Abstract
Cotton is the most widely used natural fabric in daily life owing to its excellent properties. However, cotton fabrics are highly flammable and easily cause fire events, thus putting consumers in a tremendously unsafe living environment. Flame retardants (FRs) for cotton fabrics have been widely studied by scientists, including silicon, boron elements, biomolecules and nanomaterials with phosphorus and nitrogen-based compounds. This work summarizes various FRs applied for cotton fabrics and the flame retardant mechanism of different FRs is analyzed. The flame retardancy, combustion behaviors and other properties of the treated cotton fabrics are reported. The treatment methods with the advantages and disadvantages for FRs cotton fabrics are also discussed.
Introduction
Cotton fiber is one of the most widely used natural fibers, which has excellent properties such as renewability, biodegradability, softness, and breathability, 1 and has a wide range of applications in the clothing, house furnishing, industrial and aerospace, military, and other industries. However, the limiting oxygen index (LOI) of cotton fabrics is only 18%, which makes it flammable. 2 Textile combustion causes fire threats to people’s lives and properties, so the flame-retardant treatment of cotton fabrics has important practical significance.
The earliest flame retardants (FRs) applied in cotton fabrics were halogen-containing FRs, which provide cotton fabrics with a good flame-retardant effect with a small cost and at the same time have little impact on the mechanical properties of fabrics. However, halogen-containing FRs produce toxic substances during application, which has a negative impact on the environment and personal safety. At present, halogen-containing FRs are banned by the European Union and many other countries. Therefore, halogen-free and environmentally friendly FRs have become a the research topic of scientists. Many other elements have shown effective flame suppression properties, such as phosphorus, nitrogen, magnesium, aluminium, boron, sulfur, silicon, etc. Varieties of halogen-free FRs containing the above elements have been developed and classified into different types. The major FR types applied for cotton are nitrogen and phosphorous-based FRs 3 and silicon-based FRs. 4 The combination of different elemental components showed an outstanding synergistic effect on flame retardancy, which has been extensively studied by many scientists. For instance, Deh et al. studied FRs with phosphorus, nitrogen and silicon elements on cotton fabrics. The test results showed that the combination of these three elements provided better flame-retardant properties. 5
Currently, Proban® and Pyrovatex® are commercially available phosphorus-based flame retardants. Cotton fabric treated with Proban® and Pyrovatex® showed better laundering resistance. Although halogen-free, formaldehyde is released during use. 6 In recent years, more researchers have focused on the development of green flame retardants.
Recently, new and environmentally friendly FR for cotton have been investigated, such as a high-molecular-weight FR, bis[tetrakis(hydroxymethyl)phosphonium] (THPS)-urea n -(PO3) (NH4)2, prepared by Diao et al. The treated cotton fabric has a high LOI value of 46.8% and good laundering resistance. Additionally, the FRs are free from formaldehyde. 7 Ammonium salt of neopentyl glycol phosphate acid (ASNGPA), 8 a novel FR prepared by Feng et al., had high LOI values of more than 40%, and satisfactory durability. After 50 laundering cycles, the char length of ASNGPA treated cotton fabric was 48 mm. The ASNGPA FRs were eco-friendly with a formaldehyde-free synthesis process and no harmful halogenated gases released in the combustion process. Junekar et al. explored a graphite-functionalized pigment (EGFP) related to the phenazine pigment by an easy process. The treated cotton fabrics have excellent flame retardant properties so that they do not smoke at the onset of combustion and do not burn for more than 760 s. In contrast, the blank cotton caught fire within 5 s. 9 Moreover, other environmental FRs were also explored, such as a formaldehyde-free bis-triazine ring ammonium phosphonate, ASBSMP, as prepared by Yang et al. 10
There are several methods used to endow cotton fabrics with flame retardancy, such as layer-by-layer (LbL) assembly, 11 sol–gel technique, 12 UV treatment, 13 plasma treatment, 14 impregnation and drying method. 15 Different methods were chosen according to different types of FRs and substrates. For instance, Hribernik et al. grew a layer of silicon dioxide (SiO2) on the surface of regenerated cellulose fibers by a sol–gel technique at a very early stage. The test results showed that both the decomposition temperature and the glowing combustion temperature of the residual char were increased. 16
In our previous research, we developed several FRs for cotton fabric.17–20 The cotton fabrics treated with these FRs showed better flame retardance performance. A novel phosphorus–nitrogen–silicon-containing flame retardant (DTP) was designed. The test results showed that a synergistic effect exists among phosphorus, nitrogen, and silicon. Additionally, the environmental friendliness, smoke suppression and durability of the flame retardant were also taken into consideration.
