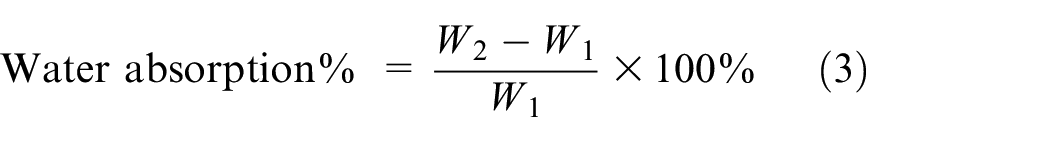Abstract
Due to the increasing use of laminated safety glass, the issue of polyvinyl butyral (PVB) resource waste has become increasingly serious. The complex composition of waste PVB presents significant challenges for recycling and application. In this paper, PVB was first grafted with phthalic anhydride (PA) to investigate the effect of the modification process on the coating film and coated fabric, thereby guiding the modification research on recycled PVB (rPVB) and successfully preparing rPVB-P. The results indicate that the optimal modification process for PVB involves a modification temperature of 70°C, a modification time of 6 h, and 6 g of PA. The feasibility of applying the prepared coated fabric in the field of trademark ribbon fabric was examined. The surface structure of the coated fabric, prepared with a kaolin mass fraction of 30%, was found to be uniform, with an ink contact angle of 20.32°, and the color fastness to washing and rubbing reached levels 4–5. The overall performance of the coated fabric prepared with rPVB was similar to that of the PVB-coated fabric, demonstrating that rPVB can effectively replace PVB for coating preparation.
Introduction
Polyvinyl butyral (PVB) is produced by polymerizing polyvinyl acetate (PVAC) or polyvinyl alcohol (PVA) with n-butyl aldehyde (BA) under acidic conditions. The polymerization process involves the condensation reaction of hydroxyl aldehyde with the raw materials and n-butyraldehyde (BA) under acidic conditions. Due to its exceptional transparency and impact resistance, 1 PVB finds extensive applications in safety glass for aviation, automotive, and high-rise buildings. Additionally, it is widely utilized in industrial sectors such as photovoltaics and textile manufacturing.2,3
With the increasing demand for safety glass, the recycling of waste polyvinyl butyral (rPVB) has become a research focus. It has been demonstrated that rPVB can be utilized in the preparation of textile coatings and electromagnetic shielding materials, indicating its significant application potential. 4 However, the composition of waste PVB film is relatively complex, often containing plasticizers, pigments, thermal oxidants, light stabilizers, antioxidants, and other chemical additives, which poses certain challenges for recycling. 5 Therefore, this paper begins with the modification of pure PVB to guide the research on the modification and application of rPVB.
As a natural non-metallic mineral material, kaolin is extensively utilized in coated fabrics, owing to its exceptional properties, which render it an ideal filler. In the domain of coated fabrics, the incorporation of kaolin has been demonstrated to enhance the coverage ability and ink absorbency of coated fabric, thereby enabling the fulfillment of diverse performance requirements across various application scenarios. Compared to traditional inorganic fillers such as calcium carbonate, talc, or silicon dioxide, kaolin is associated with notable environmental advantages. Its production process generates lower levels of pollution, aligning with contemporary principles of green manufacturing and sustainable development.6,7
Currently, organic solvents such as N,N-dimethylformamide (DMF) and dimethyl adipate (DPA) are commonly utilized for dissolving PVB to form coating pastes applied onto fabric surfaces. 8 However, these solvents, although effective, are environmentally harmful, thereby restricting the broader application of PVB. Hence, the development of environmentally friendly recycling technologies for PVB is deemed imperative. 9
Trademark fabric is a specialized textile product designed for the identification (printing) of various information, such as brand, size, price, composition, and care instructions, for textile goods. It is widely used in clothing, footwear, headwear, bags, home textiles, and other products as a primary means of product identification. Currently, traditional trademark fabrics are typically produced using a coating finish process, which imparts a combination of paper-like and cloth-like characteristics to the fabric. 10
Phthalic anhydride (PA) is recognized as a crucial chemical intermediate and is extensively employed in modification research on polymer materials due to its distinctive chemical properties. 11 In this paper, PVB was modified through PA grafting (PVB-P) to introduce new functional groups, with the structure characterized using infrared spectroscopy (FTIR), nuclear magnetic resonance (NMR), and X-ray diffraction (XRD) techniques. This modification aims to enhance the physical and chemical properties and expand the application range of PVB. The PA-modified PVB (PVB-P) was then utilized to prepare a coating slurry. By testing performance indicators such as acid value and conversion rate, a deeper understanding of the modification mechanism of PVB-P and its optimal modification process was achieved, providing a theoretical basis for the preparation of high-performance coating materials. By comparing the polymerization properties of the PVB coating slurry before and after modification and analyzing the mechanical properties of the coating film the coating formulation and preparation process were further optimized. The effect of kaolin on the properties of coatings was investigated, and its feasibility for application in trademark ribbon fabric was evaluated through tests on ink absorption, color fastness, and fabric style. This research not only aids in solving environmental pollution problems by applying PVB-P in the recycling and reuse of rPVB but also paves the way for the development of high-performance composite materials.
Experimental
Materials
Poly (vinyl butyral) (PVB) was purchased from Guangzhou Dingying Trading Co., Ltd (Guangdong, China). Phthalic anhydride (PA) and p-toluenesulfonic acid (p-TSA) were purchased from Aladdin (Shanghai) Reagent Co., Ltd (Shanghai, China). Recycled polyvinyl butyral (rPVB) was purchased from Yongsheng Chemical Co., Ltd (Zhejiang, China). Kaolin was purchased from Shanghai Maclin Biochemical Technology Co., Ltd (Shanghai). Polyester fabric (50 g/m2) was purchased from Huzhou Siny Label Material Co., Ltd (Zhejiang, China). Water based ink purchased from Epson (China) Co., Ltd (China).
Experimental Methods
Preparation of PVB-P/rPVB-P
First, 6 g of PVB, 0.6% p-toluenesulfonic acid (relative to the total amount of PVB and PA), and 100 mL of DMF were placed into a three-necked flask at room temperature. A certain amount of PA was then added, and the temperature was raised to a specific temperature. The mixture was allowed to react fully for a certain period to obtain PA-modified PVB (PVB-P) and a DMF mixture (the reaction equation is shown in Figure 1). Next, distilled water was added to the mixture to precipitate the PVB-P solid, which was then dried. Finally, PVB-P solid and 50 mL of ethanol were added into a three-necked flask and stirred to dissolve at 40°C. After complete dissolution, 2 mL of N-methyldiethanolamine was added for neutralization, resulting in a PVB-P alcohol solution. The preparation method of rPVB-P is mainly to replace PVB in the above scheme with rPVB.
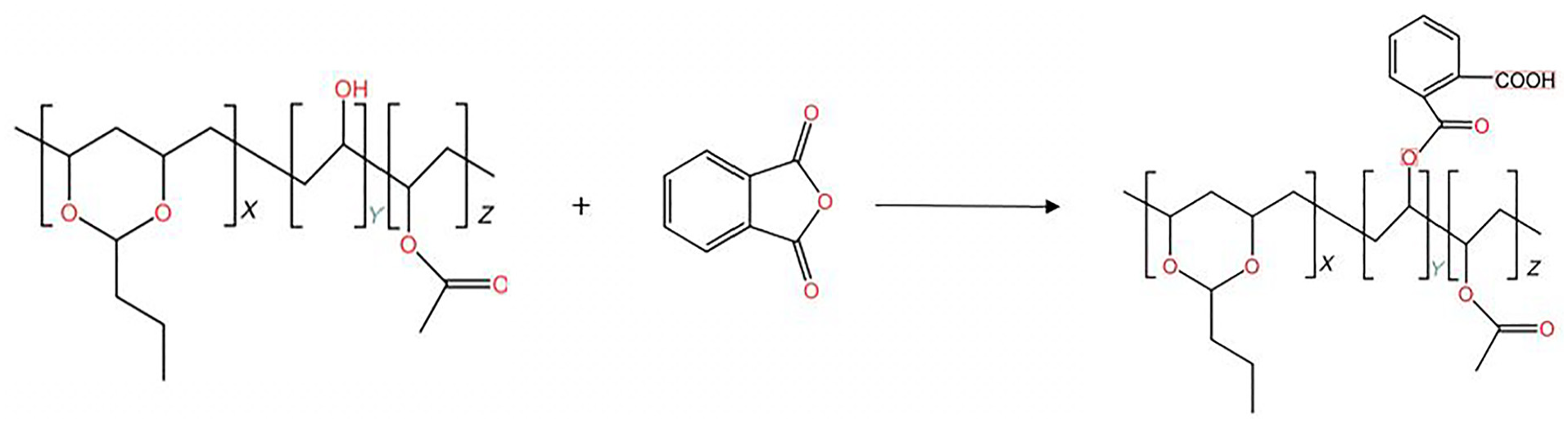
Equation for the reaction of PVB with PA.
Preparation of PVB-P/rPVB-P Emulsion
The highest solid content of PVB-P/ rPVB-P emulsion was achieved by adding an appropriate amount of Span 20 to PVB-P/ rPVB-P and stirring. Distilled water was then slowly added until no solid precipitation was observed.
Preparation of PVB-P/rPVB-P Latex Films
A certain amount of PVB-P/rPVB-P emulsion was poured into a polytetrafluoroethylene plate and dried at 120°C for 30 min to form a film, which was then removed for later use.
Preparation of Coated Fabrics
The RVB-P coating paste was formulated by incorporating a specified amount of kaolin into the previously prepared RVB-P emulsion. The resulting paste was applied onto polyester fabric and evenly spread using two glass rods. Subsequently, the coated fabric underwent a thermal curing process at a designated temperature for a specific duration, yielding the final RVB-P-coated polyester fabric.
