Abstract
Textiles are one of the basic needs of humankind. Although textiles satisfy many human needs, they are implicated in the loss of life and property since many fibers and fabrics are inflammable. Many chemical finishes are applied to improve the flame resistance of textile materials. Unfortunately, most of the flame retardants are synthesized chemically, need large quantities to be used, cause adverse impact on the textiles and also expensive. In this research, a novel approach to using DNA from herring sperm as flame retardant for cotton was explored. Surface of the textiles was modified with citric acid to increase free carboxyl content and hence absorption of DNA. It was observed that DNA was absorbed into the cotton fabrics using chemical adsorption according to the quasi-second-order-kinetic model. Flammability of the cotton before and after the addition of DNA was checked. It was observed the length of fabrics damaged due to flames was only 29 mm compared to the original length of 350 mm. Such a high increase in flame resistance has not been reported for natural materials. In addition, the chemical reaction between DNA and cotton fabric can improve the flame retardant–washing resistance of the finished cotton fabric. In this article, the strategy of using biological macromolecules to flame-retardant fabrics can provide research basis for the design and development of efficient and environmentally friendly flame retardants.
Introduction
With the increasing consumption of various industrial and household textiles, the number of fires caused by textiles is also increasing year by year. About 2.5–3.5 million fires are reported every year with 65,000–75,000 casualties. 1 Cotton fabrics are preferred over synthetics or even other natural fibers because of their soft and comfortable feel, good hygroscopicity, and good air permeability. Cotton continues to be the predominant natural cellulose fiber use in the field of clothing, household decoration, and industrial textiles.2,3 One of the major limitations of cotton is the limiting oxygen index (LOI) of cotton fibers at only 18.0% compared to 21% for polyester, 24% for wool, and 20.5% for nylon. Hence, cotton is readily flammable and poses fire hazards. 4 Therefore, it is of great importance to impart flame resistance to cotton fabric. 5 At present, the flame-retardant finishing of cotton fabrics mainly uses dip-rolling-baking method to finish inorganic materials (metal hydroxides, bentonite, etc.), halogen compounds, organophosphorus compounds, and other flame retardants. 6
Inorganic metal hydroxides used as flame retardants have several positive attributes such as good stability, ability to suppress smoke, and hence are widely used.7,8 However, metal hydroxides such as aluminum hydroxide and magnesium hydroxide do not have high efficiency and often require nearly 50% of the weight of the fabric to achieve good flame retardancy. Such a high content not only increases cost but also reduces the wearability of the fabrics.9,10 Compared to the hydroxides, halogen-based flame retardants have low dosage and high flame-retardant efficiency. 11 Nevertheless, over the past decades, many researchers have reported the long-term toxicity and hazards of halogen-based flame retardants. For instance, when burned, halogens produce carcinogenic dioxins and toxic compounds, such as HCl and HBr, which would cause damage to life. Therefore, many European countries have phased out some halogen-based flame retardants containing chlorine and bromine.12,13 Phosphorus-based flame retardants are widely used to impart flame retardancy to cotton textiles. Phosphorous-based flame retardants produce less smoke, have low toxicity, are halogen-free, and also have good thermal stability. They can be used with other flame retardants and have significant synergistic effects.14,15 However, this kind of flame retardant mainly synthesizes phosphorus-containing high polymers, which brings great pressure to environmental protection in the process of production and use.16,17 Since most of the available flame retardants have several limitations, there is a need for developing efficient and more importantly environmentally friendly flame retardants for treating cotton.
Some people’s attempts have been made to develop bio-based solutions for imparting flame retardancy. Examples of such natural flame retardants include mineral clay, 18 lignin, 19 banana pseudostem sap (BPS), 20 spinach juice (SJ), 21 green coconut shell extract (GCSE), 22 and even DNA.23,24 In addition, ammonium starch phosphate carbamates and composites containing attapulgite (ATP) have also been studied for their potential to decrease flammability.25,26
DNA is a double helix chain molecule, where the chains are linked together by intermolecular and intramolecular hydrogen bonds, which provide good heat resistance. 27 Recently, many studies have shown that the addition of DNA can improve the heat resistance of fabrics.28–30 Alongi et al.31,32 had focused on the thermal degradation process of DNA and found that different DNA dosages have a certain impact on the heat resistance of cotton fabric. The higher the DNA dosage, the better the heat resistance of cotton fabric. Carosio et al. 33 applied DNA to cotton fabrics through layer-by-layer self-assembly (LBL) technology, the results showed that the LOI of treated cotton fabrics increased to 24.0%. However, the method of applying DNA and the amount of DNA that can be sorbed by the cotton fabrics is limited.
