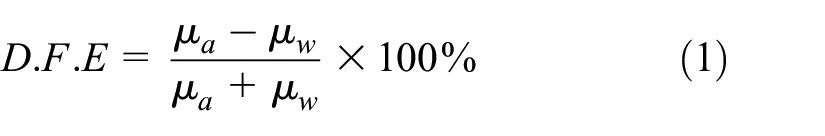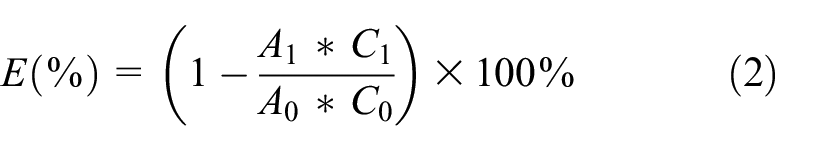Abstract
Superfine wool fiber has a soft and luxurious texture, excellent moisture absorption and is warmer and more comfortable than ordinary wool. Liquid ammonia, which has exceptional permeability, exerts a certain modification effect on the surface scales of wool in the finishing and processing of wool fabric, thereby enhancing its wear performance. However, limited research has been conducted on the impact of liquid ammonia on valuable wool fibers such as superfine wool and cashmere. In this article, superfine wool tops were treated with continuous liquid ammonia finishing equipment and the effects of liquid ammonia on the microstructure and surface morphology of superfine wool fiber were investigated. In addition, the changes in length, fineness, anti-felting, and dyeing properties of superfine wool fiber after liquid ammonia treatment were examined. The results demonstrated that the interstitial integrity of intercellular substance of superfine wool was compromised following treatment with liquid ammonia, which had a greater tendency to infiltrate cortical cells and induce alterations in wool structure and performance. Furthermore, the surface scales of superfine wool were damaged after treatment with liquid ammonia, leading to improved anti-felting and dyeing properties.
Introduction
Wool is an eco-friendly natural protein fiber that is both renewable and 100% biodegradable, capable of decomposing within a span of 3–4 months. 1 The growing demand for eco-friendly and sustainable development has resulted in the widespread utilization of wool fiber in textiles, especially those that prioritize high count, light weight and comfort, as well as those for sports and leisure activities. 2 The quality of wool is contingent upon the fineness of its fibers, and the finer the fibers, the better the spinning process is at producing high-count yarn. Therefore, the investigation into the structure and physical properties of superfine wool holds significant guiding implications for the advancement of processing technology and the development of innovative products.
The classification of Australian Merino wool fiber indicates that the diameter of superfine wool fibers is below 18.5 µm. The superfine wool fibers possess a silk-like luster, exceptional moisture absorption, remarkable elasticity, and thermal insulation. These outstanding characteristics, coupled with their scarcity, make them highly sought after by consumers. However, the unique scale structure of wool not only affects the spinning, weaving, dyeing, and finishing processes of superfine wool fiber but also significantly influences the properties of dyeing, shrink resistance, anti-pilling performance, and wettability. Hence, mitigating the impact of surface scale on fiber properties has become a focal issue in the wool textile industry in recent years.
The current methods for wool modification in environmental protection include the utilization of biological enzymes, plasma treatment, and ultrasonic techniques. The protease method, commonly used in biology, exhibits strong catalytic activity and enhances the anti-felting performance of wool. However, it primarily affects the cell membrane complex of wool fibers, resulting in a higher loss of weight in the fibers.3,4 The plasma method primarily acts on the surface of wool. After undergoing plasma treatment, the roughness of the wool surface is increased, resulting in improved apparent depth and dyeing concentration of dyed fabric, as well as enhanced color brightness of wool fibers.5–7 The ultrasonic wave possesses the characteristics of a strong and focused beam, which enables it to etch the surface of fibers. In addition, it also exerts a certain influence on the structure of wool fibers and enhances its dyeability. The application of ultrasonic treatment technology is primarily employed in wool cleaning and modification processes. 8 However, the application of plasma and ultrasonic treatment technology in industry is limited by equipment, making it difficult to popularize.
Liquid ammonia, as a non-aqueous medium, exhibits characteristics such as low molecular weight, reduced surface tension, and excellent permeability. Moreover, it shares numerous physical properties with water. 9 The utilization of liquid ammonia has proved to be effective in the modification and mercerization processes of cellulose fibers, thereby enhancing their tactile smoothness, sheen, and crease resistance.10–12 The application of liquid ammonia in wool was primarily concentrated in the 1980s. During this period, foreign scholars conducted preliminary theoretical research on the surface characteristics, thermal properties, and dyeing properties of wool by impregnating it with a small batch liquid ammonia pump in laboratory settings.13–15 However, this treatment method proved to be inefficient and prone to ammonia leakage, resulting in insufficient attention at that time. Subsequently, following the successful application of liquid ammonia finishing technology in cotton fabrics, researchers began to investigate the impact of liquid ammonia on wool fibers.16–18 However, to date, there is no research on the impact of liquid ammonia on superfine wool fibers.