Measurement
Characterization of Basic Properties
The infrared spectra of PVB before and after modification were measured using the ATR method with a Fourier transform infrared spectrometer (Tensor 27, Germany). A sample of 30 mg of PVB before and after modification was taken and dissolved in 0.5 mL of deuterated dimethyl sulfoxide (DMSO-d6). The hydrogen spectrum of the sample was then tested at 25°C using a nuclear magnetic resonance spectrometer (AV 400, Switzerland). The relative intensity of PVB before and after modification was tested at a scanning angle of 2θ ranging from 10° to 60° using an X-ray diffractometer (ARL, USA). The thermal stability of PVB/rPVB before and after modification was tested using a thermogravimetric analyzer (TGA/DSC2, Switzerland). The test conditions were as follows: a heating rate of 10°C/min, a temperature range of 25–700°C, a nitrogen atmosphere, and a flow rate of 20 mL/min.
Acid Value of PVB/rPVB
The acid value method was used to determine the content of carboxyl groups in PVB-P. The specific test method was as follows: 1 g of PVB-P sample was weighed using an analytical balance then transferred to a 150 ml conical flask, 50 ml of acetone was added to the conical flask, three to five drops of phenolphthalein indicator were added, and the titration was carried out immediately using a potassium hydroxide standard solution until the solution became reddish in color and remained there for 30 s. At this point, the end point of the titration was reached. The acid value was calculated according to formula 1:
where
A·V is the acid value, KOHmg/g PVB-P;
V is the volume of potassium hydroxide standard solution consumed in the titration, mL;
C is the concentration of the standard solution of potassium hydroxide, mol/L; and
M is the sample mass, g.
Conversion rate of PVB/rPVB
A certain quantity of PVB/rPVB and MA, denoted as W1, was taken into account. After the modification reaction, the mixed system was precipitated in distilled water. Simultaneously, the solid on the beaker wall and stirrer blade was collected. After drying, the solid was weighed as W2. The conversion rate C was calculated according to formula 2:
where
C is the conversion rate;
W 1 is the mass of the raw material before modification, in grams (g); and
W 2 is the mass of the product after modification, in grams (g).
Ink Absorbency of Coated Fabrics
The following tests are carried out with water-based ink.
(1) Contact angle
The surface of the coated fabrics was tested for the contact angle of ink drops using 2 μL ink drops, and the contact angle value was measured using the DSA-20 video contact angle tensiometer. The contact angle data of the coated polyester fabrics were obtained by averaging the values from five different positions respectively.
(2) Ink length
The coated fabric was cut into 15 × 4 cm thin strip specimens, placed in a constant temperature and humidity box [(23±2)°C, (50 ± 2%)RH] equilibrium for 4 h. After removing and fixing it on a rectangular wooden frame with a needle plate, 3 μL of ink was dropped on the surface of the coated fabric, and the rectangular wooden frame was quickly placed vertically in the constant temperature and humidity box to test the length of ink on the coated fabric for 4 h. The length of ink on the coated fabric was measured. The average value was taken after 5 repetitions. The smaller the length value, the better the ink absorption.
Measurement of Color Fastness to Rubbing and Color Fastness to Soaping
The color fastness to rubbing was tested according to GB/T 3920-2008 “Textiles–Tests for color fastness–color fastness to rubbing.”
According to GB/T 3921-2008 “Textiles–Tests for color fastness–Color fastness to washing with soap or soap and soda” the color fastness to soaping of the coated fabrics was tested. The samples were cut into 40 × 100 mm, pieces, placed in a sealed container with a diameter of 75 mm and a height of 125 mm. Ten stainless steel beads and an appropriate amount of detergent were added, and then the samples were taken out after stirring for 1 h at 50°C.
Handle Analysis
The handle of the coated trademark fabrics was determined using a Fabric Style meter (Nucybertek Phabromet, USA). Each sample was tested three times and the average value was taken.
Water Absorption of Latex Film
The weight of the prepared latex film was measured and recorded as W1, and it was soaked in deionized water for 48 h. At the end of soaking, the surface water was gently wiped off with filter paper, the sample was weighed again, and the weight was recorded as W2. According to formula 1.3, the water absorption rate of the latex film can be calculated as follows.
Mechanical Performance of the Light-cured Film
The mechanical properties of the light-cured film were tested using an Instron 2380 tensile testing machine, following the national standard GB/T 16491-2008. The test conditions were as follows: temperature of 23±2 °C, relative humidity of 50 ± 5%, clamping distance of 20 mm, and a tensile rate of 10 mm/min.
Results and Discussion
PVB-P Structural Analysis
The structure of PVB-P was analyzed by FT-IR, 1H-NMR, XRD, and TG. The structural changes of PVB before and after modification were analyzed using infrared spectroscopy (FTIR), and the results are presented in Figure 2.
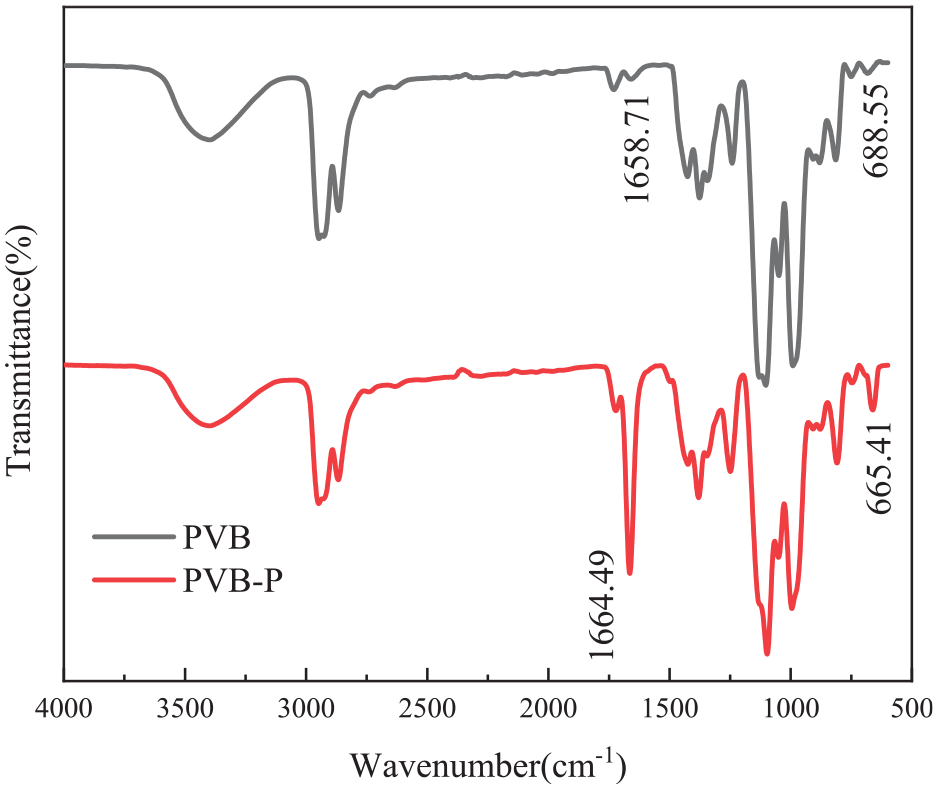
Infrared spectra of PVB and PVB-P.
It was observed that the modified PVB-P exhibited enhanced -C=C- stretching vibration peaks in the range of 665–689 cm-1 compared to unmodified PVB. Additionally, an increase in the -COOH stretching vibration peak was observed in the range of 1644–1673 cm-1. This enhancement can be attributed to the cleavage of the C–O bond of phthalic anhydride and the subsequent esterification reaction with the -OH group in PVB, resulting in the formation of a carboxylic acid group. As a result, the peaks corresponding to-C=C- and -COOH in PVB-P were enhanced, 12 thereby indicating the successful grafting of phthalic anhydride onto PVB.
The chemical structural changes of PVB and PVB-P were analyzed using nuclear magnetic resonance hydrogen spectroscopy (NMR), and the results are presented in Figure 3. Compared with the characteristic peaks of PVB, the peak values of PVB-P at chemical shifts δ of 7.34, 7.45, 7.59, and 13.47 ppm are significantly enhanced. The chemical shifts δ = 7.34, 7.45, and 7.59 ppm correspond to different protons in the benzene ring, while δ = 13.47 ppm corresponds to the protons in the -COOH group. This enhancement can be attributed to the breakage of the C–O bond in PA and the esterification of the -OH group in PVB, resulting in the introduction of -COOH and benzene rings. As a result, the content of benzene rings and -COOH in PVB-P increased, further enhancing the proton peaks at these positions. These results indicate that PVB and PA underwent a successful esterification reaction, completing the grafting modification process.
