Abstract
The textile printing process’s use of artificial thickeners has a number of detrimental effects on the environment. Researchers use starch-based thickeners, which are the most popular natural thickeners for cotton printing due to their availability and low cost. However, the use of native starch-based thickenera is not recommended for reactive printing due to the presence of many accessible hydroxyl groups. Here, the main aim of this study has been the extraction of protein-based thickener derived from trimmed fleshing waste of animal hide for better colour fastness properties of reactive dye print on cotton. The physicochemical properties and the application of this thickener with sodium alginate in different proportions were studied. In addition, after printing was performed on the cotton fabric, the physical and mechanical properties of the printed fabric were evaluated. The results show that the properties of the printed fabric, namely, overall fastness and softness, are dependent on the percentage of gelatin thickener. Moreover, the analytical result showed that the colour strengths and colour fastness properties of printed cotton fabric were enhanced. Therefore, the developed protein-based gelatin is suitable to be applied as a sustainable, eco-friendly thickening agent for the application of reactive dye printing on textiles.
Introduction
Reactive dye printing is the most common and traditional method of printing on cotton fabric because of its reactivity towards the cotton substrate, excellent washing fastness, vibrant colour and range of hues. One of its significant advantages is obtained by employing a wide range of chromophores. In cotton fabric printing, 75% of printed items are produced using reactive dye printing techniques due to their obvious advantages, which include better colour fastness and print durability. 1 In order to control the printing quality, the printing paste should be controlled. For example, with controlled viscosity the print paste can be applied uniformly to the desired area of the fabric. 2 Therefore, the quality and characteristics of the thickener are crucial components for applying the colour paste without inking other parts of the fabric because the thickener reduces the printing paste fluidity and prevents the colourant effluence from reaching the borderlines. 3 In addition, the selection of an appropriate thickening agent for controlling the viscosity of paste is essential for producing high-quality printed fabric within the desired design and without printing defects. 4 For instance, high viscosity of the paste will cause trouble in the process and not only increase the price of the finished good but also cause more problems with regards to environmental wastewater. While low viscosity within the paste can be separated from the factors considered, such as the speed of the printing machine and the type of fabric used, it also greatly affects the spread of the printing paste and eventually reduces the delicateness of the borderlines in the print design. 5
There are different types of thickeners depending on the employed natural and synthetic polymers, namely, natural thickeners, modified natural thickeners and synthetic thickeners. Natural thickeners are polysaccharides obtained from natural sources such as plant exudates, seaweeds, seeds and roots. Some of them are suitable for prints using a specific dye category, but they must be chemically customized to meet the printing requirements. 6
Basically, the use of native starch-based thickeners at industrial levels has many limitations, such as low water solubility, easy thermal decomposition, high retrogradation and low shear stress resistance. 7 In addition, there are chemical interactions with reactive dyes, which result in them being poorly washed off from the surface of the fabric and forming complex crosslinking with the reactive dye due to the presence of a huge quantity of hydroxyl groups on the starch molecule. 8 This leads to enhanced hydrolysis of the dye molecules, which results in their being incapable of reacting with the fibre and the dyes being removed during washing of the printed fabric in the end use, which causes the fading of the printed colours. Therefore, nowadays, most textile industries use sodium alginate in place of starch. 9 This is due to its better printing performance and minimum reactivity towards dyes due to the limited reaction between alginate and dye by mutual anion repulsion of the carboxyl groups, even if sodium alginate contains hydroxyl groups on the molecule.10,11 However, sodium alginate has become rare and expensive due to the increased usage of reactive dyes in textile printing, which has reduced its application.11,12 However, the high cost and limited availability of natural thickeners, along with the growing demand for them, motivates the search for eco-friendly locally available materials that can be substituted for conventional thickeners.13,14
In order to get around this restriction, it is essential to substitute thickeners or stabilizers, which are produced from environmentally friendly natural based materials such as gums or gels, for use in the printing of natural and synthetic fabrics.
In this investigation, gelatin material was used as a thickening agent because of its bioadhesive, tasteless and odourless properties.15,16 Besides, it had consistenct properties; even when stored at room temperature for long periods, it is easy to dissolve it and make gels at lower temperatures. 17 Moreover, it has the capacity to have acid, base, or amphoteric properties in a solution because it consists of carboxylic and amide groups. Furthermore, research conducted by Abdou and Ahmed 18 investigated novel chitosan–starch blends as thickening agents and their application in the textile printing treatment of textiles with chitosan. The results indicated that the developed chitosan starch-based gelatin thickening agent was considered a multifunctional finish that not only contributes to antimicrobial properties but also results in an enhancement of colour strength in printing.
Overall, there has been little study done, thus there is not enough information on using biodegradable, environmentally friendly thickening agents in printing processes that minimize pollution in effluent. Moreover, to the best of the authors’ knowledge, there are no studies that examine protein-based natural thickeners for the application of reactive dye textile printing. The main objective of this study was to use water-soluble gelatin and sodium alginate thickeners in the preparation of a printing paste for the printing of cotton fabric with reactive dyes to improve the colourfastness and strength of the printed fabric. Finally, the physical and mechanical properties of the printed fabric were investigated.
Methodology
The methodological approach for this research contains two stages. First, the trimmed edge of a limed hide was used to create the protein-based gelatin printing thickening. After that, it was characterized. Second, various mix ratios of gelatin thickener and sodium alginate were used to print onto cotton cloth.
A preliminary experimental investigation revealed that the paste ratio of gelatin and sodium alginate affected the physical and mechanical properties of the printed cotton fabric during final use.
Finally, the physical and mechanical characteristics, namely, colour fastness and flexural rigidity of printed cotton fabrics, were investigated. The various paste ratios of the thickening agent and sodium alginate are presented in Table 1.
Different paste ratio of sodium alginate and gelatin thickening agent.

Materials
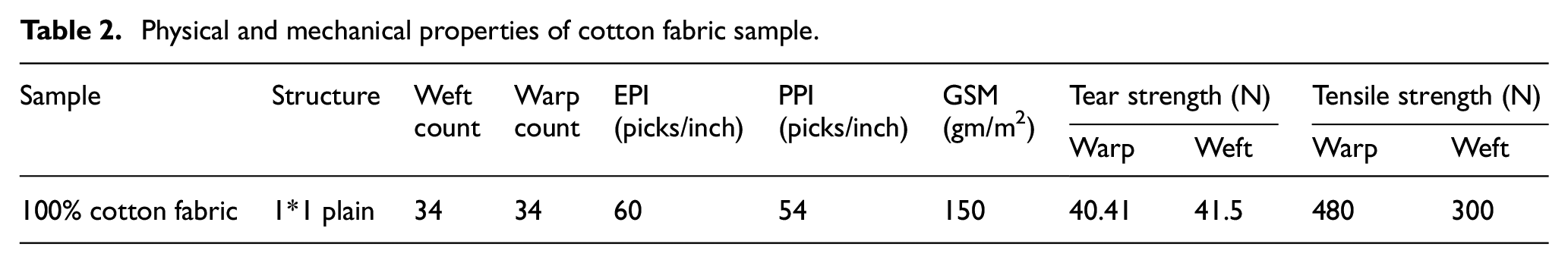
For this research, 100% cotton plain, scoured and bleached fabric was purchased from a local supplier, Kombolcha Textile Share Company. The physical and mechanical properties of the raw cotton fabric are displayed in Table 2. In addition, chemicals such as high-demand brands (Reactive Red 5 dye, mono-chlorotriazine C.I.) were used to prepare the printing paste. In addition, urea, sodium carbonate, sodium hydroxide, a wetting agent, sodium chloride and acetic acid were used. Furthermore, protein-based gelatin thickener was synthesized and used as a thickening agent in printing with reactive dyes on 100% cotton substrate.
