Abstract
Two novel halogenated triazine amine
Introduction
Bacteria exist in nature, with some causing disease, which can negatively impact human health. This has led to the development of antimicrobial materials, some of which can be applied in the field of textiles.1–3 Cotton fabric is one of the most common textile materials which can provide a suitable environment for bacteria reproduction under desirable external conditions (including humidity, temperature, nutrition, etc.). 4 Pathogenic bacteria are an important source of cross-infectious diseases. Therefore, it is extremely important to develop cotton fabrics and textiles with bactericidal functions.4,5
There are several kinds of antibacterial agents, such as quaternary ammonium salts, metal and metal oxide, chitosan, biguanide, and
Cyanuric chloride could react with reactive functional groups such as-NHR, -NH2, -SH, -OH, through the nucleophilic substitution reaction, and three chlorine atoms of cyanuric chloride could be gradually replaced by controlling the reaction temperature to form monosubstituted, disubstituted, trisubstituted triazine derivatives. One type of reactive dye contains triazine derivatives which could react with cellulosic fibers under low temperature and alkaline conditions, and the dyed fabrics maintain good tensile strength. Cyanuric chloride is used as the active group to prepare a highly reactive
In this study, two novel structures of
Experiment
Materials
The cotton fabrics were bought from Zhejiang Guandong Textile Dyeing Garment Co., Ltd. Cyanuric chloride, 2,2,6,6-tetramethyl-4-piperidinol, nicotinic acid, and 2-chloroethylamine hydrochloride were obtained from J&K Chemical Co., Ltd.
Instruments
Fourier-transform infrared (FTIR) spectra of PT and CPT were characterized by a Nicolet NEXUS 470 spectrometer (Nicolet Instrument Corporation, USA) in the optical range of 500–4000 cm−1. An AVANCE III 400-MHz Digital NMR Spectrometer (Bruker AXS GmbH, Germany) was applied to test PT and CPT. The surface morphologies of untreated and treated cotton fabrics were tested by SU1510 field emission SEM (Hitachi, Tokyo, Japan).
