, economic and operational quantitative characterization method to determing the surface oligomer content is of great significance for regulating the precipitation of oligomers to further solve the major engineering problems. Some qualitative and quantitative methods have been investigated to characterize surface oligomers of PET fibers. Qualitative characterization based on Scanning Electron Microscopy (SEM), a promising method to determing the content and attachment state of surface oligomers,5,6 is limited for wide use due to the high cost of the equipment and lack of a quantitative analysis function. Extraction combined with weighing the oligomers requires a quantitative characterization method. Soxhlet extraction, selecting tetrachlorethylene as the extractant (Te-Ex) to achieve the extraction, combined with high-efficiency liquid chromatography5,14,25 or gravimetric analysis, 9 is an effective method to determine the content of PET surface oligomers. Ultrasonic-assisted stirring extraction, 26 with the chopped sample placed in dioxane and stirred at high speed, is operated at room temperature. After the extractant is distilled off, the oligomers and impurities are firstly separated by thin-layer chromatography, and then the oligomer content is measured using a spectrophotometer.27,28 However, the chopped sample promotes the precipitation of oligomers from inside the fibers, resulting in the a measured value that is higher than the actual value. Hydrothermal extraction is an eco-friendly method, which also leads to PET hydrolysis and destroys the PET body structure to some degree. 29 Supercritical carbon dioxide extraction is a cleaner and less expensive method compared with other conventional methods, 30 which need a much higher pressure of 22 MPa during extraction. Therefore, compared with the methods of ultrasonic-assisted stirring extraction, hydrothermal extraction and supercritical carbon dioxide extraction, Soxhlet extraction, especially combining with gravimetric analysis, is a more effective method for quantitative characterization, without excessive extract, destruction of the structure, high pressure and expensive equipment.
However, the extractant of tetrachloroethylene, used in traditional Soxhlet extraction, easily to condenses due to its high boiling point, which makes it difficult for tetrachloroethylene vapor to enter the extraction tube at low room temperature, and even results in inoperability of experiment. Far greater consumption of heat energy and safety hazards could result from the use of a high temperature. In addition, the gravimetric method isn’t accurate enough, on account of some oil impurities adhering to fibers during processing have that are not removed. As a result, constructing an available quantitative characterization method with advantages of strong operability, low heat consumption, high accuracy and greater safety, has become an important issue.
In this paper, an effective quantitative characterization strategy for determining surface oligomers of PET staple fibers (PETS) based on Tr-Ex Soxhlet extraction, a pre-wash process and a gravimetric method was developed. In this method, trichloroethylene with a relatively low boiling point was selected as a novel extractant to improve the experiment operability and decrease heat consumption, and a new process of pre-washing was devised to increase the experimental accuracy by removing oil impurities. Quantitative characterization with various contents of surface oligomer was conducted by this method, and the experimental accuracy and process feasibility were systematically discussed. The quantitative dates and SEM images indicated that the Tr-Ex method has better extraction effect than that the traditional Te-Ex method, and dipole–dipole interaction between trichloroethylene and oligomer molecules was speculated to be responsible for the effect. Infrared thermal images revealed that the Tr-Ex method has lower heating and extracting temperature than the of Te-Ex method. This investigation makes a major contribution to surface oligomer quantitative measurement by demonstrating an operational and effective method based on Soxhlet extraction with lower-temperature extraction.
Experimental
Materials
PETS (linear density of 1.33 dtex, length of 38 mm) was purchased from a supermarket (Wuhu, China). Trichlorethylene (AR) and tetrachloroethylene (AR) were bought from Aladdin Reagent (Shanghai, China) Co., Ltd.
Preparation of Samples
Preparation of Fibers With Various Content of Surface Oligomer (TPETS)
Thermal-induction was used to obtain PET fibers with various contents of surface oligomers by heat-treating the fibers at 200°C for various time. Due to the oligomer mainly being formed at about 300°C during polymerization and post-processing, 20 it could only migrate from interior to surface of the fiber without producing new oligomers at a thermal-induction temperature of about 200°C. The PETS was dried for 60 min at 100°C in an oven first. Then, it was treated in a dryer (GT-7017-BL1) at 200°C for 5, 10, 15, 20, 25, 30 and 60 min (Figure 1(a)), respectively. It was cooled naturally to room temperature afterwards. For convenience, the heat-treated PETS for different times was named Tn-PETS, where n is the heat-treatment time (T5-PETS means PETS heat treated for 5 min).