Physical and mechanical properties of cotton fabric sample.
Methods
Synthesis of Gelatin
The method of extraction of gelatin from the raw hide trimming used in this study was the alkaline treatment–thermal hydrolysis technique 19 that does not need any chemicals that can affect the environment. The chemicals that are needed for pre-treatment before extraction are also eco-friendly. The main steps followed during the extraction of gelatin from hide trimminsg and fleshing waste are presented in Figure 1, and the steps are explained below.
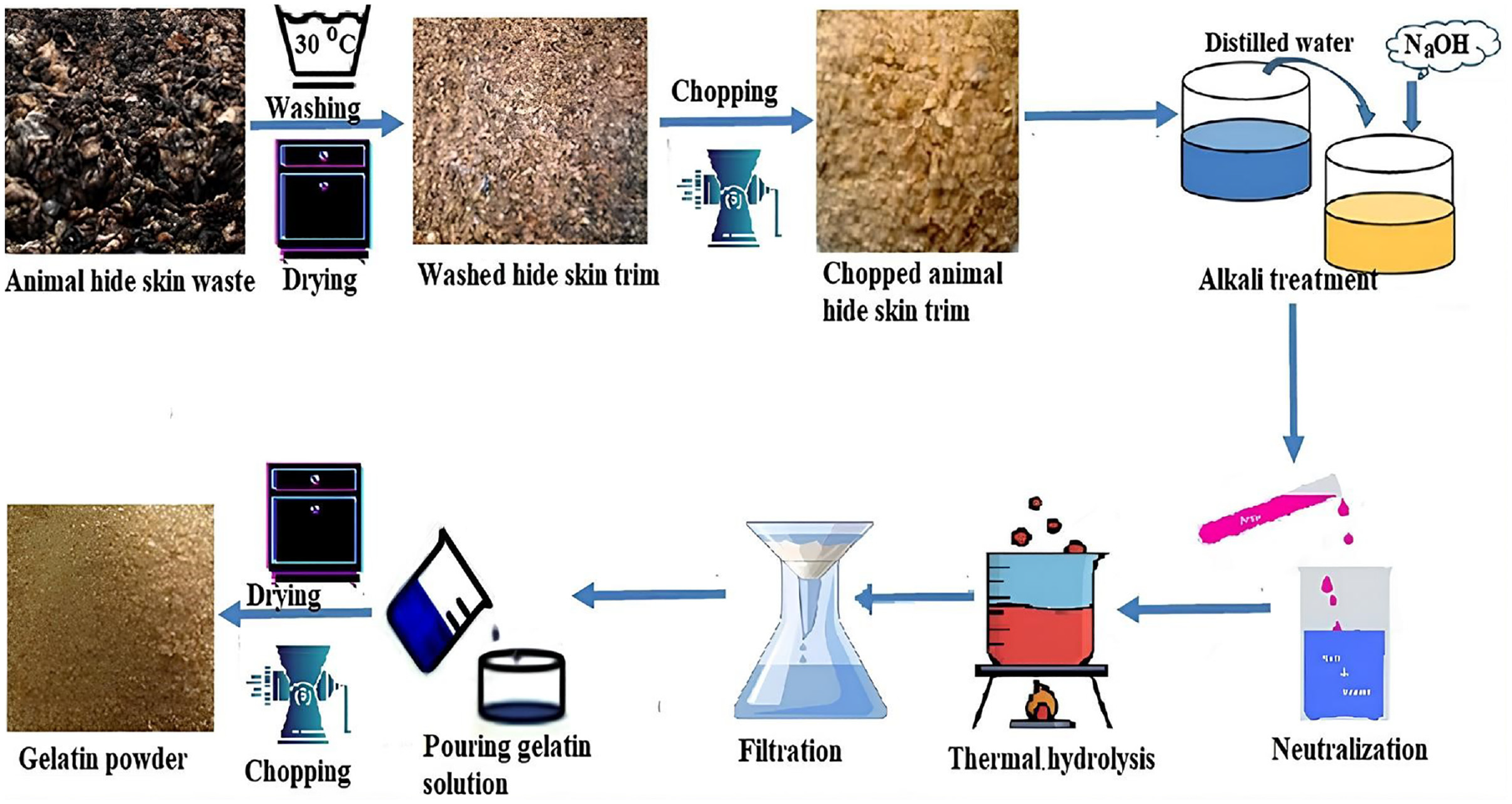
Schematic diagram of the synthesis of gelatin thickener from waste animal hide trim.
The animal hide trim and fleshing waste were collected from a nearby tannery in Addis Ababa, Ethiopia. After collection, it was extensively cleaned with distilled water to get rid of any extraneous material and contaminants that were stuck to the hide’s surface. 16 Subsequently, the hides were immersed in 1000 mL of water containing 0.3% (w/v) degreasers and 1% (w/v) sodium chloride for 2 h at 30°C to eliminate the fat components. The skins were once again cleaned with water for 30 min at 40°C, and drying was performed using an oven-drying machine. The washed and dried hide skin trim was also chopped and ground. After being chopped into tiny pieces, the hides were once again cleaned with water under high pressure. Then, the chopped hides were pre-treated for 24 h in 100 cm3 of 0.4% (w/v) sodium hydroxide. Afterwards, the alkali-treated hide was neutralized using HCl acid, and the gelatin was extracted via a thermal hydrolysis process using distilled water at a material liquor ratio of 1:5 (g/mL) at an optimal temperature (90°C) for an optimal duration (6 h), resulting in an optimal yield efficiency of 92.4%. To create ultra-pure gelatin, the extracted gelatin was first filtered through plain cotton fabric to remove larger, undesirable particles that were naturally present in the hides. After the gelatin was centrifuged using a 5810R ultrafiltration to remove any remaining colloids and turbidity, the gelatin solution was dried using an oven at 105°C and ground with a grinder machine to get gelatin powder.
Printing Paste Preparation
The gelatin thickener powder was immersed in water at 45°C prior to the printing paste preparation. Subsequently, the dye was combined with 2 g of urea and a small quantity of water. Then, the combination was stirred to guarantee that the dyes were thoroughly dissolved. To create consistent printing pastes, all the materials, namely, a beaker containing a mixture of 93 g of reactive dye, 2 g of sodium carbonate, 1 g of wetting agent which used to improve irrigation efficiency, 0.2 g of sequestering agent /ethylenediaminetetraacetic acid (EDTA) and 2 g of acetic acid, were mixed together. Then, the mixture was stirred continuously for 10 min.
After the preparation of the printing paste, manual screen-printing was performed with varying recipe components and varying curing times and temperatures on the surfaces of bleached cotton fabric. Then, the fabric was dried for 3 min at 100°C, and the printing paste was fixed using the thermo-fixation method for 3 min in a mini dryer at different temperatures (130°C, 145°C and 160°C).
The protein molecules have reactive functional groups such as amino and carboxyl groups, which can efficiently react with the reactive dye molecules. So, the presence of these reactive functional groups in proteins allows them to form strong covalent bonds with the dye, resulting in efficient dyeing. In addition, under acidic conditions, the protein in the gelatin becomes positively charged and it reacts with anionic dyes (i.e. contain negatively charged), whereas under basic conditions the protein in the gelatin becomes negatively charged and will be react easily with cationic dyes. This efficient dyeing mechanism ensures that the protein effectively thickens the ink, enhancing its viscosity and printability for reactive dye printing processes.
Therefore, the selection of a protein as a suitable thickening agent for reactive dye printing is based on its ability to efficiently react with the reactive dye molecules and improve the overall quality of the printed material.
Experimental Setups: Physicochemical Properties of Gelatin
Fourier Transform Infrared
The Fourier transform infrared (FTIR) spectra were collected in total reflectance mode with FTIR 4000. The FTIR test was performed on gelatin powder. The investigated wave number range was 4000–500 cm –1, and the resolution was 2 cm –1, with 64 scans performed on each sample. The spectra were acquired by absorbance.
