Abstract
Available and clean water resources in the world are decreasing day by day. Textiles is one of the industries that are consuming the highest volumes of water. For this reason, it is necessary to develop environmentally friendly textile processes within the scope of sustainability. Reactive dyeing is followed by a washing process consisting of many steps to remove unfixed/hydrolyzed dyes, which is the most preferred dyeing in the coloring of cotton products. A high amount of water is consumed in these processes. In this study, an alternative washing process was developed instead of the conventional reactive washing process. The baths, which were decided to be used according to the transmittance values of the washing baths after dyeing in light, medium, and dark shades, were used again as the first two baths of the next washings. Alternative washing did not have a negative effect on the quality parameters, namely washing and rubbing fastnesses. As a result, it was observed that the last two baths of the washing cycle could be used as the first two baths of the next washing cycle. In this case, it was predicted that approximately 40%−50% water savings could be achieved.
Introduction
Since the mid-1990s, cotton has been the most consumed natural fiber, even though it has fallen to second place overall since synthetics took the lead. Fiber production had a volume of 111 million metric tons in 2019, 23% of which was cotton. It is expected that the demand for textile products will continue to increase rapidly due to factors such as rapidly changing fashion, improving living standards, and so on, to reach 146 million metric tons, an increase of 30%, by 2030.1,2
In textiles, most of the cotton fabrics are used colored and the coloration of fabrics is usually performed by the dyeing process. 3 The consumption of cotton dyes in the world is about 360,000 tons, making up a large part of the dye industry with a value of $6 billion. 4 Reactive dyes, which have the highest share in this volume, have many advantages such as wide color gamut, versatility of application methods, and high level fastness to washing.5,6 Reactive group(s) of reactive dyes that give high washing fastness by reacting with the hydroxyl groups of cellulose under alkaline conditions also react with hydroxide ions in the bath to produce non-reactive hydrolyzed dyes. These hydrolyzed dyes, which remain on the fiber in the dyebath, are removed from the fiber by a time-consuming, energy-intensive, and expensive washing procedure consisting of many steps to obtain a high level of fastness.7–9
Within the scope of sustainability, textile wastewater containing high concentrations of numerous chemicals, auxiliary compounds, and dyes has attracted worldwide attention as one of the most polluting industrial wastes.10–12 While the decrease in photosynthesis due to the reduced light transmission because of the presence of these chemicals in the aqueous environment harms aquatic life, they also negatively affect human health due to their toxic effects. 13
In the literature, there are many articles about the treatment and decolorization of textile effluents.14,15 In these studies, the cleanability of wastewater was investigated by various methods. Some of these methods were chemical oxidation,16–19 photo-oxidation,20,21 biological treatments,22–25 and adsorption.26–28 Although the reuse of textile wastewater without any treatment is one of the methods considered within the scope of cleaner production, previous research efforts on this have been limited.29,30 Therefore, this study fills an important gap in the literature. In this study, it was shown that a basic and simple method that many textile mills could easily implement and save water was applicable and useful. There are many studies on the use of wastewater that became reusable after different processes.31–34 For example, in one article, similar color strengths were obtained from dyeings with photo-Fenton-treated water and fresh water. 35 In another study, it was observed that reverse osmosis concentrate became suitable for reuse in dyeing after being treated with combined persulfate oxidation and lime-soda softening methods. 36
In this study, an alternative washing process was developed to save the high amount of water consumed in the washing process consisting of many steps to remove the hydrolyzed and/or unfixed dyes. For this purpose, conventional and alternative washing processes after dyeing in light, medium, and dark shades were examined. To determine which of the reactive washing baths could be reused, the transmittance and absorbance values of these baths were measured in the UV-Vis spectrophotometer. The last two washing baths determined to be suitable for reuse were used a second time as the first two baths of the next washing. The effects of this alternative washing processes on fabric quality were examined.
Experimental
Material
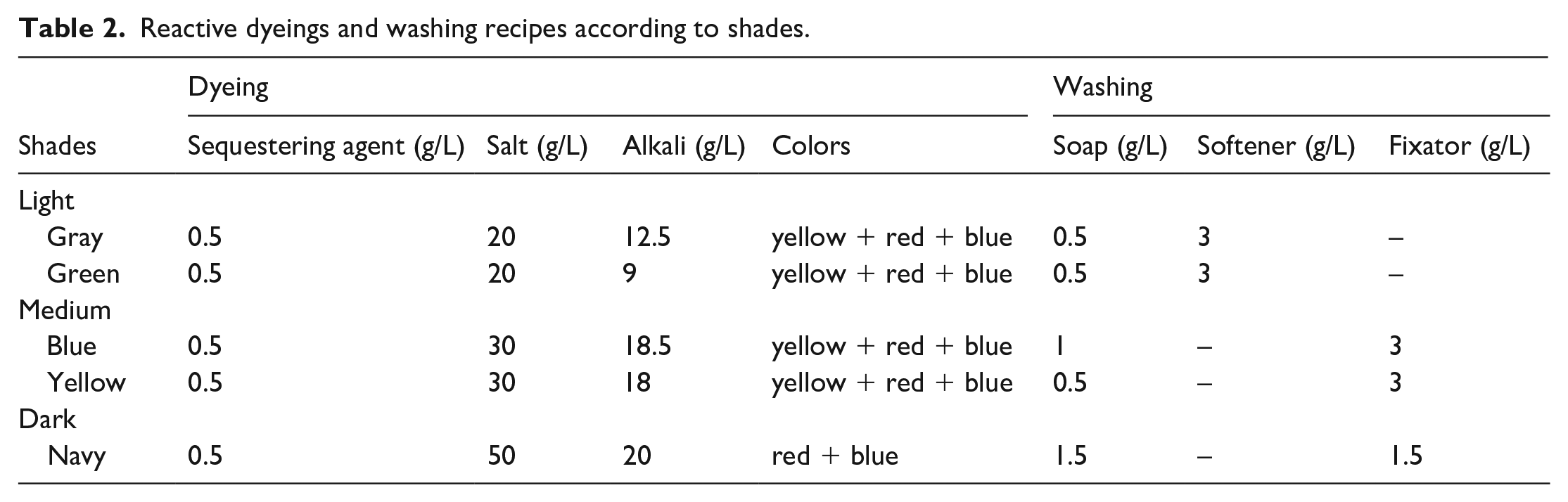
The weight of the cotton towel used in the study was 370 g/m2. The ground yarn and pile warp yarn numbers were Ne 20/2 and Ne 16/1, respectively. The number of the weft, the third thread, was Ne 16/1. Dyeing auxiliaries—alkali (sodium carbonate-Na2CO3), acetic acid, and salts (sodium chloride-NaCl)—were of technical grade and provided by TEKKİM Kimya (Bursa, Turkey). The Securon Bath 3, Aquasoft Nass and Securon 1420 CT from Pulcra Chemicals were an acid buffer, softener, and sequestering agent, respectively. Isopon HDS-T, one of the products of the Bozzetto group, was used in the soaping-off. Akfix P obtained from Akkim was used as a fixator in the washing steps. The names and mixing ratios of reactive dyes classified according to shades are given in Table 1.