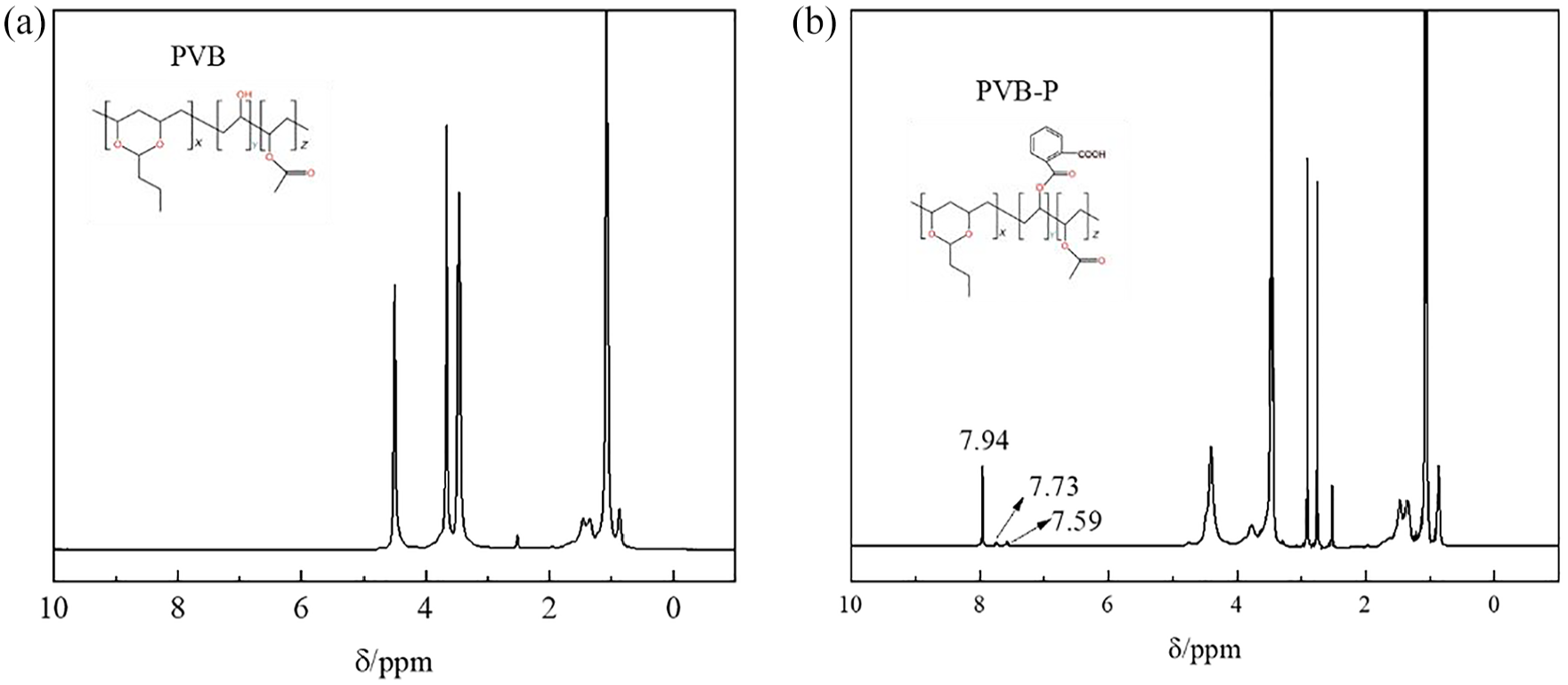
1H NMR spectra of PVB and PVB-P: (a) PVB and (b) PVB-P.
The XRD patterns of PVB before and after modification are presented in Figure 4. It can be observed that the main diffraction peak of unmodified PVB is located at approximately 19.60°, while a strong diffraction peak of modified PVB-P is evident at 20.44°, with both peaks approximately at 2θ = 20°. This shift in diffraction peaks is attributed to the esterification reaction, which may alter the molecular spacing. Additionally, the introduction of carboxylic acid groups reduces the crystallinity of PVB. 13 PVB exhibits low crystallinity due to the presence of numerous vinyl acetate units in the macromolecular chain. Consequently, the polymer chain segments of PVB typically exist in a disordered state with limited crystallinity. 14 After modification, the spectral peaks become broader, and no distinct diffraction peak is observed, indicating the amorphous nature of the material. The degree of crystallinity decreases, while the degree of amorphousness increases. It can be observed that the crystalline morphology of PVB modified with phthalic anhydride remains unchanged.
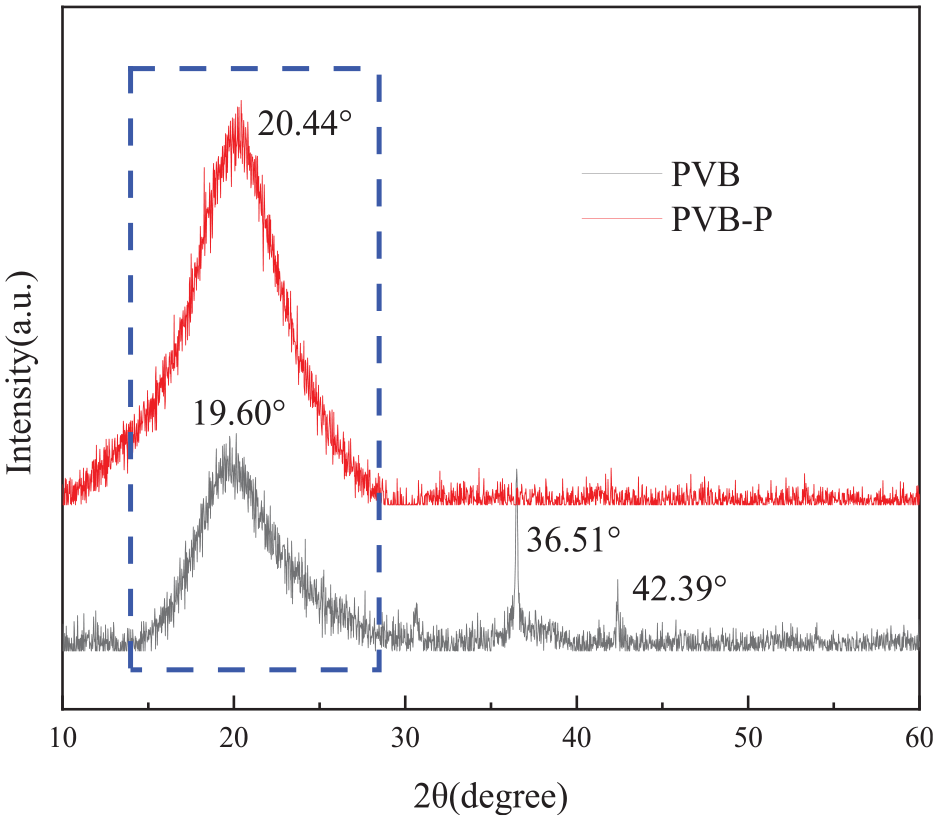
XRD patterns of PVB before and after modification.
The TGA and DTG curves of PVB before and after modification are depicted in Figure 5. Unmodified PVB exhibits a single rapid weight loss stage in the temperature range of 370°C–500°C, with a weight loss of 99.65%. The thermal decomposition during this stage is primarily initiated by the cleavage of the C–C bond in the PVB main chain. In contrast, modified PVB-P displays two rapid weight loss stages. The first stage occurs between 200°C and 370°C, primarily due to the cleavage of the grafted branched chain, resulting in a weight loss rate of 30.78%. The second stage, between 370°C and 500°C, is mainly attributed to the breakage of the carbon-carbon bond in the main chain, with a weight loss rate of 67%. As observed in the figure, the -COOH stretching vibration peak is located at 1720.22 cm-1, and the benzene ring stretching vibration peak is located at 744.40 cm-1. With an increase in the modification temperature, the intensity of both peaks first increases and then decreases, reaching maximum intensity at 70 °C modification temperature.
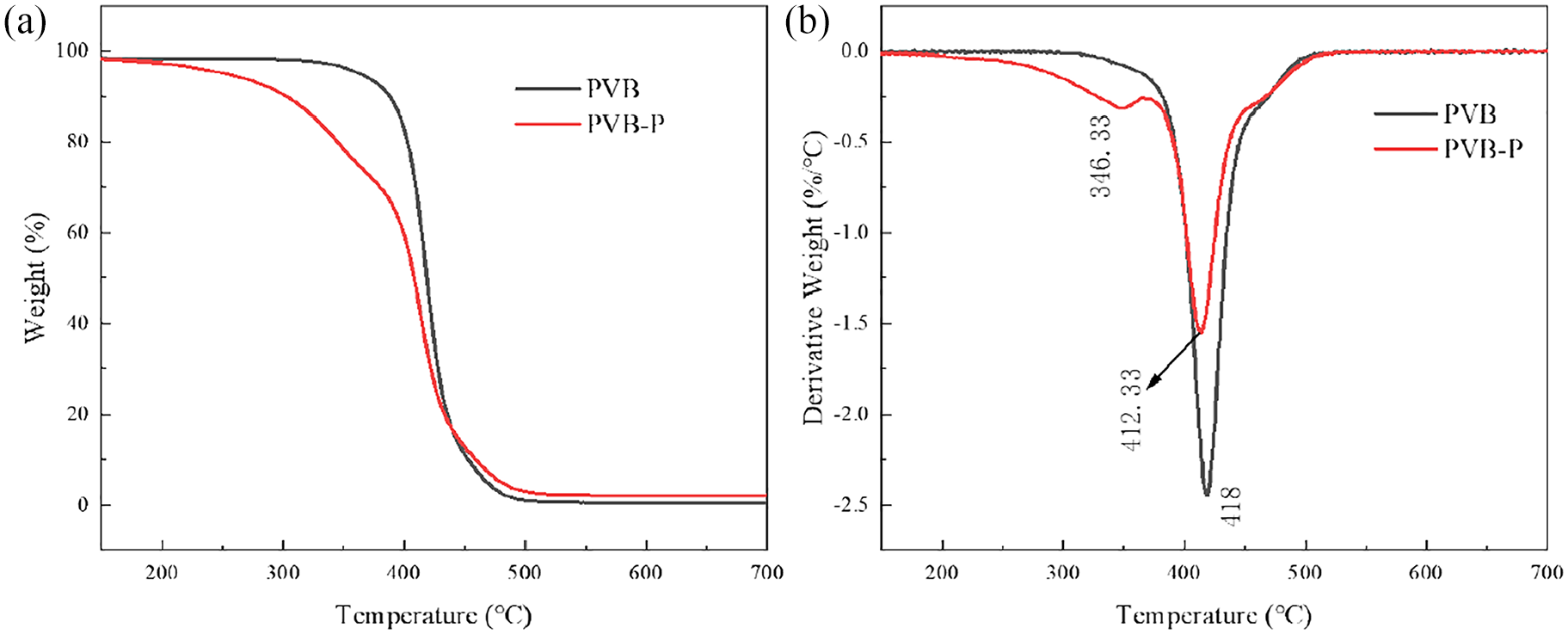
TG/DTG spectra of PVB before and after modification: (a) TG and (b) DTG.
