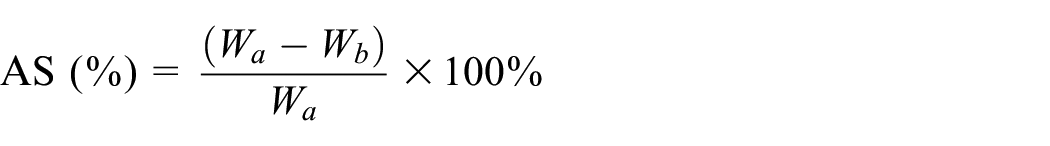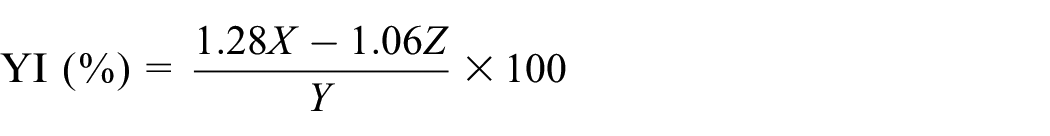Abstract
In order to enhance the shrink-resistant properties of wool to achieve a machine washable effect, the effect of water-soluble polyurethane polymer on a worsted wool fabric which was modified by liquid ammonia and protease was investigated. The worsted wool fabric was pretreated by a continuous liquid ammonia finishing machine and then treated by protease, followed by polyurethane nano-emulsion coating. The results showed that, after liquid ammonia and protease treatment, the surface scales of wool were seriously damaged, even partly peeling off, and the disulfide bond content of the wool decreased while the active group content of the wool increased. Furthermore, after polyurethane finishing, the surface scale and the gap between the scales were covered with a thin film, and the area shrinkage reached 3.1% when the concentration of polyurethane was 20 g/L, showing an effective improvement in the shrink resistance of the worsted fabric. As far as our knowledge goes, this is a systematic report on the synergistic effect of liquid ammonia, protease, and polyurethane on the shrink resistance of wool fiber, and provides a new method for the commercial application of shrink-resistant finishing of wool fabric.
Introduction
Wool fiber is a natural renewable protein fiber with excellent warmth, elasticity, and moisture properties. 1 However, due to the directional friction effect of the wool scale layer wool fabrics are susceptible to felting and shrinking during the washing process, which affects the fabric handle and the dimensional stability. Therefore, many anti-felting methods have been developed to reduce the directional friction effect of wool. At present, the most commonly used method of anti-felting finishing in industry is the Chlorine–Hercosett method. However, the method can release the carcinogenic and teratogenic absorbable organic halogen AOX, and it has been restricted in the European Union.2–4 Therefore, the chlorine-free ecological anti-felting finishing has become a new research hotspot in recent years. The common chlorine-free anti-felting methods include protease and polyurethane finishing.
Protease treatment aims to decrease the directional friction effect of wool by catalytically hydrolyzing its peptide bonds.5,6 However, poor anti-felting properties were obtained due to powerless hydrolysis of the surface scale. Furthermore, the protease mainly acts on the cell membrane complex (CMC) between surface scale and cortical cells, causing serious damage to the fiber strength. 7 To improve the effect of protease, methods such as multi-enzyme synergy,8–10 ultrasonic treatment, 11 plasma radiation, 12 and chitosan pre-adsorption 2 were used to increase the effect of protease on the surface scale, and reduce the damage to the fibers.13,14 However, the ultrasonic and plasma technologies limited by equipment are difficult to apply in industry, and the chemical reagents will seriously affect the feel and strength of the fabric.