Reactive dyes and mixing ratios.

Method
Conventional Reactive Dyeing
The conventional reactive dyeing process was started at 50°C. Asetic acid, salt, and sequestering agent were added to the dyeing bath. Towel fabric was treated within this bath for 15–20 min. After the bath temperature was increased to 60°C, reactive dye and alkali were added to the bath. The dyeing process was done at 60°C for 60 min at a 10:1 liquor ratio. Dyeing recipes used for dark, medium, and light shades are shown in Table 2. The details of the colors can be found in Table 2. Conventional reactive dyeing process details are given in Figure 1.
Reactive dyeings and washing recipes according to shades.
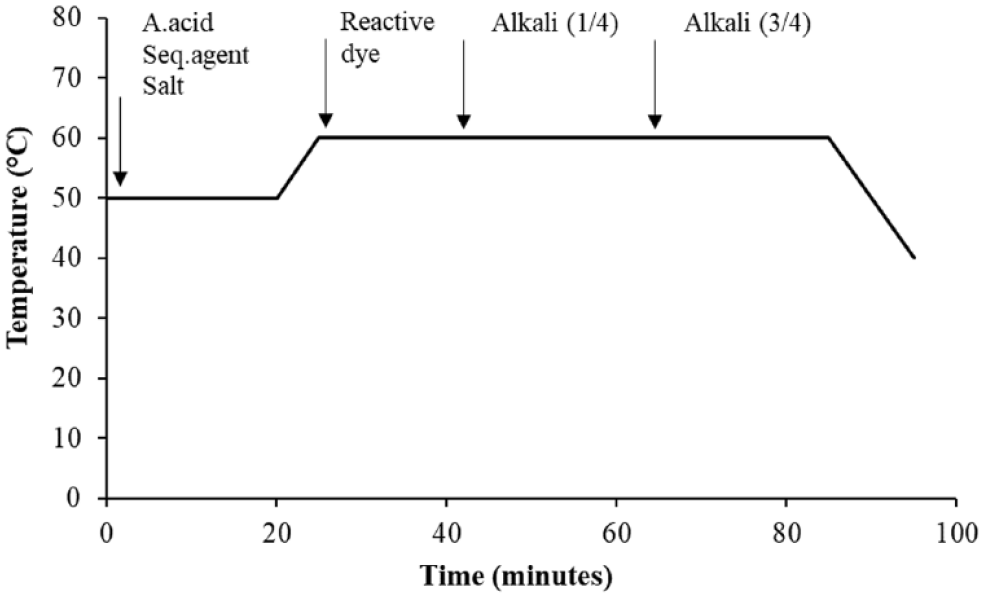
Conventional reactive dyeing temperature-time diagram.
Conventional Washing Processes
The light shade washing process consisted of five washing steps. After rinsing at medium temperature (50°C for 10 min) followed by a second wash with acetic acid (50°C for 10 min), soaping at 85°C for 10 min was conducted. The process was terminated with hot rinsing as the fourth step (75°C for 10 min) and washing with softener and buffer acid as the fifth step (50°C for 10 min). Details and the recipe of the process are given Figure 2 and Table 2, respectively.
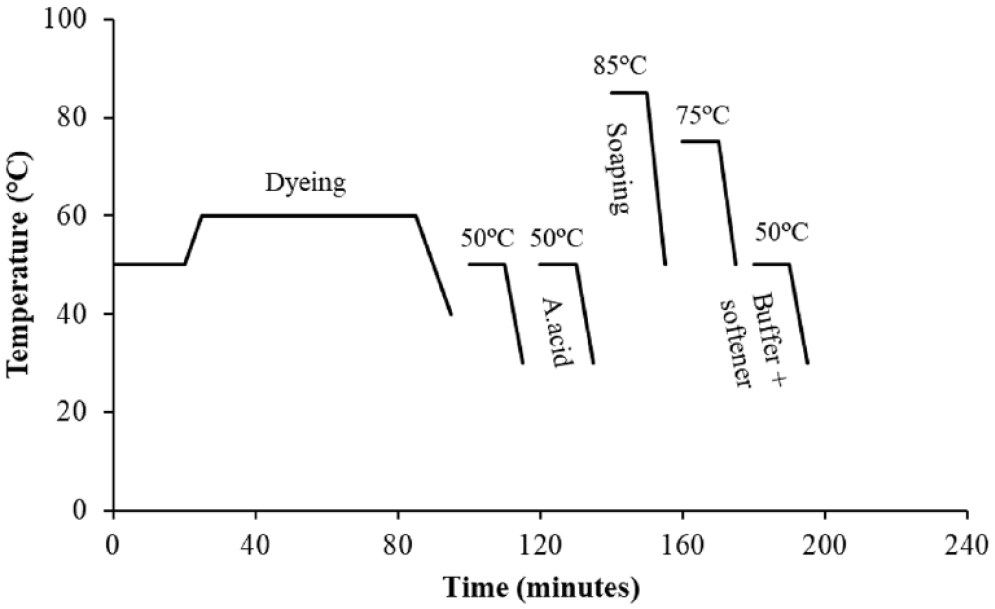
Conventional light shade washing.