The objective of this study was to investigate the impact of liquid ammonia on the physical and chemical properties of superfine wool fibers by a continuous liquid ammonia finishing machine. In this study, wool tops with diameters of 19.5 µm (W-19.5 µm), 16.6 µm (W-16.6 µm), and 15.5 µm (W-15.5 µm) were subjected to treatment using a continuous liquid ammonia finishing machine, respectively. After liquid ammonia treatment, a comprehensive investigation was conducted on the microstructure, surface morphology, physical properties, anti-felting characteristics, and dyeing properties of three different diameters: 19.5 µm (LAT-19.5 µm), 16.6 µm (LAT-16.6 µm), and 15.5 µm (LAT-15.5 µm). The research establishes a solid foundation for the development and processing of wool products, particularly providing an essential theoretical basis for the production of superfine wool fibers.
Experimental
Materials
Australian wool tops with diameters of 19.5, 16.6, and 15.5 µm were provided by Ruyi limited, China.
Metal-complex dyestuff Lanaset Red G (C.I. 18800, λmax = 497 nm) was provided by Huntsman Company. The chemical structure is shown in Figure 1.
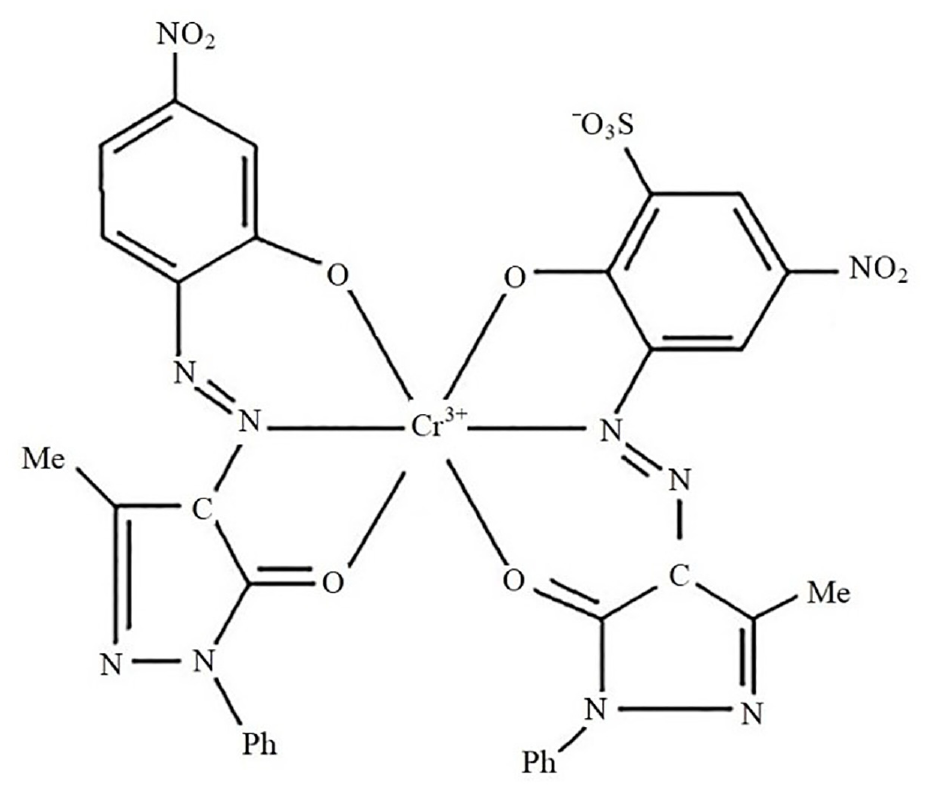
Chemical structure of Lanaset Red G. 19
Sample Pretreatment
In order to enhance the cleanliness of the fiber, 120 kg of wool tops were subjected to a 20-min rinsing process at 75°C in a high temperature–pressure package dyeing machine (Thies, Germany) with a liquor ratio of 1:10. The solution employed was SP-2 (Feijian, China) at a concentration of 1% based on fabric weight (o.w.f), and it was adjusted to a pH range of 5–6 using a sodium acetate/acetic acid buffer solution.
Liquid Ammonia Finishing
A continuous liquid ammonia finishing machine (Kyoto Machinery, Japan) was utilized for the liquid ammonia treatment of wool. The machine is equipped with an automatic ammonia recovery device (Maekawa Seisakusho, Japan) to mitigate ammonia emissions and minimize environmental pollution. The schematic diagram illustrating the structure and operational principles of a continuous liquid ammonia finishing machine is presented in Figure 2.
