Abstract
Four novel methoxy- and hydroxy-triphenylamine-based azo dyes were synthesized and applied on polyester and nylon fabrics. Photophysical properties of the dyes were studied in different organic solvents. Lightfastness and sublimation fastness properties were evaluated. Level dyeing with intense red and yellow colors was observed for these dyes on polyester fabric. Comparatively, brighter color and higher color strength (
Keywords
Introduction
Azo dyes are the largest class of synthetic dyes—they cover the entire spectrum of colors. 1 Azo dye colors can be easily tuned by varying different auxochromic groups in the dye. 2 They have advanced applications 3 and are used as food colors, sensors, and as nonlinear optical materials.4–6 They are used for dyeing wool, nylon, cotton, and silk materials, as well as synthetic materials like polyester, acrylic, and cellulose acetates.7–9
The photophysical properties of azo dyes are dependent on the donor and acceptor groups and strength of the substituents present on either side of the azo system.
10
Various donor groups such as carbazole,
11
phenol,
12
and
Azo disperse dyes are widely applied on polyester fabrics.20,21 Thisclass of dyes is mostly water-insoluble and is small in molecular weight. The small size has limitations for textile dyeing—these dyes can sublime, limiting their retention on fabric at higher temperatures. 22 To avoid such problems, comparatively larger or more polar molecules are desired. There is a need for larger molecules with bulky substituents that can easily dye polyester.
Herein, we synthesized methoxy and hydroxy triphenylamine-based azo dyes. Different substituents were used to study changes in color shade and dyeing properties. Systematic changes in photophysical properties and color on dyed polyester and nylon were demonstrated for these new azo dyes.
Experimental
Materials and Methods