The washing process after dyeing in medium shades consisted of six steps. Unlike light shade washings, after soaping, a step was added (80°C for 10 min) to the process before the final rinsing, increasing the number of baths. Figure 3 and Table 2 show the details and recipe of this washing process, respectively.
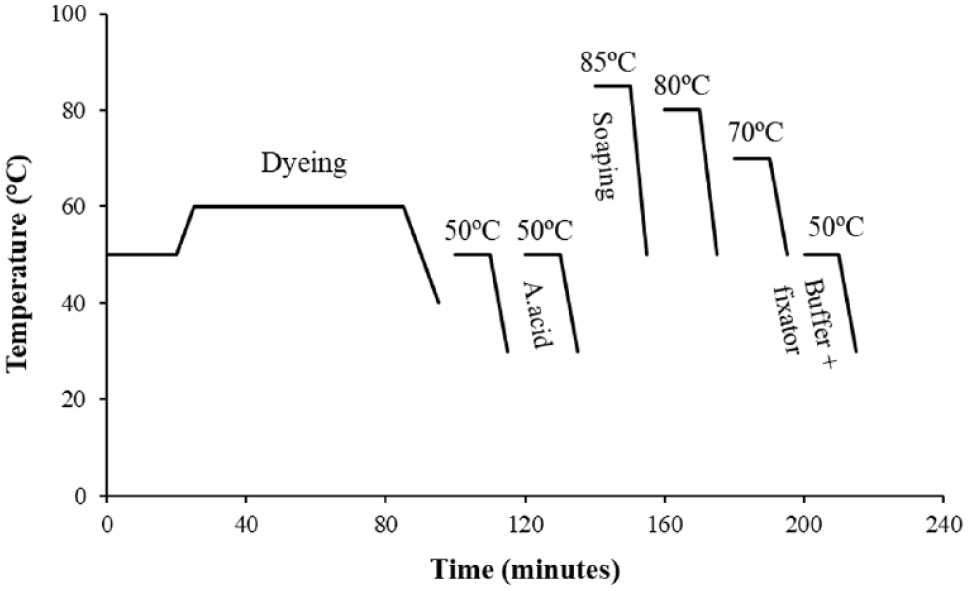
Conventional medium shade washing.
The washing process carried out after dark shade reactive dyeings consisted of more steps (seven) than the others. The differences from the medium shades were the increased temperature of the soaping step (95°C for 10 min) and the second soaping process performed at a lower temperature (80°C for 10 min) rather than soaping at boiling temperature. Details and recipe of this process are given in Figure 4 and Table 2, respectively.
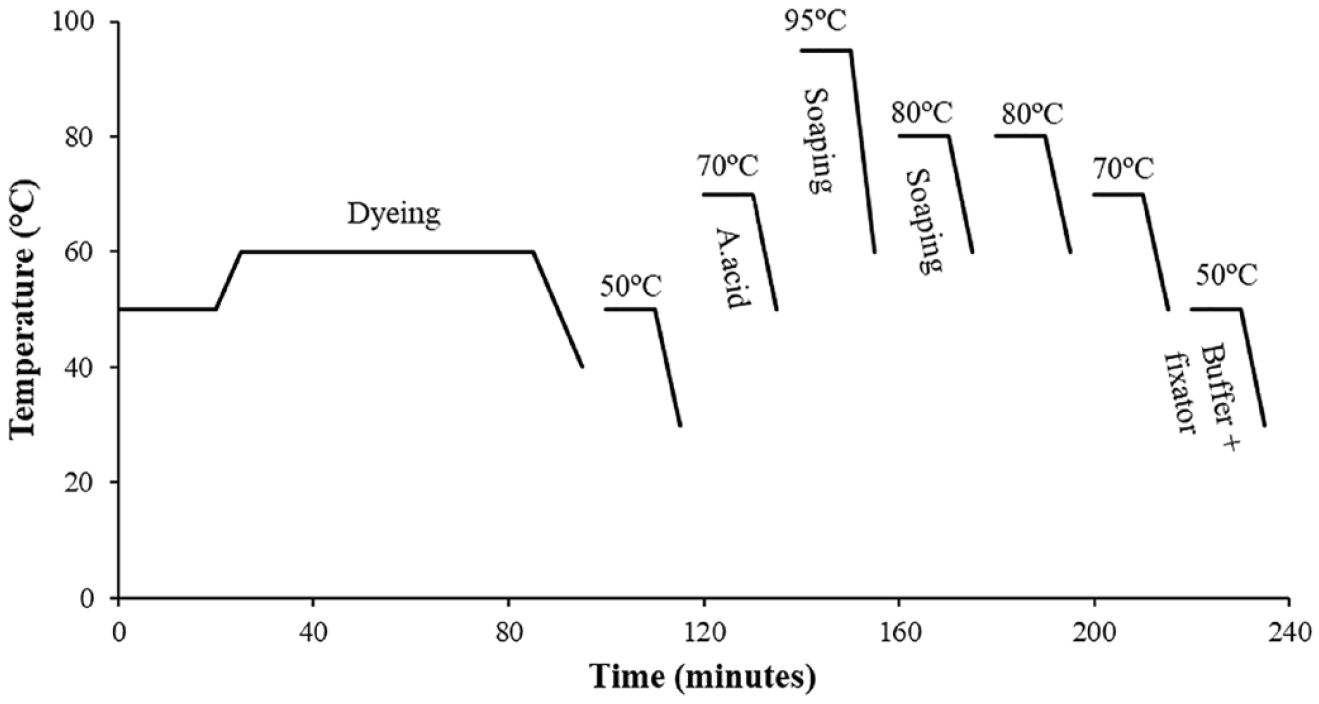
Conventional dark shade washing.