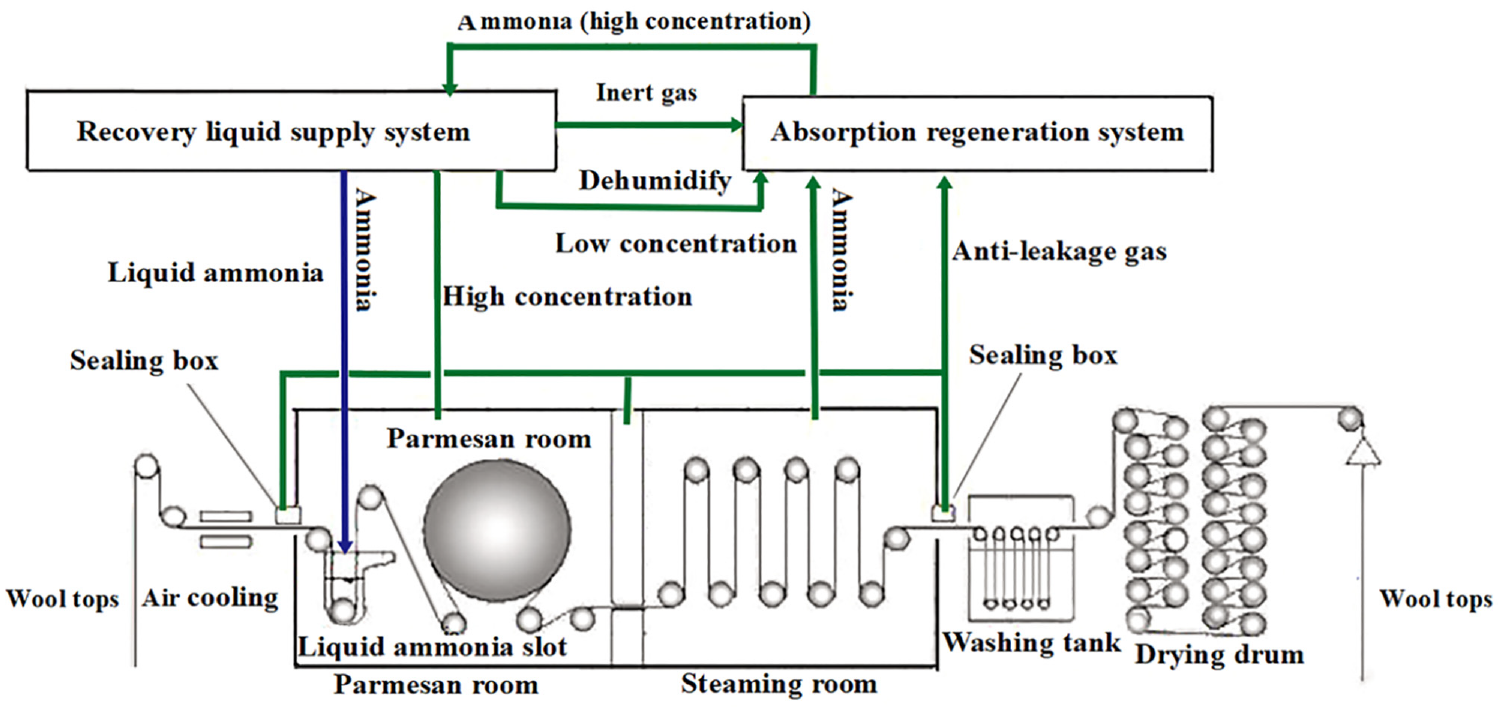
Schematic diagram of a continuous liquid ammonia finishing machine.
The main processes involved in liquid ammonia finishing include prebaking, air cooling, padding with liquid ammonia, blanket finishing, steaming, washing, and drying. The wool breakage under high tension was minimized by implementing a flexible finishing process at a low speed of 15 m/min. The wool tops were immersed in pure liquid ammonia at a temperature of −33.4°C for 30 s, while being subjected to a roller pressure and tension of 0.16 MPa and 0.18 kg (cm−2), respectively. The ammonia in wool tops was removed using blanket finishing at a cylinder pressure of 0.6 MPa and a blanket tension of 0.29 MPa. Subsequently, the elimination of residual ammonia was further facilitated by steaming and washing processes. The washing tank was modified by incorporating a set of rolls and enhancing the roller pressure, in response to the higher volume of wool tops compared to that of wool fabrics. The pressures of the first to the fourth rollers are 5–6 MPa, 6–7 MPa, 8–9 MPa, and 9–10 MPa, respectively. The second washing tank is an acid neutralization tank with a pH of 5.5. The schematic diagram of reformed washing tank is shown in Figure 3.
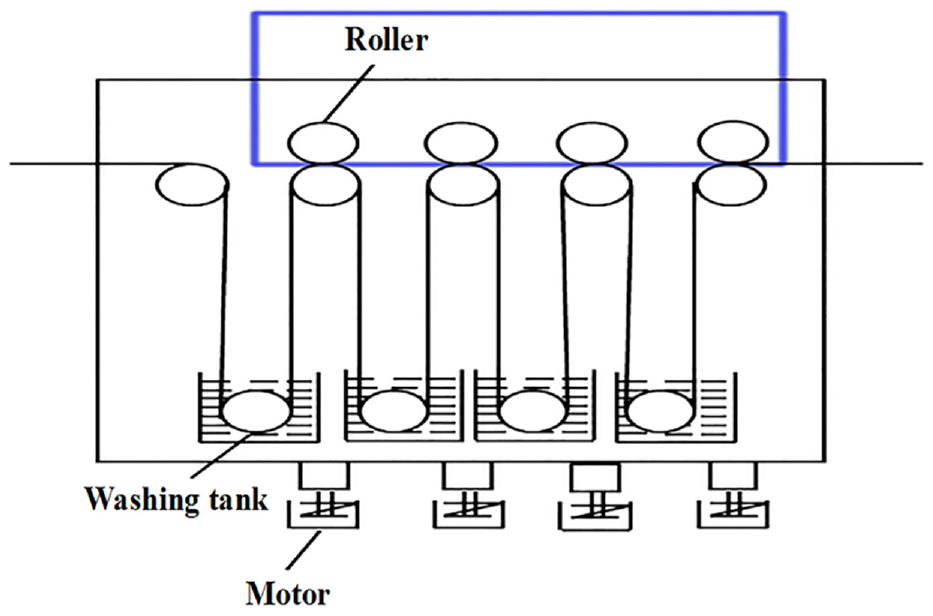
Schematic diagram of reformed washing tank.