Synthesis of PT
Aqueous solution (50 mL) containing 8.55 g of 2,2,6,6-tetramethyl-4-piperidinol was put in acetone solution (80 mL) containing 9.10 g of 2,4,6-trichloro-1,3,5-trizine in three round bottom flasks (250 mL) and stirred at 0–5°C for 3 h. The pH of the solution was 6–7 which was adjusted using 1.25 M Na2CO3. An equivalent of sodium salt of
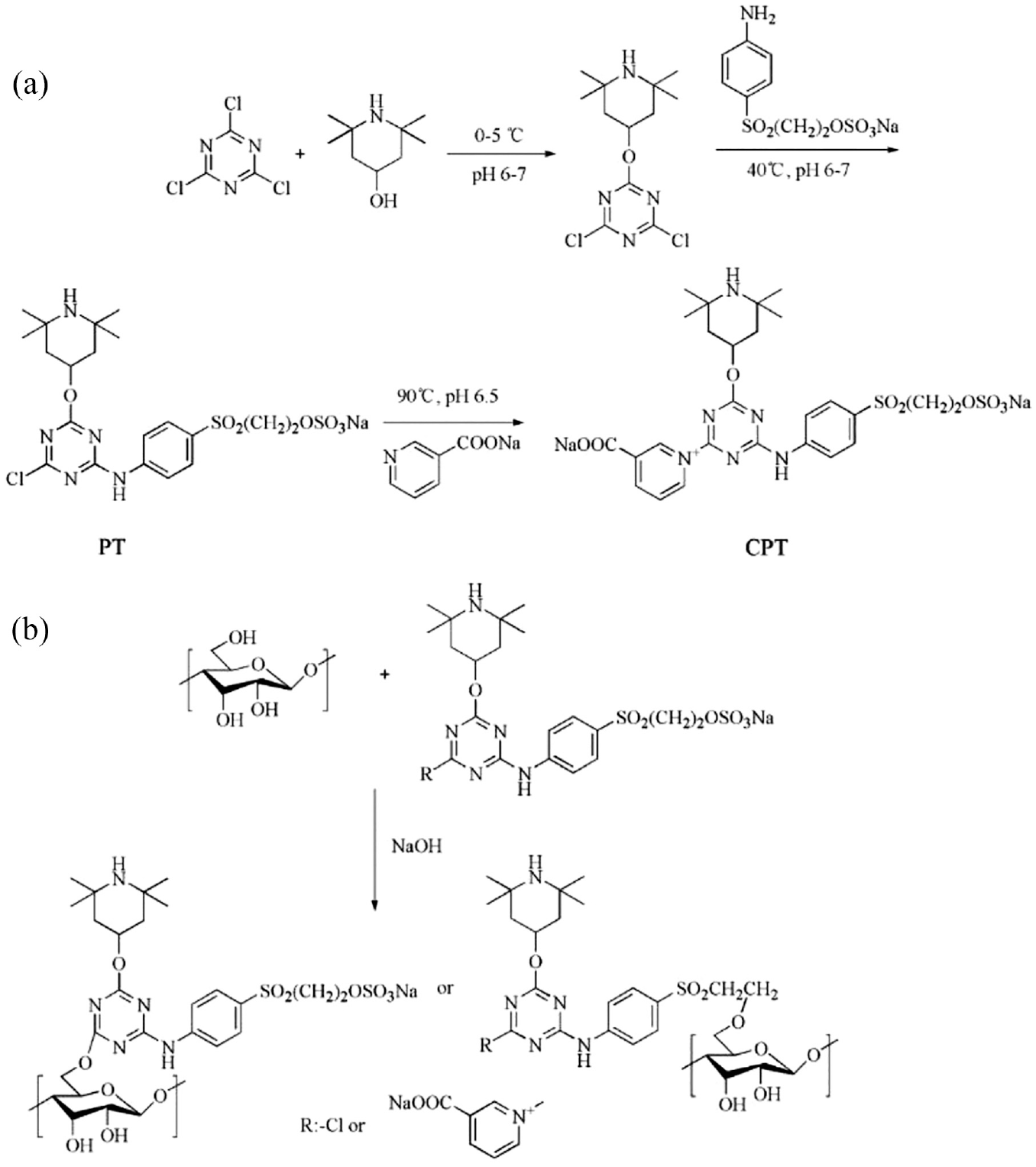
(a) The synthesis schemes of PT and CPT and (b) preparation of PT- and CPT-coated cotton fabrics.
Synthesis of CPT
A double active group containing nicotinic acid triazine and vinyl sulfone group of a halogen antibacterial precursor to CPT was synthesized with the introduction of nicotinic acid in the original PT structure. After the reaction of PT was completed, 50 mL of aqueous solution of 3-pyridyl sodium (made from nicotinic acid (0.05 mol) and sodium carbonate (0.025 mol) dissolved in water) was slowly put dropwise into the system at 90°C, while the solution was kept at 90°C with a pH 6.5 for 3 h. Acetone was removed by rotary evaporation when the reaction was completed. After suction filtration and salting out, a brown solid was obtained, washed with ethanol, and dried in a vacuum oven at 45°C for 24 h to get the reaction product CPT (63% yield). The synthesis of CPT is shown in Figure 1(a).
Preparation of Antibacterial Cotton Fabrics
A certain amount of PT (30% owf) with Na2SO4 (100 g/L) or CPT (20% owf) with Na2SO4 (50 g/L) was added to a beaker, and then stirred well. The fabrics were dipped into the solution for 0.5 h. NaOH of 1 g/L was added in the mixture when the temperature was 60°C. The products were taken out after 3 h and removed unfixed PT or CPT with water. The preparation of PT- and CPT-coated cotton fabrics is shown in Figure 1(b). The schematic illustration of the antibacterial process of the PT- or CPT-coated cotton fabrics including the finishing process and chlorinating procedure is shown in Figure 2.