Polyurethane finishing could be used to decrease the directional friction effect of wool by cross-linking with wool fiber by polymer macromolecules or coating a polymer film on the surface of the wool. 15 As early as in the 1970s, polyurethane emulsion containing free isocyanate groups (Synthappret LKF, Bayer AG, Germany) was produced. However, due to the use of organic solvents and the active groups that are easy to hydrolyze, the finishing has shortcomings with regards to its environment friendless and poor shrink resistance. 16 Subsequently, polyurethane with bisulfite adducts was investigated and commercialized quickly, such as Synthappret BAP in Bayer. 17 However, normal polyurethane finishing of wool usually had some shortcomings like stiff feeling, yellowing, and poor long-term durability.18,19
Therefore, to meet the needs of industrial production, a new and operable chlorine-free shrink-resistant finishing becomes an inevitable trend. Liquid ammonia finishing has become a rapidly developing technology in recent years, especially widely used in the cotton textile industry.20–22 Liquid ammonia has super permeability and fluidity, and easily penetrates into the fiber to change the fiber crystallinity. 23 In this article, liquid ammonia was introduced to partially peel off the surface scales of wool,24–26 so as to promote the modification of wool by protease and reduce the hydrolysis of CMC layer. However, the water-soluble polyurethane with good permeability and elasticity19,27,28 was used in the shrink-resistant finishing of wool. The finishing method overcomes the deficiency of single enzymatic and resin finishing through the synergistic effect of liquid ammonia, protease, and water-soluble polyurethane; in particula, the continuous liquid ammonia treatment has not been reported.
In order to discuss the synergistic effect of wool by liquid ammonia, protease, and polyurethane, the untreated wool fabric (W) was first pretreated with liquid ammonia by a continuous liquid ammonia finishing machine, and then the fabric (LAT) treated with protease. Finally, the modified wool fabric (LAPT) was treated by polyurethane nano-emulsion (LPPT). The amount of polyurethane was optimized by testing the breaking strength, area shrinkage, and weight loss. The surface morphology, molecular structure, and crystallinity were analyzed by scanning electron microscopy (SEM), attenuated total reflectance Fourier-transform infrared spectroscopy (ATR-FTIR), X-ray photoelectron spectroscopy (XPS), and X-ray diffraction (XRD). The shrink resistance and mechanism of liquid ammonia, protease, and water-soluble polyurethane were determined and discussed.
Experimental
Materials
A worsted fabric with 2/3 twill and mass per unit area of 232 g/m2 was used in this study (Ruyi, China). Savinase 16L was subtilisin protease produced by Bacillus tarda, which was supplied by Novozymes, China. The activity of the protease was 20,000 U/mL. One unit of enzyme activity is defined as the production of 1 µmol tyrosine per minute at pH 7.5 and 40°C, expressed in U/mL. BAYPRET® NANO PU was nanoscale polyether water-soluble polyurethane with self-cross-linking and was supplied by TANATEX Chemicals in the Netherlands. The diameter of the polymer was less than 100 nm.
Wool Treatment with Liquid Ammonia, Protease, and Polyurethane
The experiment was carried out on a continuous liquid ammonia finishing machine (Kyoto, Japan), and the worsted wool fabric was impregnated in the liquid ammonia at −33.4°C for 15 s. The excess ammonia was removed by steaming at 110°C for 15 s, followed by washing at 50°C for 1 min, and drying at 110°C for 1 min. After liquid ammonia pretreatment, the LAT fabric was treated by protease of 2% o.w.f. (on weight of fabric) at 45°C and pH 8 with a liquid ratio of 1:25 for 1 h in a high-temperature overflow dyeing machine. Then, protease was deactivated at 90°C. After that, LATP fabric was treated with 20 g/L polyurethane at pH 5 (adjusted by acetate and sodium acetate buffer solution) using the VNE6 model drying and shaping machine (Bruckner, Germany) at a speed of 20 m/min. Then, the fabric was dried at 105°C for 1 min and then baked at 160°C for 3 min. The combined processing of liquid ammonia, protease, and polyurethane on wool fabric is shown in Figure 1.

Schematic diagram of combined processing of liquid ammonia, protease, and polyurethane on wool fabric.
Surface Morphology
The surface morphology of wool was observed by a SU1510 scanning electron microscope (Hitachi, Japan) with an accelerating voltage of 5 kV, a current of 10 µA, and a 3000 magnification.
ATR-FTIR
Total reflection infrared spectroscopy was determined by a Nicolet iS10 FTIR (Thermo Fisher Scientific, Waltham, MA, USA). The scanning range was from 4000 to 650 cm−1 with 16 times, and the scanning resolution was 4 cm−1.