This work aims to introduce the FRs on the basis of phosphorus and nitrogen FRs and their treatment methods for cotton fabrics. In the case of phosphorus and nitrogen FRs, the effect of the addition of silicon-containing, boron-containing biomaterials and other compounds on the flame retardant is summarized and new types of flame retardants developed in recent years are introduced. What’s more, the advantages and disadvantages of the flame-retardant treatment methods are compared. This work will provide ideas and a theoretical basis for suitable flame retardant selection for cotton fabrics under different application requirements.
Phosphorus and Nitrogen-based Flame Retardants
Phosphorus–Nitrogen Compound Flame Retardants
As FRs for cotton fabrics, phosphorus and nitrogen-containing FRs have been studied by many scholars. Phosphorus and nitrogen-containing FRs are the most widely used FRs for cotton fabrics in recent studies. Halogen-free phosphorus compounds are often used as flame retardants for cotton textiles as environmental requirements increase. Low manufacturing costs, good thermal stability, low volatility, and promotion of char formation are their advantages. 21 Moreover, several studies have shown that phosphorus and nitrogen flame retardants are more efficient flame retardants due to increased char formation. More important, these FRs are generally environmentally friendly and highly effective. 22
Sun et al. 23 prepared an ammonium salt of N,N-dimethylene-p-benzenesulphonic acid (ADBSPA) and applied onto cotton fabrics. The LOI of cotton treated with 350 g/L ADBSPA increased from 18% to 31.7%. The results of the cone calorimetry test (CCT) showed that 25.1% of the char residue remained at 250 s and the peak of heat release (PHRR) was greatly reduced. Zhao et al. 24 prepared a phosphorus–nitrogen FR, 6-chloro-4-(diethylamino phosphorate phosphoryl chloride)-2-(sodium 4-aminobenzensulfonate)-1,3,5-triazine (APPCABT), and applied it to cotton fabrics with P–O–C bonds. Cotton fabrics treated with 25% APPCABT obtained a high LOI value of 28.2%. Thermogravimetric analysis (TGA) showed that the treated cotton fabrics were endowed with high initial decomposition temperature and good char-forming abilities.
Based on phosphate, nitrogen-containing compounds are selected and reacted with it to synthesize phosphorus–nitrogen FRs. Gao et al. 25 explored an FR, which was an named ammonium salt of hexamethylenediamine N,N,N′,N′-tetra (methylphosphonic acid; AHDTMPA). AHDTMPA reacted with the O-6 hydroxyls of glucose in the structure of cotton fabrics to form P–O–C covalent bonds and the AHDTMPA-treated cotton showed better durability. The LOI value of AHDTMPA-treated cotton was 36%. An ammonium salt of ethylenediamine tetram ethylenephosphonic acid (AEDTMPA) for cotton fabrics was prepared by Zheng et al. 26 The AEDTMPA-treated cotton fabrics showed more efficient flame-retardant performance than the other typical phosphorus-containing flame-retardant agents due to the higher phosphorus content.
An ammonium salt of ethylene glycol diphosphoric acid (AEGDP) was synthesized by Jia et al. 27 under moderate and solvent-free conditions. Moreover, no formaldehyde was used in the synthesis of AEGDP compared with the synthesis of AEDTMPA and AHDTMPA. Compared with Proban® and Pyrovatex®, AEGDP will not release formaldehyde. Moreover, an ammonium salt of triethanolamine phosphoric ester acid (ATEPEA) 28 solves the disadvantage of whiteness reduction of AEGDP.
Zhang et al. 29 also prepared an ammonium salt of melamine hexa (methylphosphonic acid; AMHMPA), a new halogen-free and formaldehyde-free FR. Compared with AHDTMPA, AEDTMPA, AEGDP and ATEPEA, 1 mol of AMHMPA contained 12 mol of -P=(NH4)2 groups and -PO(OH)2 groups, which showed strikingly more reactive compounds and can promote the formation of P–O–C covalent bonds. The test results showed that AMHMPA-treated cotton showed highly effective fire resistance, outstanding durability, low cost, facile preparation and no in vitro cytotoxicity toward the environment and humans.
A new active FRs with a serrated structure, 1,3-diaminopropane tetraammonium salt (methylphosphonic acid; ADDTMPA), 30 was prepared by Xu et al. ADDTMPA is halogen-free and formaldehyde-free, and has high contents of phosphorus. In addition, the treated cotton fabrics have both good flame retardant properties and softness.