Alternative Washing Process
Reuse of the light, medium, and dark washing baths according to the color shades are given in Figures 5–7, respectively.
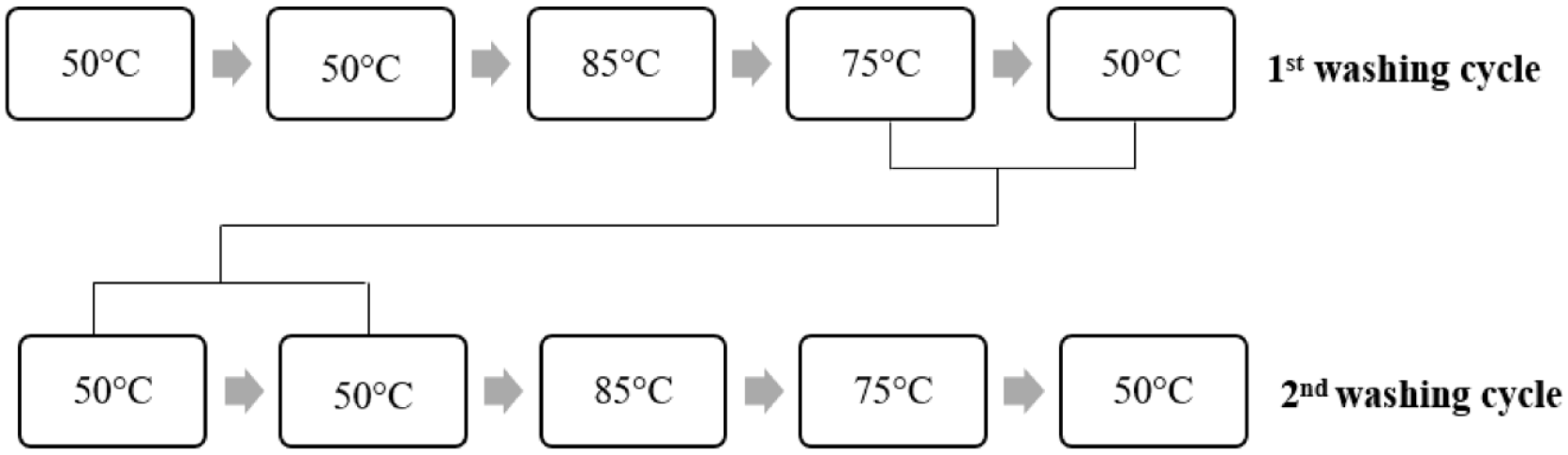
Reusing light shade washing baths.
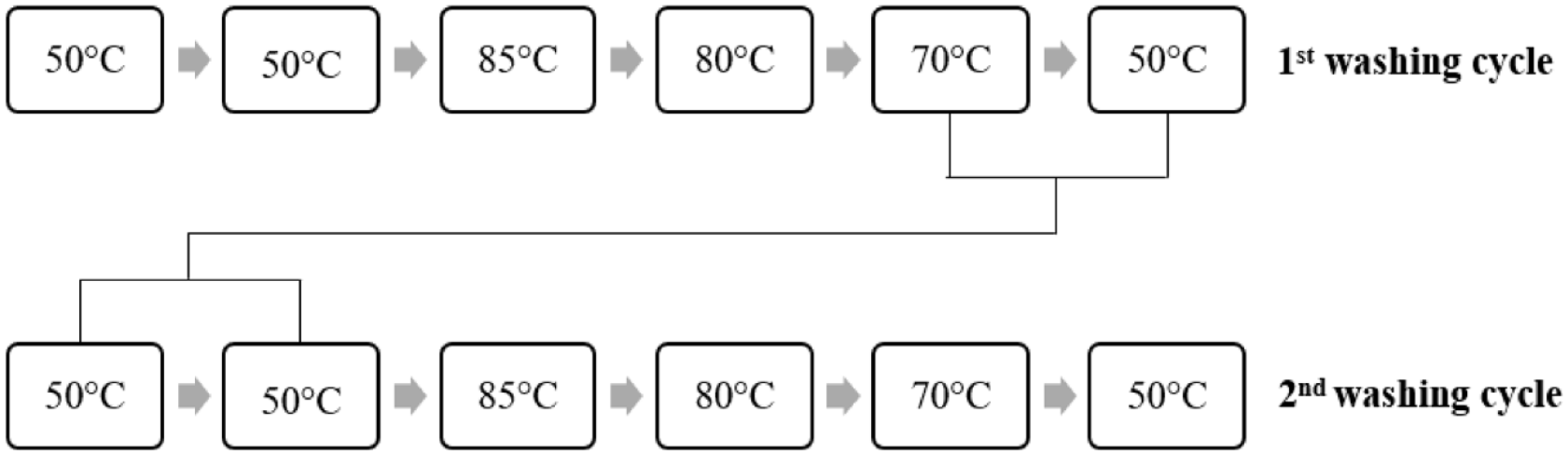
Reusing medium shade washing baths.