Thermal Properties
The thermal degradation of the developed gelatin can be detected with differential thermogravimetric analysis (TGA 4000). These machines were used to determine how much energy the gelatin sample was absorbing or releasing at a heating rate of 10°C/min in a temperature range of 25–600°C.
Moisture Content, Viscosity and Gel Strength Test
The moisture content properties of the gelatin samples were conditioned at 25°C and 53% relative humidity (RH) for 48 h. Then the moisture content of the gelatin samples was determined through weight loss after drying at 105°C using an oven drying machine until a constant weight was obtained. The moisture content was calculated using Equation 1 as a loss in the weight of the original sample and expressed as a percentage:
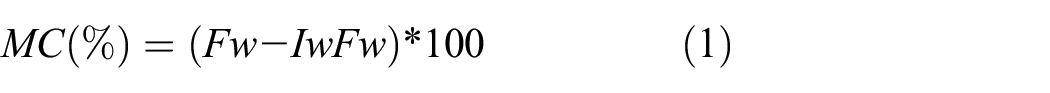
where MC stands for moisture content, Fw represents the final weight and Iw is the initial weight.
Furthermore, a rotational rheometer (RheolabQC, Anton Paar Instruments Ltd, Japan) fitted with a concentric cylinder (CC 39, 40 mm diameter, Austria) and a temperature control system was used to ascertain the rheological characteristics of the film-forming solutions (TEZ 150P-C). At 25°C, the shear rate was adjusted using the Ramp log process between 1 and 300 s, and the flow behaviour was recorded every 30 s. The Cross model further simulated the rheological properties of films.
Moreover, the strength of the developed protein-based gelatin thickener was measured using a bloom test based on international standards. 20 The gel strength of gelatin was measured by the rigidity of a gel formed from a 6.67% solution prepared at 60°C and cooled to 100°C for 17 h. The bloom was a measure of the force (weight) required to depress a standard plunger into the gel sample at a distance of 4 mm.
Percentage Yield of Extracted Gelatin
The yield of the gelatin extraction depends on the extraction temperature and extraction time. Gelatin was extracted from the collagen at various temperatures of 70°C, 85°C and 100°C, with extraction times of 120, 240, 360 and 480 min with maintaining constant conditions for other variables. The effects of the extraction time and extraction temperature on the gelatin yield were analysed using a two-way analysis of variance (ANOVA).
The extracted gelatin yield was calculated as a percentage using the following equation

where
Printed Fabric Characterization
Measurement of Colour Strength
The colour strength (

where
The colourfastness properties with washing, rubbing, light, and perspiration of the cotton-printed samples were evaluated as described below.
Testing of Rubbing Fastness
Rubbing fastness was investigated according to the ES ISO 105-X12:2018 standard. 21 A specimen of 220 mm × 80 mm was prepared. The specimen was mounted on the base board of the crock metre, putting the long direction of the specimen parallel to the rubbing track. The white rubbing cloth was mounted flat over the end of the peg on the crock-metre and held by means of the spring clip provided. The specimen was then rubbed back and forth over a straight track for 10 complete cycles at a rate of 1 s for each cycle. Finally, the white rubbing test cloth was removed, and the degree of staining on the undyed fabric was evaluated with a grey scale.
Testing of Washing Fastness
The washing fastness was measured with accordance in ES ISO 105 C06: 2018 22 standards. A specimen measuring 100 × 40 mm was sandwiched between cotton and polyester fabric and sewn along all four sides to form a composite specimen. A washing solution containing 5 g/l soap and 2 g/l sodium carbonate was taken in the launder metre with a liquor ratio of 1:50. The specimen was treated for 45 min at 60°C at a speed of 22 revolutions per minute. The specimen was removed and rinsed in running water without applying friction to the cold water. The stitch was opened on three sides and dried at room temperature. The change in colour and degree of staining were evaluated using geometric grey scales.
Testing of Light Fastness
Light fastness was tested according to the ES ISO 105 B02:2015 23 standard. A fabric sample, half of it exposed and half of it covered, was mounted in a frame together with a standard blue scale reference fabric, and it was placed in a testing chamber. This blue-scale wool fabric, consisting of eight dark to light blue shades, was used to record the colour change of the fabric. The test sample was exposed to continuous light from xenon-arc lamps at a radiance of 5 for about 72 h. The change was compared with the original unexposed sample, and assessed by blue scales 1–8.
Testing of Perspiration Fastness
Perspiration fastness was tested according to the ISO 105F10 standard. 24 Specimens of 40 × 100 mm were attached to the same size of multifibre to form a composite fabric. The composite fabric was wetted in acidic and basic solutions and placed between acrylic-resin plates under a pressure of 12.5 kPa separately. The test devices containing the composite specimens were placed in the oven for 4 h at 37 ± 2°C. The change in colour of each specimen and the staining of the adjacent fabrics were assessed and compared on a grey scale.
Determination of Flexural Rigidity
The stiffness of fabric was tested according to ASTM D1388-2007 25 using a Shirley Stiffness Tester (SDL International Ltd, UK). A rectangular sample of 2.5 cm × 20 cm (template size) in the warp and weft directions, respectively, was prepared. The sample was placed on the stiffness tester and moved gently until the sample coincided with the inclined indicator. The length to which the specimen moved was measured. The flexural rigidity was calculated using equation
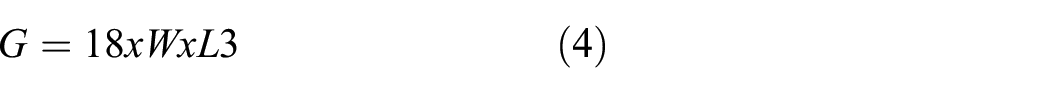
where
Determination of Tear Strength
The tear strength was tested according to the ASTM D2261-13 standard method. 26 The tear strength is the capacity of a material to withstand the tearing force required to propagate a tear after its initiation. The test specimen was cut according to the standard template size, and the required dimensions are specified in relevant test standards. The tearing strength was tested in the warp and weft directions using a digital tear tester with a C-type load. An initial cut of about 1 cm in length was made part way down the centre of a strip, and then the two tails thus formed were pulled apart so that a tear proceeds through the uncut portion of the fabric. The force used to tear the fabric was measured and recorded.
Determination of Tensile Strength
The tensile strength of the fabric sample was determined using the ASTM D5035-95 standard test method. 27 A fabric sample measuring 100 × 150 mm was prepared for both the warp and weft direction tests separately. The Universal Strength Tester was used to measure the tensile strength of the fabric in the warp and weft ways. The gauge length between the two jaws was adjusted to 75 mm.
Results and Discussion: Chemical Properties of Gelatin
Functional Group (FTIR) of Extracted Gelatin
The functional groups identified from the FTIR spectra of the developed gelatin sample are presented in Figure 2.