Dyeing of Superfine Wool Fiber
The wool tops weighing 120 kg were dyed in a high temperature–pressure package dyeing machine (Thies, Germany) with a liquor ratio of 1:10. The Lanaset Red G was utilized for dyeing, with a concentration of 3% (o.w.f). The pH value of the dyeing solution was adjusted to 5 using an acetic acid–sodium acetate buffer solution. The initial dyeing temperature was set at 40°C and then it was gradually increased to 85°C at a rate of 1.0°C/min. The dyeing curve of Lanaset Red G is shown in Figure 4.
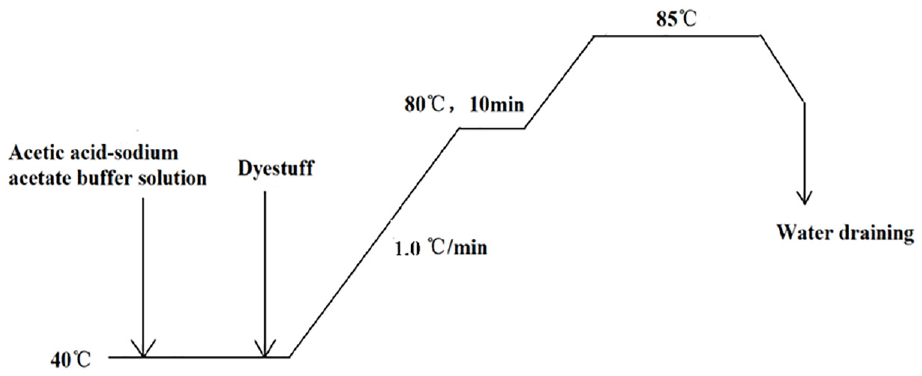
Dyeing curve of Lanaset Red G at 85°C.
Microstructure Analysis
The microstructure of wool was examined using a transmission electron microscope (TEM). The wool fibers were washed with acetone and ethanol in a Soxhlet extractor to remove oils, and then immersed in a 3% (o.w.f) OsO4 solution for 120 h at the chemical hood to completely dye the wool fibers black and enhance the contrast of the wool fibers in TEM imaging. Subsequently, extensive rinsing with distilled water was carried out iteratively to remove any potential electronic staining. Afterward, wool fibers were longitudinally placed within the directional embedding capsule and subsequently immersed in epoxy resin for injection. The resin was subjected to a temperature of 30°C for 6 h, followed by 60°C for 12 h and finally at 90°C for another 12 h to ensure complete solidification and polymerization. Subsequently, the sample was equilibrated in a desiccator for 24 h in preparation for slicing. The embedded fibers were sliced into ultrathin sections with a thickness of 70 nm using an ultrathin microtome EM UC7 (Leica, Germany). Finally, the fiber cross-section was observed and the microstructure of the wool was analyzed using an HT 7800 TEM.
Scanning Electron Microscope
The surface morphology of superfine wool fibers was examined using a Regulus 8100 scanning electron microscope (SEM) (Hitachi, Japan) at a voltage of 5 kV and a magnification of 5000 times.
Cross-Section Test
The Y172 fiber slicer (Darong, Wenzhou, China) was utilized to produce wool fiber cross-sectional slices with a specific thickness range of 10–20 µm. Subsequently, the cross-sectional slices of wool were examined using a DMM-300C optical microscope (Caikang, Shanghai, China) with a magnification of 1000 times.
Allwörden Reaction Test
The bubbles formed on the surface of the superfine wool fiber, after being exposed to saturated bromic water for 2 min, were observed using a DMM-300C optical microscope (Caikon, Shanghai) at a magnification of 400 times.
Fiber Fineness and Curvature Test
The average diameter and curvature were measured by a laser scanning fiber diameter analyzer (AWTA, Australia) following the IWTO-12-00 test method. Each sample underwent 2000 measurements, and the results were averaged.
Fiber Length Test
The fiber length was determined in accordance with the IWTO-17-04 method, using a minimum sample weight 2 g. The length variation and its discrete distribution were measured by Hauteur (H, mm), coefficient of variation (CVH, %), and undercoat content (≤30 mm) (UH, %) respectively.
Surface Friction Properties Test
The Roeder method 20 was used to measure the coefficient of friction between the fiber and metal roller using an XCF-1A fiber friction coefficient tester (Xinxian, Shanghai, China), with a tension clamp load of 200 × 10−3 cN and a speed of 30 rap/min. The average value was obtained by randomly testing 30 fibers of each type, and the mathematical expression for the directional friction effect (D.F.E) is as follows:
where μa represents the coefficient of friction against the scale direction, and μw represents the coefficient of friction along the scale direction.