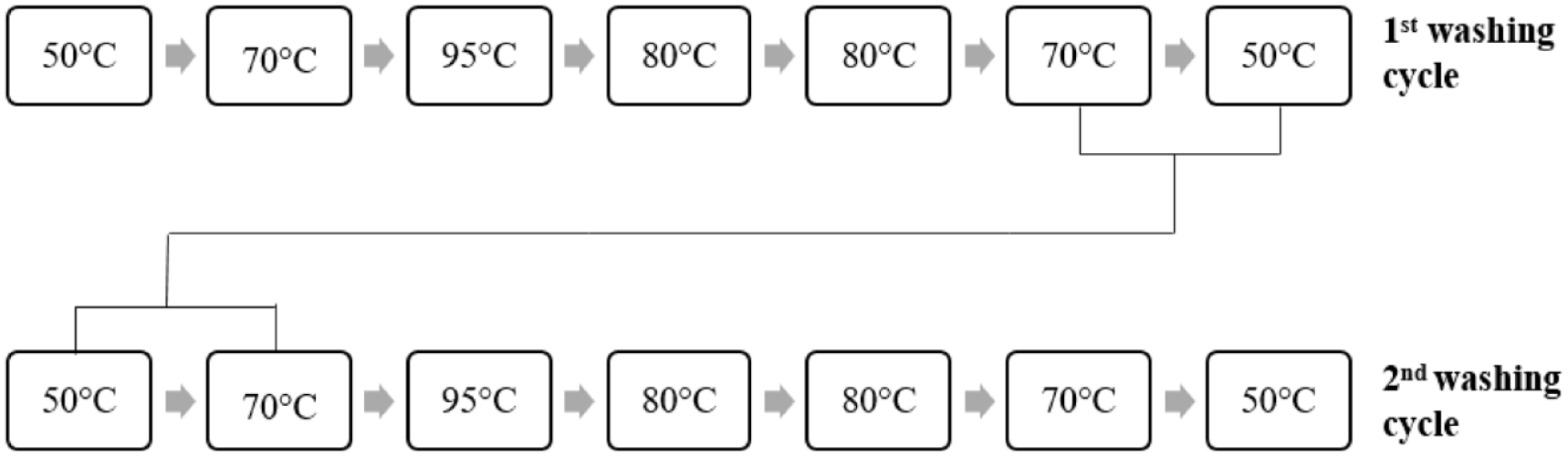
Reusing dark shade washing baths.
Measurements
A UV-vis spectrophotometer (Rayleigh VIS-723G) was used to determine the absorbance-transmittance values of the washing baths. Measurements were performed in the 320–800 nm wavelength range and at 5 nm intervals. The absorbance values of the washing baths were determined as the peak points in the absorbance graph of the first baths. As the transmittance value, the value at which the wavelength of the peak in the absorbance graph coincides in the transmittance graph was taken. Since the absorbance graphs of the dyes varied according to the colors, the wavelengths at which the maximum absorbance values of the first baths were taken in this study were as follows: 380 nm for gray/green, 420 nm and 425 nm for yellow and blue, and 555 nm for navy.
Color coordinates (L*, a*, b*, C*, and h°) and color strengths were measured using a Konica Minolta CM-3600D spectrophotometer (10° standard observer and under D65 illuminant). The K/S values were calculated using the Kubelka–Munk equation. The color strength (K/S) formula 37 is presented in equation (1):
The washing fastness of the dyed fibers was determined using a TEST color fastness tester, using the color fastness to domestic and commercial laundering standard test method EN ISO 105-C06/C2S. The rubbing fastness of samples was determined according to EN ISO 105-X12:2002-12.
Statistical Analysis
An independent samples t-test was preferred to compare means of color strengths of conventional and alternatively washed fabrics. A value of p < 0.05 was considered to be statistically significant.
Results and Discussion
Color Coordinates and Strengths
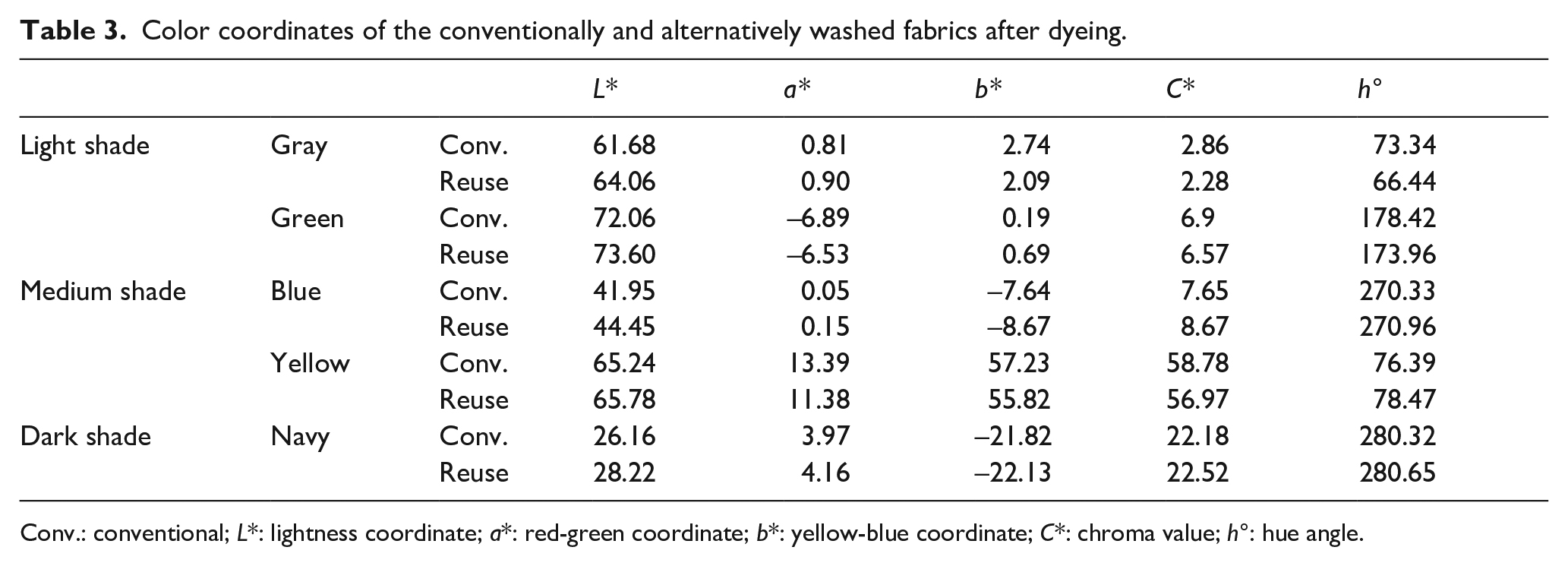
The color coordinates and color strengths of the conventionally and alternatively washed fabrics after dyeings are given in Table 3 and Figure 8, respectively. In Figure 9, photographs of these fabrics are given.