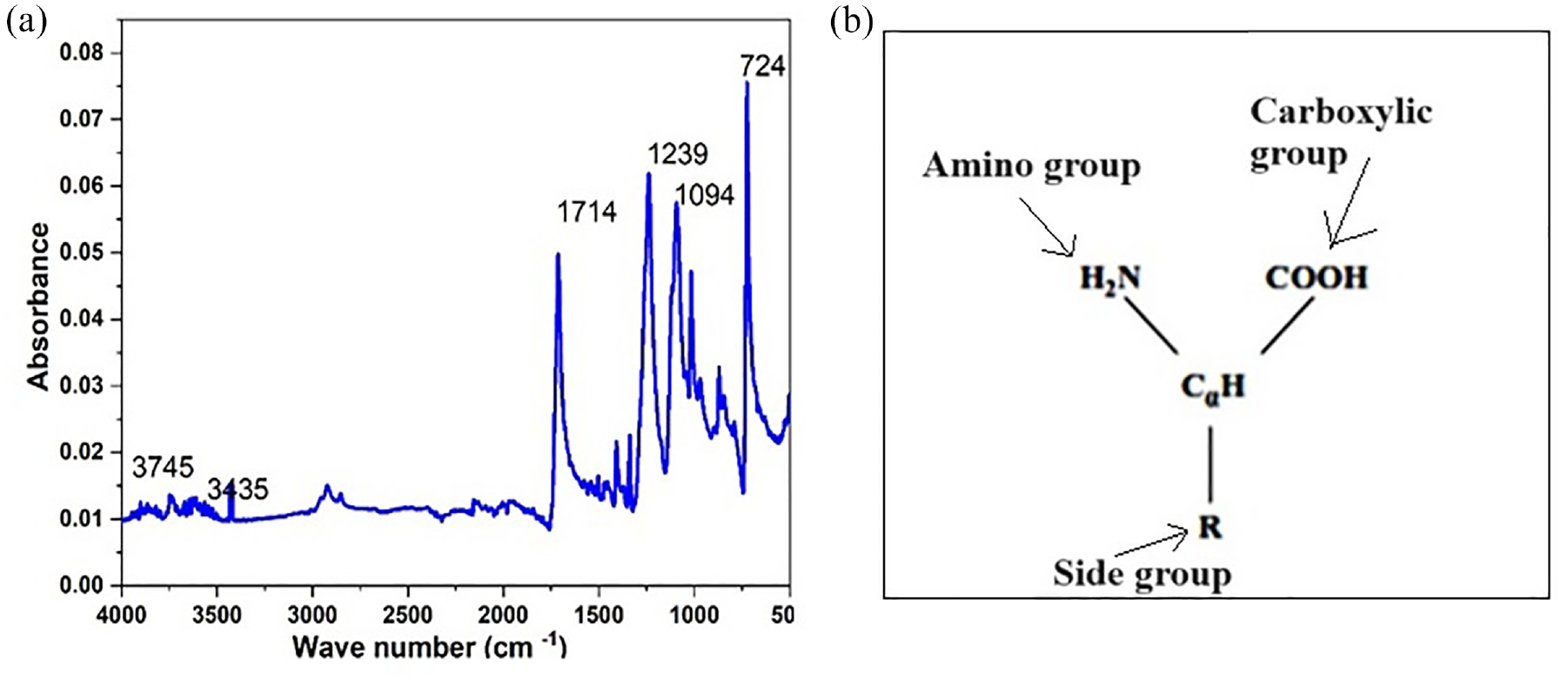
Functional group (FTIR) spectra of extracted gelatin (a) and its structure (b).
Gelatin has amphoteric properties. Gelatin in solution is amphoteric, capable of acting either as an acid or as a base. In acidic solutions, gelatin is positively charged and migrates as a cation in an electric field. In alkaline solutions, gelatin is negatively charged and migrates as an anion.
In acidic solutions, the gelatin develops positive charge and migrates as a cation in an electric field, and the dyes have a negative charge in aqueous solution, thus electrostatic attraction makes the dye easily react with gelatin, which causes hydrolysis of the dyes. However, in alkaline solutions, the gelatin develops negative charge and migrates as an anion. The dyes also have a negative charge in aqueous solution, thus leading to electrostatic repulsion between the dye and the gelatin, which makes the gelatin stable. When the pH of the printing paste becomes more alkaline, the pH of the charge that develops on gelatin is anionic, which is similar to the dyes. As we know that same charges repel each other rather than attract, this phenomenon is suitable for printing to reduce the hydrolysis of reactive dyes as the gelatin becomes stable. 28
The gelatin samples had major peaks in the amide region (I, II, and II). The amide I band at 17,600–1714 cm−1 was similar to that in the report by Xu et al. 29 The amide I vibration mode was primarily a C=O stretching vibration coupled to contributions from the CN stretch, to CCN deformation and in-plane NH bending modes. Besides these, the samples also had peaks at 1238–1240 cm−1, which was the amide II band, and at 1094 cm−1, the amide III band. The amide II band was generally considered to be much more sensitive to hydration than to secondary structure change.
In addition, the peak at 3524 cm−1 represents NH stretching coupled with hydrogen bonding. A free NH stretching vibration normally occurs in the range of 3400–3440 cm−1 while the position is shifted to lower frequency and amide A is found at 3435 cm−1. Moreover, at 3745 cm−1, the amide B peaks are found.
TGA (Thermal Properties) of Gelatin
The thermal properties of the extracted gelatin are displayed in Figure 3.
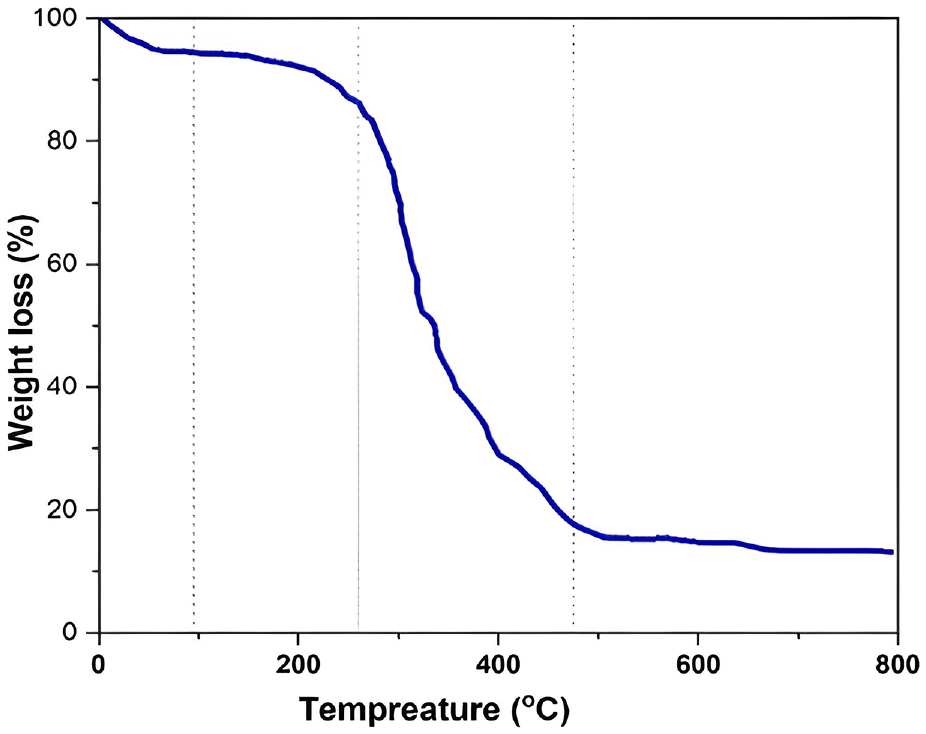
Thermal behaviour (TGA) graph of extracted gelatin.
From Figure 3, it can be shown that the thermogravimetric analyses (TGA) were conducted in the range of room temperature to 800°C to estimate the thermal stability of the developed gelatin in the control state. The TGA thermogram in Figure 3 shows a profile of weight loss with increasing temperature that can be divided into two separate regions. First is a slight degradation step with onset at around 95°C; this first stage leads to a weight variation of around −6.5%, due to loss of absorbed water. Second is a much more pronounced decline in mass close to 260°C, carrying a 14% weight reduction. In addition, at 47.5°C, a degradation occurs and 82% weight loss. This stage corresponds to the degradation of the low molecular weight protein fraction, as well as structurally bound water.