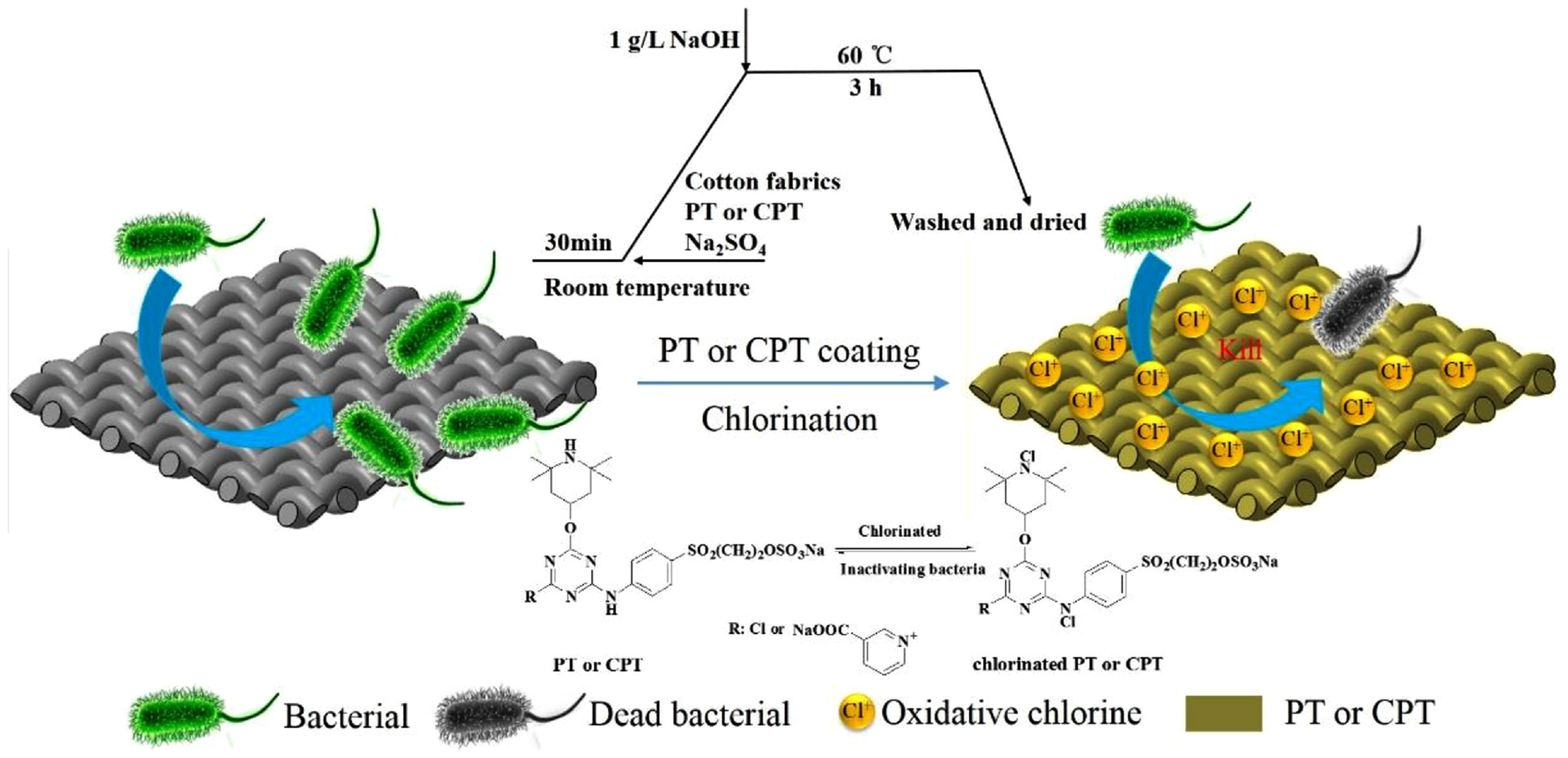
Schematic illustration of the antibacterial processes of PT- or CPT-coated cotton fabrics including the finishing process and chlorinating procedure.
Chlorination and Analytical Titration
To obtain antibacterial properties, the PT- and CPT-coated cotton fabrics were immersed in 10% aqueous sodium hypochlorite solution (pH 7) for 1 h. Then, the chlorinated fabrics were washed with distilled water to get rid of free chlorine. The active chlorine was measured by the iodometric/thiosulfate titration technique, and the concentration of sodium thiosulfate solution (Na2S2O4) was 0.001 N. The loaded chlorine was calculated from equation (1):
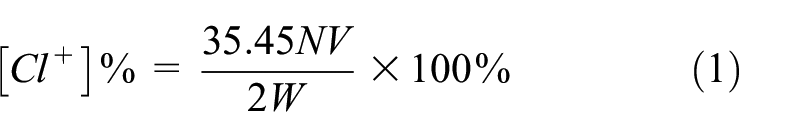
where [
Antimicrobial Test
The antimicrobial test was performed with a sandwich test according to AATCC 100-2004. The bacteria were suspended in 100 mM phosphate buffer (pH 7); then, 25 μL of bacterial suspension was put in the middle of two pieces of the prepared samples. All oxidative chlorine residues on the testing fabrics were removed after contact for 1, 5, 10, and 30 min using 5.0 mL of sterile 0.02 N Na2S2O4 solution. The surviving bacteria were rinsed from the samples, and serial dilutions of the quenched solutions were plated on Trypticase soy agar for 24 h at 37°C. The bacterial groups were calculated for antimicrobial efficacy analysis.