Raman Spectra
The Raman spectra of wool fabrics were measured using in via Microscopic Confocal Raman spectrometer (Renishaw, Britain) at 4 cm−1 resolution and with a laser power of 500 mW. The spectral scanning area was from 400 to 3500 cm−1.
Crystallinity Analysis
A D8 X-ray diffractometer (Bruker AXS, Germany) was used for crystallinity analysis with a Cu target (1.54 Å) at 40 kV and 40 mA. The scanning range was from 5 to 50° with a step size of 0.05°, and the scanning speed was 2°/min.
Weight Loss
The weight loss of the fabric was determined in terms of oven-dried weights of fabric before and after treatment using a weighing balance according to ASTM D1576-90 (2001). All samples were dried at 105°C for 2 h, then placed in a desiccator for balancing and weighted until constant weight. The average weight loss percentage (WL%) of each sample was calculated by the following equation:
where W0 is the dry weight of the untreated wool fabric (g), and W1 is the dry weight of the wool treated with liquid ammonia, protease and polyurethane nano-emulsion.
Alkali Solubility
The alkali solubility (AS%) of wool fabric was determined according to ISO 3072-1975, and the results are given as the arithmetic means of three parallel samples. The equation is as follows:
where Wa is the dry weight of the untreated wool fabric (g), and Wb is the dry weight of the wool treated with alkali (g).
Area Shrinkage
The shrinkage percentage of wool fabric was measured according to the IWS Test Method 31. The fabrics were washed in a Y(B)089E automatic shrinkage test machine (Darong, China) in one cycle of wash program 7A for relaxation and three cycles of program 5A for felting shrinkage. All samples were hang-dried dry after washing and conditioned at 25°C and a relative humidity of 60% for 24 h before measuring the area shrinkage. The results were given as the arithmetic means of three parallel samples.
Tensile Strength
The tensile strength of wool fabrics was determined by the H10K Universal tester (Tinius Olsen, Horsham, PA, USA) according to ISO5081. The results are given as the arithmetic means of three parallel samples.
Yellowing Index
The X, Y, and Z tristimulus values of the fabric were measured by a Datacolor 800 (Datacolor, Lawrenceville, NJ, USA) computer color testing and matching instrument. The yellow index (YI%) was calculated by the formula:
where X is the primary stimulus of red, Y is the primary stimulus of green, and Z is the primary stimulus of blue.
Hand Feel
Hand feel of wool fabrics was tested by the PhabrOmeter fabric style tester (Nu Cybertek, Davis, CA, USA) according to AATCC 202. The untreated wool fabric was used as a standard sample to test the softness of the fabric before and after finishing.
Results and Discussion
Chemical Characters of Wool Fabrics
FTIR was used to research the main functional groups of wool treated with liquid ammonia, protease, and polyurethane successively. The results are shown in Figure 2.
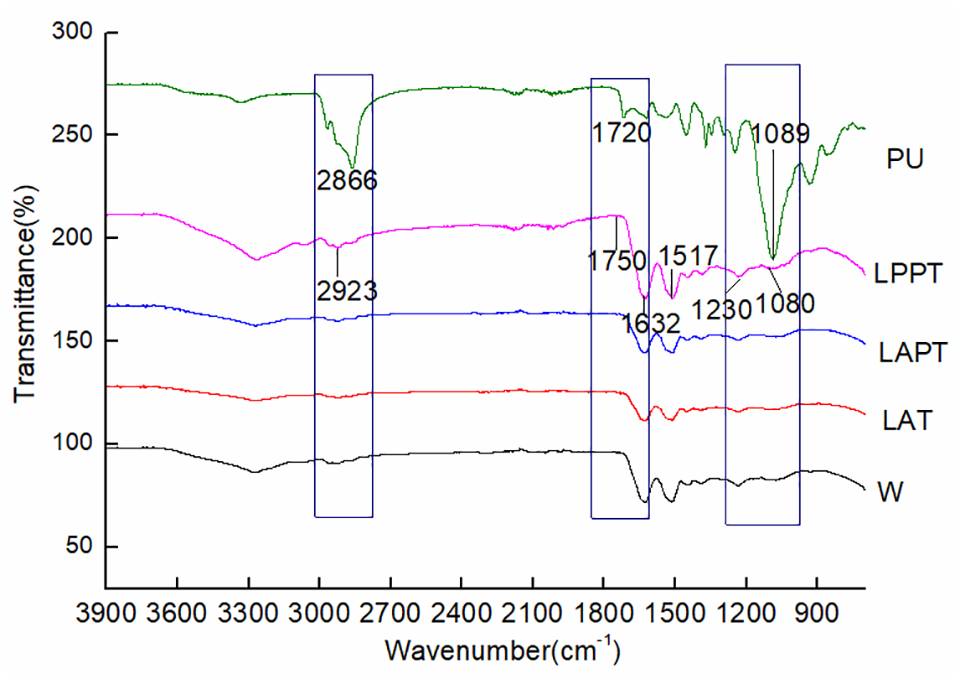
FTIR spectra of wool fabric and polyurethane nano-emulsion.