Triphenylamine and pyridine hydrochloride were procured from Sigma-Aldrich. 1,10-Phenanthroline and copper iodide (S. D. Fine Chemical Ltd.) and potassium tert-butoxide and sodium nitrite (Spectrochem Pvt. Ltd.) were of commercial grade and used without further purification. All other chemicals were of analytical grade and purchased from S. D. Fine Chemical Ltd. Hexane, ethylacetate, chloroform, dichloromethane,
All reactions were monitored by TLC (thin-layer chromatography) with detection by UV light at 254 nm. Melting point was recorded in an open capillary on a Sunder Industrial Product and is uncorrected. The absorption spectra of the synthesized dyes were recorded on a Perkin-Elmer Lambda 950 spectro-photometer. Freshly prepared solutions in solvents of different polarities at a concentration of 1 × 10–6 mol/L solution were used in 1-cm optical path length quartz cuvettes. 1 H and 13 C nuclear magnetic resonance (NMR) spectra were recorded on a Agilent 500 MHz instrument.
Ready for dyeing nylon (70 d/24 f, plain weave, 210 × 191/5 cm), and polyester substrate (weight 70 g/m2, ends/in. = 105 and picks/in. = 94) were purchased from Suresh Kumar & Bros.
Lightfastness of dyed fabric was measured on a Q-Sun Xenon Test Chamber (Q-Lab Corp.) and sublimation fastness tested on a sublimation fastness tester (RBE Electronics and Engineering Pvt. Ltd.) A computational study was performed using the Gaussian 09W software. 23 Ground state optimization modeling was done using a B3LYP functional 24 and 6-31G (d) basis set. 25
Synthesis
3-Methoxy-
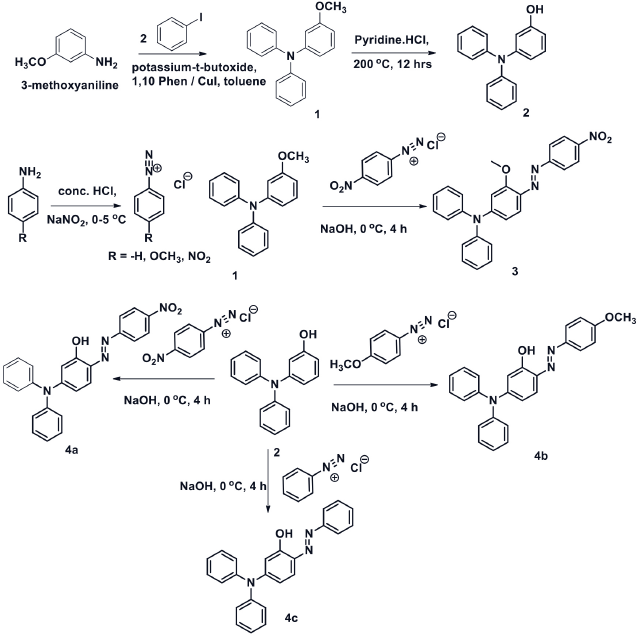
Synthetic scheme for dyes
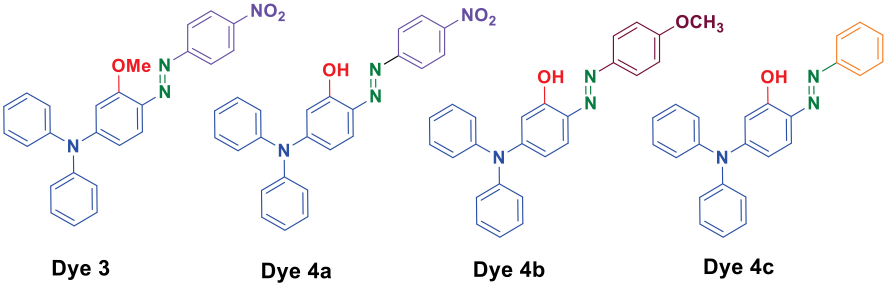
Structures of the triphenylamine-based azo dyes
3-Methoxy-4-((4-nitrophenyl)diazenyl)-N,N -diphenylaniline (3)
Synthesis of Dyes 4a-c
5-(Diphenylamino)-2-((4-nitrophenyl)diazenyl)phenol (4a )
Product
5-(Diphenylamino)-2-((4-methoxyphenyl)diazenyl)phenol (4b )
Product
5-(Diphenylamino)-2-(phenyldiazenyl)phenol (4c )
Product
General Procedure for Dyeing