Stabilities and Rechargeabilities
The coated cotton fabrics were washed to estimate the stability and durability of chlorine according to AATCC 61-1996, and the specific operation was as follows. The treated swatches (1 in × 2 in) were soaked in 0.15% detergent water solution with 50 stainless-steel balls. Then, the stainless-steel canisters were rotated at 42 r/min and 49°C for 45 min, which was equal to five machine washings. After 5, 10, 25, and 50 machine washing, the fabrics were taken out and washed with distilled water and dried. The stability and rechargeability of each type of sample were tested by an iodometric/thiosulfate titration method with/without rechlorination, respectively.
For the storage stability measurement, the chlorinated PT- and CPT-coated cotton samples were stored at normal temperature. After several days of storage, the samples were taken out and two types of experiments were carried out. The storage stability of fabrics were measured by an iodometric/thiosulfate titration technique, and other fabrics were rechlorinated and the rechargeability was measured by the same way.
Breaking Strength Testing
According to GB/T 3923-1997, the breaking strength of cotton fabrics with/without chlorination was tested by an electronic fabrics strength tester. Each tested sample was cut into 250 × 50 mm2, and the average value was recorded for analysis.
Results and Discussion
Synthesis and Characterization of PT and CPT
2,4,6-Trichloro-1,3,5-trizine is a very important ingredient of reactive dyes. The three chlorines of s-triazine rings could be substituted by many chemicals or bonds with other fabrics types at an appropriate temperature.25,26 In this study, s-triazine rings with the first chlorine was replaced with 2,2,6,6-tetramethyl-4-piperidinol as the carrier of the antimicrobial agent at 0–5°C. The second reactive group,
The structures of PT and CPT contain a double-reactive group, similar to the molecular structure of the reactive dye, which can be coated onto cotton fabric by nucleophilic substitution (monochloro triazine reactive group and nicotinic acid triazine reactive group) and nucleophilic addition (bis-sulphatoethylsolphone reactive group) at a suitable temperature and alkalinity through covalent bonds.27–29 In general, only one random reactive group could react with the materials in the finishing process and the other reactive groups would be retained (Figure 1(a)). In the application of cotton fabrics, the function of alkalinity can be used for increasing cellulose anions and dissolving antimicrobial precursors. Moreover, the addition of the neutral salt was used to reduce the rejection between cotton fabrics and the agent, which accelerated the reaction between substrate and agent. The 1H NMR spectra of PT and CPT are shown in Figure 3.
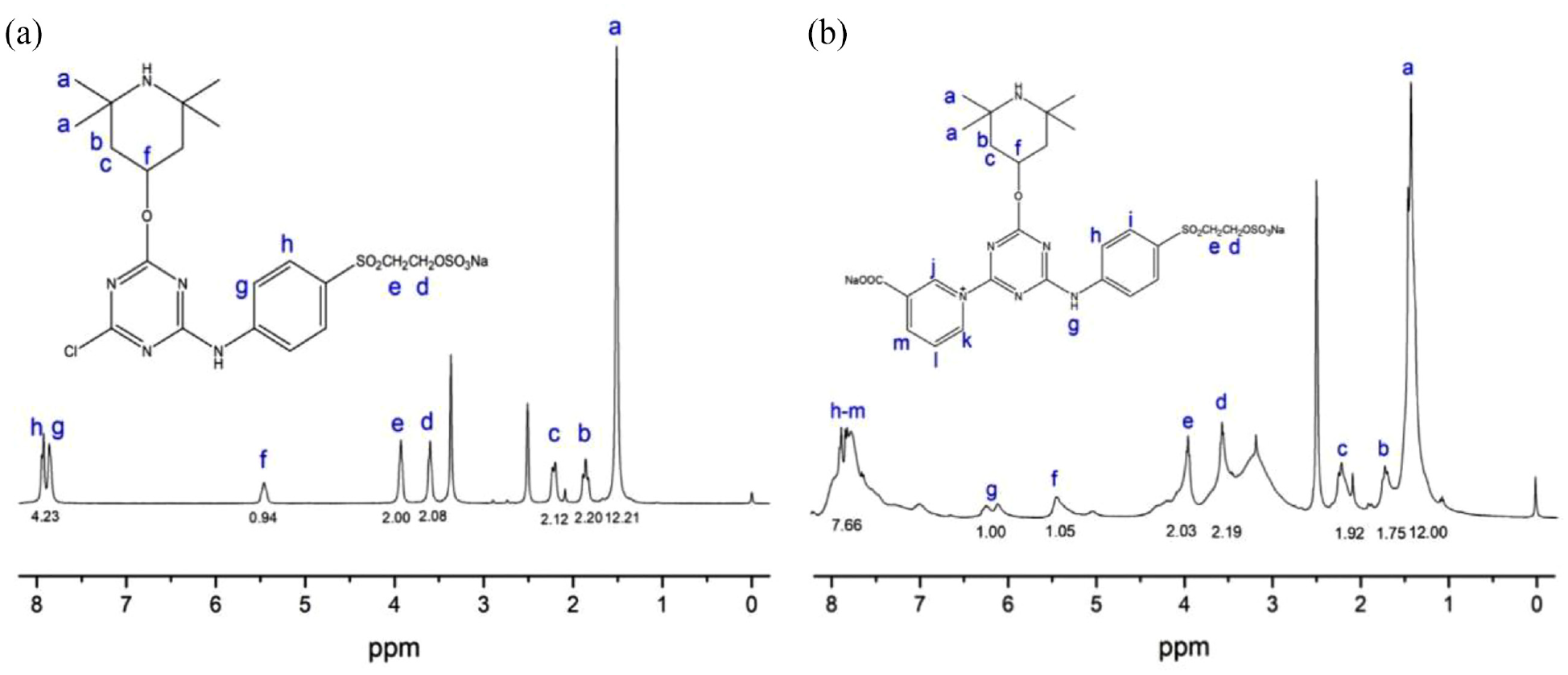
The 1H NMR spectra of (a) PT and (b) CPT.
The 1H-NMR spectrum of PT is shown in Figure 3(a). 1H-NMR(DMSO-d6): δ 1.40–1.65(12H), δ 1.86(2H), δ 2.21(2H), δ 3.60(2H), δ 3.93(2H), δ 5.46(1H), δ 7.82–7.97(4H). The 1H-NMR spectrum of PT is shown in Figure 3(b). 1H-NMR(DMSO-d6): δ 1.25–1.60(12H), δ 1.76(2H), δ 2.23(2H), δ 3.57(2H), δ 3.96(2H), δ 5.46(1H), δ 6.20(1H), δ 7.64–7.96(8H). As a result, the PT and CPT were successfully synthesized.