Hence, the objective of this research is to modify the cotton surface and enhance the absorption of the DNA. To achieve this objective, cotton fabrics were treated with citric acid (CA) to increase the carboxylic group content which will in turn lead to higher sorption of DNA. Efficiency of loading DNA and the ability of DNA-treated textiles to improve flame resistance, increase washability, and other properties has been studied, which can provide ideas for the future design and development of simple, efficient, green, and environmentally friendly textile flame retardants.
Experimental section
Materials
Scoured and bleached 100% cotton fabrics were supplied by Shandong Huafang Co., Ltd. China with specifications of thread size (count) being 40S × 40S × 96 × 96 × 120 and weight per unit area of 115 g/m2. DNA powder (from herring sperm) was purchased from Beijing Solarbio Reagent Co., Ltd; DNA was stored at 2°C–4°C before its application to the fabrics. Both trisodium citrate (TSC) and sodium chloride were purchased from Shanghai Joint Test Chemical Co., Ltd. CA was supplied by Jiangsu Qiangsheng Functional Chemistry Co., Ltd. 1-Ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC) was purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. In addition, calcium acetate, phenolphthalein, and sodium hydroxide were supplied by Shanghai Silian Chemical Plant Co., Ltd. Deionized water was used for all the experiments in this study.
CA modification of cotton fabrics
Prior to modification, cotton fabrics were cut into pieces of fixed size and washed with water and then diethyl ether, in order to remove the grease and wax on the surface of the fabrics. Later, fabrics were dried in an oven at 70°C for 1 h. With CA as modifier and TSC as catalyst, deionized water was added in 1:50 bath ratio (w/v) to prepare modified solution. The amount of CA in the modified solution was varied from 1 to 9 wt% (the mass ratio of CA to TSC is 3:2). Cotton fabrics were impregnated in the modified solution by two dipping and two rolling processes. After the modification, cotton fabrics were dried at 80°C for 5 min and cured at 150°C for 150 s. Modified fabrics should be conditioned for more than 12 h for the fabrics to achieve constant weight.
Flame-retardant finishing of modified cotton fabrics
Various concentrations of herring sperm DNA solutions (2, 4, 6, 8, and 10 o.w.f %) were prepared by dissolving in sodium chloride solution (2 mol/L) using a bath ratio (w/v) of 1:50. And 0.01 wt% EDC solution was added; the reaction was carried out for 30 min at pH between 4 and 5. After that, cotton fabrics were immersed in the prepared solution for different time periods (5, 10, 15, 30, 60, 180, and 300 s). Flame-retardant finishing was carried out by DNA treatment with two dipping and two rolling processes. Finally, the treated cotton fabrics were dried at 40°C for 300 s and re-dried at 50°C for 30 s.
Analysis of adsorption mechanism
In order to analyze the adsorption mechanism of DNA molecules on cotton fabrics more accurately, the (1) Lagergren first-order rate equation and the (2) quasi-second-order kinetic model 34 were used
where qɛ is the equilibrium adsorption amount of DNA on cotton fabric, mg/g; qt is the DNA content on cotton fabrics at T-time, mg/g; k1 is the first-order adsorption rate constant and k2 represents the secondary adsorption rate constant.
A straight line graph can be obtained by plotting ln(qe – qt) (as shown in Figure 5). Comparing the regression coefficient R2 obtained by linear fitting, it can be seen that the degree of first-order reaction kinetics is consistent and k1 can pass through a straight line. The slope is found similarly, by t/qt plotting t and a line graph is obtained (as shown in Figure 6) and the degree of conformity to the second-order adsorption kinetic model was judged by linearly fitting the regression coefficient R2.