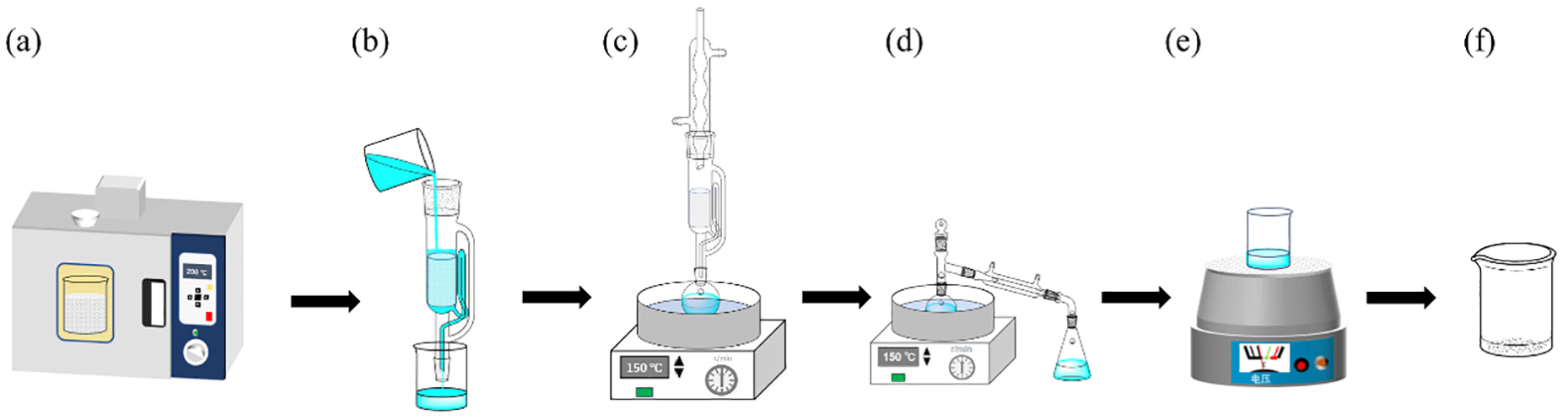
Flow chart of surface oligomer extraction of PETS/TPETS: (a) preparation of TPETS, (b) pre-wash, (c) extraction, (d) distillation, (e) evaporation and (f) PET oligomer.
Pre-wash of PETS/TPETS
To remove the oil impurities, the dried ∼10 g of PETS/TPETS wrapped in filter paper was placed into a Soxhlet -extractor and pre-washed with trichloroethylene three times at room temperature. It was soaked for 5 min each time (Figure 1(b)). The pre-washed samples were dried for 1 h at 100°C for use.
Extraction for Surface Oligomer of PETS/TPETS and Quantitative Analysis
The extraction of surface oligomers of PETS/TPETS is shown in Figure 1(c)–(f), where the oil-bath heating temperature was adjusted with the extractant. Extraction and quantitative analysis based on Tr-Ex method are was shown below.
A∼10 g sample of pre-washed PETS/TPETS wrapped in filter paper was placed into Soxhlet extractor, and extracted with trichloroethylene extractant at an oil-bath temperature of 120°C for 10 h, with a flow rate of 2 mL/min (Figure 1(c)). The PETS/TPETS after extraction was rinsed three times with trichloroethylene and the cleaning liquid was placed in a dry flask for distillation (Figure 1(d)). The obtained oligomer concentrate was transferred to a dry beaker. The cleaning liquid was also poured into the beaker after the flask was rinsed with trichloroethylene three times. Finally, the beaker was heated to evaporate the liquid in an oven at 87°C for 1 h (Figure 1(e)), then cooled to room temperature and weighted (accurate to 0.0001 g). The evaporation was repeated until the mass difference between the latst two times is weighings was less than 0.0003 g to remove the extractant completely. Finally, surface oligomer was achieved (Figure 1(f)). Tr-PETS/Tr-TPETS were obtained after the extracted PETS/TPETS was dried in a fume hood. The experiment was repeated four times to ensure the accuracy of the extraction.
The calculation of the surface oligomer content is shown in equation (1):
where Y is surface oligomer content (%); m1 is the total mass of the beaker and surface oligomers after extraction (g); m2 is the mass of the dry baker (g); and m is the mass of the pre-washed PETS/TPETS (g).
To verify the rationality of the Tr-Ex method, extraction and quantitative analysis based on conventional Te-Ex method were employed for comparison. The process and the calculation accord agree with the above Tr-Ex method except for the following operation. Trichloroethylene was replaced by tetrachloroethylene, and the oil-bath temperature was set to 150°C. The beaker was heated to evaporate liquid in an oven at 121°C for 1 h. Finally, Te-PETS/Te-TPETS was achieved after tetrachloroethylene-extracted PETS/TPETS fibers were dried in a fume hood.