Characterization of PT- and CPT-Coated Cotton Fabrics
The FTIR spectra of different cotton fabrics are shown in Figure 4. The distinctive vibrational bonds of PT-coated cotton fabrics were found at 1542 cm−1 (Figure 4(b)), which amounts to the phenyl vibrational modes of PT. This bond was not observed in unfinished cotton (Figure 4(a)). The characteristic vibrational bonds of CPT-coated cotton samples appeared at 3291 and 1248 cm−1 (Figure 4(d)), which is equal to the methyl and C–N bonds in nicotinic acid vibrational modes of CPT, respectively. It provided evidence that PT and CPT were coated onto the cotton fabrics. After chlorination, these vibrational bonds moved to 1551 (Figure 4(c)), 3296, and 1254 cm−1 in Figure 4(e), respectively, due to the electron withdrawing effect of oxidation.30,31
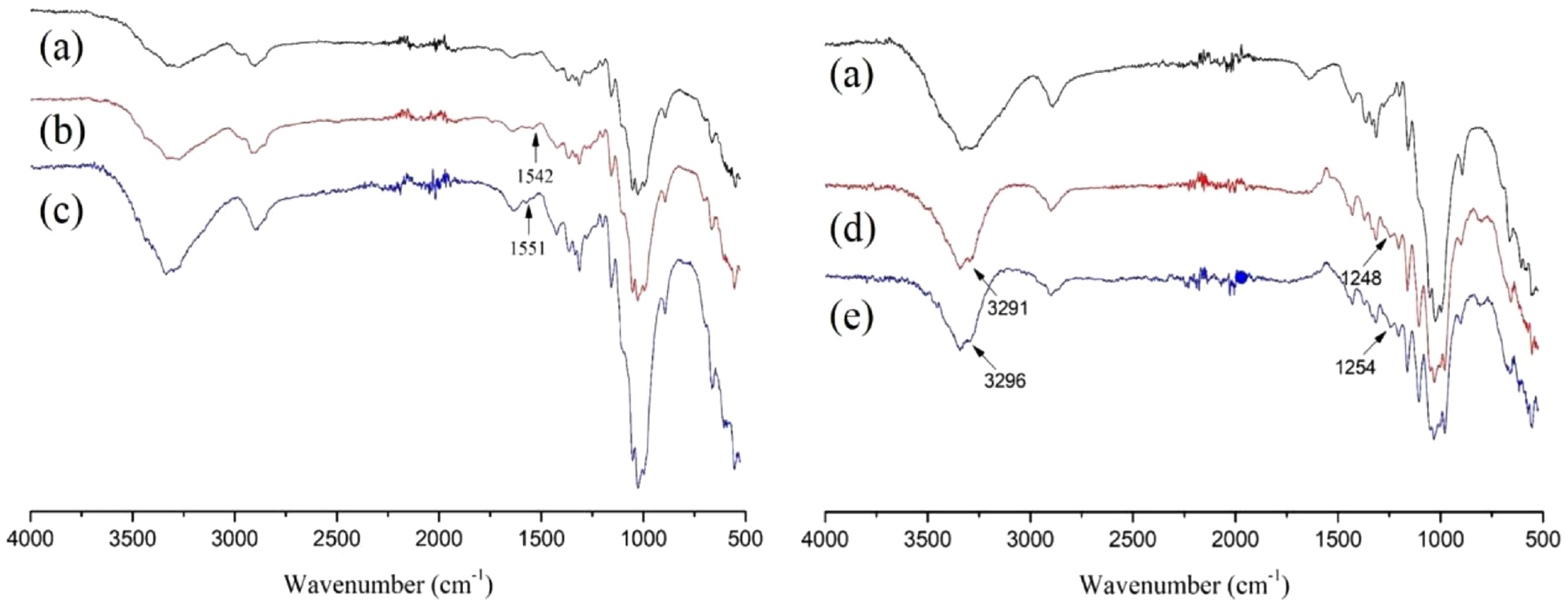
FTIR spectra of (a) cotton fabrics, (b) PT-coated cotton fabrics, (c) chlorinated PT-coated cotton fabrics, (d) CPT-coated cotton fabrics, and (e) chlorinated CPT-coated cotton fabrics.
Figure 5 reveals the SEM images of the surface topographies of cotton, PT- and CPT-coated cotton fabrics. It was observed that the uncoated cotton fabric surfaces were smooth. The surface of PT- and CPT-coated fabrics became rough compared with the uncoated cotton fabric.

SEM images of cotton fabrics: (a) Uncoated, (b) PT-coated, and (c) CPT-coated.