The Preparation Process of PVB-P
To further investigate the optimal preparation process of PVB-P, PVB-P emulsion was prepared according to the Section “Preparation of PVB-P/rPVB-P Emulsion”. The effects of modification temperature, modification time, and PA content on the properties of the emulsion were investigated, and the optimal modification process for PVB-P was determined. The result is shown in Figures 6–8.
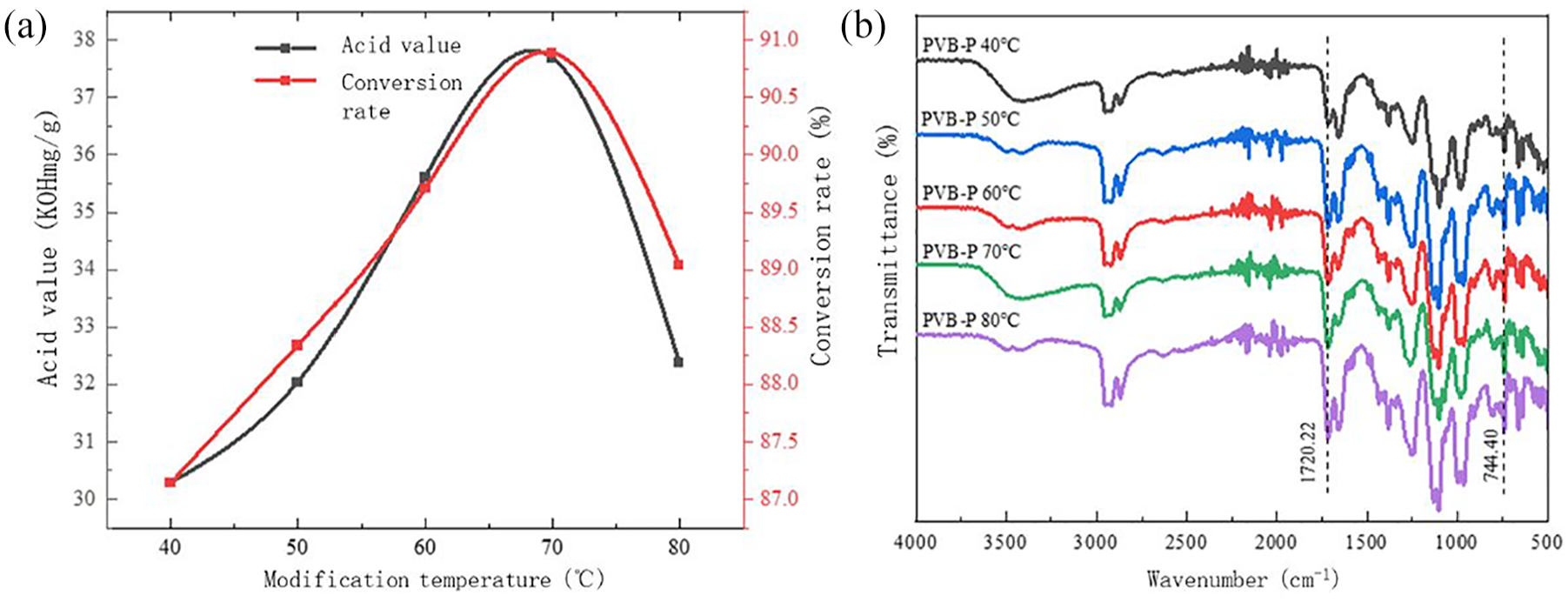
Effect of modification temperature on the modification effect of PVB-P: (a) acid value and conversion rate and (b) infrared spectra.
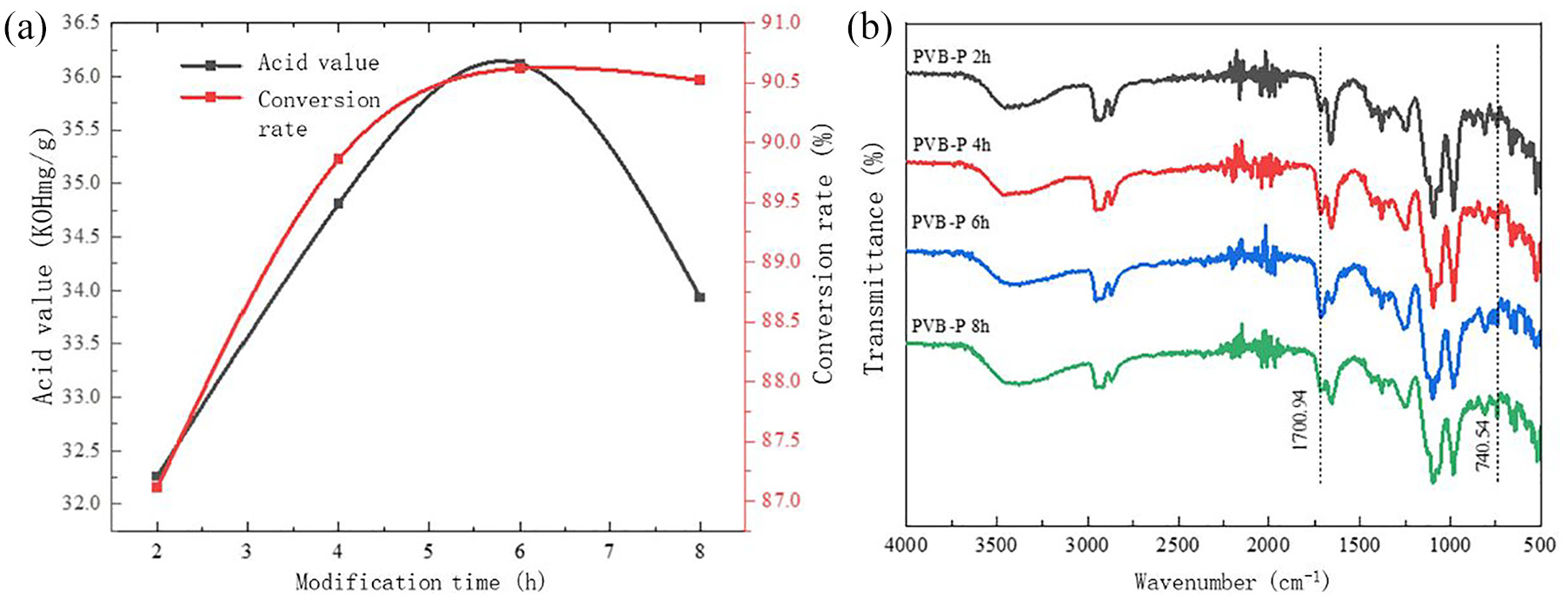
Effect of modification time on the modification effect of PVB-P: (a) acid value and conversion rate and (b) infrared spectra.
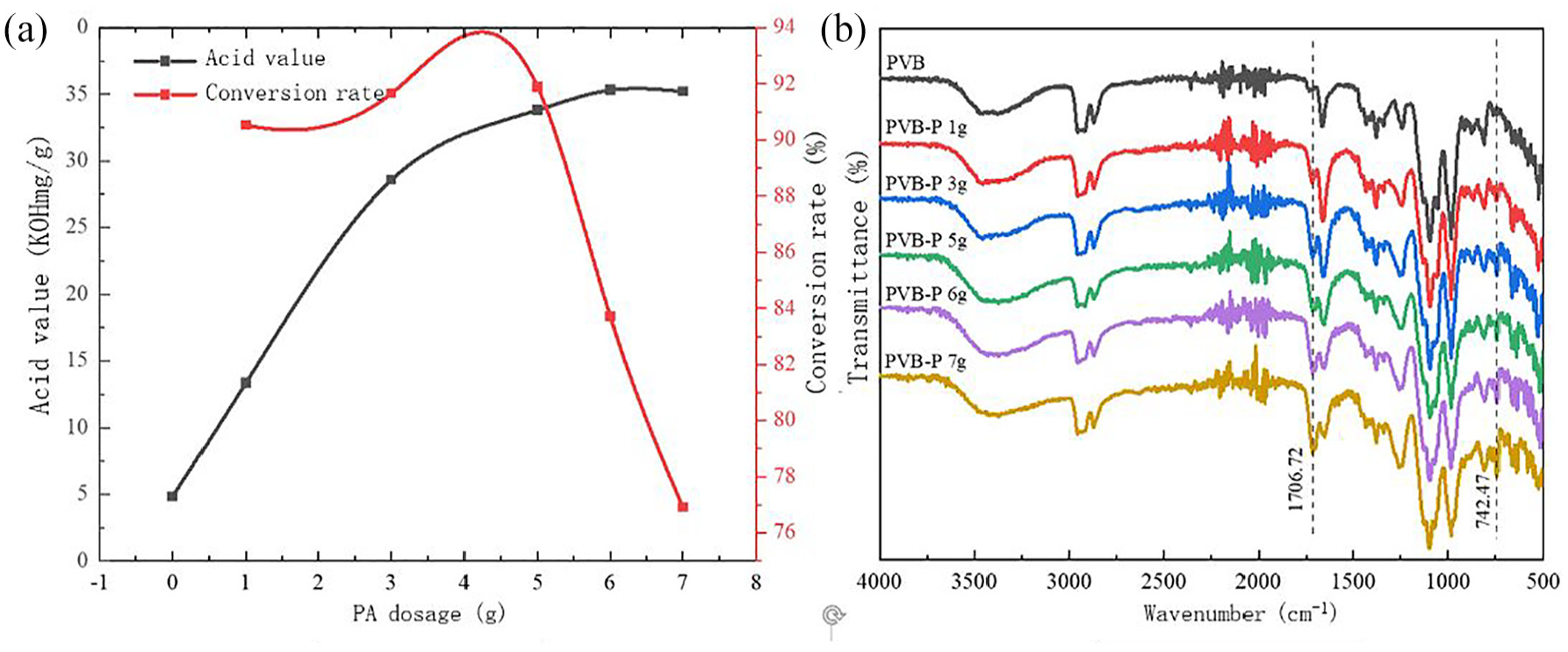
Effect of PA content on the modification effect of PVB-P: (a) acid value and conversion rate and (b) infrared spectra.