Dye-Uptake
The absorbance of the dye solution before and after dyeing at its maximum absorption wavelength (497 nm) was measured using a UV1800 ultraviolet spectrophotometer (Shimadzu, Japan). The formula for calculating the dye-uptake efficiency (E, %) is expressed as follows:
where A0 represents the absorbance of the initial dye solution and C0 denotes its dilution factor, while A1 corresponds to the absorbance of the residual solution after dyeing, and C1 signifies its dilution factor.
Color Depth
The K/S value was determined using a Datacolor800 spectrophotometer. Each group underwent 10 repeated tests, and the average value was obtained. The Kubelka–Munk equation 21 in formula (3) was employed to calculate the K/S value:
where K is the adsorption coefficient, S is the scattering coefficient, and R is the reflectivity of the maximum wave strength.
Color Fastness
The rubbing fastness was tested by standard of ISO 105-X12-2016 (E). The potting fastness was tested by standard of ISO 105-E09-2010; samples were boiled in boiling water for 30 min.
Bundle Strength Test
The bundle strength of wool was measured using the Sirolan Tensor bundle strength tester (Sirolan, Australia) in accordance with the IWTO-82 (E) standard method.
Results and Discussion
Microstructure Analysis
TEM was used to observe the microstructure of wool. In this study, the research focused on investigating the TEM images of 19.5 µm wool fibers before and after liquid ammonia treatment, and the results are shown in Figure 5.
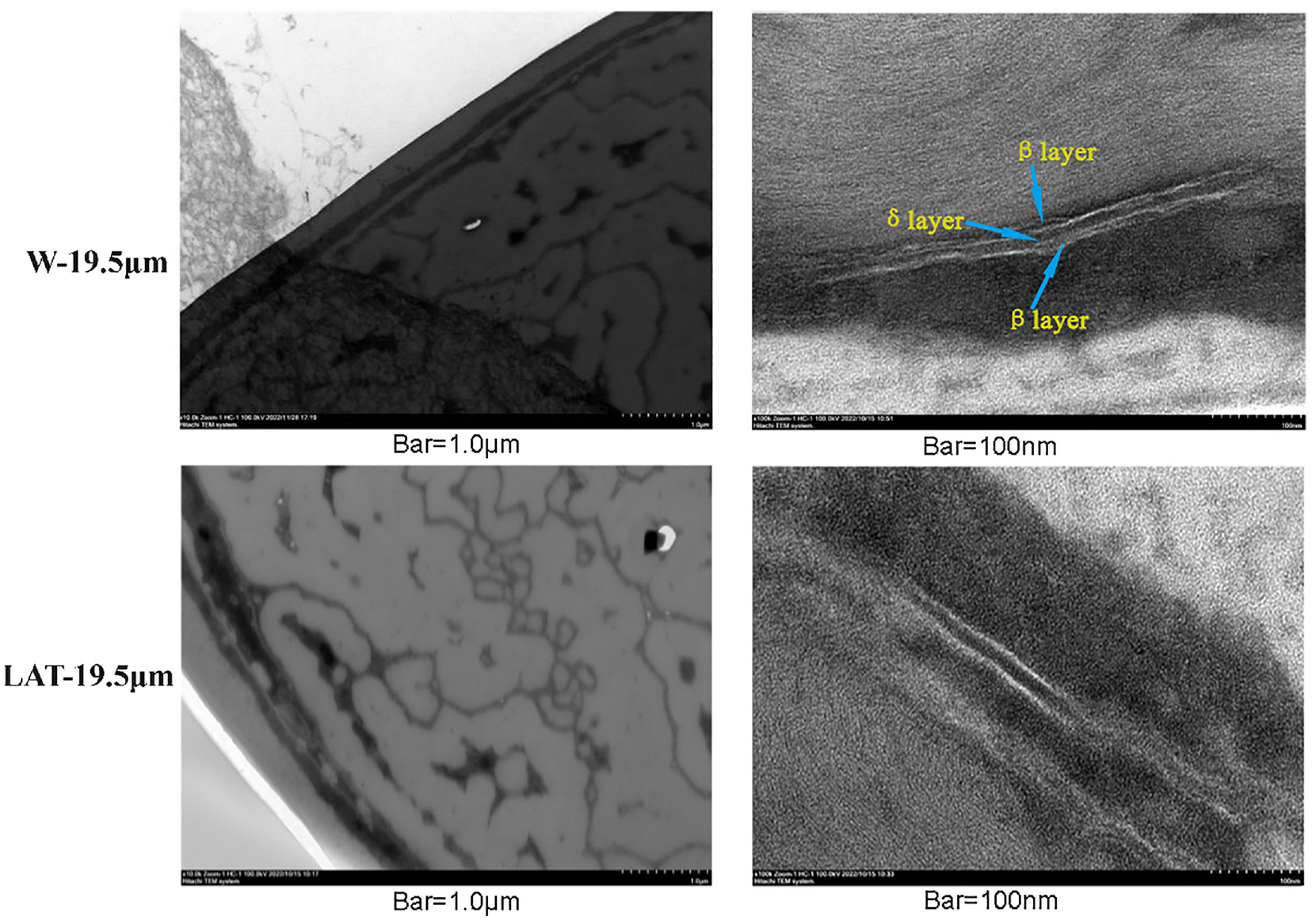
TEM images of W-19.5 µm and LAT-19.5 µm.