Yield
The gelatin yields at different extraction temperatures, that is, 70°C, 85°C and 100° C, with various extraction times (2, 5 and 8 h) are presented in Figure 4.
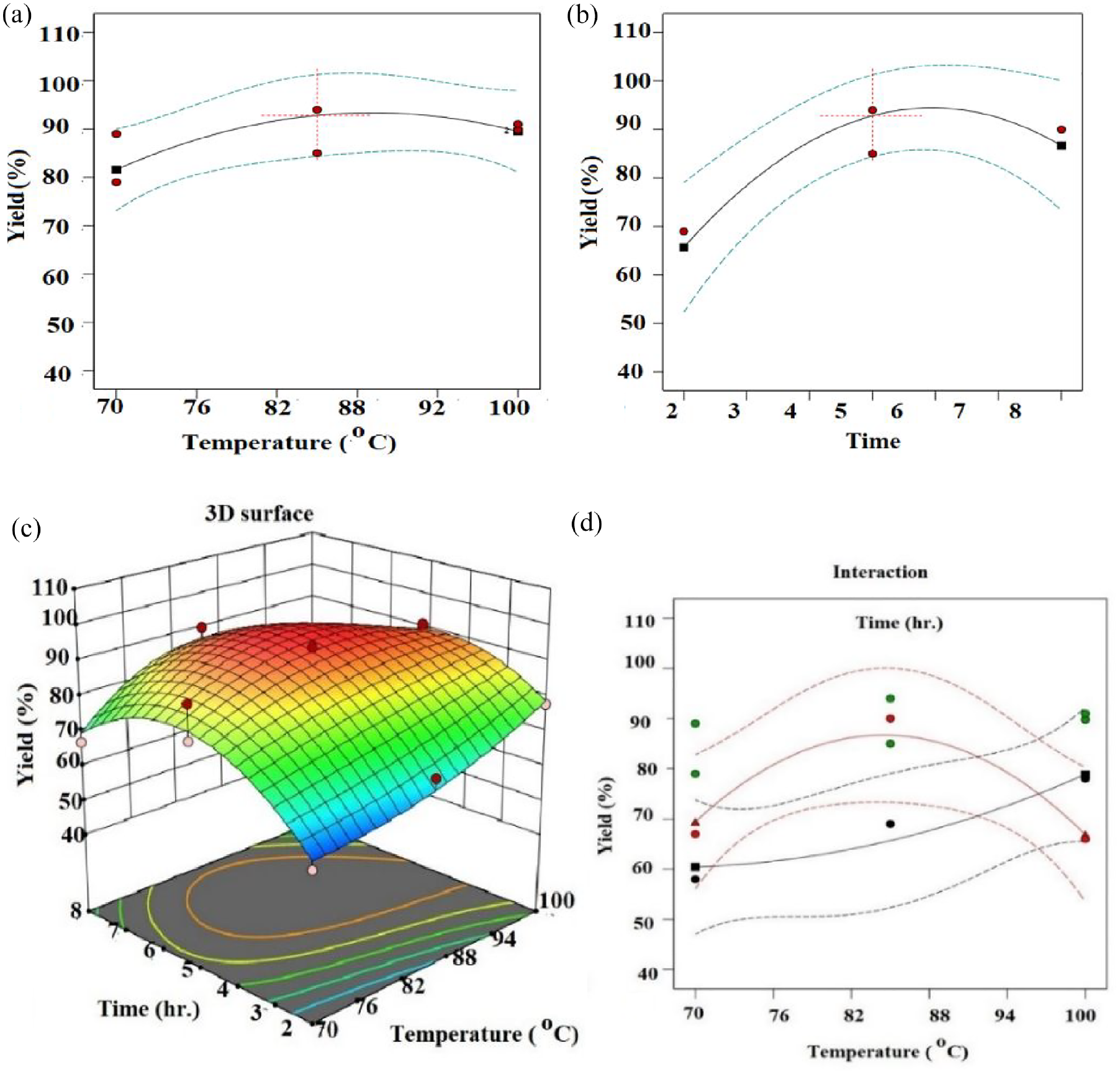
Effects of temperature (a) and time (b) on yield percentage. 3D surface plot graph (c) and interaction effects of temperature and time on yield percentage (d).
From Figure 4(a), it can be seen that as the temperature increases from 70 to 85°C, the yield percentage is slightly increased, while when the temperature increases beyond 85°C, the yield of the graph shows a downwards parabola and the yield drops down. In addition, time is also another factor contributing to yields.
Figure 4(b) demonstrates that increasing the duration from 2 to 6 hours results in higher yields, followed by declining trends. Furthermore, the interaction effects of the temperature and time on the extraction yield were also investigated.
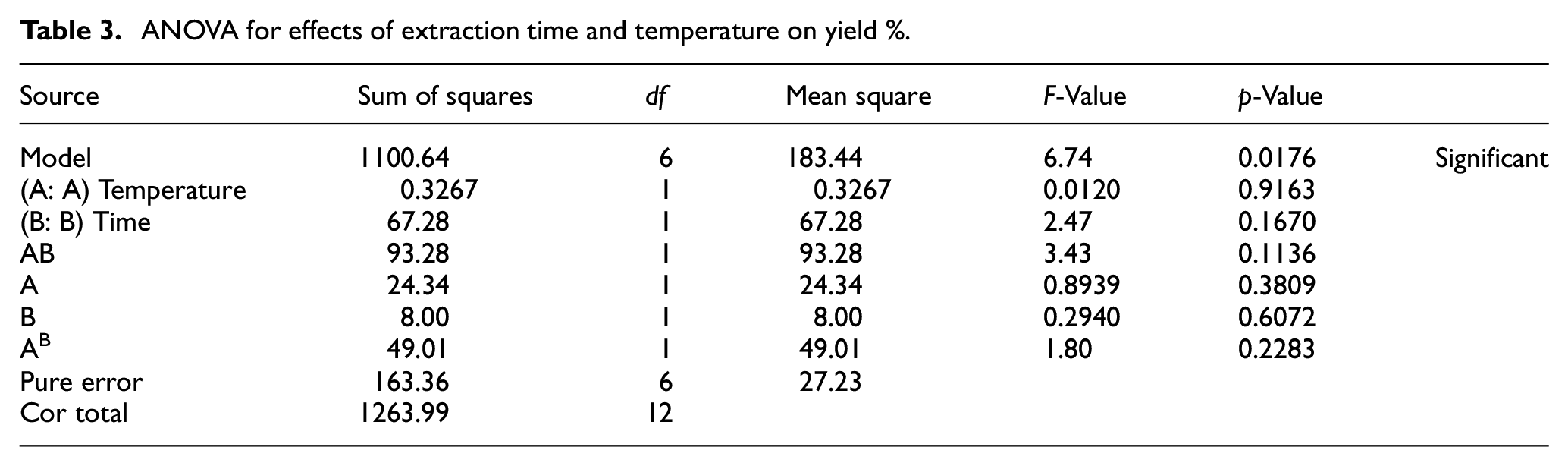
From Figure 4(d), it can be seen that both factors had an interaction effect. The highest yield of extraction was achieved from heating to a temperature of 84.6°C for 5.5 h, when about 94.04% of collagen was converted to gelatin. Moreover, Table 3 (ANOVA) shows that, at
ANOVA for effects of extraction time and temperature on yield %.
Moisture Content, Water Absorption, Viscosity and Gel Strength
The physical characteristics, namely, the moisture content, water absorption, viscosity and gel strength of the developed gelatin, are shown in Table 4. The test results show that the moisture content, viscosity and gel strength of the developed gelatin have values of 12.65%, 7 and 266 g, respectively. Furthermore, the experimental results show that the pH value of the developed gelatin is 7.6. (i.e. the properties of the developed gelatin are weakly basic). This result indicates that there was no chance of forming a covalent bond between the gelatin and reactive dye. Meanwhile a viscosity of 7.85 cp indicated that there was more bonding among and between its molecular structures, which led to the final product resisting the impact force. Moreover, from the table, it can be shown that the developed gelatin was superior to other sources of gelatin because it had superior gel strength and viscosity.