Figure 6 shows the effect of modification temperature on acid value and conversion of PVB-P. It shows that with increasing reaction temperature, the acid value and conversion of PVB-P exhibited a trend of initially increasing and then decreasing, reaching a peak at 70°C. This trend can be attributed to insufficient cleavage of the C–O bond of phthalic anhydride at lower temperatures, limiting the esterification reaction and resulting in lower acid value and conversion rates. As the temperature increased, the reaction rate accelerated, leading to an increase in the acid value and conversion rate. However, above 70°C, excessive temperature inhibited further progress of the reaction due to the exothermic nature of the esterification reaction. Therefore, the optimal modification effect was achieved at a reaction temperature of 70°C. As observed in Figure 6(b), the intensity of the -COOH stretching vibration peak at 1700.94 cm-1 initially increases and then decreases with an increase in the modification time. Conversely, the intensity of the benzene ring stretching vibration peak at 740.54 cm-1 gradually increases with the modification time and eventually stabilizes. The peak intensity of both peaks is highest when the modification time is 6 h.
Then, the effect of modification time on the acid value and conversion rate of the coating slurry was investigated.
In Figure 7, with increasing modification time, the acid value and conversion rate exhibited a trend of initially increasing and then decreasing, with the acid value reaching its maximum when the modification time was 6 h. The highest acid value of PVB was observed when the modification time was 6 h. When the modification time was less than 6 h, an increase in time resulted in enhanced activity in the reaction between the cleavage of the C–O bond of phthalic anhydride and the hydroxyl group in PVB. This increased activity promoted the generation of carboxyl groups and the grafting degree, thereby enhancing the modification effect. Additionally, the amount of double bonds introduced during this process increased with the extension of time. After the modification time exceeds 6 h, the esterification reaction between the hydroxyl group and the newly generated carboxyl group within the PVB molecule may become dominant. This could inhibit further grafting reactions and lead to a decrease in the grafting rate and material properties. Therefore, the optimal modification effect is achieved when the reaction time is 6 h.15–17
The effects of PA content on the acid value of the coated slurry and the conversion rate were investigated, and the results are presented in Figure 8.
As can be seen from Figure 8, with the increase of phthalic anhydride content, the acid value of PVB-P gradually increased, and slightly decreased after the content reached 6 g. The increase in acid value with increasing PA content can be attributed to the enhanced degree of esterification reaction, resulting in an increase in the acid value. However, the slight decrease observed after reaching 6 g may be due to the hydrolysis of the anhydride or the esterification of newly formed carboxylic acid groups with the residual hydroxyl groups of PVB. This process leads to a reduction in the number of free acidic groups, resulting in a decrease in the acid value. 18 The conversion rate exhibited a trend of increasing and then decreasing after reaching 6 g, which was attributed to the saturation of reaction sites on PVB. When the content of phthalic anhydride reached 6 g, the combined effect on both acid value and conversion rate was optimal. As depicted in Figure 8(b), the peak intensities of -COOH at 1706.72 cm-1 and the benzene ring at 742.47 cm-1 increased with the amount of PA. However, when the PA content reached 6 g, there was no significant change in the peak intensities. Therefore, it can be concluded that the optimal content of PA for modifying PVB is 6 g.
Effect of PA Content on the Properties of PVB-P Emulsions and PVB-P Latex Films
During the PVB modification process, the increase in the amount of PA enhances the degree of esterification reaction, significantly impacting the performance of the PVB emulsion. Consequently, the effect of PA amount on the viscosity, maximum solid content, and emulsion particle size of the PVB emulsion was studied. The results are shown in Figures 9 and 10.
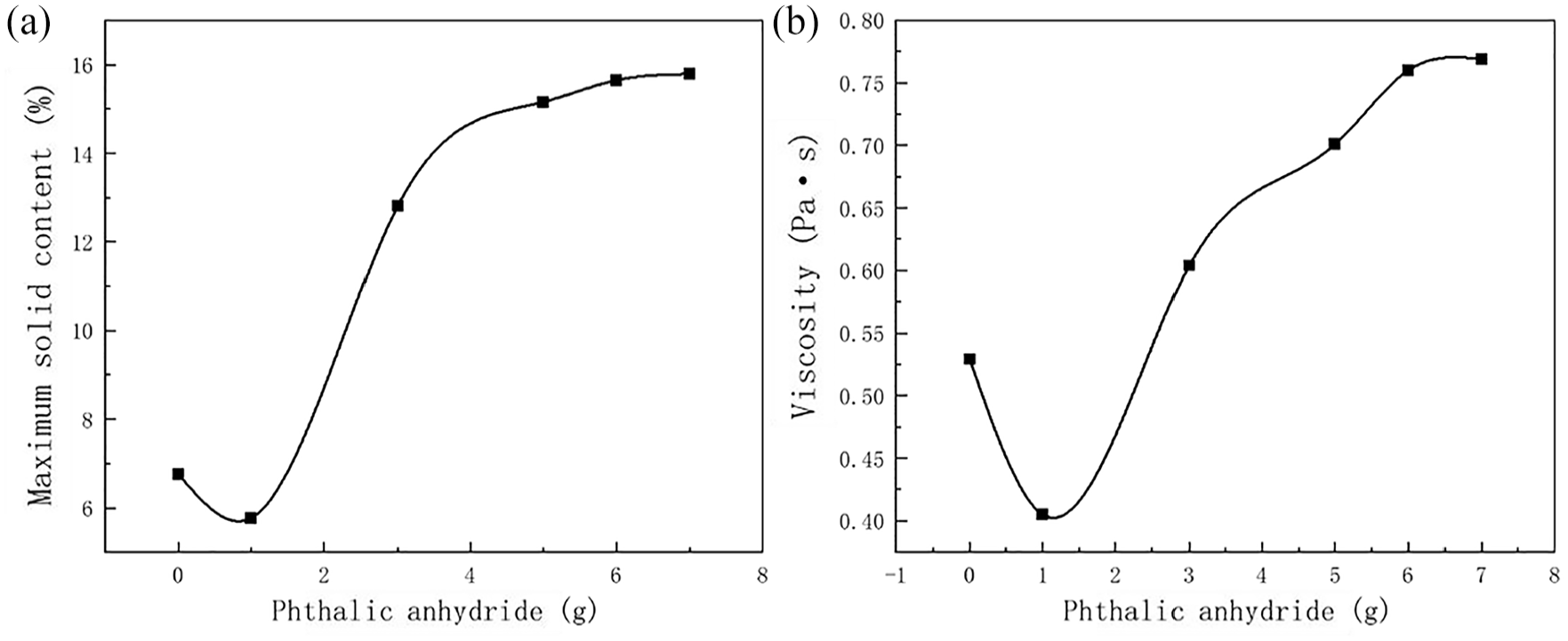
Effect of PA content on the viscosity of PVB-P emulsion: (a) maximum solid content and (b) viscosity.
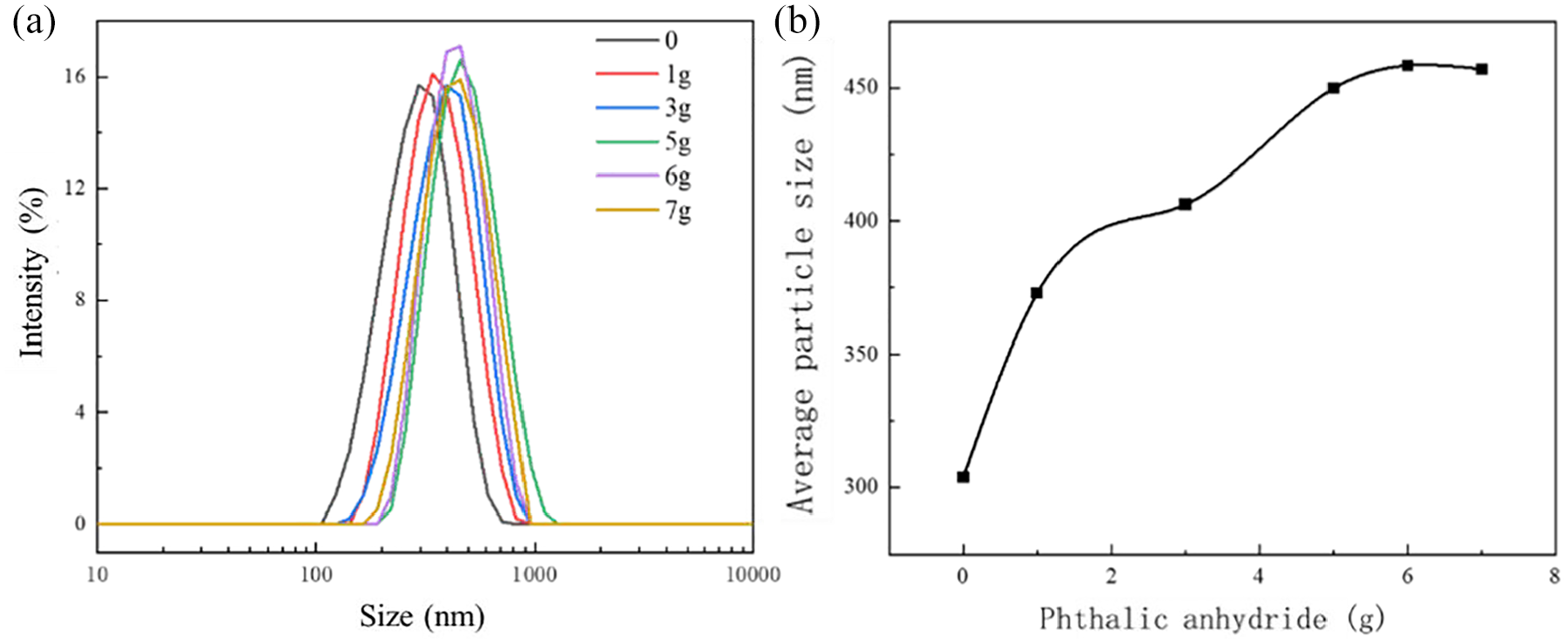
Effect of PA content on the particle size of PVB-P emulsion: (a) particle size distribution and (b) average particle size.