Color coordinates of the conventionally and alternatively washed fabrics after dyeing.
Conv.: conventional; L*: lightness coordinate; a*: red-green coordinate; b*: yellow-blue coordinate; C*: chroma value; h°: hue angle.
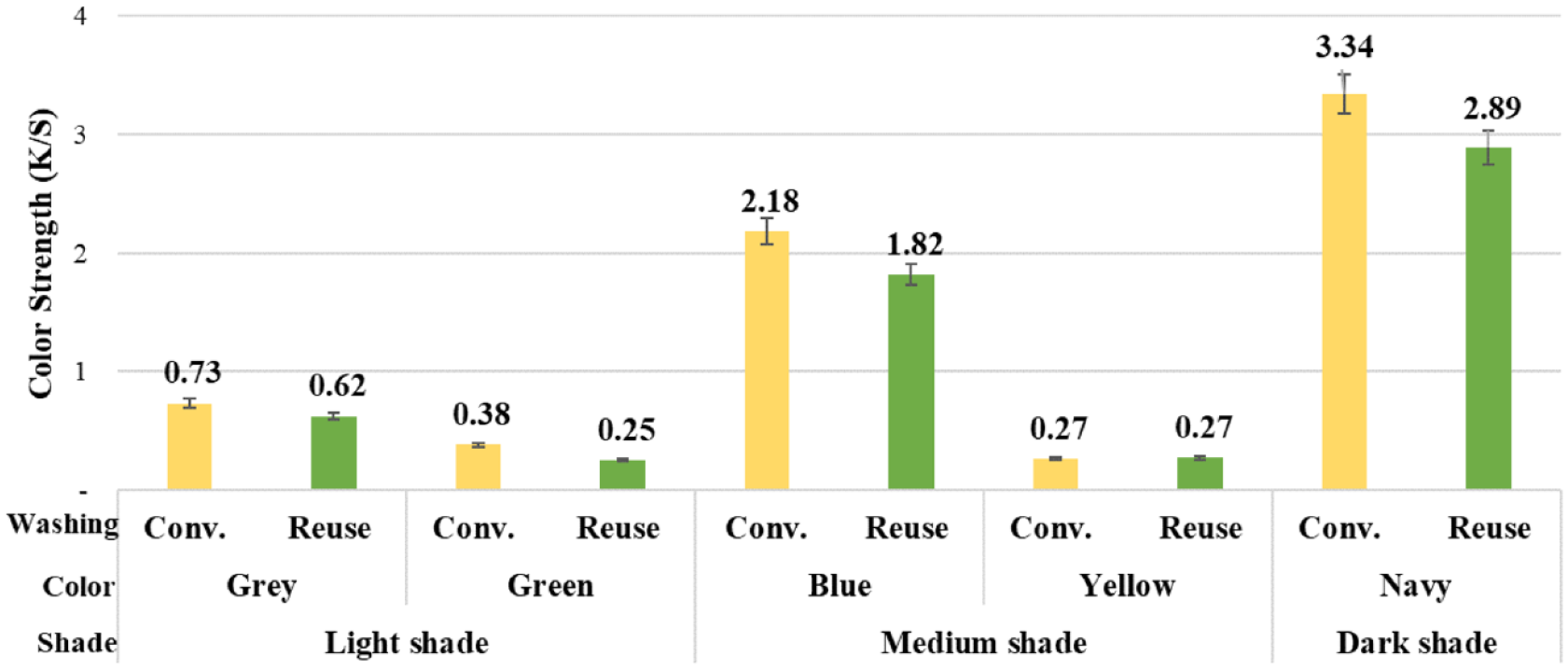
Color strengths of the conventionally and alternatively washed fabrics after dyeing.

Photographs of the conventionally (a) and alternatively (b) washed fabrics after dyeing.
The comparison after dyeing the towel fabrics with different colors and subsequently two different washings showed that repeated use of the washing baths resulted in lighter colors compared with conventional ones for each color. The chroma values of conventional dyeings and washings were also higher for each color than those of the conventional dyeings and alternative washings. No evaluation could be made about the effects of different washings on a* and b* values since the colors were different.
It was revealed that for each color, samples with conventional washings after dyeing had higher color depths than those of the alternative washings. These changes were correlated with the L* values, that is, higher L* values correspond to lower color strength. While different washings caused a higher color difference in light tones, that is, 15% for gray and 34% for green, the color difference decreased toward dark ones, that is, navy-dyed (13%). The color difference, which was seen to be high for green, was not remarkable considering the color was light.
The statistically evaluation of the differences between the color strengths of the alternative and conventionally washed samples after dyeing revealed that the difference between the color strengths of the yellow- and navy-dyed towels was not significant, while the others were significant.