Characterizations
PETS, pre-washed PETS/TPETS, and extracted PETS/TPETS were sprayed with a thin layer of gold, and their micro-morphology and distribution of surface oligomer were observed by SEM (Hitachi S-4800, Japan). The qualitative analysis for surface oligomers of PETS/TPETS was conducted by SEM. An infrared heat imager (Fluke-TC01, USA) was used to obtain temperature photos for various zones of the extraction device.
Results and Discussion
Morphology and Distribution of Surface Oligomers on PETS and TPETS
The morphologies and distribution of surface oligomers on PETS and TPETS are shown in Figure 2. It shows that with the increase of thermal-induction time from 5 to 15 min, both the amounts and the size of surface oligomers on the TPETS showed a gradually increasing trend (Figure 2(a)(d)). In Figure 2(a), there were fewer point-like particles (∼0.47 μm) on the surface of PETS, and most of them are deposited in a loose state. As thermal-induced for 5 min, the surface oligomers on the fiber appeared as irregular particles (Figure 2(b)). The surface oligomers on the fiber gradually showed an irregular lamellar structure in Figure 2(c) and (d), showing a gradual transformation from particles (Figure 2(a)) to irregular lamellae. Meanwhile, when PETS was heat-induced for 15 min, the size (∼1.92 μm) and number of oligomers structure were the largest. The reason for this was that, at 200°C, with the increase in thermal-induction time, more and more internal oligomers continued to migrate to the surface resulting from the high content of interior oligomers in PETS. As a result, the content of surface oligomers on the fiber gradually increased, especially at 15 min, where the interior oligomers and PET polymer reached a new equilibrium. With longer thermal-induction times, the interior oligomers no longer migrated to the surface, resulting in the surface oligomer content on the fiber reaching the highest value. When the thermal-induction time was further prolonged to 20–60 min, the lamellar size continued to increase, especially at 60 min with the size reaching 2.66 μm, and more lamellar layers gradually changed to regular hexagonal (Figure 2(e)–(h)), which is consistent with the results of Schreier et al. 7 and Colombo et al. 8 This was due to the motion and rearrangement of molecules under the heat effect are being conducive to the increase in the lamellae size and the regularization of the oligomer structure.

Surface oligomers of PETS (a) and TPETS with various thermal-induction times (b) 5 min, (c) 10 min, (d) 15 min, (e) 20 min, (f) 25 min,(g) 30 min and (h) 60 min. The insets are the corresponding diameters of surface oligomers in (a–h).
Effect of Removing Oil Impurities for PETSby a Pre-wash Treatment
The PETS fiber morphology and surface oligomer distribution before and after pre-wash and extraction with trichloroethylene are shown in Figure 3. As presented in Figure 3(a) and (b), it is obvious that many irregular particles with different sizes (Figure 3(b), marked with the red arrow) and some oil films (Figure 3(b), marked with the yellow dotted circle) were distributed on the surface of the PETS. The PETS fiber surface after pre-wash became smoother with some oligomer particles and without any oil films (Figure 3(c) and (d)), which proved that pre-washing with trichloroethylene was an effective pathway to remove oil impurities on PETS. The quantitative contents of oil impurities removed by the pre-wash process are shown in Table 1. As illustrated in Table 1, the mean mass difference of PETS before and after the pre-wash was 0.0885% ± 0.0001%, which meant that about 0.09% oil impurities were removed, improving the accuracy of subsequent extraction results. In addition, after extraction with trichloroethylene, the surface of the extracted PETS became smooth with very few irregular particles (Figure 3(e) and (f)) compared to pre-washed PETS (Figure 3(c) and (d)). This further demonstrated that the pre-wash process could effectively remove oil imurities but retain the surface oligomers of the PETS, which verifies the usability of the pre-wash process.

Micro-morphology of PETS before (a, b) and after pre-wash (c, d) and extraction (e, f) with trichloroethylene and distribution of surface oligomers.
The contents of oil impurities for PETS removed by the pre-wash process.

m is the mass of PETS, Δm is the mass of oil impurities removed from PETS.