Figure 9 shows the effect of PA content on the viscosity of PVB-P emulsions. As observed in Figure 9(a), the maximum solid content of the emulsions increased significantly with the increase in phthalic anhydride content. It decreased slightly at lower contents of phthalic anhydride, then increased rapidly with further increase in content, eventually leveling off. This phenomenon can be attributed to the reaction between -COOH in PVB-P and -OH in Span 20, resulting in the formation of ester groups. These ester groups are hydrophobic, leading to a decrease in the hydrophilicity of the emulsion and consequently a decrease in the solid content. With the continued increase in phthalic anhydride content, the effect of -COOH surpasses that of the ester group, resulting in an increase in the hydrophilicity of the emulsion. This makes it easier to form dispersions with water, consequently increasing the solids content of the emulsion. When the phthalic anhydride content was increased to 6 g, further increases did not significantly enhance the solid content. This is because the emulsion reached saturation, and further reaction was not possible. As the content of phthalic anhydride was gradually increased, the viscosity of the emulsion first decreased, reaching a minimum value, then increased rapidly with further content increase, and finally leveled off. This pattern shows a certain correlation with the maximum solid content of the emulsion. When Span 20 reacts with a small amount of phthalic anhydride, the solid content of the emulsion decreases. The maximum solid content of the emulsion is the largest, and the viscosity is also the largest when the content is 6 g.
Then, the effects of PA content on the emulsion particle size were investigated by varying the amount of phthalic anhydride from 0 to 7 g; the results are presented in Figure 10.
The average particle size of the PVB-P emulsion increased with the increase in phthalic anhydride content and leveled off after reaching a content of 5 g.This may be attributed to the cross-linking of -COOH in PVB-P with -OH in Span 20, resulting in a change in the internal structure of the emulsion particles. As more reactions occur, the particle size increases due to the interconnection of the internal structure. 19 With a further increase in the amount of phthalic anhydride, the reaction reached its saturation point, leading to stabilization of the particle size.
Further, the effect of PA content on the properties of PVB-P latex films was investigated. The results are shown in Figures 11 and 12.
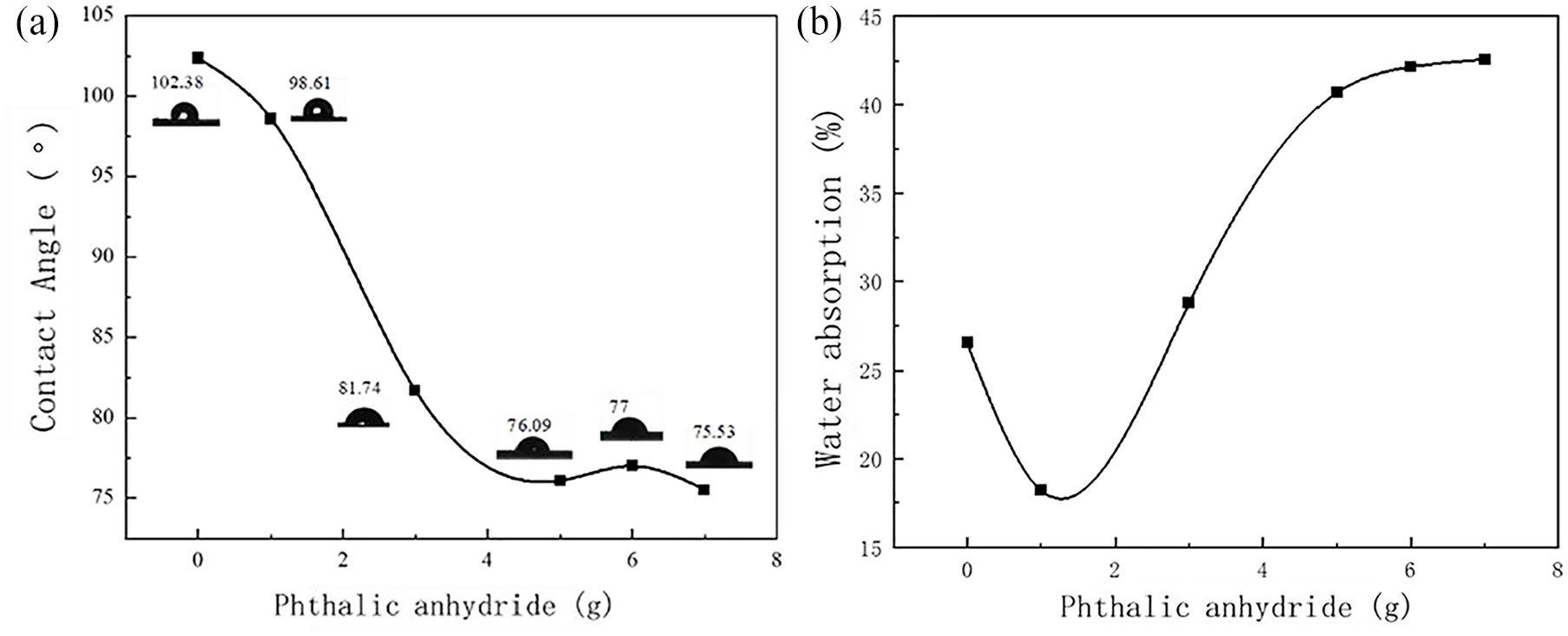
Effect of PA content on the contact angle and water absorption of PVB-P latex film: (a) contact angle and (b) water absorption.
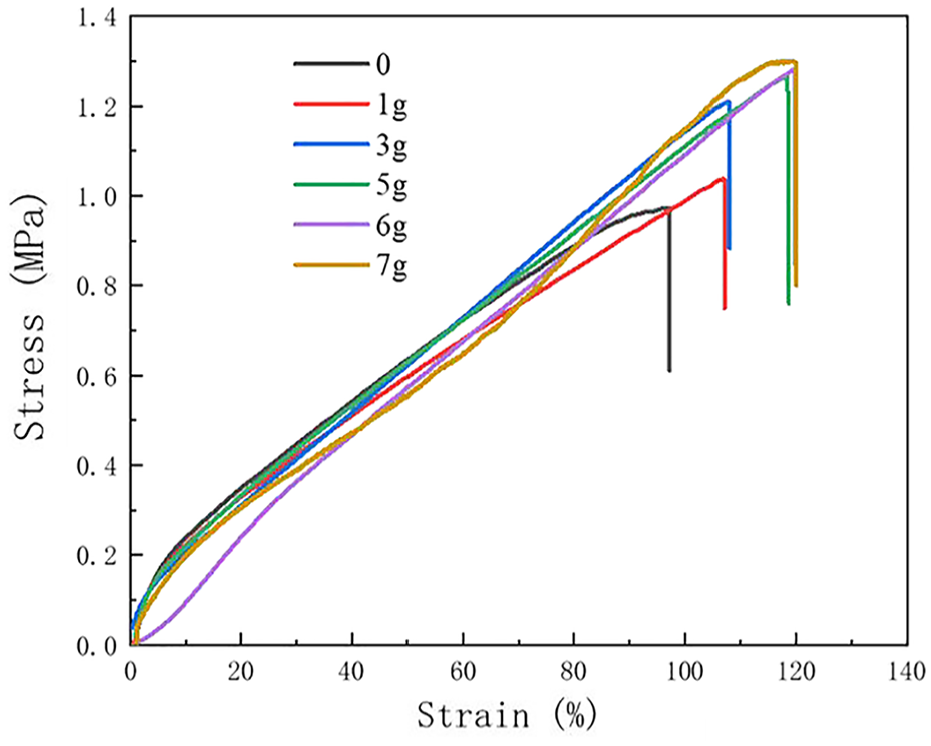
Effect of phthalic anhydride content on mechanical properties of PVB-P latex films.
Figure 11 shows the effect of PA content on contact angle and water absorption of PVB-P latex films. With the increase in phthalic anhydride content, the contact angle exhibited a significant decrease and then leveled off. This phenomenon can be attributed to the reaction between -COOH in PVB-P and -OH in Span 20 to form ester groups, which are hydrophobic. Consequently, this leads to a decrease in the hydrophilicity of the emulsion. As the content of phthalic anhydride continues to increase, the effect of -COOH exceeds that of the ester group. This results in an increase in the hydrophilicity of the emulsion film, leading to a decrease in the contact angle. The contact angle stabilizes when the reaction tends toward saturation. After the reaction reaches saturation, the contact angle tends to stabilize. Figure 11(b) illustrates the effect of PA content on the water absorption of PVB-P latex film. As observed, the water absorption of the PVB-P latex film decreases initially with the increase in the amount of PA, then increases, and finally remains relatively stable. This trend corresponds to the results obtained from the contact angle tests of the PVB-P latex film. These results suggest that the modification of PVB by PA enhances its hydrophilicity.