Biocidal Efficacy
The biocidal efficacy of PT- and CPT-coated cotton fabrics against bacteria is shown in Figure 6. The chlorinated PT-coated fabrics could inactivate 97.49% (1.60 log) within 1 min and all (5.97 log) of
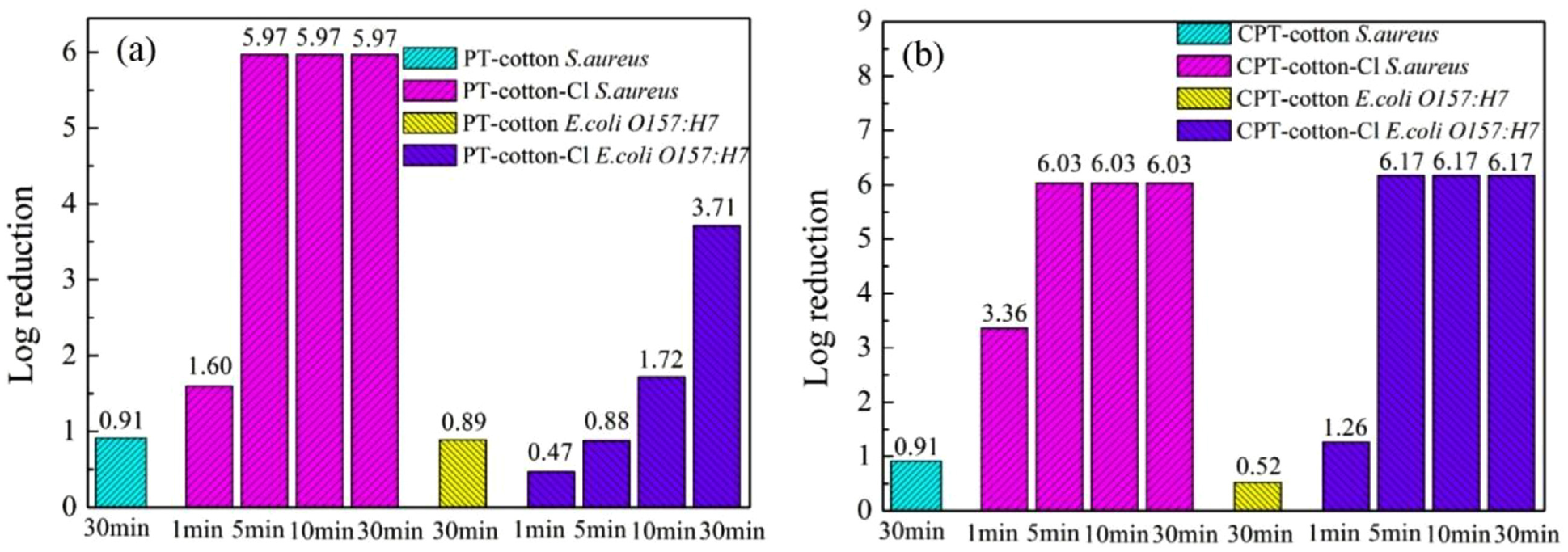
Biocidal efficacy of (a) PT- and (b) CPT-coated cotton fabrics against
Washing and Storage Stability
The washing stability of the coated fabrics is demonstrated in Figure 7(a). For the chlorinated PT-coated cotton fabrics, the retained chlorine decreased rapidly with the increase of washing cycles. Almost all chlorine was lost (from 0.12% to 0.02%) and 83% of the chlorine could be regained after rechlorination after 50 washing cycles, which demonstrated that 17% of the coating was lost after multiple machine washings. Meanwhile, after 50 washing cycles, 75% of chlorine on CPT-coated fabrics was lost due to the hydrolysis of the N–Cl bonds, 34 and over 93% chlorine could be renewed after rechlorination, which indicated that CPT-coated samples were more stable than PT-coated samples. An additional coulomb force was formed between cellulose anion and unreacted nicotinic acid triazine reactive groups in CPT, and the larger molecular weight prompted higher affinity between CPT and cotton fibers.
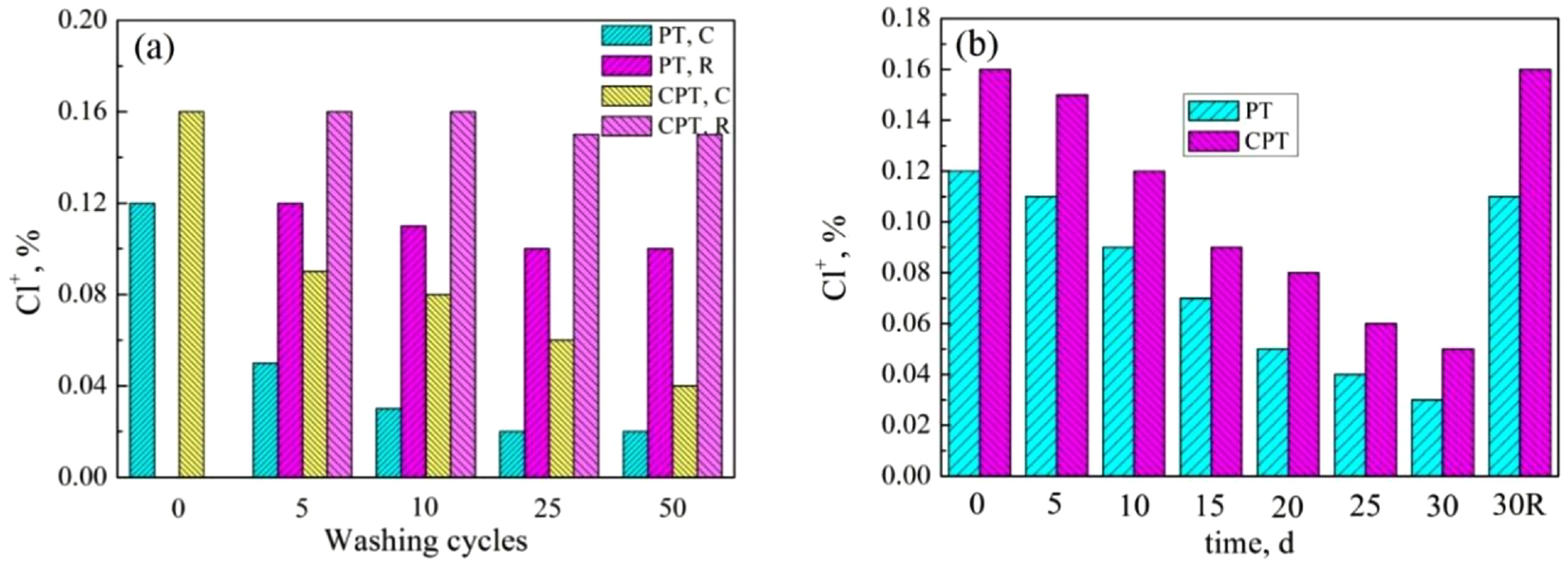
(a) Washing and (b) storage stability of the cotton coated with PT and CPT. C was chlorination before washing (Cl+%); R was rechlorinated after each washing cycle (Cl+%). Each washing cycle is identical to five machine washings in this method.