Accuracy of Quantitative Analysis for PETS Surface Oligomers by Tr-Ex Method
It is obvious that the qualitative content of surface oligomer was adjustable depending on thermal-induction time (Figure 2), gradually increasing until 15 min while remaining stable with particle size growing after 20 min (i.e. surface oligomer concentration was higher with a thermal-induction time of 60 min than with one that of 10 min). In this context, PETS and TPETS thermal-induction times of respectively 10 min and 60 min were selected to quantitatively characterize the surface oligomer content by this new Tr-Ex method, while the conventional Te-Ex method was conducted to verify the accuracy of the new method. As shown in Figure 4, the content of surface oligomers extracted by Tr-Ex increased from 0.320% (PETS) to 0.527% (T10-PETS) and 0.876% (T60-PETS), which was consistent with the SEM result in Figure 2(a), (c), (h), where the surface oligomer increased gradually with thermal-induction time. Similarly, the content of surface oligomers extracted by Te-Ex increased from 0.307% (PETS) to 0.516% (T10-PETS) and 0.828% (T60-PETS). As a result, the increase in the quantitative content for various samples by Tr-Ex was consistent with that of Te-Ex, and the content discrepancies (0.320–0.307%, 0.527–0.516%, 0.876–0.828%, respectively) for the same samples by the Tr-Ex and Te-Ex methods were small, which indicates that the Tr-Ex method had similar accuracy to the Te-Ex method. Interestingly, the contents of surface oligomers of PETS extracted by trichloroethylene (Tr-PETS) were all slightly higher than that of PETS extracted by tetrachloroethylene (Te-PETS) for samples with various thermal-induction times (Figure 4), which confirmed the stability of the Tr-Ex method and the higher extraction efficiency (improvement of ∼4.10%) than that of Te-Ex. Therefore, the combination of trichloroethylene extraction and quantitative characterization of surface oligomers is an accurate and effective test method.
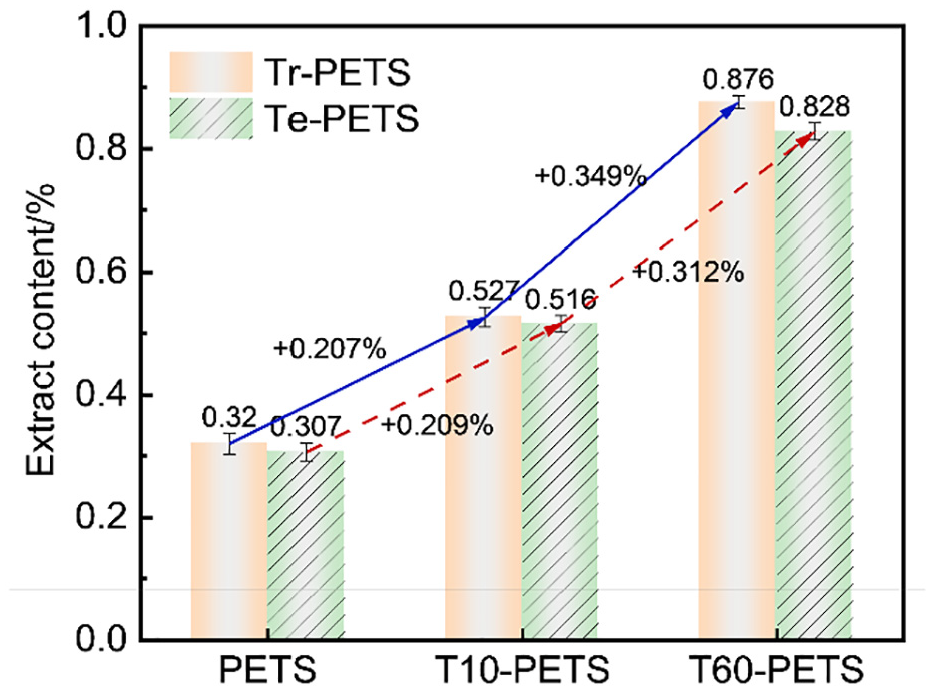
Surface oligomer content of PETS/TPETS based on Tr-Ex and Te-Ex.
To further evaluate the effectiveness of extraction with trichloroethylene compared to tetrachloroethylene methods, the surface morphology and the distribution of oligomer on surface of PETS, TPETS before and after the extraction were observed and are shown in Figure 5. It was obvious that there were granular oligomers, irregular flake oligomers and hexagonal flake oligomers on the surfaces of PETS, TPET with thermal-induction for 10 min (T10-PETS) and 60 min (T60-PETS; Figure 5(a)–(c)). Interestingly, after extraction with tetrachloroethylene, the surfaces of the Te-PETS, Te-T10-PETS and Te-T60-PETS became relative smooth (Figure 5(d)–(f)), and the surface oligomer concentrations were greatly reduced. Compared with the surface of PETS, T10-PETS and T60-PETS extracted by tetrachloroethylene, the surface of that extracted by trichloroethylene was cleaner and smoother and surface oligomers were removed with only trace residual particles (Figure 5(g)–(i)), which is consistent with the result in Figure 4 and demonstrates the better extraction effect of Tr-Ex than Te-Ex.