Characterization
Calcium acetate method was used to determine the carboxyl content on the surface of cotton fabric modified by CA. The content of phosphorus in fabrics was determined by inductively coupled plasma atomic emission spectrometry (Leeman Company, USA), then the DNA content on the fabric is calculated by dividing the molecular weight of phosphorus and multiplying by the molecular weight of DNA. LOI tests were carried out using a FIRE oxygen index apparatus according to ASTM D2863 standard. The flame retardancy and flame retardant–washability properties of cotton fabrics were tested in vertical configuration and carried out by flame-retardant tester. These tests were repeated three times for each specimen. The tensile strength and elongation at break of fabrics before and after flame-retardant treatment were tested by fabric strength tester.
Results and discussion
Surface modification of cotton fabric by CA
After modification with CA, there will be a large number of –COOH groups on the surface of cotton fabric. Moreover, chemical reaction between cotton fabric and CA can produce ester bond, which increases initially and then tends to stabilize, as shown in Supplemental Figure S1. The change of free carboxyl content on surface of cotton fabric is shown in Figure 1. It can be seen from Figure 1 that the free carboxyl content of the original cotton fabrics is about 17 mmol/kg, which mainly comes from the by-products produced during the oxidative bleaching of cotton fabrics. With the introduction of CA, the free carboxyl groups in cotton fabric increased (from 16.67 to 727 mmol/kg). When the amount of CA reached a certain level, saturation on the surface of the fabric occurs and the content of free carboxyl group maximizes at ~1070 mmol/kg. The saturation of the samples is indicated by the changes in the hydroxyl content on the surface of cotton fabrics. Combined with the whiteness values of fabric (Supplemental Table S1), we choose cotton fabric modified by CA with 8% mass fraction for further studies.

Effects of CA mass fraction with free carboxyl content on surface of cotton fabric.
After the CA/TSC treatment, CA could be attached on the surface of the fibers through the covalent bonding. CA and all its decomposition products were able to form ester linkages with cellulose. The specific modification mechanism is shown in Figure 2.35,36

Modification mechanism of CA on cotton fabric.
Adsorption of herring sperm DNA on cotton fabric surface supported by CA
The adsorption capacity of herring sperm DNA on cotton fabrics at different concentrations is shown in Figure 3. It is obvious that with the increase of herring sperm DNA concentration, the content of DNA on the fabric also increases and gradually tends to saturate at 6%, which indicates that there is an adsorption equilibrium between herring sperm DNA and CA groups on cotton fabric. This is because there is chemical adsorption between herring sperm DNA molecules and cotton fabrics, mainly through –NH2, –OH on DNA molecules and –COOH on cotton fabrics. Also, dehydration and condensation reactions occur to form chemical bonds under the action of EDC. When the concentration of herring sperm DNA is low, the probability of interactions between DNA molecule and cotton fabric surface is low, so the rate of chemisorption is slower (2%~4%). When the concentration of DNA increases gradually, the concentration of –NH2 and –OH in the chemical reaction process increases. Under the action of catalyst, the chemical adsorption of DNA molecule on the surface of cotton fabric proceeds rapidly, which results in the obvious increase of DNA on the fabric at this stage. However, because the surface area of cotton fabric and the content of free carboxyl group are relatively constant, when the concentration of herring sperm DNA increases again (6%–10%), the reaction rate slows down and the reaction reaches equilibrium. Therefore, herring sperm DNA concentration of 6% was selected for subsequent experiments.

DNA content in cotton fabric at different concentrations of DNA.
Adsorption kinetics of DNA on cotton fabrics
The DNA content of cotton fabrics at different adsorption time is shown in Figure 4. It can be seen from Figure 4 that the adsorption of herring sperm DNA on cotton fabric is a fast process. The sorption reaches saturation within 30 s and no significant increase in absorption is seen with further increase in reaction time. When the adsorption time is short (5 s), DNA molecules could not diffuse through the surface of the fabric and reach functional groups, so the adsorption capacity of DNA molecules on the fabric is very small. With the increase of adsorption time, the reaction rate of DNA molecule with cotton fabric modified by CA accelerated, and the free carboxyl groups on the surface of cotton fabric reacted with –NH2, –OH. Because the essence of chemical adsorption is the formation of chemical bonds, the adsorption on the surface of cotton fabrics is monolayer. However, when the time is long enough (>30 s), there is sufficient time for all the DNA and functional groups to react and the sorption reaches saturation.