An ammonium salt of teraethyiene pentamine heptamethylphosphonate (ATEPAHP) 31 was prepared by Tian et al. In this study, 1 mol of ATEPAHP FR contains 7 mol of reactive groups that can achieve high grafting content. The ATEPAHP-treated cotton also showed outstanding durability and efficient flame-retardance. Moreover, an ammonium salt of pentaethylenehexamine octa (methylene-phosphoric acid; APHOMPA) 32 with more reactive groups was prepared by Chen et al. Bio-resourced mannitol was employed to synthesize an ammonium salt of mannitol hexaphosphate acid ester (AMHPE). 33 Zhang et al. investigated the performance of AMHPE-treated cotton fabrics which showed high flame retardancy and excellent durability.
From the above studies, it can be concluded that FRs and cotton fabrics can form stable chemical bonds (P–O–C covalent bonds) and greatly improve the durability of flame-retardant cotton fabrics. Moreover, FRs can be bonded onto cotton fabrics not only with P–O–C bonds, but also with other chemical bonds. For example, a novel bio-based FRs ammonium salt of sodium glutamate tetramethylene phosphonic acid (ASGTMPA) can combine with cotton fabrics through P–O–C and -COOC covalent bonds. 34
The formation of P–O–C covalent bonds between ammonium salt of tris-(hydroxymethyl) - aminomethane- penta (methylphosphonic acid; ATPMPA) and the cotton fabrics endowed the flame-retardant cotton fabrics with improved washing performance. 35 An ammonium salt of guanidine acetate tetram ethylenephosphonic acid (AGATMPA) can also be grafted onto cotton fabrics via C-O-C covalent bonds. 36 The physical properties of the AGATMPA-treated cotton were retained well. Chen et al. 37 prepared a new ammonium salt of citrulline-penta (methylphosphonic acid; ACPMPA) FRs and grafted it onto cotton via -P=O(–O–C) and -COOC bonds. The LOI of the treated cotton decreased by 15% after 50 LCs. 2,7-bis (Dimethoxyphosphoryl)-3,7-dimethyl-3-phosphonic acid (FR-LO) 38 with two phosphoric acid ester groups and one ammonium phosphorus acid group was synthesized by Liu et al. The durability of the treated cotton fabrics was enhanced due to the replacement of ammonium phosphorus acid groups with phosphate ester groups in FRs.
The specific test data of FRs mentioned above are shown in Table 1. The CCT test results are under an irradiative heat flux of 35 kW/m2. In the table, the percentage concentration was used as a unit of FRs and the char length was tested by vertical flammability tests (VFT).
Summarised data of different flame retardant cotton fabrics.
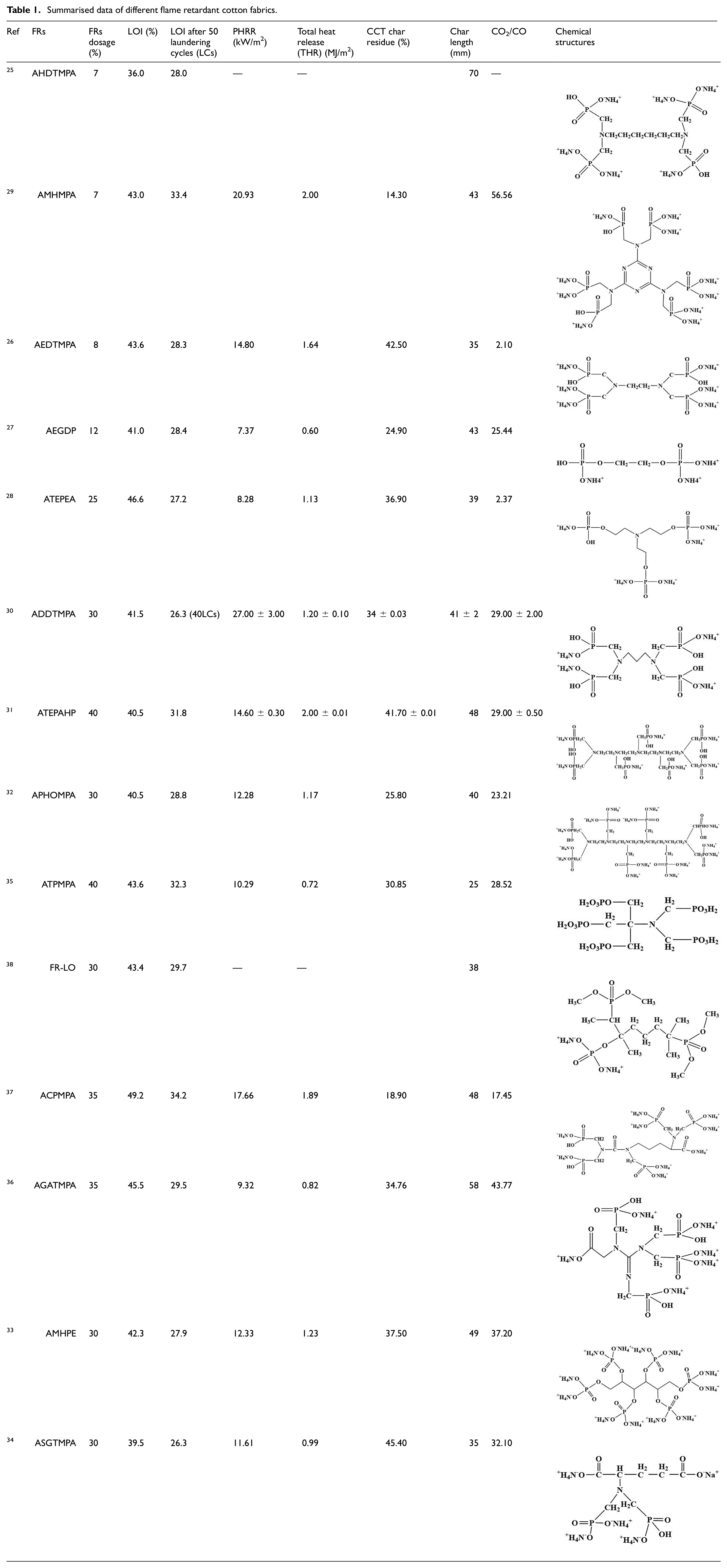
From the above data, it is clear that the higher phosphorus content of FRs showed highly efficient flame-retardant performance. The cotton treated with 8% AEDTMPA showed an LOI of 43.6%, while the 7% AHDTMPA treated cotton showed an LOI of 36.0%. The LOI of 7% AMHMPA and 40% of ATEPAHP treated cotton remained at 33.4% and 31.8% after 50 LCs, respectively, which showed outstanding durability. The main reason is that AMHMPA and ATEPAHP contained more reactive groups, which can promote the formation of P–O–C covalent bonds and achieve high grafting content.