As shown in Figure 2, the spectra of wool treated with liquid ammonia and protease were basically consistent with that of untreated wool. However, the intensity of the absorption peak decreased compared with untreated wool. After the polyurethane finishing, the band intensity of the modified wool fabric was higher than that of untreated wool, especially at 3275, 2923, 1517, and 1630 cm−1. The wide and strong absorption band at 3275 cm−1 is the characteristic absorption band of amide A, which is the coupling of –NH– stretching vibration with frequency doubling stretching vibration of amide II. The absorption band is associated with the force resulting from the breakage of hydrogen bond between molecular chains, and the band intensity increased after polyurethane finishing. The absorption peak at 2923 cm−1 is the C–H stretching vibration, with two weak absorption peaks nearby, which reflect the vibration of –CH2 and –CH3 on the side group of the macromolecule main chain of wool.29–31 After polyurethane finishing, the intensity of absorption peaks increased, relating to the strong absorption peaks induced by the stretching vibration of –CH2 and –CH3 of polyurethane at 2866 cm−1. The absorption peak at 1517 cm−1 is amide II, induced by the bending vibration of N–H. The absorption peak of β-sheet structure of amide I was at 1630 cm−1, and the intensity of the peak increased after polyurethane finishing, indicating that the content of β-sheet increased. Two new characteristic absorption peaks were observed at 1080 and 1750 cm−1. The absorption peak at 1080 cm−1 was induced by the C–O–C absorption peak of polyurethane, while the peak at 1750 cm−1 was induced by allophanate of polyurethane, all indicating that the group of polyether polyurethane was found on the wool. 32
Raman Spectroscopy
The Raman spectrum of modified wool fiber after polyurethane finishing is shown in Figure 3.
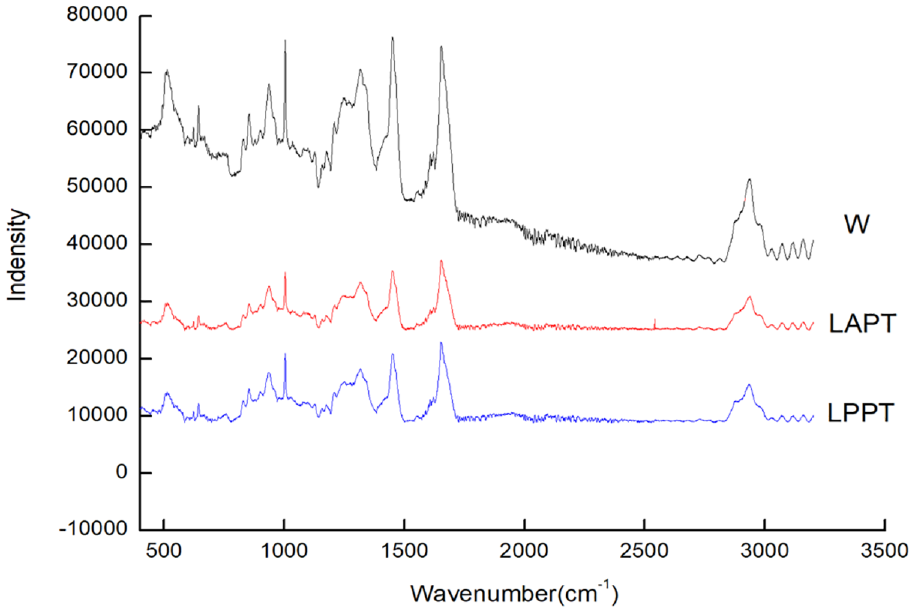
Raman spectra of modified wool fabrics.