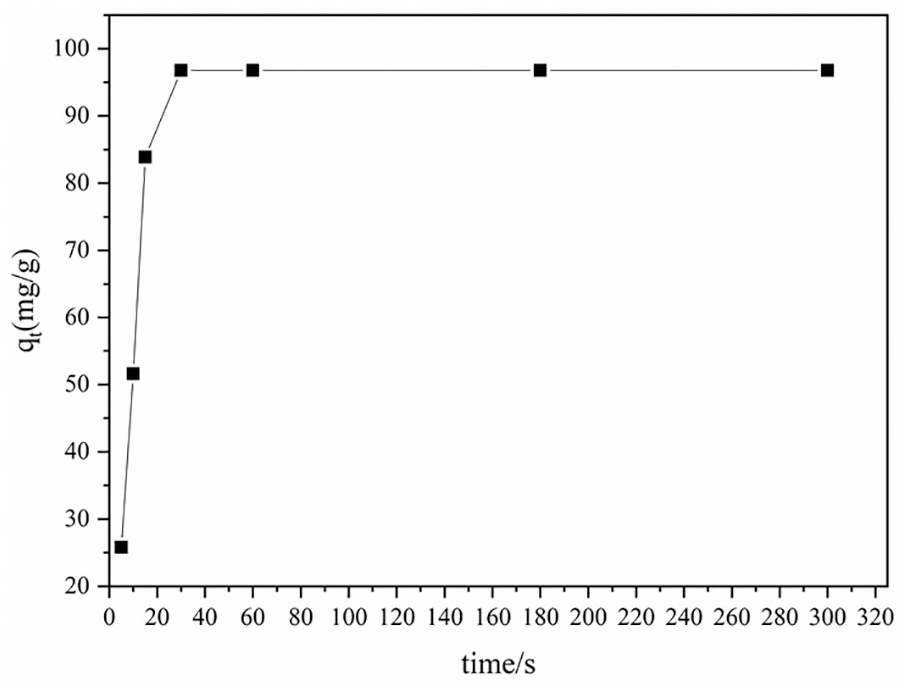
Effect of adsorption time with DNA content on cotton fabric.
Figures 5 and 6 are the fitting curves of the first-order and the second-order adsorption kinetics of DNA molecules on cotton fabrics, respectively, and the calculation results are shown in Table 1. It is apparent that the regression coefficient R2 of the second-order kinetics fitting is much higher than that of the first-order kinetics, which is 0.99778. The high fitting value indicates that the adsorption of DNA molecules on cotton fabrics is more in line with the quasi-second-order kinetic model, and the saturated adsorption capacity is 97.66 mg/g.

The pseudo-first-order model.
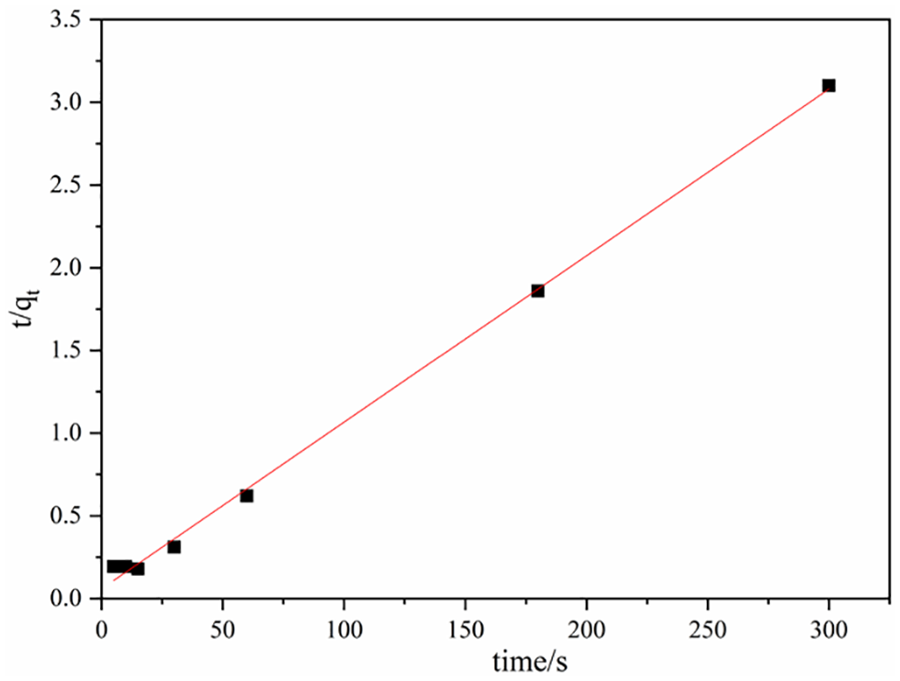
The pseudo-second-order model.