For the development of flame retardants for cotton fabrics, many scholars have explored FRs with good flame retardant properties. From the data in the above table, it can be seen that the PHRR and THR of AEGDP treated cotton decreased significantly, 7.37 kW/m2 and 0.60 MJ/m2, respectively. The char residues of CCT can achieve values as high as 45.40% and the char length of VFT can achieve values as high as 70 mm. In addition, AEDTMPA had a lower CO2/CO ratio of 2.1, which means less inefficient combustion. However, most of the FRs in Table 1 are used in high volumes. In order to develop more efficient FRs, many scholars have begun to study FRs with silicone-containing materials.
Phosphorus–nitrogen FRs are often used for flame retardant treatment of cotton fabric and exhibit good flame retardant properties. The flame retardant mechanism of phosphorus and nitrogen FRs has also been explored. Firstly, phosphorus compounds produce reactive species such as H- and OH- which can be trapped during combustion, reducing the amount of heat generated and slowing down the combustion process.39,40 Secondly, nitrogen-containing molecules promote the formation of a protective layer of char when heated. This char acts as a thermal barrier to protect the underlying fabric from further thermal degradation and oxygen ingress, thereby inhibiting flame spread. The addition of phosphorus and nitrogen to the polymer then improves the thermal stability of the polymer, delays the onset of decomposition and reduces the release of volatile fuels, thereby inhibiting flame spread.41,42 In addition, the phosphorus and nitrogen polymers form a viscous melt that covers the surface of the fabric, creating a physical barrier that prevents oxygen diffusion and heat transfer. This molten film solidifies into a ceramic-like residue as it cools, further inhibiting combustion. Some of these phosphorus–nitrogen polymers swell when heated, filling the gaps between the fiber and forming a dense, cohesive layer that prevents flame penetration.43,44 The above flame retardant mechanism of phosphorus and nitrogen FRs is can be applied not only to cotton fabrics, but also to other materials, and have a good flame-retardant effect. 45
Silicon–Phosphorus–Nitrogen Flame Retardants
Based on phosphorus and nitrogen elements, FRs added with silicon elements have shown better flame-retardant properties. The synergistic effect of silicon and phosphorus has a great influence on flame-retardant performance.
Deh et al. 5 studied cotton fabrics treated with phosphorus-based FRs in the presence of nitrogen or silicon elements, and the effect of three elements, phosphorus, nitrogen, and silicon on LOI was investigated. The results showed that the LOI value of the treated cotton was higher, and higher residual masses were obtained when phosphorus/nitrogen/silicon combination was used instead of phosphorus/nitrogen element. Therefore, it can be seen that the addition of silicon can increase the flame retardancy of FRs. Recently, many scientists began to study the flame-retardant properties of silicon-containing FRs using different methods.