Surface morphology and oligomer distribution of PETS, T10-PETS and T60-PETS before (a, b, c) and after tetrachloroethylene (d, e, f) and trichloroethylene (g, h, i) extraction.
The Action Mechanism Analysis for Better Extraction of Tr-Ex Method
As described in the above discussion, trichloroethylene is an effective extractant for surface oligomers on PET fibers and it has better extraction than tetrachloroethylene. To clarify the action mechanism for the effectiveness, a solubility parameter of the compounds is proposed for the explanation. It is well known that solubility parameter of polymers is used to predict the compatibility between polymer and solvent; the smaller the difference, the more easily the polymer dissolves. As the literature shows, the solubility parameter of trichloroethylene (9.2 (cal/cm3)1/2) is similar to that of tetrachloroethylene (9.3 (cal/cm3)1/2), 31 which is slightly lower than that of PET oligomers (9.8 (cal/cm3)1/2) 32 and much lower than that of PET (10.7 (cal/cm3)1/2). 31 Thus, both trichloroethylene and terephthalate present good compatibility and diffusion performance with oligomers but poor compatibility and diffusion performance with PET, which means both trichloroethylene and terephthalate have an extraction effect for PET surface oligomers with little influence of on the interior oligomer.
To further investigate the action mechanism for the better extraction of Tr-Ex than Te-Ex, the structure and polarity of trichloroethylene, tetrachloroethylene and cyclic oligomer, and the dipole–dipole interaction between polar molecules33,34 are presented in Figure 6. As can be seen in Figure 6(a) and (b), trichloroethylene is composed of polar molecules with asymmetric polar bonds, while tetrachloroethylene is a nonpolar molecule with symmetrical polar bonds. Cyclic oligomer, the main components of surface oligomers, is a polar molecule as shown in Figure 6(c). It is the dipole–dipole interaction between the polar molecules of trichloroethylene and cyclic oligomers (Figure 6(d)) that facilitates the penetration of trichloroethylene into the oligomers, which is beneficial for a much better extraction effect.
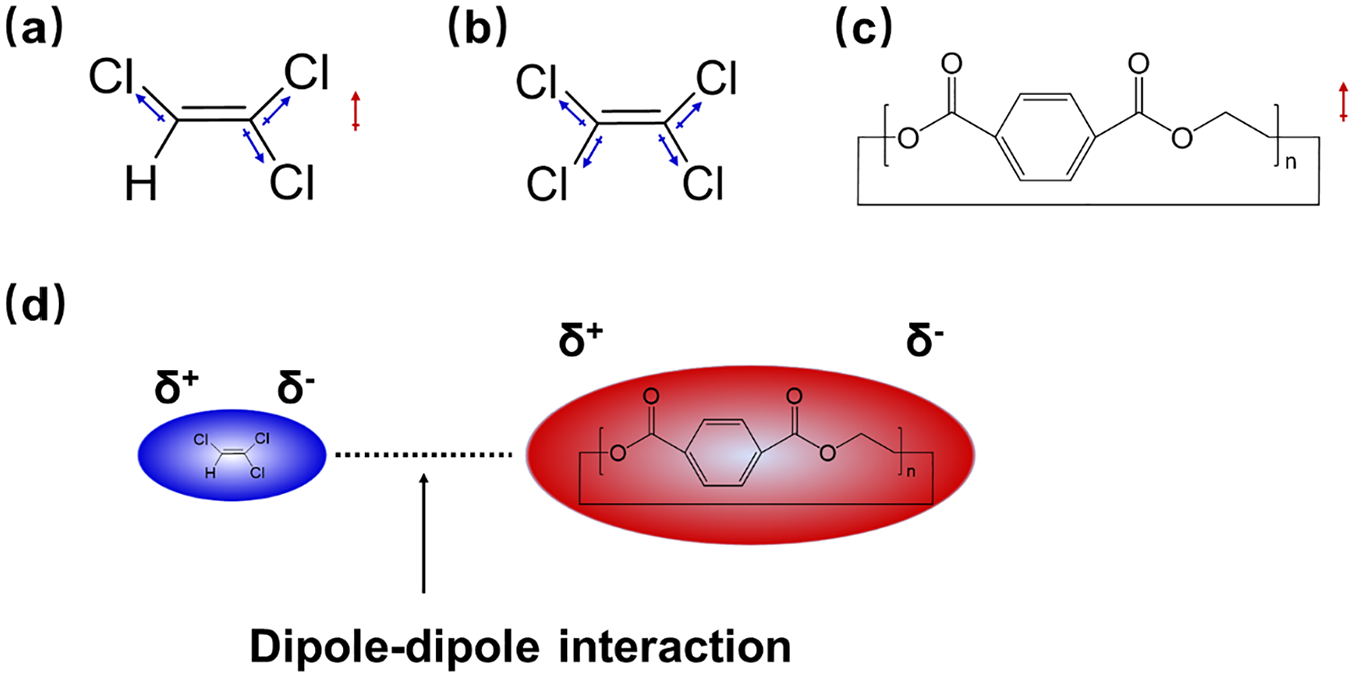
The structure and polarity of trichloroethylene (a), tetrachloroethylene (b), cyclic oligomer (c) and schematic diagram of dipole–dipole interaction between trichloroethylene and cyclic oligomer molecules (d).