Then, the mechanical properties of PVB-P latex films with different PA content were tested; the results are shown in Figure 12. The increase in the amount of phthalic anhydride resulted in an increase in the stress of the latex film. When PA is 6 g, the stress is 1.84 MPa and the strain is 119.97%. This increase can be mainly attributed to the introduction of unsaturated double bonds (-C=C-) in the modified PVB during the polymerization process. These unsaturated double bonds increased the resistance of molecular movement space and enhanced the rigidity of the molecular chain. Consequently, this led to an increase in the stress of the latex film and an improvement in its mechanical properties.
Preparation and Properties of rPVB-P
The modification of rPVB was carried out using PA, with the structure and modification process of rPVB-P being studied. The performance of rPVB-P and PVB-P emulsions was compared, and the feasibility of preparing rPVB-P emulsion was explored.
Structural Analysis of rPVB-P
Figure 13(a) and (b) presents the 1H-NMR spectra of rPVB and rPVB-P. As illustrated in Figure 13(a), the peaks of rPVB-P at chemical shifts δ of 7.39, 7.56, 7.67, and 13.36 ppm are significantly enhanced compared to the characteristic peaks of rPVB. The chemical shifts at δ = 7.39, 7.56, and 7.67 ppm correspond to different protons in the benzene ring, while δ = 13.36 ppm corresponds to the proton in -COOH. These enhancements indicate that the grafting modification of rPVB with PA has been successfully completed. The structure of the modified rPVB was characterized using infrared spectroscopy, and the results are presented in Figure 13(c). Compared with pristine rPVB, the vibrational peaks of the double bond C=C in the 1644–1669 cm-1 range and the carboxylate COOH in the 1731–1734 cm-1 range of the modified rPVB-P were significantly enhanced. This indicates that esterification occurred between phthalic anhydride (PA) and rPVB, resulting in the formation of carboxyl groups. Consequently, the intensities of the characteristic peaks of C=C and COOH in rPVB-P increased. These results suggest that the grafting chemical reaction between rPVB and PA was successfully carried out.
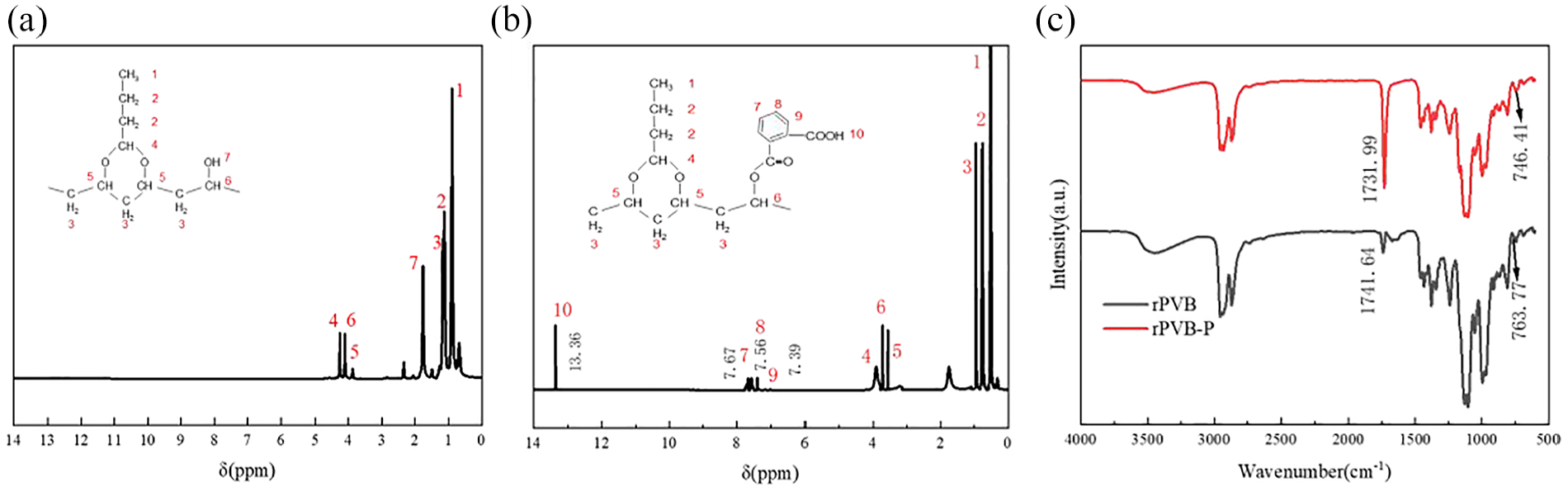
Structural analysis of rPVB and rPVB-P: (a) 1H-NMR spectrum of rPVB-P, (b) 1H-NMR spectrum of PVB-P, and (c) infrared spectra of rPVB and rPVB-P.
Emulsion Properties of rPVB-P
The particle size and stability of the rPVB-P emulsion were characterized, as shown in Figure 14.
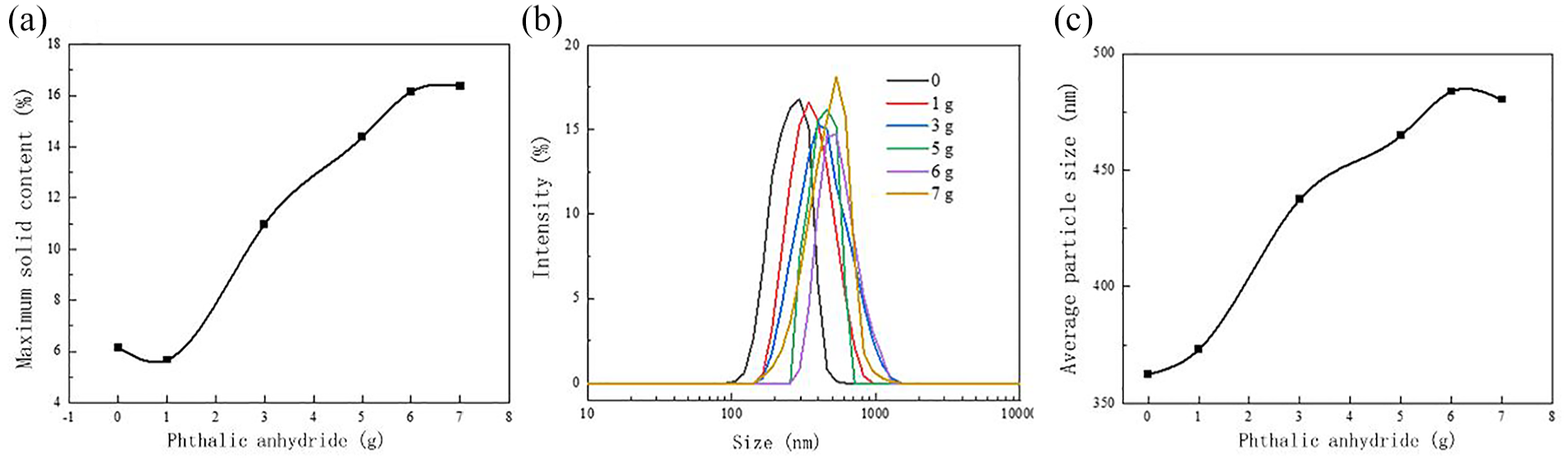
Properties of rPVB-P emulsion: (a) solid content, (b) particle size distribution, and (c) average particle size.
As observed in Figure 14(a), the solid content of rPVB-P initially decreases and then increases with the increase in PA dosage, reaching a maximum solid content of 16.37%. Figure 14(b, c) illustrates the effect of PA dosage on the particle size distribution and average particle size of the rPVB-P emulsion. Figure 14(b) demonstrates that the particle size distribution of the rPVB-P emulsion remains uniform across different PA dosages. Figure 14(c) indicates that as the PA content increases, the particle size of the emulsion gradually increases and stabilizes when the PA dosage reaches 6 g.
Effect of Different Filler Amounts on the Properties of rPVB-P Coated Fabrics
Surface Morphology
To investigate the effect of kaolin content on the surface morphology of the coated fabrics, the coated fabrics were prepared in accordance with section “Preparation of Coated Fabrics.” The amount of kaolin was varied from 0% to 40%. The results, showing the effect of kaolin content on the surface morphology of the coated fabrics, are presented in Figure 15.

Surface morphology of rPVB-P coated fabrics with different content of kaolin: (a) 10%, (b) 20%, (c) 30%, (d) 40%, and (e) 50%.
When kaolin is not added, the coating slurry penetrates between the fibers, resulting in exposed fabric fibers. With low kaolin content, the viscosity of the coating slurry is low, allowing it to penetrate between the fibers, leading to exposed fibers and a small number of particles on the surface. As the kaolin content increases, the viscosity of the coating slurry increases. This results in the surface of the fabric transitioning from having a small number of exposed fibers to being completely covered. This increase in kaolin content also leads to an increase in surface roughness and the presence of more particles.
Effect of Different Kaolin Contents on Ink Absorption of Coated Fabrics
To investigate the effect of kaolin content on the ink absorbency of the coated fabrics, the coated fabrics were prepared in accordance with section “Preparation of Coated Fabrics.” The content of kaolin was varied from 0% to 40% to study its effect on the ink absorbency of the coated fabrics. The results are presented in Figure 16.