Figure 3 shows that the intensity of characteristic absorption peaks of wool decreased after polyurethane finishing. The absorption peak at 1452 cm−1 is the bending vibration spectral band of –CH3 and –CH2 on the amino acid side chain, which is very stable and not affected by the conformation of the main peptide chain. Herein, the Raman spectrum of wool was normalized by the band intensity of the peak. After polyurethane finishing, the intensity of spectral bands at 510, 1255, and 1657 cm−1 changed. Herein, we use I510/145, I1255/1452, and I1657/1452 to represent the ratio of the actual intensity of these bands to the band at 1452 cm−1.
I510/1452 reflects the content of cysteine disulfide bonds.33,34 For the untreated wool, I510/1452 was 0.847, while the modified wool after liquid ammonia and protease treatment was 0.398. The results indicated that the disulfide bond of wool was seriously broken after being treated with liquid ammonia and protease. After polyurethane finishing, water-soluble polyurethane nano-emulsion easily entered the fiber due to its excellent dispersion and permeability, and then a thin film was formed to cover the surface and between the scales after baking at high heat and humid conditions. Furthermore, the disulfide bonds may be fractured under high heat and humid conditions and form new cross-links. These may be the reasons why the content of disulfide bond further decreased a little from 0.398 to 0.378 after polyurethane finishing. I1255/1452 reflects the disorder degree of the macromolecule aggregation peptide structure of keratin fiber. For the untreated wool, I1255/1452 was 0.738, while that of LAPT was 0.573 and LPPT was 0.556, which indicated that the disorder degree was decreased after combined finishing of liquid ammonia, protease, and polyurethane.
I1657/1452 reflects the content of α-helix. For the untreated wool, it was 0.937, and that of LAPT was 1.136, while that of LPPT was 1.108. The α-helix structure of wool macromolecules was increased after being modified by liquid ammonia and protease. However, after polyurethane finishing, the α-helix content decreased which indicated that the β-sheet content may be increased. Figure 2 shows that the absorption peak intensity at 1630 cm−1 was enhanced which means the β-sheet content increased in FTIR spectra.
XRD Analysis
The X-ray diffraction was used to measure the crystallinity of wool, and the results are shown in Figure 4.
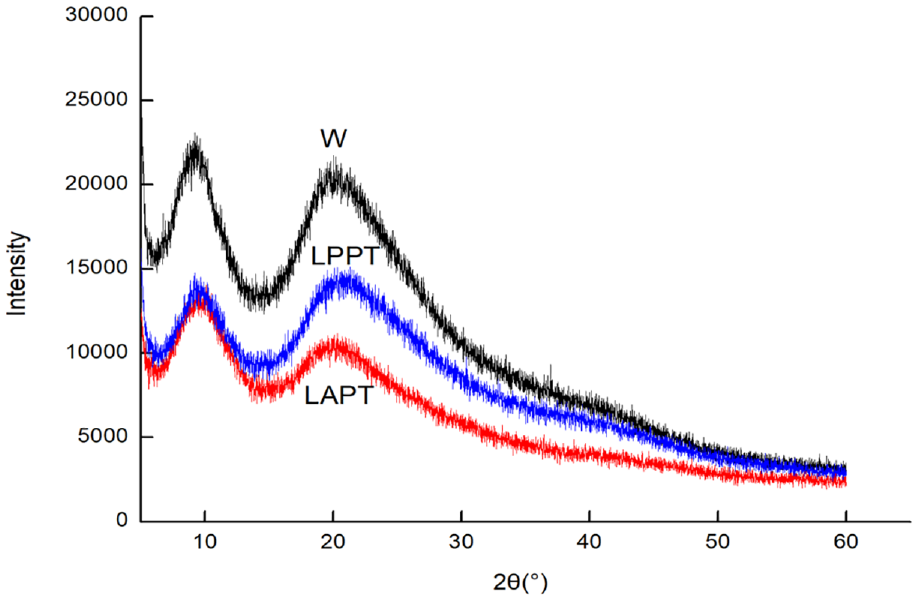
X-ray diffraction of modified wool fabrics.