Physical characteristics of gelatin.

Colour Strength
To study the influence of gelatin thickener concentration and curing temperature on the colour strength, the paste preparation was carried out using gelatin to sodium alginate concentrations of 100%/0%, 75% /25%, 50%/50%, and 25%/75% for 130°C, 145°C and 160°C temperature for 5.5 h. The effects of gelatin thickener concentration and curing temperature on the colour strength (
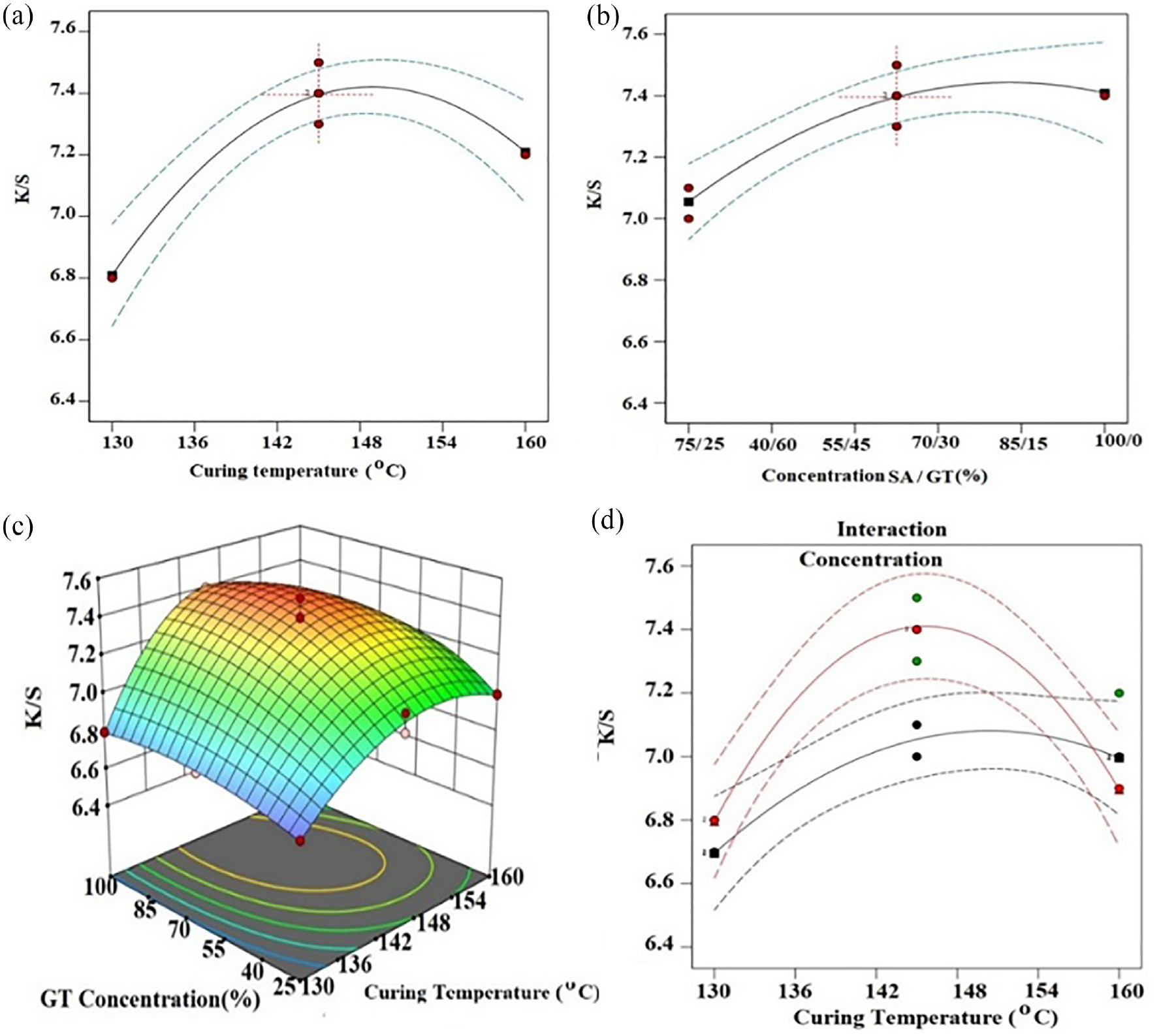
Effects of curing temperature (a), sodium alginate/gelatin concentration (b), K/S value of gelatin. 3D surface graph (c), and its interaction effects on colour strength (d).
From Figure 5(a), it can be seen that the colour strength (
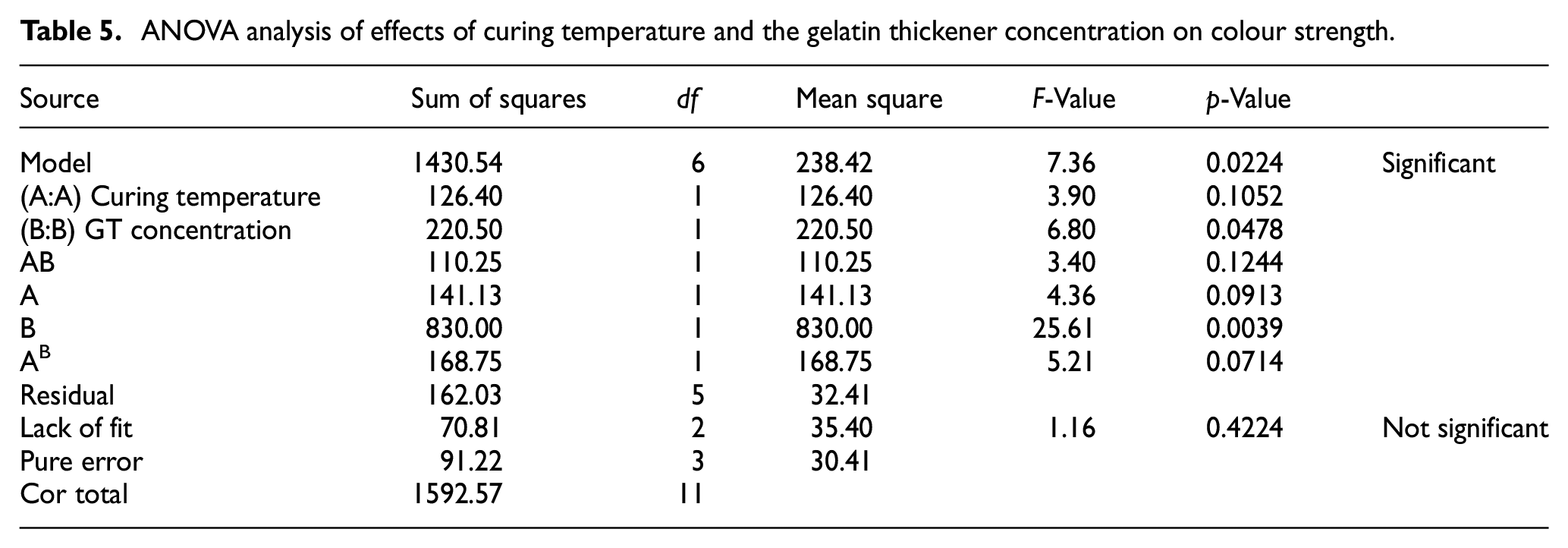
Moreover, the interaction effects of the curing temperature and concentration of gelatin thickener were investigated. From Figure 5(c), it can be seen that the colour strength was influenced by both the curing temperature and concentration of gelatin thickener. In addition, the graph shows that there was an interaction effect between the curing temperature and gelatin thickener concentration. Besides, the desired optimal colour strength (
ANOVA analysis of effects of curing temperature and the gelatin thickener concentration on colour strength.