The TEM analysis of W-19.5 μm revealed the presence of cuticle, cortex, and cell membrane complex in the wool structure. The cell membrane complex originates from both the cell membrane and intercellular substance, with the latter being the only continuous tissue throughout the wool. 22 The intercellular substance exhibits a characteristic “sandwich” structure, in which a β layer of lipid bilayer structure with low sulfur content is distributed on both sides of the cellular interstitium, while a δ layer composed of non-keratinized proteins with high sulfur content is distributed in the middle. The δ layer represents a relatively vulnerable component of the interstitium, filling the space between cells and facilitating the transmission of cortical cell deformation.23,24
After liquid ammonia treatment, the wool structure still retained the cuticle, cortex, and cell membrane complex. However, the intercellular substance between cuticle and cortex was disrupted, resulting in a discontinuous tissue structure. Individual globular proteins were observed in the intercellular matrix, potentially resulting from disruption of the intercellular substance, leading to the release of these proteins. 25 Due to the rupture of the intercellular substance, liquid ammonia can easily act on the adjacent layer of endocuticle and subsequently penetrate the exocuticle from inside to out. In addition, liquid ammonia can quickly penetrate into the fiber through the intercellular substance and impact the structure and properties of wool.
Surface Morphology
SEM Analysis
SEM images of wool with three different diameters before and after liquid ammonia treatment are shown in Figure 6.
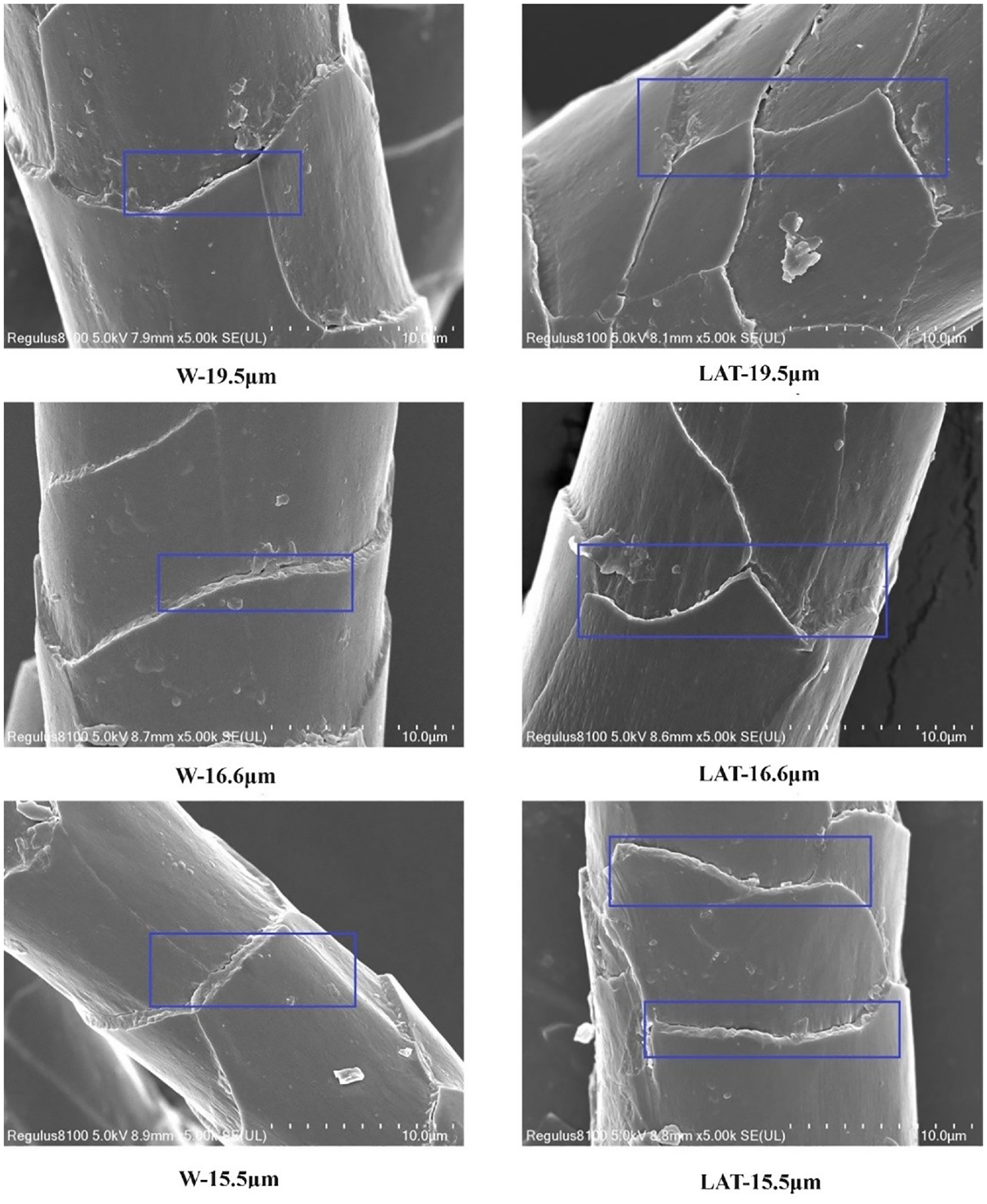
SEM (×5000) images of W and LAT with three different diameters.