Absorbance-Transmittance Values of Washing Baths and Fastness of Dyed Fabrics
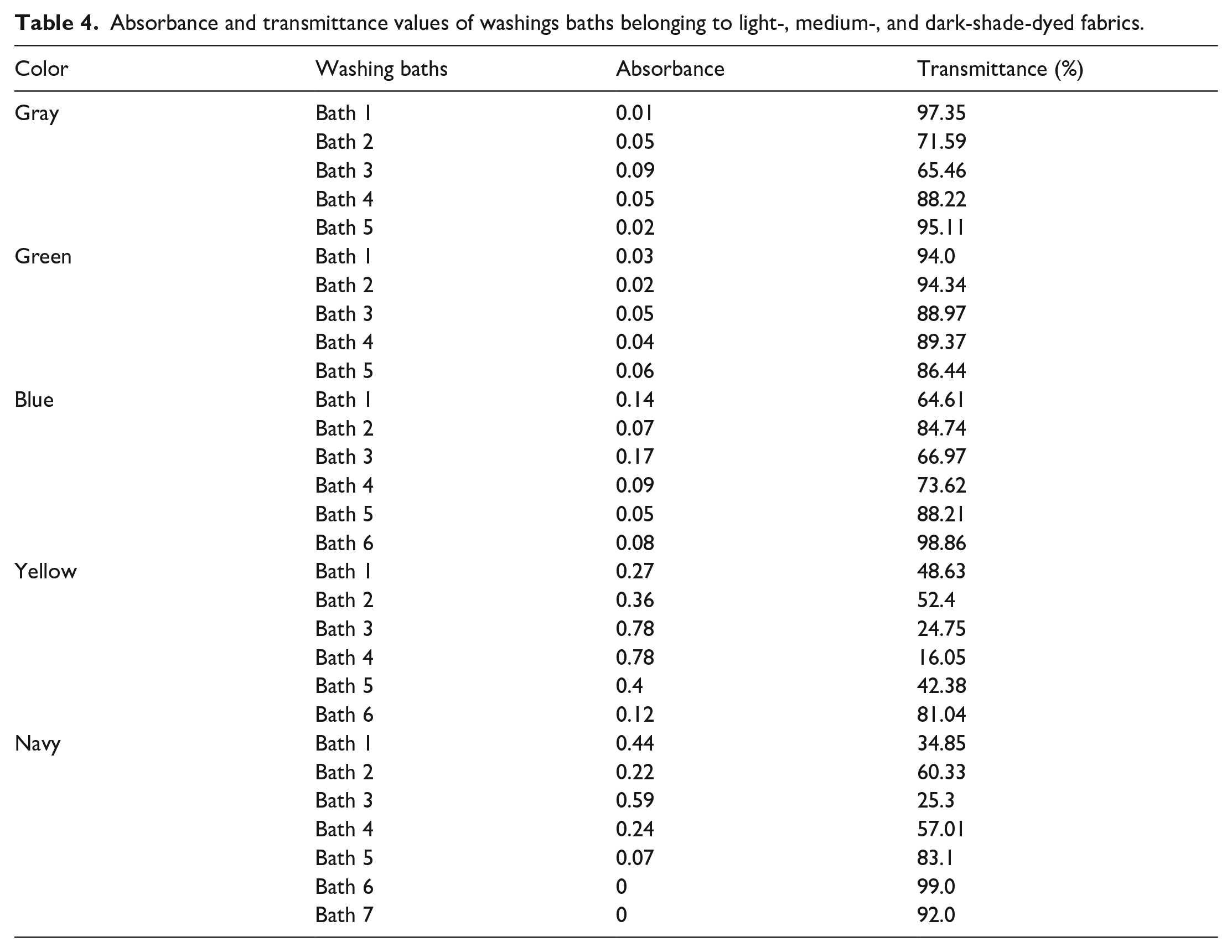
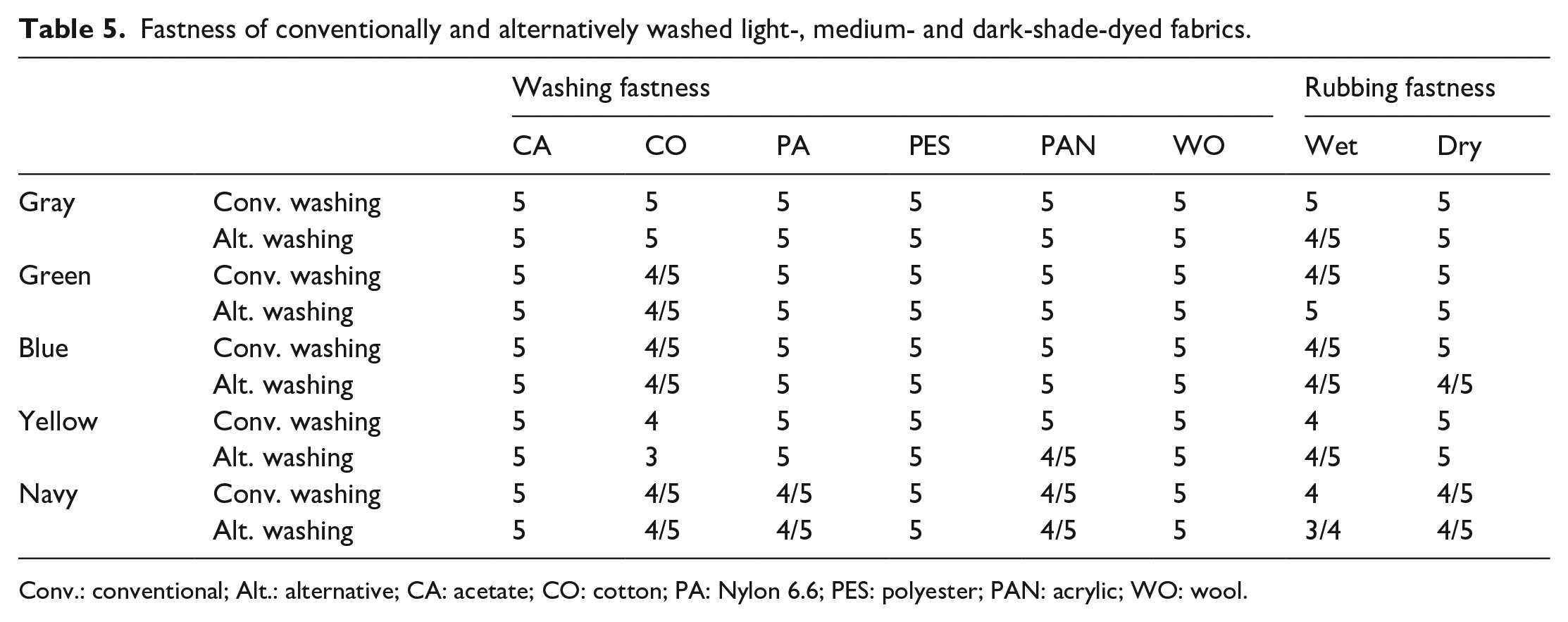
In Table 4, the absorbance-transmittance values of conventional washings are given. The fastness values of conventional and alternative washing processes are presented in Table 5. Photographs of the conventional washing process are given in Figure 10.