As shown in Figure 3, there are two distinct peaks of wool at 9.27° and 20.2°. The peak at 9.27° is the diffraction peak formed by the synergy of α and β crystals, while the peak at 20.2° is the diffraction peak of β crystals.35,36 After liquid ammonia and protease treatment, the intensity of the peaks was still decreased, which means that the crystallinity of wool was changed. Furthermore, the intensity of the peaks is still decreased after polyurethane finishing. Since it is troublesome to calculate the crystallinity of natural macromolecules, the crystallinity index (CI) proposed by Segal et al. 37 was used in this article. The crystallinity index means the crystallinity degree of wool fibers, and was measured by the following empirical formula:
where CI is the crystallinity index, I9° is the maximum diffraction intensity at about 9°, and I14° is the minimum diffraction intensity at about 14°.
According to the above formula, the crystallinity index of W is 45.93%, LAPT is 45.85%, and that of LPPT is 43.21%. This indicated that the crystallinity of wool decreased after being treated with liquid ammonia and protease, due to the catalytic hydrolysis of protease to the peptide chain of wool. However, the crystallinity of LPPT was decreased furthermore due to the changes of molecular structure after combined finishing of liquid ammonia, protease, and polyurethane.
SEM Analysis
The images of the surface scales of wool fibers after different finishing are shown in Figure 5.

The SEM photos of wool fabrics: (a) W, (b) LAPT, and (c) LPPT.
Figure 5 shows that the surface scales of untreated wool are relatively thick, with large warping angle and clear grain. However, the scales are severely damaged and partly peeling off after liquid ammonia and protease treatment. Furthermore, the effects of polyurethane on the modified wool fabric are obvious, the surface scales and warping angles are covered with a film, and the warping angles are no longer prominent, and one can hardly see the scales on the severely damaged parts.
The isocyanate group (–NCO) of polyurethane is very active. In hot and humid conditions, it can react with water to form a group with –NH2, which can react with –NCO to form a polymer film with a urea-based group (–NHCONH), and then cover the surface of the wool. For another, the –NCO can react with hydrophilic groups, such as –OH, –NH2, and –SH, forming stronger chemical bonds between fiber molecules, which remarkably decreases the directional friction effect of wool, and the anti-felting property is improved effectively.
Effect of Polyurethane Finishing on the Performance of Modified Wool
The effects of polyurethane concentration on the properties of wool modified by liquid ammonia and protease are shown in Table 1.
Effects of polyurethane concentration on properties of modified wool fabric.