Operability of the Tr-Ex Extraction withLower-temperature Extracting
To figure out the feasibility of lower-temperature extraction and the specific extraction temperature (Toe) of trichloroethylene, an infrared heat imager was employed to collect the temperatures of different zones for the extraction device, and tetrachloroethylene was used for comparison. The schematic and experimental device diagram is shown in Figures 1(c) and 7(a), respectively. The experiments were conducted under at normal atmospheric pressure at room temperature of about 16°C, with flow rate of condensate droplets of 2 mL/min. Based on much experimental experience, the optimal oil-bath temperatures of trichloroethylene and tetrachloroethylene were set as 120°C and 150°C, respectively. The instantaneous temperatures are shown in Figure 7(b) and (c), while the specific statistical temperatures of different zones of the device, boiling point and heating temperatures of trichloroethylene and tetrachloroethylene are listed in Table 2.
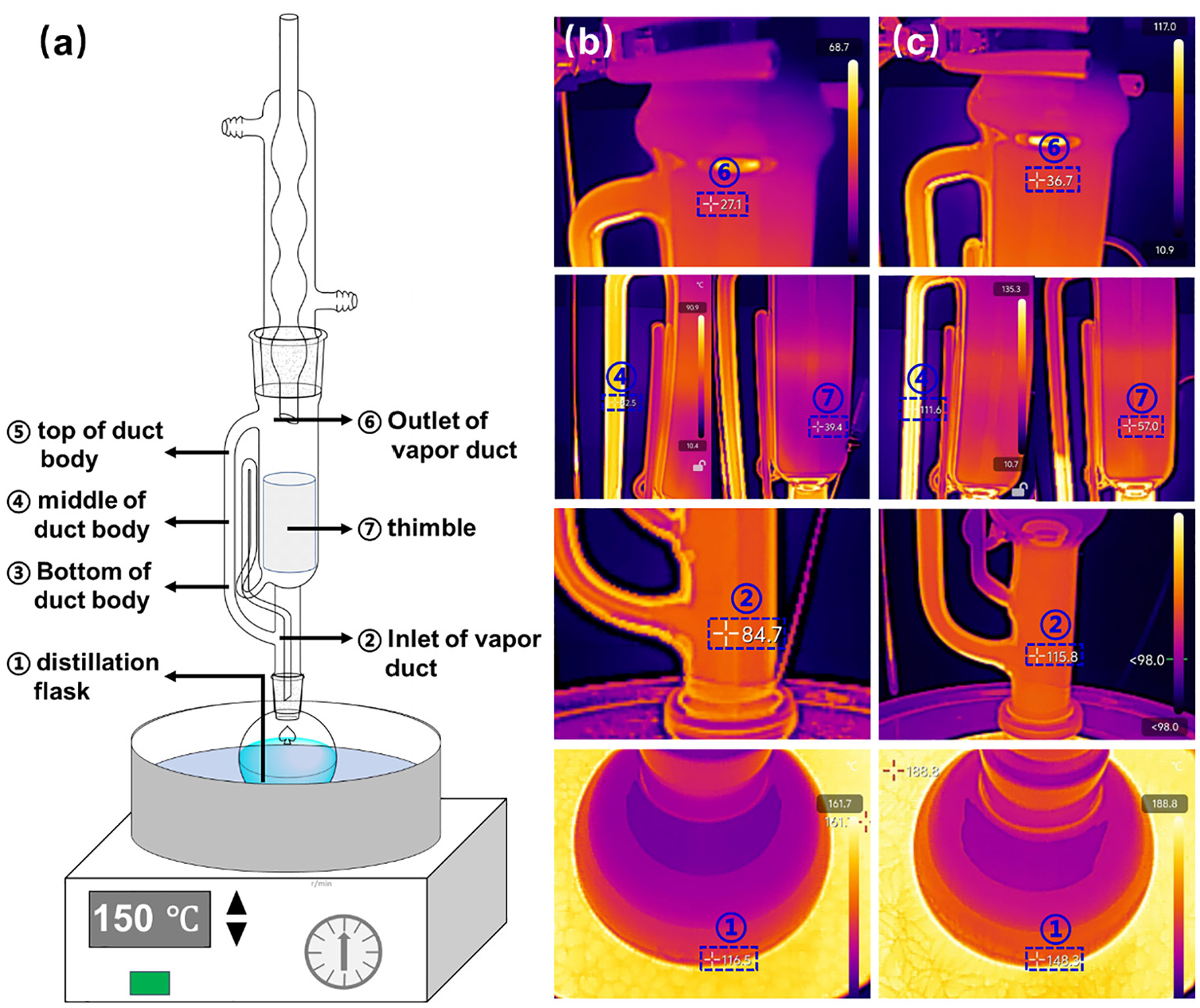
Photo of the extraction experimental device (a), and infrared thermal images of extraction with trichloroethylene (b) and tetrachloroethylene (c).