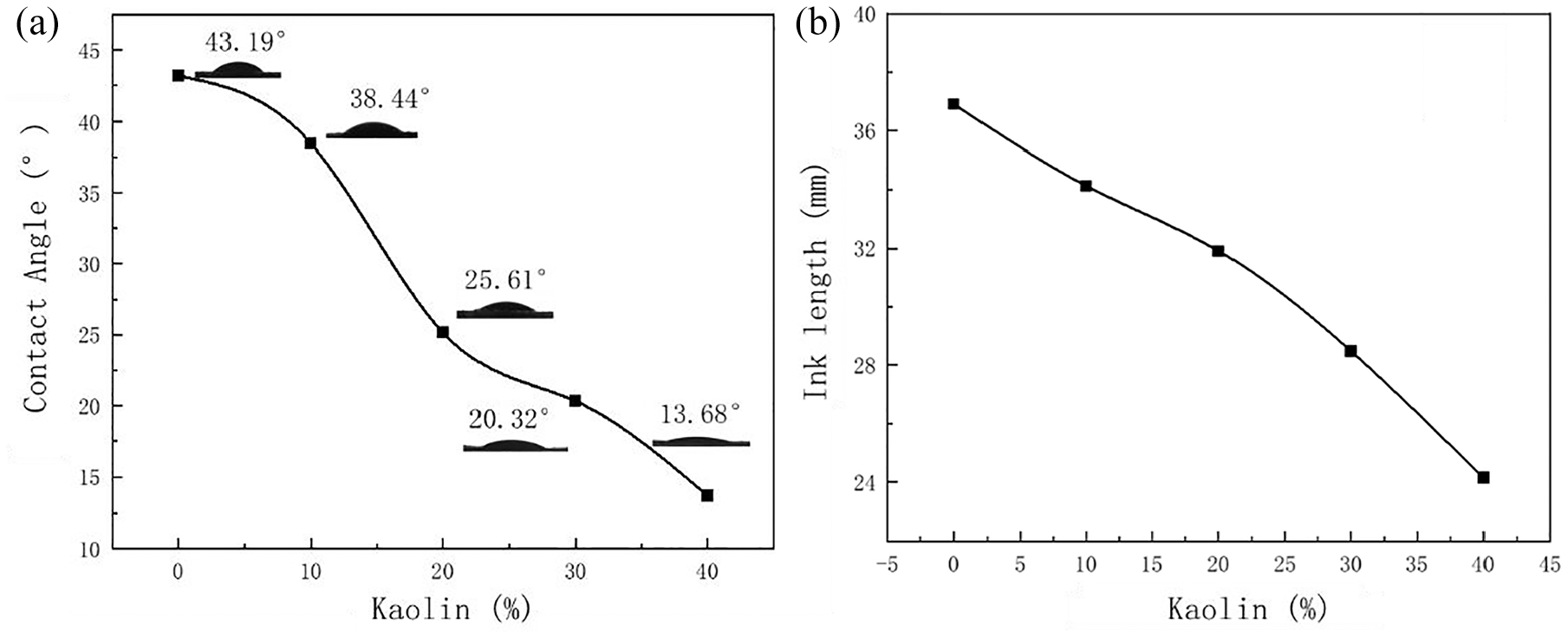
Effect of different kaolin content on the ink absorption of rPVB-P coated fabrics: (a) contact angle and (b) ink length.
With the increase in kaolin content, there is a gradual decrease in the contact angle of the ink and the length of the ink stain. This phenomenon can be attributed to the increased kaolin content, which leads to increased roughness on the fabric surface and the formation of more pores. Consequently, the ink absorption is enhanced.
Effect of Different Kaolin Contents on Color Fastness of Coated Fabrics
To investigate the effect of kaolin content on the color fastness of the coated fabrics, the coated fabrics were prepared in accordance with section “Preparation of Coated Fabrics.” The content of kaolin was varied from 0% to 40% to study its effect on the color fastness of the coated fabrics. The results are presented in Table 1.
Effect of different kaolin content on the color fastness of coated fabrics.
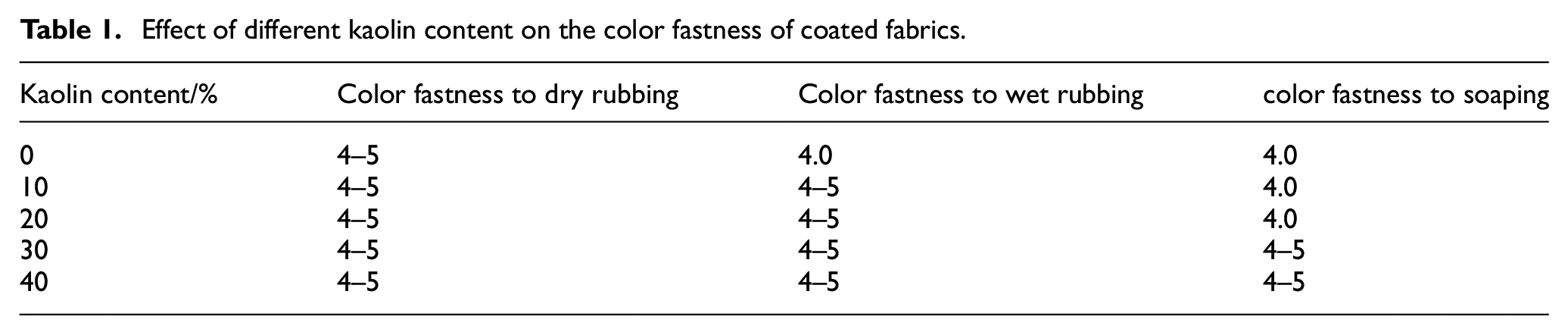
With the increase in kaolin content, the overall color fastness of the fabrics gradually increased, and at a content of 30%, all of them were able to reach grade 4–5. This improvement can be attributed to the increase in the number of pores, resulting in a larger surface area and enhanced capillary action, which promotes the initial absorption of ink. Additionally, the increased number of pores helps to lock the ink particles within the fabric structure during and after drying, thereby allowing for better penetration and retention of the ink.
Effect of Different Kaolin Content on the Fabric Handle of Coated Fabrics
To investigate the effect of kaolin content on the fabric style of the coated fabrics, the coated fabrics were prepared in accordance with section “Preparation of Coated Fabrics.” The amount of kaolin was varied from 0% to 40% to study its effect on the fabric style of the coated fabrics. The results are depicted in Figure 17.
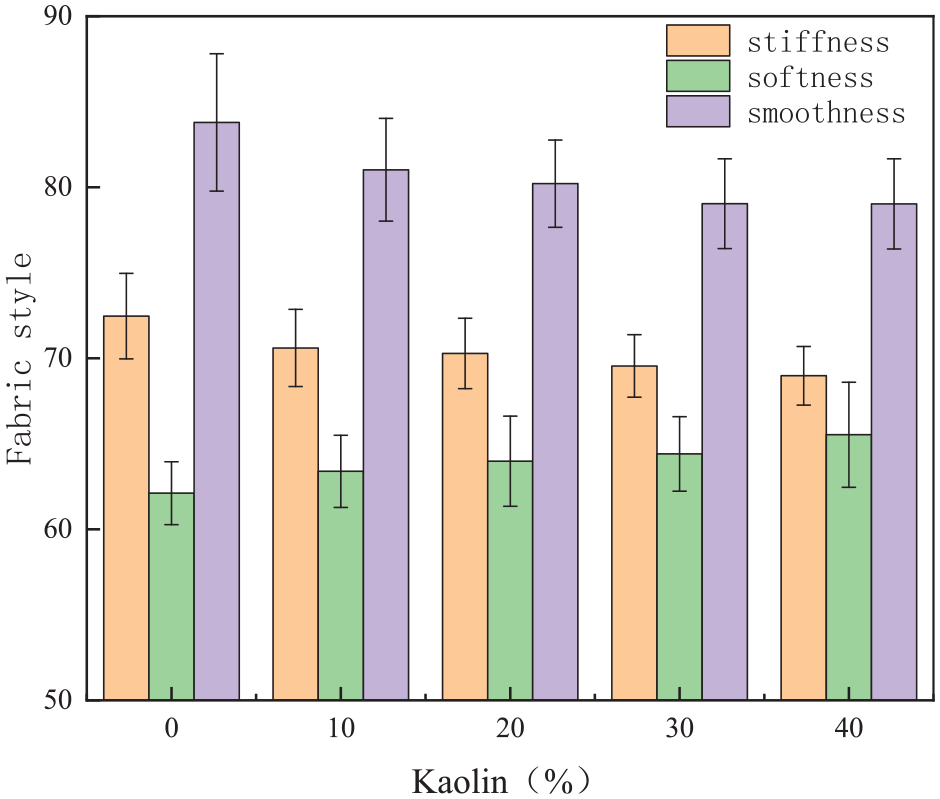
Effect of different kaolin content on the fabric handle of coated fabrics.
As the kaolin content increased, the stiffness of the fabric gradually decreased, while the softness and smoothness exhibited a trend of decreasing and increasing. The increase in filler content resulted in the formation of a denser coating of kaolin particles on the fabric, leading to increased stiffness. Additionally, more particle agglomerates appeared on the surface, reducing smoothness.
Conclusion
In this paper, pure PVB was initially modified with PA to successfully prepare PVB-P. On this basis, rPVB was modified, resulting in the successful preparation of rPVB-P and its emulsion, which were subsequently applied to coated fabrics. By examining the effects of the modification process and PA dosage on the properties of PVB-P/rPVB-P emulsion and latex film, the optimal process for PVB-P/rPVB-P was determined to be a temperature of 70°C, a reaction time of 6 h, and a PA dosage of 6 g. With the addition of kaolin, the ink absorption and color fastness of rPVB-P coated fabrics were found to be satisfactory, and the fabric exhibited a soft feel. The presence of plasticizers in rPVB had minimal impact on the modification effect, indicating that rPVB can be used as a substitute for PVB to prepare water-based polymer emulsions for textile coatings.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Basic Public Welfare Research Program of Zhejiang Province (LGF21E030004).
Data availability
All data have been reported in the article.