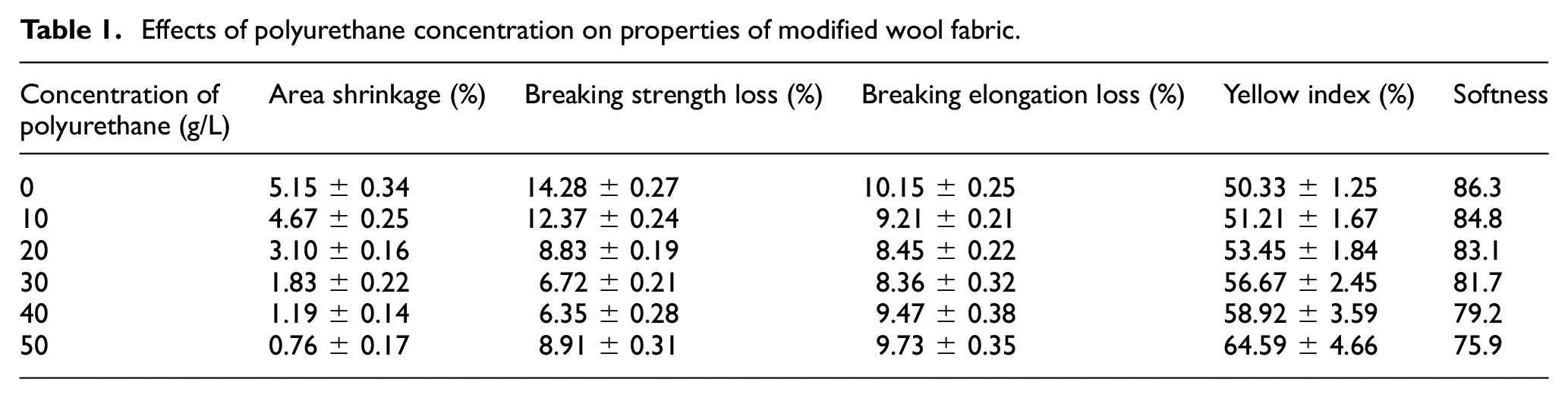
The felting shrinkage of the untreated wool is 24.34%, and the yellowing index is 57.57%. The softness of untreated wool is 81.5, while that of the wool treated with liquid ammonia and protease is 86.3.
Table 1 shows that the area shrinkages of wool gradually decrease with increased polyurethane concentration. The area shrinkage can reach 0.76% at a dosage of 50 g/L, but the yellowing index increases significantly. After the liquid ammonia and protease treatment, the cuticle of wool was damaged, and the scale no longer protruded from the surface as prominently as in untreated wool; some parts of surface scales even fell off. For the above reasons, the polyurethane was more conducive to deposition on the surface of the wool. Furthermore, more reactive groups were formed due to the disulfide bond breaking, which improved the cross-linking between –NCO and the reactive groups of the wool. All of this effectively prevented the directional friction effect of surface scales, so that an excellent anti-felting property was gained under a lower concentration of polyurethane. After polyurethane finishing, both breaking strength and elongation were increased, because of the deposition of polyurethane on the wool surface accompanying the reaction with its reactive groups to form new cross-linking, reducing the stress concentration point of the fabric during the stretching.
In order to study the mechanism of polyurethane finishing on the fabric modified by liquid ammonia and protease, weight loss, AS, breaking strength and elongation, and area shrinkage of wool were discussed, as shown in Table 2. The WPT means the wool only treated by protease.
Effect of polyurethane finishing on properties of modified wool fabric.
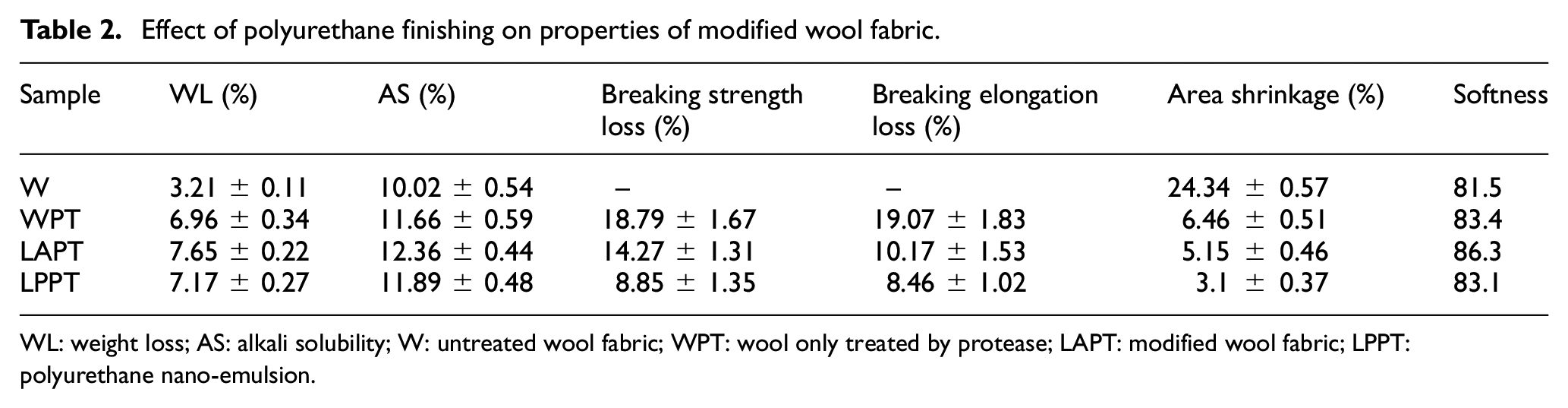
WL: weight loss; AS: alkali solubility; W: untreated wool fabric; WPT: wool only treated by protease; LAPT: modified wool fabric; LPPT: polyurethane nano-emulsion.