In order to explore a novel molecular form containing 9,10-dihydro-9-oxa-10-phos-phaphenanthrene-10-oxide (DOPO) with synergistic flame retardancy of phosphorus/nitrogen/silicon, cotton fabrics were treated with 1-(9,10-dihydro-9-oxa-10- phosphaphenanthrene-10-oxide)-4-(trimethoxysilyl-methyl) piperazine (DOPO–PiP–Si) synthesized by Zhang et al. 46 Another novel high-efficiency and durable FRs DOPO–ammonium phosphate (DOPO–AP) was prepared by Wan et al. 47 The LOI of DOPO–PiP–Si treated cotton fabrics and DOPO–AP treated cotton fabrics were 27.6% and 40.2%, respectively. It is worth mentioning that the LOI of DOPO–AP treated cotton fabrics was maintained at 26.4% even after 50 LCs. The cotton fabrics treated with DOPO–AP showed higher LOI values than those treated with DOPO–PiP–Si while even using lesser amounts of flame retardants. The reason for this phenomenon is that DOPO–AP was synthesized by two phosphorus containing substances, DOPO and H3PO4, which have a higher phosphorus content. On the other side, dicyandiamide was chosen to act as a cross-linker between DOPO–AP and fabrics, which can play a flame retardant role in the gas phase and play a synergistic effect with phosphorus. During combustion, silicon-based fragments have the ability to form a film on the surface of cellulose to further suffocate the flame; however, sufficient carbonization capacity of the system is required.
Liu et al. 48 synthesized di-(trimethoxysilylpropyl) spirocyclic pentaerythritol bisphosphorate (SPDP–PTMS) by the sol–gel technique. Cotton fabrics treated with 500 g/L SPDP–PTMS showed an LOI of 29.5% and maintained it remained at 26.2% after 20 launderings and 8.8 cm char length with no after-flame or after-glow. In addition, SPDP–PTMS treated cotton released non-flammable gases during combustion and formed a stable char layer, indicating the good flame retardancy of treated cotton.
Based on 3-aminopropyltriethoxysilane (APTES) and sodium phytate, flame-retardant cotton fabrics were prepared by Li et al. 49 Cotton fabrics treated with 15 bilayers extinguished immediately, while the untreated cotton fabrics burned out, leaving only a little ash in the VFT. Phosphoryl chloride and neopentyl glycol,2, 2-dimethyl-1, 3-propanediol-(3′-triethoxysilanepropyl)phosphoramidate (DPTP) was synthesized by the sol–gel technique. 50 Cotton fabrics treated with DPTP showed good flame-retardant properties with increased LOI value from 28.6 to 30.1% with 18.1–25.8% weight gain. The release of combustible gases decreased (CO, ether and carbonyl), and the generated nonflammable gas improved the flame retardancy. A novel reactive phosphoramidate siloxane (DTSP) containing silica, phosphorus and nitrogen was prepared and applied to cotton fabrics. 51 DTSP can improve the flame retardancy of cotton fabrics by promoting the formation of char and releasing non-flammable volatiles. The LOI of cotton fabrics with 16% weight gain can reach 30.3% and remain at 27.0% after 20 LCs. Moreover, the tensile strength and breaking elongation of the treated cotton fabrics showed a slight decrease.
Zhang et al. prepared intumescent FRs and hydrophobic coatings by using sodium montmorillonite (MMT), ammonium dihydrogen phosphate (ADP), and methyltrimethoxysilane (MTMS) as raw materials. The Si–O–Si network structure was formed via the hydrolysis–condensation reaction of MTMS induced by the catalysis of ADP. 52 Cotton fabrics treated with 2.0 wt% MTMS showed significant flame retardancy, hydrophobicity, improved thermal stability, and heat release behavior.