Dyeing of polyester and nylon was carried out in a Rossari Labtech Flexi Dyer Machine using a material to liquor ratio (LR) of 1:30. The depth of the shade was 2% owf.
28
All synthesized azo dyes were solubilized in 5 mL of
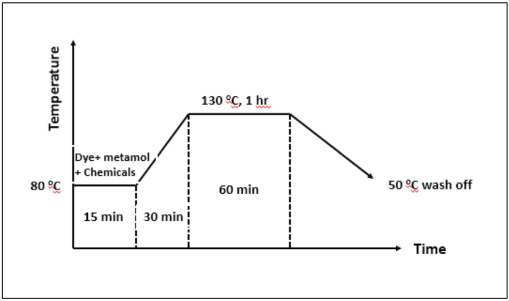
Dyeing curve.
Results and Discussion
Photophysical Properties
A photophysical study of the synthesized dyes (
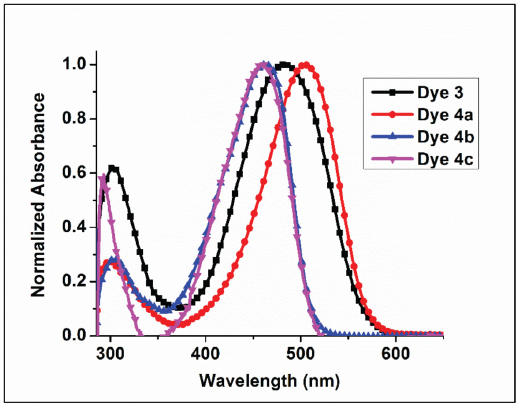
Normalized absorption spectra of dyes
Absorption peak characteristics were evaluated using full width half maxima (FWHM). FWHM is the wavelength difference between the two points on either side of the maximum at which the intensity is at half of the peak's maximum value. FWHM of dyes
Absorbance Maxima, FWHM, and Molar Absorptivity of Dyes in Various Solvents
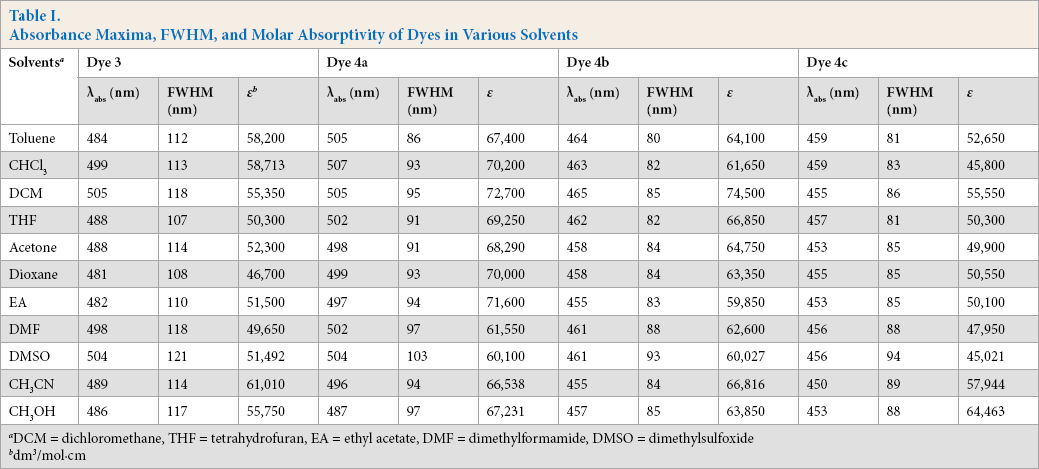
DCM = dichloromethane, THF = tetrahydrofuran, EA = ethyl acetate, DMF = dimethylformamide, DMSO = dimethylsulfoxide
dm3/mol∙cm
Intramolecular Hydrogen Bonding
To study the effect of intramolecular hydrogen bonding on the photophysical properties of these dyes, we have selected dye
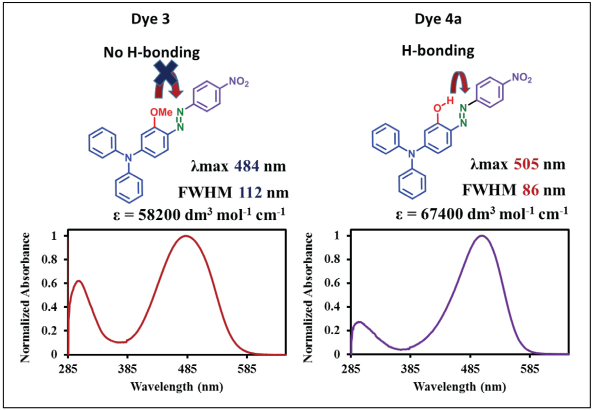
Comparison of properties of dyes
pH Effect
The presence of hydroxyl group in these dyes suggested that pH studies would be of value. Studies were carried out from pH 2 to 12. Aqueous DMF solutions were prepared and pH 2, 4, 6, 8, 10, and 12 solutions were created using 5% hydrochloric acid and 5% sodium hydroxide solutions. Fig. 5 shows the effect of various pHs on the new synthetic dyes. Dyes containing hydroxyl groups (
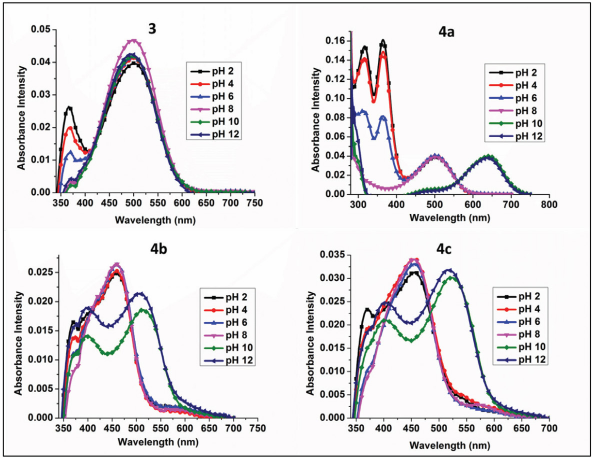
Effect of different pH on the absorption of dyes
Dye Optical Properties
When we compared the synthesized dyes
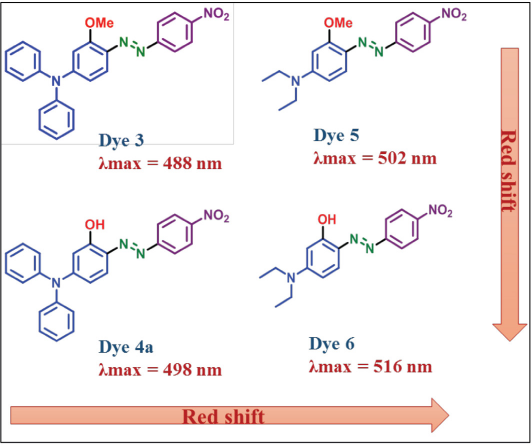
Absorption of synthesized dyes
Polyester and Nylon Dyeing
Color Properties
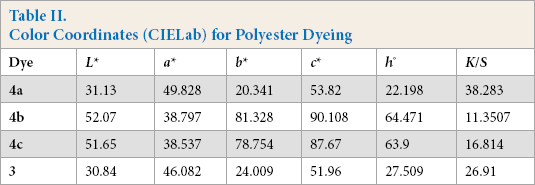
The colorimetric properties of the fabric dyeings were expressed in terms of the CIELab system. The
Color Coordinates (CIELab) for Polyester Dyeing
Color Coordinates (CIELab) for Nylon Dyeing