Colour Fastness of Printed Fabric
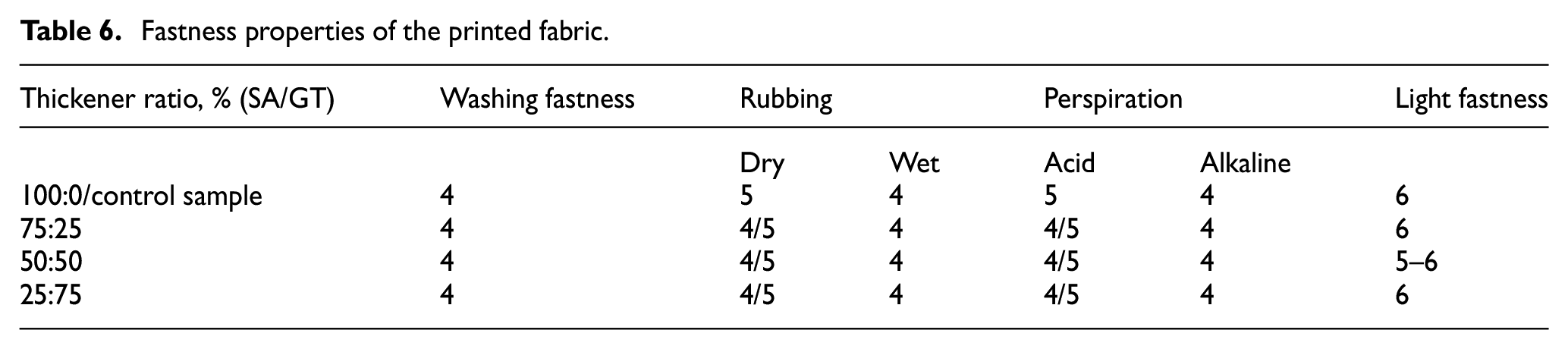
Colour fastness is a critical factor in all textile dyeing and printing processes. Thus, here, the results of the printed samples using controlled gelatin and sodium alginate concentration as thickeners are assessed. The colour fastness test, namely, the washing fastness, rubbing fastness, perspiration and light fastness of the printed fabric, was investigated based on the described standards, and the results are presented in Table 6. From Table 6, it can be seen that the printed samples which measured the rubbing and light fastness had very good colour fastness properties. Besides, both the washing fastness and perspiration also had the same colour fastness properties compared to each other. The higher the number is, the better the fastness.
Fastness properties of the printed fabric.
Flexural Rigidity of Printed Fabric
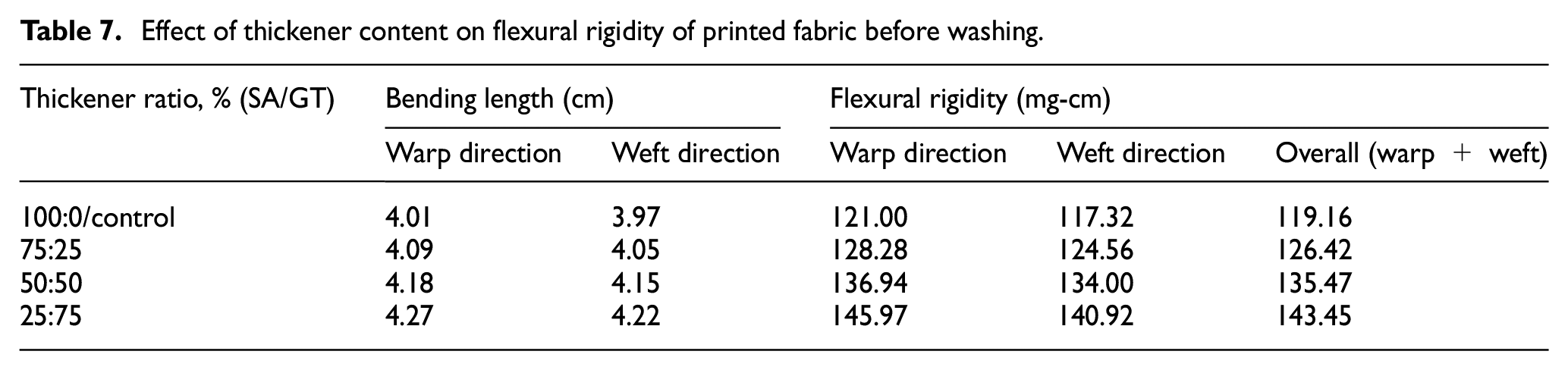
The flexural rigidity properties of the printed sample before and after washing are displayed in Tables 7 and 8, respectively.
Effect of thickener content on flexural rigidity of printed fabric before washing.
Effect of thickener content on flexural rigidity of printed fabric after washing.
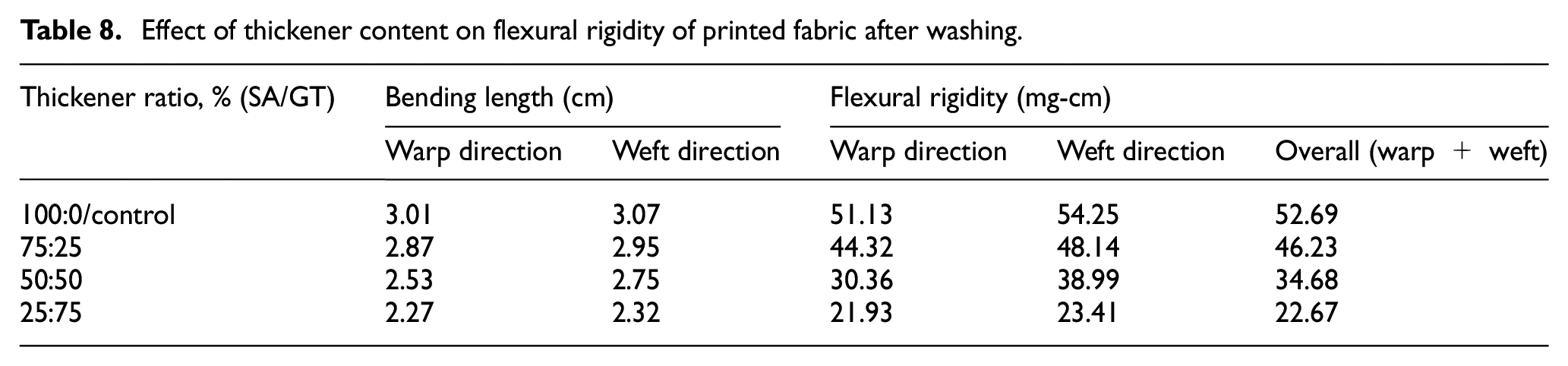
Table 7 details the effects of gelatin thickener concentration on flexural rigidity (bending stiffness) of printed cotton fabrics. From the table, it can be indicated that as the concentration of gelatin thickener increased from 0% to 75%, the flexural rigidity (mg-cm) of the printed fabric increased both in the warp and weft directions. This happened because the chemical bond between the reactive dye with gelatin thickenier and cotton fabric was affected, not only by the number of free hydroxyl groups but also by the concentration of the dye. These phenomena ware reflected in the fabric stiffness properties.
The bending length and flexural rigidity of the printed materials dramatically decreased after washing, as shown in Table 7. This demonstrates the great washability of the thickener employed in this investigation when washed after printing.