The flame-retardant mechanism can be considered as silicon provides heat shielding, while the phosphoric acid source can promote the formation of carbon,53,54 which can improve the overall flame-retardance performance. Silicon element promotes the formation of a dense char layer on the surface, which acts as a heat and oxygen barrier. The addition of silicon components can further improve the hydrophobic properties of flame-retardant cotton fabrics. 55
Boron–Phosphorus–Nitrogen Flame Retardants
Besides the three commonly used flame-retardant elements, phosphorus, nitrogen, and silicon, boron has also attracted a lot of research attention because of its synergistic effect. Boron-based compounds have been used to enhance the flame-retardant properties of various cellulosic materials due to their low toxicity, smoke suppression, and char-promoting properties. 56 Boron and its derivative compounds exhibited flame retardancy in both condensed and gas phase. 57
Hydroxyethyl methylene phosphate, pentaerythritol diborate, and cyanuric chloride were used to prepare an “H” shaped flame retardant (TBSA). 58 The TBSA treated cotton fabric showed a high LOI value of 48.8%. The treated cotton fabrics exhibited excellent performance in terms of durability and strength due to the “H” shaped flame retardant being able to form covalent bonds with multiple cellulose fibers. Moreover, it can enter the interior of the fabric fiber to fill in the cracks and holes, thus increasing its tensile strength potential. A nitrogen–phosphorus–boron intumescent coating was prepared by Fang et al. 59 using sodium polyborate (SPB) and polyhexamethylene guanidine phosphate (PHMGP). A neotype boron–nitrogen–silicon synergistic FR, BTDU–PTMS, was successfully synthesized by Wang et al. The LOI and char length of the treated cotton (weight gain 30%) tested by VFT were 32.7% and 85 mm, respectively. Moreover, excellent smoke suppression ability was demonstrated by CCT. 60
The FRs contain various flame retardant elements that can play a flame retardant role in different phases, such as the gas and condensed phases. Boron-containing flame retardants act as a protective layer that prevents contact between oxygen and combustibles, as well as the diffusion of combustible gases from combustion decomposition to act as fuel for combustion. The boron acid could catalyze dehydration to form char. However, there is little research on the use of boron-based flame retardants on cotton fabrics and most of the research has been focused on silk 61 and wood. 62 It is expected that there will be more research and development on cotton fabrics in the future.
Biobased Phosphorus–Nitrogen Flame Retardants
The addition of bio-based materials can solve the problem of poor compatibility between some FRs and cotton fabrics, as well as the issue of environmental unfriendliness. Bio-based FRs include deoxyribonucleic acid (DNA), proteins, phytic acid (PA), etc. The flame-retardant properties of bio-based materials with other components showed better performance than bio-based FR systems alone. DNA has an intrinsic intumescent feature due to the presence of the classical three ingredients of intumescent formulations in its structure. 63 Proteins such as casein, 64 whey protein 65 and chicken feather protein 66 have a certain ability to promote carbon formation when fabrics are ignited. PA also showed flame retardancy due to its rich phosphorus contents. 67
DNA from herring sperm as a novel, green, natural flame retardant and suppressant for cotton fabrics has been explored by Alongi et al. 68 The DNA-treated cotton fabrics showed good flame-retardant properties. Alongi et al. 69 further studied the effect of different additions of DNA on flammability and the combustion behavior of treated cotton fabrics under a heat flux of both 35 and 50 kW/m2 was investigated. When a methane flame is used, only 19% add-on by weight can confer resistance to a radiant heat flux of 35 kW/m2 and not burn under CCT. The combination of DNA with other materials such as chitosan (CS), 70 modified MMT, silver nitrate and octadecyltriethoxysilane (ODTS) 71 has been explored. All the combinations showed good flame-retardant properties. Moreover, the DNA/silver nitrate/ODTS treated cotton became superhydrophobic with a water contact angle (WCA) of 157°. Bosco et al. 15 explored the role of different parameters on the overall flame retardancy of three different molecular sizes, and found that the coatings containing low molecular size DNA exhibited superior fire protection on the fabric substrates.
Indeed, DNA can be considered an intrinsic intumescent flame retardant because it contains the three main components of an intumescent flame retardant, an acid source, a carbon source and a gas source. The acid source catalyzes dehydration into char, the char source forms a char layer in the presence of the acid source, and the gas source produces non-combustible gases during combustion, resulting in a porous foamy char layer.
Other types of proteins such as caseins and hydrophobins applied on the surfaces of cotton fabrics were also investigated by Alongi et al. 72 Caseins were found to promote carbonization, inhibit the formation of volatile species and achieve self-extinction with the increased LOI. Xu et al. 73 attempted to investigate the flame-retardant properties of the coatings of casein-based FRs and found that coatings were durable, since casein-based FRs were bounded onto cellulose by P(=O)−O−C and C(=O)−O−C bonds. The TGA, energy-dispersive X-ray (EDX) and TG-IR tests showed that the casein-based FRs mainly modified the thermal decomposition pathway of the cellulose while cotton fabrics retained tensile strength and whiteness after treatment.