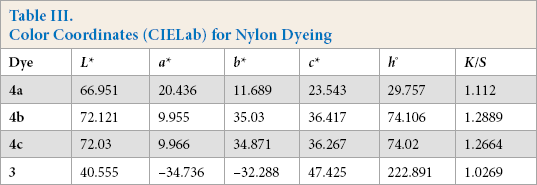
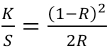
For CIELab determinations,
All dyes gave uniform colors on the test fabrics. Dyes
Lightfastness
Lightfastness is the degree to which a dye resists fading due to light exposure. The lightfastness of the dyed samples was measured by AATCC Test Method (TM) 16-200428 in which half of the sample was exposed to a xenon lamp for 17 h while the other half was covered. The samples were then compared with the Blue Wool standard scale and fastness ratings were given. Lightfastness of dyes on polyester was better than on nylon fabrics. All dyes showed moderate lightfastness on both polyester and nylon (Table IV).
Sublimation Fastness
Sublimation fastness is the most significant requirement of dyed fabrics. It is determined by standard method ISO 105-F04. 28 The dyed samples were sandwiched between two undyed cloth pieces (polyester and nylon depending on which fabric was tested) and were subjected to 150 °C, 180 °C, and 210 °C for 30 s. Two parameters, color change (cc) and color staining (cs, on undyed cloth) were rated.
Sublimation fastness ranged from low to moderate. It was a half scale better for dyed polyester than for the dyed nylon samples (Table IV).
Lightfastness and Sublimation Fastness Rating for Dyed Polyester and Nylon Fabrics
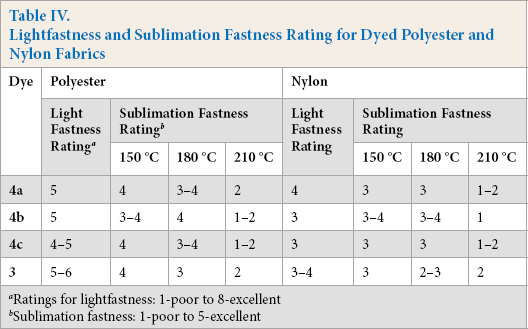
Ratings for lightfastness: 1-poor to 8-excellent
Sublimation fastness: 1-poor to 5-excellent
Percent Dye Exhaustion and Fixation
The percent exhaustion of the dyebath plays an important role in the kinetics of the dyeing process. The amount of dye exhausted from dye bath during dyeing is calculated using Eq. 2. 29
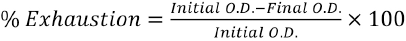
Initial O.D. is the optical density of dyebath before dyeing and final O.D. is the optical density of dyebath after dyeing.
Percent fixation gives the amount of dye fixed on the fabric. It is calculated by taking the ratio of fabric weight before and after dyeing (Eq. 3).