Figure 6 shows the unique scale structure of wool with irregular scales. After liquid ammonia treatment, the surface scales of wool fibers exhibited varying degrees of disruption, accompanied by the presence of fragments and flakes on the surface. In addition, the cuticle edges underwent flattening and thinning, while the spacing between the cuticle scales decreased. The LAT-19.5 µm fiber exhibited more severe damage compared to the other two fibers. The damage to the surface scale was caused by the disruptive effect of liquid ammonia on the cuticle scale. In addition, the pressure exerted by rollers during liquid ammonia treatment may further fracture the surface scales.
Cross-Section Analysis
After liquid ammonia treatment, the cross-sections of wool with three different diameters are shown in Figure 7.

Cross-section (×1000) images of W and LAT with three different diameters.
After liquid ammonia treatment, the cross-section of wool exhibited enhanced regularity and compactness, particularly for LAT-15.5 µm, which was almost round. Liquid ammonia, being a non-aqueous medium, possesses inherent characteristics such as low surface tension and high permeability. Consequently, the fibers swelled and the orientation increased after liquid ammonia treatment. The cross-section became more uniform in shape, especially for LAT-15.5 µm, where it tended toward a perfectly circular form.
Allwörden Reaction Analysis
The unique scale structure of wool fibers makes them susceptible to the well-known Allwörden reaction with saturated bromic water. 26 The results of wool with different diameters are illustrated in Figure 8.

Images of the Allwörden reaction of W and LAT with three different diameters.
The results depicted in Figure 8 demonstrate a substantial coverage of bubbles across the entire surface of W, while a noticeable reduction in bubble presence was observed for LAT, particularly for LAT-19.5 µm where a significant decrease occurred. The number of scales per unit area increased for superfine wool fibers, while the volume of bubbles decreased, particularly with regards to LAT-15.5 µm where the bubbles were almost covered on the surface of hair shaft. After liquid ammonia treatment, the surface scales of wool exhibited partial disruption, resulting in a reduction in the number of bubbles formed with bromine water. However, the impact of liquid ammonia on the surface scales of superfine wool fibers was less than that of W-19.5 µm; therefore, there were fewer changes in bubble formation on the surface of superfine wool fiber after treatment with liquid ammonia.
The Fineness of Wool Fiber
The fineness of wool fiber is a crucial physical parameter for assessing the value of wool, as it directly influences the quality of yarn and the fabric’s wearability.27–29 The state of fineness distribution is quantified by the coefficient of variation, as illustrated in Figure 9.
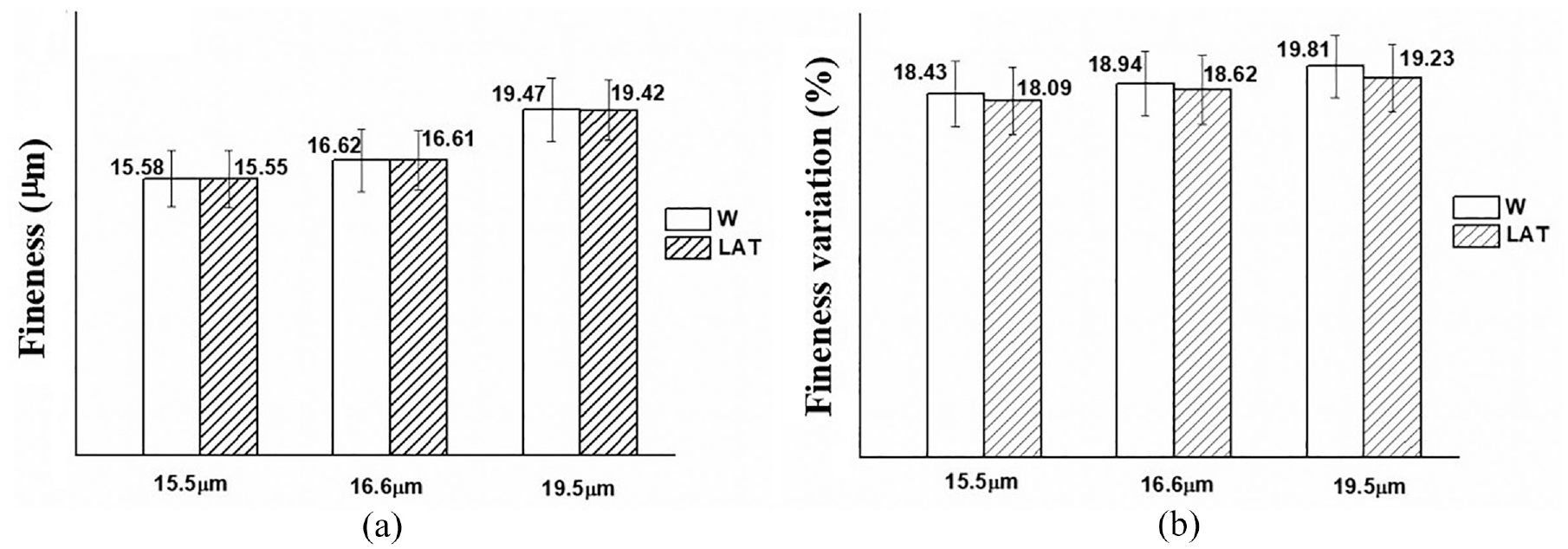
Fineness (a) and coefficient of variation (b) of W and LAT with three different diameters.