CS and PA as bio-based materials are also commonly used with other materials as part of the FRs for cotton fabrics. The flame retardancy of cotton treated with CS, sodium phytate and APTES was investigated by Liu et al. 74 It is worth noting that the addition of CS increased the LOI and improved the HRR and THR, while the total smoke release (TSR) and smoke production rate (SPR) of treated cotton fabrics decreased. Zhang et al. 75 synthesized FRs using bio-based CS, PA and divalent metal ions. The increase of metal ions improved the flame retardancy and has had little effect on the softness of fabrics.
The chemical structure of PA comprises 6 phosphate groups and 12 hydroxyl groups, which confer the ability to bind to a wide range of metal ions and positively charged compounds.
Polymeric Phosphorus–Nitrogen Flame Retardants
The addition of polymeric materials for the preparation of FRs based on phosphorus and nitrogen content has been widely studied due to their durability and environmental friendliness. The most commonly used materials in polyelectrolyte FRs for cotton fabrics are phosphate-based such as ammonium polyphosphate (APP), thiourea–phosphoric acid polymer, and phosphate ammonium salts. Among them, APP is often used as an FR with other materials to provide a flame-retardant effect, such as metal–polyphosphate ionomer ammonium. 76 Davies et al. found that the presence of metal salts was useful for catalyzing the decomposition of APP. Metal salts have an important effect on the LOI, but play an insignificant role in carbon generation. The LOI value reached 35% for the addition of metal salts. Chen et al. 77 investigated the flame-retardant effect of APP and CS on knitted cotton fabrics, and it was found that the designed FRs worked in both condensed phase and gas phase. The flame retardancy of APP and casein was also investigated by Faheem et al. 78 Different ratios of APP and casein were studied, and the results showed that the physiological comfort of cotton fabrics would increase when the addition of casein was above 5 wt%.
Considering the durability of FRs on cotton fabrics, SiO2/APP @polydimethylsiloxane (PDMS) 79 and the combinations of branched poly(ethyleneimine; bPEI), APP and fluorinated decyl polyhedral oligomeric silsesquioxane (F-POSS) 80 were prepared by Zhao et al. and Chen et al., respectively. The treated cotton fabrics not only showed good flame-retardant properties but also super-hydrophobicity. The SiO2/APP@PDMS treated cotton fabrics exhibited WCA of 154.2°, and the bPEI/APP/F-POSS treated cotton showed a super-hydrophobic surface with self-healing function. The flame-retardant and self-healing superhydrophobic bPEI/APP/F-POSS coatings can withstand more than 1000 abrasion cycles at 44.8 kPa pressure without losing the original properties.
Nano Phosphorus–Nitrogen Flame Retardants
It is reported that inorganic nanomaterials are alternatives to traditional flame-retardant materials. 81 Nanomaterials such as nanoclays and metal oxides have the advantages of abundant resources and environmentally friendly properties. However, it is difficult for nanomaterials alone to perform well as flame retardants, and they require combination with other materials for flame-retardant treatments. Li et al. 82 reported on cotton fabrics treated with nanosilica (SiO2)–polyethyleneimine (PEI)/polyphosphoric acid (PPA)/REPELLAN FF, and found that the char lengths after igniting for 12 s, the char yield at 600°C, and the LOI value were 9.2 cm, 8% and 28.2% respectively. The treated cotton fabrics exhibited good flame-retardant properties and excellent hydrophobicity with WCA of 142 ± 1°.
Improving the thermal stability and flame retardancy of cotton fabrics using polyhedral oligomeric silsesquioxane (POSS) and boehmite nanoparticles 83 was investigated by Alongi et al. The TTI increased and the HRR reduced by up to 40% after nano FRs treatment. Ortelli et al. 84 coated cotton with two different self-assembled TiO2@DNA systems. The performance of the cotton fabrics with the addition of nanoparticles was compared to phosphorus-based flame-retardant those for fabrics and DNA-treated fabrics. The nanoparticle-containing flame-retardant performance and the washing durability were better than the fabrics treated with only DNA. Li et al. 85 studied an intumescent all-polymer multilayer nanocoating via LbL assembly with the ability to extinguish the flame on the treated fabric. The flame retardancy of cotton fabrics prepared with polyelectrolytes, cationic CS and anionic poly(sodium phosphate; PSP), was investigated by Guin et al., 86 and the CS-PSP nanocoating promoted charring of cotton, making the fabric self-extinguishing.
Flame Retardant Treatment Methods
In general, different flame retardants were applied onto cotton fabrics by different treatment methods including the impregnation and drying method, LbL assembly, sol–gel technique, UV treatment, plasma treatment, etc., since the different methods have their own advantages and disadvantages.