Temperatures in different zones of the device, boiling point and heating temperatures with trichloroethylene and tetrachloroethylene during extraction.
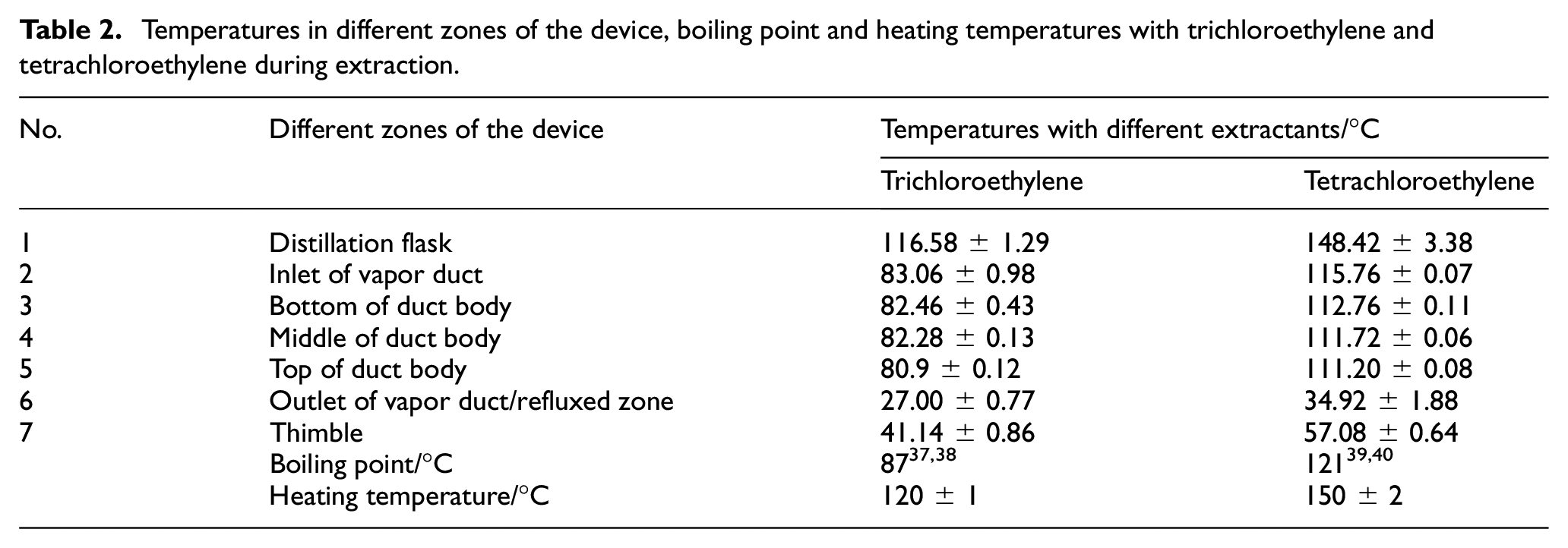
As shown in Figure 7(b) and (c) and Table 2, the heating temperatures were higher than the boiling points, which facilitated extractant boiling and evaporation into the extraction tube, achieving the purpose of extraction. From the data in Table 2, it is apparent that the heating temperature of ∼120°C and refluxed temperature of ∼27°C for trichloroethylene were lower than those of tetrachloroethylene (∼150 and ∼35°C, respectively), which verified that the new Tr-Ex method was of operational at lower-temperature extraction compared to Te-Ex. Moreover, the extractant temperatures at the inlet, body and outlet of the duct decreased successively (83→82→27°C and 116→112→35°C, respectively), and the temperature difference between the inlet and outlet of the duct, was smaller for trichloroethylene (56°C, equals to 83–27°C) than for tetrachloroethylene (81°C, equals to 116–35°C). This further indicated that trichloroethylene with less heat escape could have a lower heat temperature and wider operability compared with tetrachloroethylene, especially at the same low ambient temperature.