Tear Strength of Printed Fabric
The tear strength of cotton printing fabric using different concentrations of gelatin thickener is shown in Figure 6. As presented in Figure 6, the tear strength of printed cotton samples (warp-wise and weft-wise) decreased as the proportion of gelatin concentration increased in the printing paste. This result may have occurred due to different reasons, such as the increased cross-linking of gelatin with the substrate, reducing yarn slippage, or the flexural rigidity of fabric increasing and tear strength decreasing.
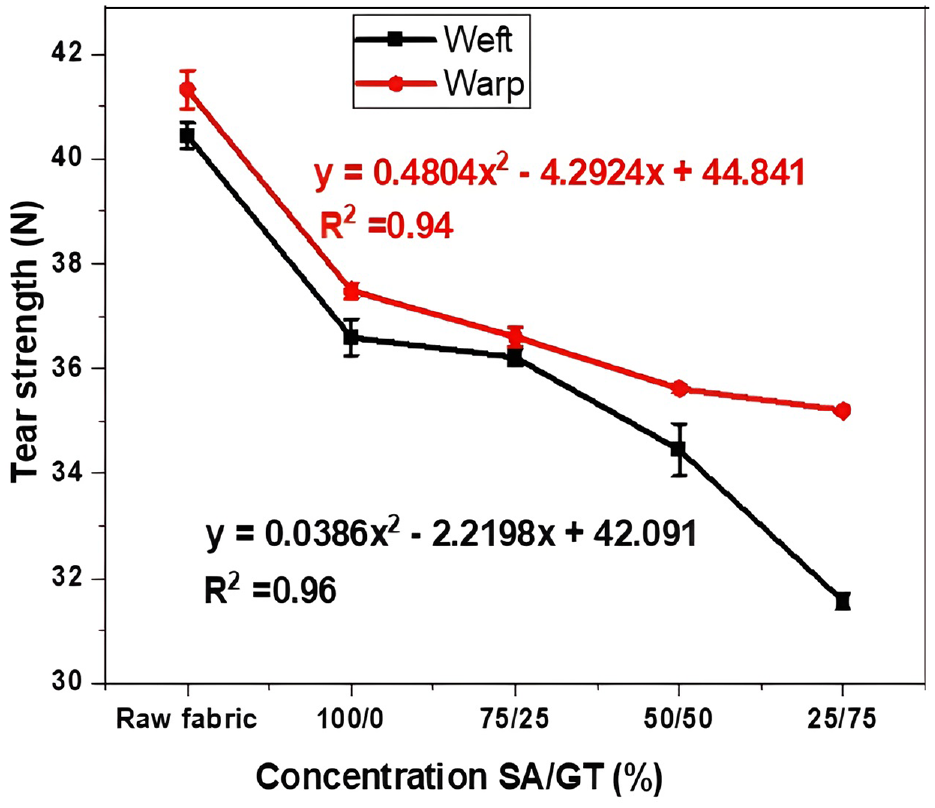
Effect of thickening agent concentration on tear strength.
Tensile Strength of Printed Fabric
The tensile strength of the cotton fabric printed using reactive dye with gelatin thinker is shown in Figure 7. The tensile strength of the raw fabric (unprinted) and the printed fabric was investigated in both warp and weft directions; then, the change in strength percentage was calculated.
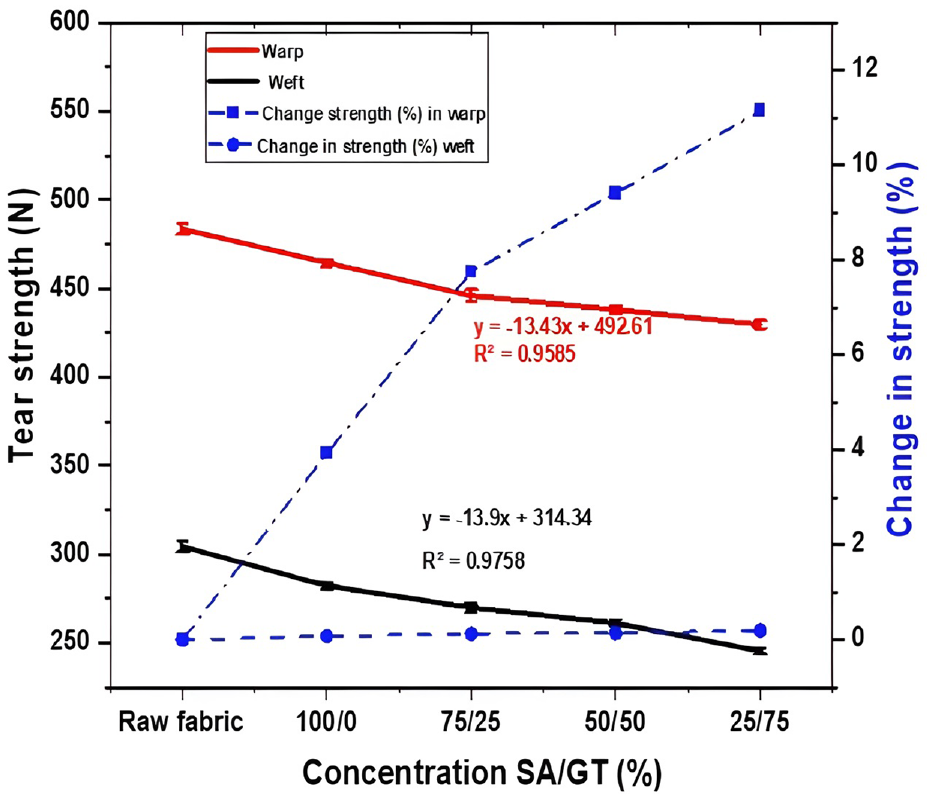
Effect of thickening agent concentration on tensile strength.
The tensile strength of the printed fabric decreased both warp-wise and weft-wise when the proportion of gelatin in the printing paste was increased, as indicated by the results shown in Figure 7. The hydroxyl groups of the cellulose may have formed cross-links, which could be the cause of the tensile strength decrease. The cross-links prevent the cellulose chains in cotton fibre from sliding, which lowers the fiber’s tensile strength. The tensile strength of the unprinted samples is greater than that of the printed sample textiles.
Conclusion
In this study, the synthesis of gelatin material from animal hide skin trim was performed using the thermal hydrolysis technique for the application of textile printing as a thickening agent. Then the developed gelatin was used as a thickning agent for reactive dye printing on cotton fabric with various concentrations of sodium alginate. Optimisation was also performed at various curing temperatures. From the result, it can be seen that the effects of temperature and time on the yield percentage are statistically significant. Besides, the maximum yield percentage synthesized was achieved at 85°C extraction temperature and 5 h extraction time. Regarding to the printed fabric quality, the results show that excellent printing was achieved. In addition, the analytical findings show that the optimum value for augmenting the proportion of gelatin to sodium alginate as a thickening agent was a gelatin/sodium alginate (GT/SA) concentration of 70%/30%. Moreover, the effects of the GT to SA concentration on physical and mechanical properties of the printed fabric were evaluated. Furthermore, the results of the colour values, colour fastness characteristics, and physical attributes of reactive dye-printed cotton fabric samples show that extracted protein-based gelatine with sodium alginate as a thickening agent is a promising eco-thickening agent for the application of textile printing .
Footnotes
Acknowledgements
The authors are grateful to Kombolcha Textile Share Company (KTSC) and Ethiopian Institute Textile and Fashion Technology (EITEX) for providing basic laboratory facilities. In addition, the authors thank Kombolcha Institute of Technology staff for support and delivering relevant comments.
Author contributions
Conceptualization – A.M.N. and A.S.A.; methodology – A.M.N. and A.S.A.; formal analysis – A.M.N. and A.D.G.; investigation – A.M.N. and A.S.A.; resources – A.S.A.; writing – original draft preparation – A.M.N. and A.D.G.; writing – review and editing – A.S.A. and A.D.G.; visualization – A.S.A. and A.D.G. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