The fitting data of dynamic model.
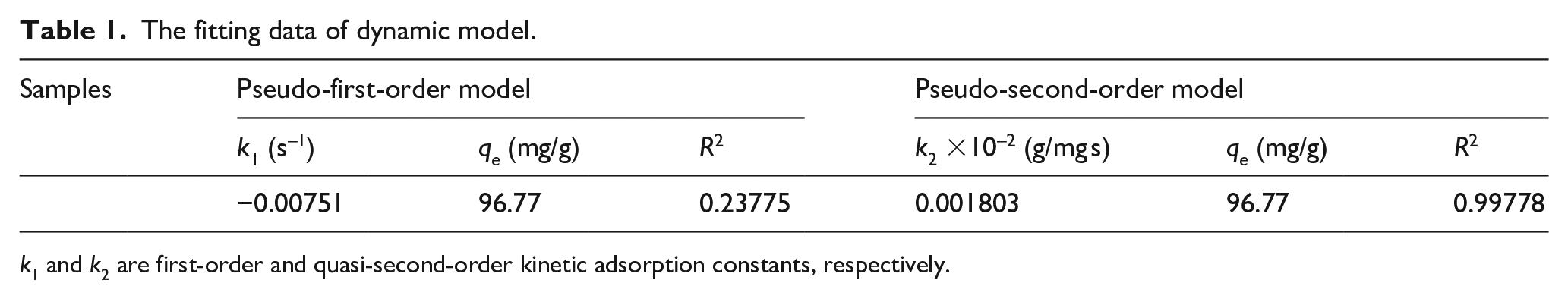
k1 and k2 are first-order and quasi-second-order kinetic adsorption constants, respectively.
Vertical combustion properties of cotton fabrics
Effect of adsorption time on the combustibility of cotton fabrics
The vertical combustion properties and LOI value of cotton fabrics treated with DNA flame retardant at different adsorption times are shown in Table 2. From Table 2, it can be seen that cotton fabrics without flame-retardant treatment were fully burnt, the damage length was 350 mm, and LOI value was only 17. After applying DNA on to the fabrics, with adsorption time varying from 5 to 300 s, there was considerably higher flame retardancy. The fabric damage length was far less than that of the original sample, with an average damage length of only 29.5 mm more than 10 times lesser compared to the untreated fabrics. The LOI value increased from 17 to 23, indicating that the flame-retardant property of cotton fabric has been improved to a certain extent. The damage length of fabric treated with traditional cotton flame retardant is 330 mm, and the burning picture is shown as follows. It was also observed that the cotton fabric after DNA finishing can self-extinguish, with no flame propagation. As a cellulose fiber, the combustion process of cotton fiber is shown in Figure 7(a).37,38 The excellent flame retardancy imparted by DNA is mainly due to the presence of pentose, nitrogenous base groups, and phosphoric acid groups. These groups constitute a system similar to intumescent flame retardant. 33 At the beginning of combustion, DNA contacts with flame, and the phosphate groups are able to produce phosphoric acid, the deoxyribose units can act as a carbon source and as blowing agents and the nitrogen-containing bases (adenine, guanine, cytosine, and thymine) may release ammonia, similar to the flame-retardant mechanism of ammonium polyphosphate. 39 When the temperature of combustion zone increases gradually, the residue of DNA molecule combustion has thermal stability similar to that of ceramics, which makes it to have a stable carbon layer, isolates air and greatly reduces the flammability (shown in Figure 7(b)).40–42 The mechanisms of flame retardancy for DNA are to influence the pyrolysis reaction to produce less flammable volatiles and more residual char; the specific reaction is shown in Figure 7(c). 43
Vertical combustion properties of cotton fabrics treated with DNA at different absorption times and traditional flame retardants at 300 s.

LOI: limiting oxygen index.

Mechanisms of flame retardancy: (a) a diagram of the current model of combustion of textile fiber, (b) flame-retardant mechanism of DNA finished cotton fabric, and (c) chemical reactions in flame-retardant process.