As a significant parameter during extraction, the Toe has a crucial impact on extraction efficiency and compound thermal-decomposition performance. The temperatures of the two zones of the device are generally defined as the Toe in the Thimble method and the Distillation Flask method. 35 The boiling temperature of extractant is seen as the Toe in the Thimble method, based on the assumption that it is the condensed extractant with similar temperature to the boiling point in the Thimble method. The set heating temperature was seen as the Toe in the Distillation Flask method, because the temperature of the extractant in the flask is close to the constant heating temperature which facilitated normal operation of the experiment. However, as shown in Table 2, temperatures in the refluxed zone and in the thimble were not similar, and also not close to the boiling temperature. In fact, these temperatures vary depending on the change in the ambient temperature, condensate temperature and extraction time. Therefore, the Thimble method is not appropriate for determining the Toe. As can be seen from the data in Table 2, the temperature of the extractant in the flask was constant and indeed close to the set heating temperature, 116.58–120°C of trichloroethylene and 148.42–150°C of tetrachloroethylene, which indicated that the Distillation Flask method is suitable for determining the Toe. As a result, according to the Distillation Flask method, the Toe of trichloroethylene was determined as 120°C, higher by of about 30°C than its boiling temperature of 87°C.36,37 This method was consistent with that of trichloromethane with a Toe of 80°C,38-40 which is higher by about 20°C than its boiling temperature of 61.3°C. 41
As explained in the above discussion, the Toe of tetrachloroethylene was determined as 150°C, higher by approximately 20°C than its boiling temperature of 121°C.42,43 However, it’s interesting that the Toe of tetrachloroethylene was determined as around 70–80°C while the Toe of trichloromethane was 80°C in the literature. 44 It is obvious that the Toe of trichloromethane was determined according to the Distillation Flask method while tetrachloroethylene was not, which was strange – not employing the same method in the same study. And as demonstrated in Table 2, the tetrachloroethylene temperatures of the refluxed zone and the thimble were respectively ∼35°C and ∼57°C, which was far from 70–80°C. A possible explanation for this might be that the Toe values above 70–80°C of tetrachloroethylene were miswritten or estimated.
Conclusions
An effective quantitative characterization method for surface oligomers of polyester fibers with lower-temperature extraction, high accuracy, excellent extraction effect and wider operability was developed. The method mainly consists of Soxhlet-extraction, pre-wash process and gravimetric analysis. Benefiting from a trichloroethylene extractant, the extraction temperature decreases by about 30°C compared to conventional extractant of tetrachloroethylene. The extraction effect for surface oligomers with trichloroethylene was systematically better than that of tetrachloroethylene; maybe dipole–dipole interaction between trichloroethylene and oligomer molecules was responsible for that. The pre-wash process with trichloroethylene could ideally remove oil impurities but retain the surface oligomers on the fibers, endowing the method with promising accuracy. This quantitative characterization method has potential for application in industrial online measurement of surface oligomers on PET fibers, films and related materials.
Footnotes
Author contributions
Conceptualization, Y.X.; methodology, W.Z. and Y.X. (Yaning Xu); software, S.Z.; validation, W.Y., W.Z. (Wenqiang Zhang) and X.H.; formal analysis, Y.X. and Y.X.; investigation, Y.X. and W.Z.; resources, L.Z. and Y.X.; data curation, Y.X. and W.Z; writing—original draft preparation, Y.X. and W.Z.; writing—review and editing, H.Z. and L.Z.; visualization, Y.X.; supervision, H.Z. and L.Z.; funding acquisition, L.Z. and Y.X. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by Youth the Key Research and Development Plan Project of Wuhu (No. 2022cg18 and 2019yf55), the Joint Open Fund Project of Key Laboratory of Universities of Anhui Province (No. 2018AKLTF13), the National Innovation Program for College Students(no. 202310363051), the Natural Science Foundation of Anhui Province (2208085QE141,2024AH050105), Anhui Polytechnic University Scientific Research Project (nos. FFBK202344, FFBK202367, FFBK202238 FFBK202368) and the Research Project with Chuzhou Seeker Solution Dyed Co. Ltd (no. HX2021-11-002).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon request.