The impregnation and drying method is a common treatment method for cotton fabrics. This method is very simple to operate. With the development of flame-retardant finishing methods and the exploitation of FRs, the LbL assembly method is more suitable as the number of flame retardant components increases. The LbL assembly involves the formation of multilayer films by successive adsorption of oppositely charged polyelectrolytes and/or nanoparticles onto substrates, and the positively and negatively charged layers piled up on the surface of substrates and bonded by the electrostatic attraction at the nanometric level. Each pair of positive and negative electrodes is referred to as a bilayer. These layers are usually applied to the substrates by dipping or spraying.87,88 Simplicity, versatility and nanoscale thickness control are the advantages of LbL assembly over other conventional coating methods.
The sol–gel technique used as a treatment method has been widely applied for functional textiles, such as flame-retardant treatment, antibacterial protection, anti-wrinkle treatment, super-hydrophobicity, etc. 50 The technique is based on two steps involving hydrolysis and condensation reactions starting from metal alkoxides, such as tetraethoxysilane (TEOS), tetramethoxysilane, and titanium tetraisopropoxide.89,90 This method has the advantages of simple operation, ease of meeting the required conditions, and environmentaly friendliness. 91 However, the application of the sol–gel technique in the treatment of fabrics has the disadvantage of yellowing or decreasing the tensile strength of fabrics, which limits the potential for application. 92
The UV treatment method is another finishing method to apply organic–inorganic hybrids onto fabrics, which has the advantage of low level of energy consumption, fast curing rate and solvent-free polymerization, and is used in combination with the sol–gel technique to produce substrates with different properties.93,94 However, this technique has some limitations in flame-retardant materials and substrates.
In addition to the above commonly used treatments methods, there are also some methods used for flame-retardant treatments. Plasma treatment does not require solvent and produces contaminants. This technique is divided into two types: high temperature and low temperature treatments. Low temperature treatments are generally applied for cotton fabrics. 95 A flame-retardant diethyl methacryloylphosphoramidate containined phosphorus and nitrogen. The synthesized compound was grafted onto cotton fabrics using electron beam irradiation. 96 The electron beam treated cotton exhibited efficient flame retardancy which was proved by the LOI value of 30.2%.
Przybylak et al. 97 reported flame-retarded cotton fabrics with strongly hydrophobic properties by a two-step modification process. In the first step, guanidine carbonate and/or ammonium dihydrogen phosphate were immobilized in aminopropyltriethoxysilane using the sol–gel technique. In the second step, the modified fabrics were impregnated in a hydrophobizing composition by the bath method. The two-step modification makes it possible to produce multifunctional fabrics.
Summary
There are many classes of FRs mainly based on phosphorus and nitrogen elements developed for cotton fabrics, and based on which combination properties can be achieved by adding or selecting some other components. First, the FR cotton fabrics can be endowed multifunctionalities with excellent performance, e.g. silicon elements could help achieve hydrophobicity and further improve the durability, boron-containing compounds always provide smoke suppression properties, and some biological components show certain antibacterial properties. Second, the FRs can have a much more efficient flame-retardant effect using smaller amounts of usage without significantly influencing the performance of the original cotton fabrics. Third, different components have different flame retardant mechanisms and can act in gas and condensed phases. At the same time, the method used for flame-retardant treatment was selected according to the reactive groups, electric charges, and molecular structures, etc., of FRs, including LbL, impregnation and drying, sol–gel technique and UV treatment method.
However, with the development of research on FRs for cotton fabrics, the requirements for FRs and treatment methods have gradually increased. The environmental friendliness of FRs, smoke suppression and laundering resistance properties of flame-retardant cotton fabrics have become the research focus of scientists. In more detail, the key challenges of the development of flame retardants are achieving ideal flame retardancy and durability using environmentally friendly components, which are required to balance good flame resistance, environmental factors, and easy-to-produce performance at the same time. The combination of nanoparticles, and intumescent components and bio-based products could be the focus of research in the future. The practicality and efficiency of flame-retardant treatment methods are also a concern for the industry. New treatment technologies with high efficiency need to be investigated. While studying how to increase the utilization rate of FRs and improve flame retardancy and durability, cotton fabrics can be endowed other functions such as anti-bacterial, anti-ultraviolet, anti-static, etc. to increase the added value of cotton fabrics. It is hoped that future FRs for cotton fabrics will address these aspects and expand the potential application of flame-retardant cotton fabrics.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the State Key Laboratory of Wuhan Science and Technology Bureau Supported Special Fund (2022023988065193) and the Key Laboratory of Textile Fiber and Products (Ministry of Education), (Wuhan Textile University; Fzxw2024010).