Stability of flame-retardant finished cotton fabrics to washing
The ability of the flame-retardant cotton fabrics to washing was tested and the results showed that the cotton fabrics treated in DNA solution for 300 s had the shortest damage length and the best flame retardancy, as shown in Supplemental Table S2. Therefore, the flame retardancy and washability of two kinds of cotton fabrics (without CA and EDC and with CA and EDC) with 300 s adsorption time were determined after five soaping times of AATCC standard (equivalent to 25 times of household washing). The specific results are shown in Table 3. It can be seen from Table 3 that with the increase of washing times, the flame-retardant properties of DNA finished cotton fabrics decreased to a certain extent. After five washings, the damaged length of cotton fabrics changed from 31 to 350 mm, similar to the untreated fabrics suggesting that the DNA finishing could only withstand five washes. Therefore, it can be used for non-durable flame-retardant products, such as disposable protective clothing, flame-retardant gloves, decorative fabrics.
Flame retardant–washable stability of cotton fabrics after finished with washing time.

CA: citric acid; EDC: 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride.
Interestingly, after flame-retardant finishing with CA and EDC, the damaged length of the fabric increased gradually, while the damaged length of the DNA flame-retardant cotton fabric without CA and EDC changed rapidly. After two times of washing, the fabric was completely damaged. This shows that the adsorption of DNA on cotton fabric after CA modification is the chemical adsorption of carboxyl group on cotton fiber.
Testing of tensile mechanical properties of cotton fabrics
The tensile properties of cotton fabrics treated with DNA at different adsorption time and original cotton fabrics were tested. The results are shown in Table 4. As observed from the table, it can be seen that the breaking strength, breaking elongation, and breaking time of cotton fabrics treated with herring sperm DNA in different adsorption time (from 5 to 300 s) are all enhanced compared with the original fabrics. However, the increase is not high, which shows that flame-retardant finishing will not affect the mechanical properties of cotton fabric in use. This is mainly due to the chemical adsorption between DNA flame-retardant finishing agent and cellulose macromolecule on the surface of cotton fiber, among adsorption layers, between adsorption layer and cellulose macromolecule, which make slippage between the macromolecular chains of fibers hindered in some extent during the stretching process. Therefore, the breaking strength and elongation increase slightly, and the breaking time also prolongs in the process of tension; the mechanical properties of finished fabrics by DNA are improved.
Tensile properties of cotton fabrics before and after flame-retardant finishing.

Conclusion
We demonstrate that flame-retardant finishing of cotton fabric treated with CA and with herring sperm DNA and EDC by a simple and efficient process is able to impart more than 10 times higher flame retardancy compared to the untreated fabrics. The modification of cotton fabrics with CA increases the free carboxyl groups and forms ester bonds, which is helpful for increasing the absorption of DNA on to the fabrics. The adsorption equilibrium of DNA molecule occurs at a concentration of 6%, as the absorption reaches saturation. The adsorption time of DNA on cotton fabrics showed that the adsorption of DNA molecules on cotton fabrics was more in line with the quasi-second-order kinetic model, and the saturated adsorption capacity was 97.66 mg/g. Cotton fabrics treated with DNA had excellent flame-retardant properties with a damage length of 30 mm compared to 350 mm for the untreated fabrics. Treated fabrics also showed good resistance to washing. In addition, flame-retardant finished fabrics did not show any deterioration in mechanical properties or processability. Ability to impart flame retardancy to cotton fabrics without the need for chemical solvents, excellent flame retardancy, and stability of the treated fabrics makes DNA a preferred choice for imparting flame retardancy to fabrics.
Supplemental Material
Supporting_information – Supplemental material for Imparting flame resistance to citric acid–modified cotton fabrics using DNA
Supplemental material, Supporting_information for Imparting flame resistance to citric acid–modified cotton fabrics using DNA by Qiulan Luo, Pu Gao, Jie Zhou, Jian Zhang, Wen Wu, Jianda Cao, Narendra Reddy and Hui Ma in Journal of Engineered Fibers and Fabrics
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge the financial support from the National Natural Science Foundation of China (grant number 51703079), Key Laboratory of Yarn Materials Forming and Composite Processing Technology, Zhejiang Province (MTC-2020-12) and Key Scientific Research Program in college of Nanhu (N41472001-49).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