Absorbance and transmittance values of washings baths belonging to light-, medium-, and dark-shade-dyed fabrics.
Fastness of conventionally and alternatively washed light-, medium- and dark-shade-dyed fabrics.
Conv.: conventional; Alt.: alternative; CA: acetate; CO: cotton; PA: Nylon 6.6; PES: polyester; PAN: acrylic; WO: wool.

Conventional washing bath images: (a) gray-, (b) green-, (c) yellow-, (d) blue-, and (e) navy-dyed fabrics.
It was observed that the transmittance values of the washing baths belonging to the gray and green colors decreased from the first to the third bath. It turned out that these decreases in transmittance values correspond to increases in absorbance values, as expected. While the transmittance of the fifth washing bath of gray was higher than that of the fourth, the opposite was true for the green color.
The lowest transmittance value of the washing baths of the blue color was obtained from the first bath. While the transmittance of the second bath was higher than that of the first, it decreased in the third bath. From this bath, the transmittance increased again. In yellow, the decrease in the transmittance, which started in the third bath, was also seen in the fourth. The highest transmittance value was obtained from the sixth bath. The changes in the absorbance values of the baths changed inversely with the transmittance.
It was revealed that the lowest transmittance value was obtained from the third bath in the washing processes carried out after dyeing with dark-shade navy. The transmittance values, which increased until the sixth bath, decreased again in the seventh bath, but even with this decrease, the transmittance value still remained above 90%.
Reusing the washing baths had no adverse effect on the washing fastness of the colors, except for yellow. Fastnesses obtained after conventional and alternative washing cycles were similar. This could be explained by the reactive dye forming strong bonds with the fiber 38 and the low colorfullness of reused washing waters. If the colorfullness was high in the repeatedly used washing baths, the unfixed dyes remaining in the bath could form weak bonds with the fiber, causing a decrease in fastness. 38 The lower washing fastness of the alternative washing for the yellow color compared with the conventional ones could be explained by the high colorfullness of the reused fifth washing bath (Table 4).
All dyes had good rubbing fastness which may be imputed to be due to good dye–fiber bond stability and the well dye penetration into the towel. 39 It was observed that the conventional and alternative washings did not cause significant changes in the rubbing fastness, but the wet rubbing values of the alternative washings were 1/2 point lower than the dry ones. This situation could be explained by the dye molecules carried by repeated washing baths remain on the fiber surface or by the dye molecules are attached to the fibers by weak bonds. In addition, as the color shade got darker, the wet rubbing values decreased by half a point. This was interpreted as increasing dye concentration increasing the number of dye molecules that adhered but did not bind to the fibers, that is, the amount of dye molecules to be removed by friction.
Conclusion
In general, it was observed that the first washing and soaping baths were the most colored baths, and this was especially true for medium shades. For light shades, it was evaluated that only the soaping baths were colored and the colorfullness was very low in the other baths. In dark shades, this situation could not be determined exactly and it was seen that the last two washing baths could be used again, although all baths contained intense colorfullness. By the way, the light transmission properties of some reactive dyes are very good, and this does not mean that the dye concentration in the solution is very low, and it should not be considered that low-transparent reactive dyes are used in high concentrations. Therefore, as an indicator of the reusability of the last two baths, not only transmittance measurements but also other measurements that determine wastewater pollution, such as COD (chemical oxygen demand), are useful to evaluate the results as a whole. It was revealed that there was no significant difference in the colors and fastnesses of the samples washed according to the alternative washing process, in which the last two baths of the previous washing cycle were used as the first two baths of the next washing, compared with those of the conventional washings. It should also be examined whether these good results obtained with a single reuse of the last two baths can be obtained for many repeated uses of these baths because this method cannot be repeated forever. As a result of this study which was carried out within the scope of environmentally friendly production, it was predicted that approximately 40–50% water savings could be achieved by reusing reactive washing wastewater.
It is planned that this study will be further developed and evolved into sustainable wastewater recycling. In this new process, the first two baths of each washing cycle will consist of the last two washing baths of the previous washing cycle and a small amount of clean water added to it. Thus, the idea of water saving, which is the main purpose of the current study, will be further developed and transformed into sustainable waste recycling. Moreover, it is foreseen that similar results will be obtained by using other materials and dyes such as polyesters and disperse dyes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
No animal or human trials were undertaken or conducted for this study.
Funding
The authors express their sincere thanks for funding this study with the project “Yeni ve Çevreci Teknolojilerin Reaktif Boyama Sonrası Yıkamada ve Atık Suyun Dekolorizasyonunda Kullanımı” (Project Number: TEYDEB 1505 - 5180061) by the Scientific and Technological Research Council of Turkey (TÜBİTAK).