Figure 7(b) reveals the storage stability of active chlorine on the chlorinated cotton samples coated with PT and CPT at ambient temperature. After 30 days, the chlorine loading on the chlorinated fabrics coated with PT was decreased by 75% from the initial 0.12% to 0.03%. While 69% chlorine on the CPT-coated fabrics was lost (from 0.16% to 0.05%), the remaining chlorine maintained sufficient antimicrobial activity against the bacteria. 21 Almost all active chlorine on both PT- and CPT-coated cotton samples could be regenerated after rechlorination.
Breaking Strength Testing
The breaking strength of uncoated and coated fabric samples without/with chlorination is exhibited in Figure 8. Figure 8 shows that more than 90% of tensile strength of PT- and CPT-coated fabrics was retained in the warp direction before and after chlorination, and about 85% of tensile strength was retained in the weft direction. The breaking strength was decreased on account of the alkaline conditions. The hydrogen bonds were lost owing to the substitution of hydroxyl groups. 20 The oxidation of chlorine caused the small reduction of tensile strength after chlorination.
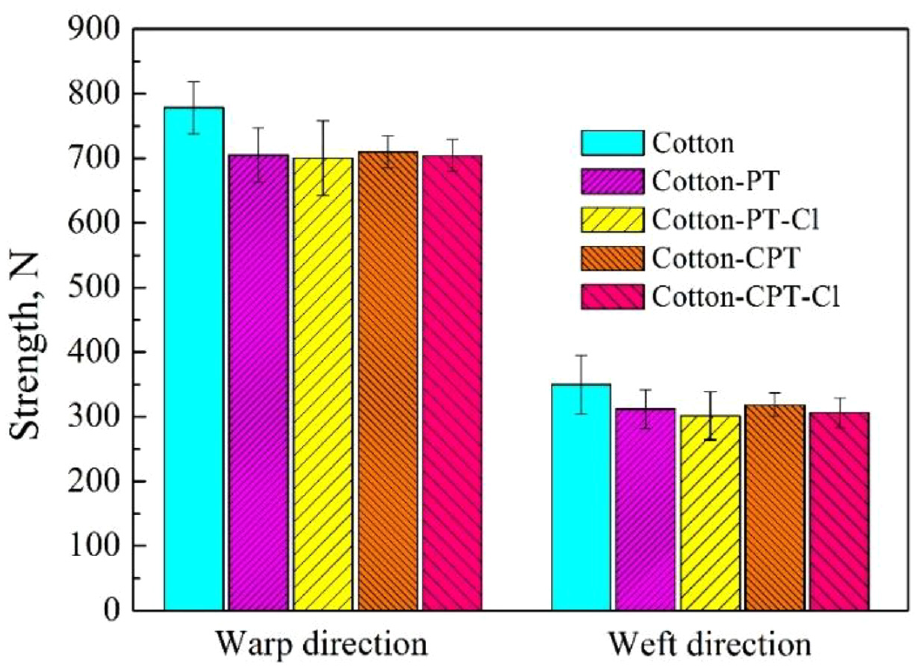
Breaking strengths of cotton fabrics and cotton fabrics coated with PT and CPT.
Conclusion
The antimicrobial
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the Hubei Provincial Central Leading Local Science and Technology Development Special Fund (2022BGE253), Jiangsu Province Industry-University-Research Cooperation Project (BY2022893), Nantong Basic Science Research Program (JCZ2022100), Jiangsu Province Vocational College Young Teachers Enterprise Practice Training Project (2022QYSJ015), Jiangsu Advanced Textile Engineering Technology Center Funds (XJFZ/2021/10, XJFZ/2021/19), Scientific Planned Projects for Jiangsu College of Engineering and Technology (GYKY/2021/6), and Science and Technology Guiding program of China Textile Industry Federation (2022032).