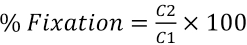
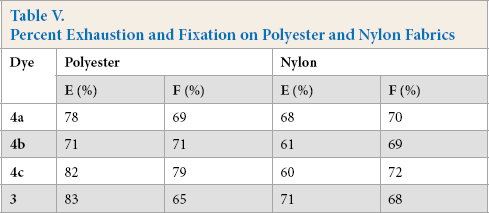
Fixation values were comparable for both fabrics. The color and exhaustion values were greater for polyester than for nylon (Table V). This was probably because polyester allowed better diffusion of the dye into the fibers than nylon.
Percent Exhaustion and Fixation on Polyester and Nylon Fabrics
Comparative Study
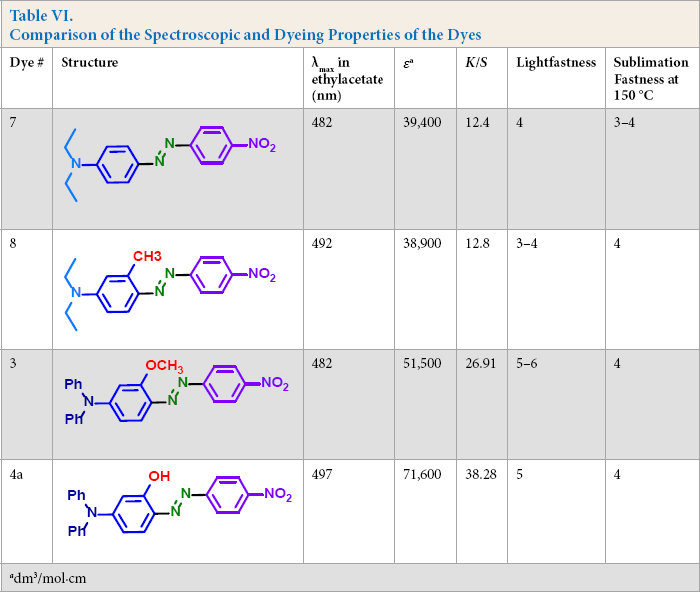
Spectroscopic and dyeing properties of the newly synthesized dyes
Comparison of the Spectroscopic and Dyeing Properties of the Dyes
Molecular Modeling Studies
Optimized Dye Geometry
Ground state geometries of
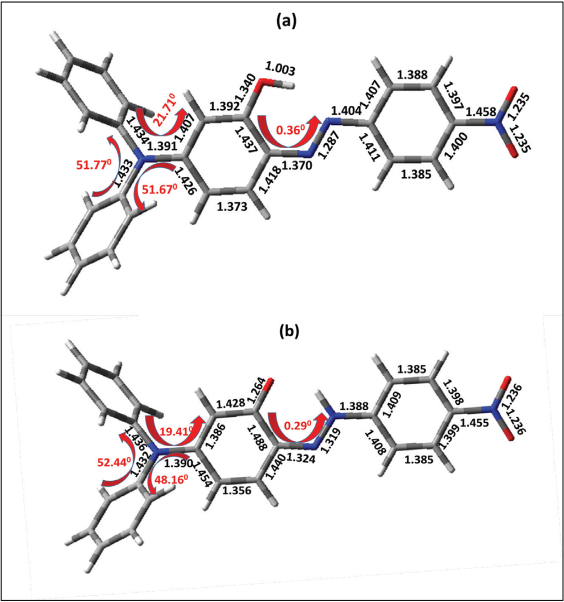
Optimized geometry of dye 4a (a) azo and (b) hydrazone tautomer in chloroform.
The computed energies of the tautomers were compared. Dyes
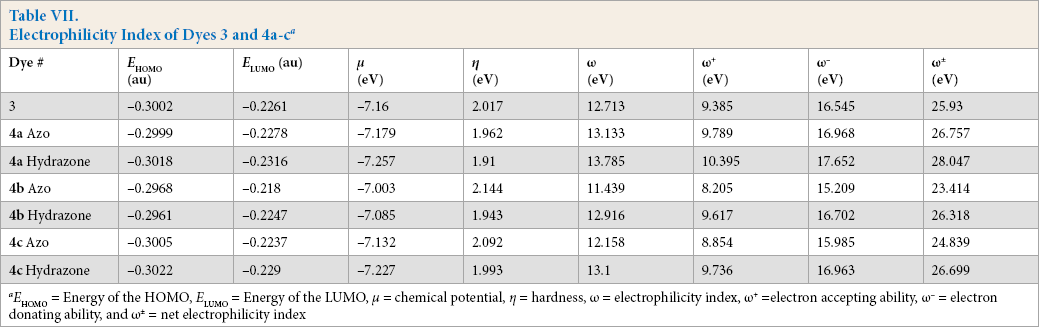
Electrophilicity Index
To understand the chemical stability of these dyes, global electrophilicity index (ω) 32 values were calculated according to Eq. 4.