Catalytic hydrolysis of protease was mainly on the cortex and CMC of wool fiber; part of protein macromolecules are hydrolyzed into small soluble molecules or amino acids, diffusing from the keratinocyte and causing weight loss. 38 The weight loss and the area shrinkage of WPT are 6.96% and 6.46%, respectively; the loss of breaking strength is 18.79%. Nevertheless, the weight loss of LAPT was slightly increased while the loss of breaking strength and the area shrinkage were much lower than those of WPT. This suggested the destruction of surface scales and disulfide bonds by liquid ammonia, increasing the action points of protease on the wool fiber surface scales, and promoting the hydrolysis of protease on wool surface scales, with a serious damage on the surface scales. The weight loss of LPPT was lower than that of LAPT, but the loss of breaking strength and area shrinkage decreased significantly; in particular, the area shrinkage decreased to 3.1%, which remarkably improved the shrink resistance of wool, and achieved a machine washable effect.
The mechanism of polyurethane finishing on the wool modified by liquid ammonia and protease is as follows. The modification of wool fabric by liquid ammonia and protease induces the damage of surface scales, and accelerates the disulfide bond breaking to expose more hydrophilic reactive groups, such as –OH, –NH2, –SH, and –COOH. However, the active group of polyurethane is the isocyanate group (–NCO) with strong activity. On one hand, –NCO can interact with the hydrophilic group of wool to form intermolecular cross-linking. On the other hand, –NCO can involve self-cross-linking reaction to form a network of urea-based (–NHCONH) polymers, and then is deposited on the surface scale. This indicates that the combined effect of liquid ammonia, protease, and polyurethane not only decreases the loss of breaking strength but also improves the anti-felting property of wool through decreasing the directional friction effect of the wool.
Conclusion
The synergistic action of liquid ammonia, protease, and water-soluble polyurethane in the shrink-resistant finishing of worsted wool fabric was investigated. Due to the damage of liquid ammonia pretreatment on the wool surface scale and disulfide bonds, liquid ammonia canpromote the catalytic hydrolysis of protease on the surface scales and inhibit the diffusion of protease into the fiber. After wool was treated with liquid ammonia and protease, the surface scale and disulfide bond were seriously damaged, and the scale was no longer protruded from the surface as prominently as in untreated wool, which made the polyurethane more conducive to deposition on the surface of wool. Furthermore, after polyurethane finishing, the crystallinity of the wool decreased further, and the molecular structure of the wool changed. The surface scale and the gap between the scales were covered with a thin film after polyurethane finishing, and the severely damaged parts of the scale were completely covered by the film, which effectively reduced the directional friction effect of the fibers. The area shrinkage of the wool reached to 3.1% when the amount of polyurethane nano-emulsion was 20 g/L, while the fabric strength less damaged. The synergistic effect of liquid ammonia, protease, and polyurethane remarkably improved the shrink resistance of the wool, and achieved a machine washable effect. It can be seen from these positive results that the combined finishing of water-soluble polyurethane nano-emulsion and liquid ammonia and protease provides a new and potential path for the shrink-resistant finishing of wool worsted fabrics. In addition, it also provides guidance for developing more functional finishing methods.
Footnotes
Acknowledgements
The authors thank the International Joint Research Laboratory for Eco-Textile Technology at Jiangnan University for their support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Key R&D Program of China (2021YFC2104000) and Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX18_1832).