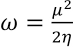
μ is the negative of the electronegativity obtained using Eq. 5.
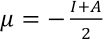
η is chemical hardness as defined by Eq. 6.
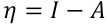
Energies of the HOMO and LUMO of the dyes were obtained from optimized geometries of the dyes by the density functional theory (DFT) method, 34 and then substituted into Eqs. 5 and 6 to give Eqs. 7 and 8.
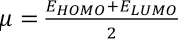
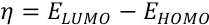
Therefore, to propose dye stabilities, the net electrophilicity index (ω±) was calculated using Eq. 9.
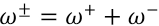
ω+ is electron accepting power and or is electron donating power and were obtained from Eqs 10 and 11.
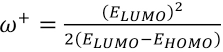
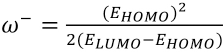
From Table VII, the azo tautomer gave greater
Electrophilicity Index of Dyes 3 and 4a-c a
Frontier Molecular Orbital (FMO) Approach
The frontier molecular orbitals diagram (Fig. 8) of the dyes shows that, for dyes
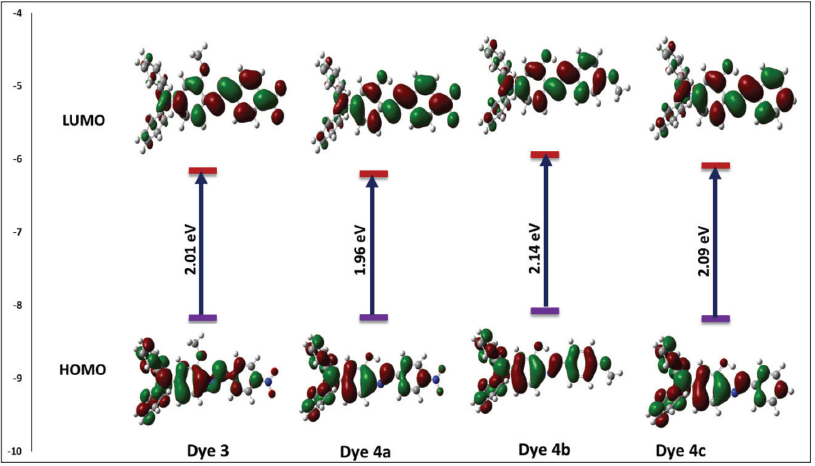
FMO diagram for dyes
Molecular Electrostatic Potential (MEP) Plots
To determine the reactive sites of the dyes, MEP analysis was performed at the B3LYP/6-31G(d) level of theory. Fig. 9 shows the MEP surface plots for dyes
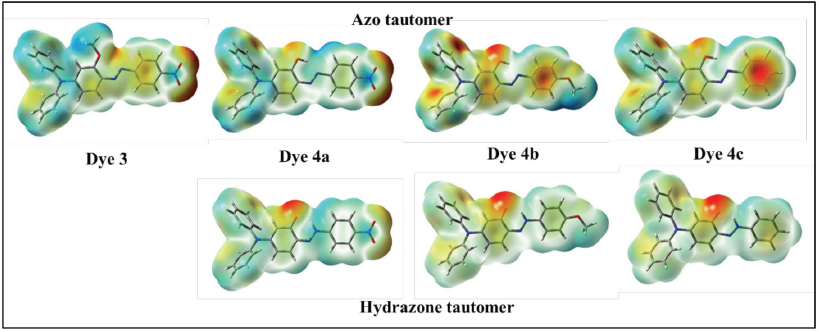
MEP plots of dyes
For the hydrazone form, the C=O group has a negative electron potential. MEP values observed for the azo tautomer of dyes
Conclusion
Four methoxy- and hydroxy-based triphenylamine azo dyes (
Supporting Information
NMR spectra and absorption plots of the dyes