The impact of liquid ammonia on the fineness of wool is minimal; however, it has a significant effect on the coefficient of variation. Figure 9(b) illustrates that the coefficient of variation for superfine wool fibers with a diameter of 15.5 µm decreased by 2.4% after being treated with liquid ammonia. Due to its strong permeability, liquid ammonia can easily penetrate into the fiber and induce swelling, resulting in a more uniform distribution of fiber diameter and reduced coefficient of variation. This facilitates the reduction of spinning difficulties associated with superfine wool fibers and enhances spinning efficiency.
The Curvature of Wool Fiber
The bilateral structure commonly observed in the wool cortex is the primary factor contributing to fiber curvature. 30 The curl of wool fibers can be classified into three types: weak curl (coarse hair), normal curl (fine hair), and strong curl (fine and ventral hair) Figure 10. The curvature of the fibers is closely associated with the elasticity and recovery properties of wool, which significantly impact the spinning process and product characteristics.31,32
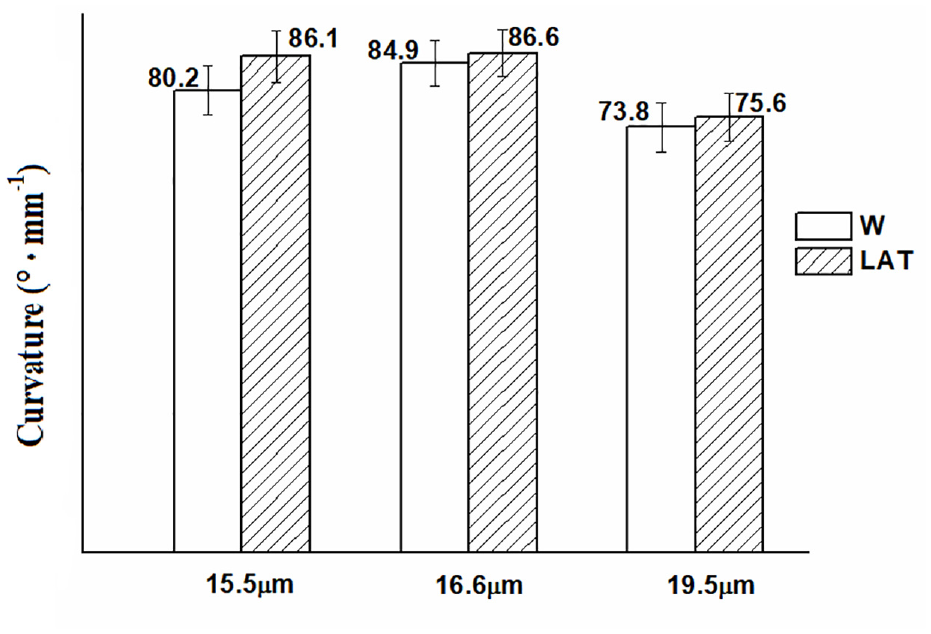
Curvature of W and LAT with three different diameters.
The curl of wool is unique and incomparable to other textile fibers, exerting a significant impact on the textile finishing. After liquid ammonia treatment, the curvature of wool was improved, especially for superfine wool fibers. This can be attributed to the disruption of the scale structure on the surface of wool cause by liquid ammonia treatment, alterations in the macromolecule structure of the wool keratin, and an increase in the α-helix content. These factors collectively result in the enlargement of non-uniform contraction of the orticortical and paracortical cells on both sides of the wool fiber, rendering it more susceptible to curling and increased bending deformation.
The Length of Wool Fiber
The length of wool fibers is also crucial for spinning quality. The wool fibers with longer lengths exhibit better inter-fiber adhesion, thereby reducing susceptibility to slippage and fracturing when subjected to external forces, consequently enhancing the strength and uniformity of the yarn. The impact of fiber length on yarn quality ranks second only to fiber fineness.33,34 The length variation was represented by Hauteur (H, mm), while the discrete distribution was represented by undercoat content (≤30 mm) (UH, %). 35
Figure 11 shows that the length of superfine wool fibers was lower than that of the 19.5 µm fibers. After liquid ammonia treatment, the length of the 19.5 µm wool fiber decreased by 14.4%, while the 16.6 µm superfine wool fiber experienced a reduction of 6.3%. Furthermore, the length of the 15.5 µm fibers was reduced from 68.8 to 60 mm, representing a decrease of 12.8%. This was because the degradation of liquid ammonia on the surface scale of wool fiber enhanced the susceptibility of the wool surface, making it prone to breakage under external forces and resulting in a decrease in strength and an increase in undercoat content. Therefore, the influence of length and fineness should be equally emphasized in the spinning process for superfine wool fibers. The coefficient of variation improved after treatment with liquid ammonia, and the increase in the 19.5 µm wool fiber was greater than that of the superfine wool fiber.
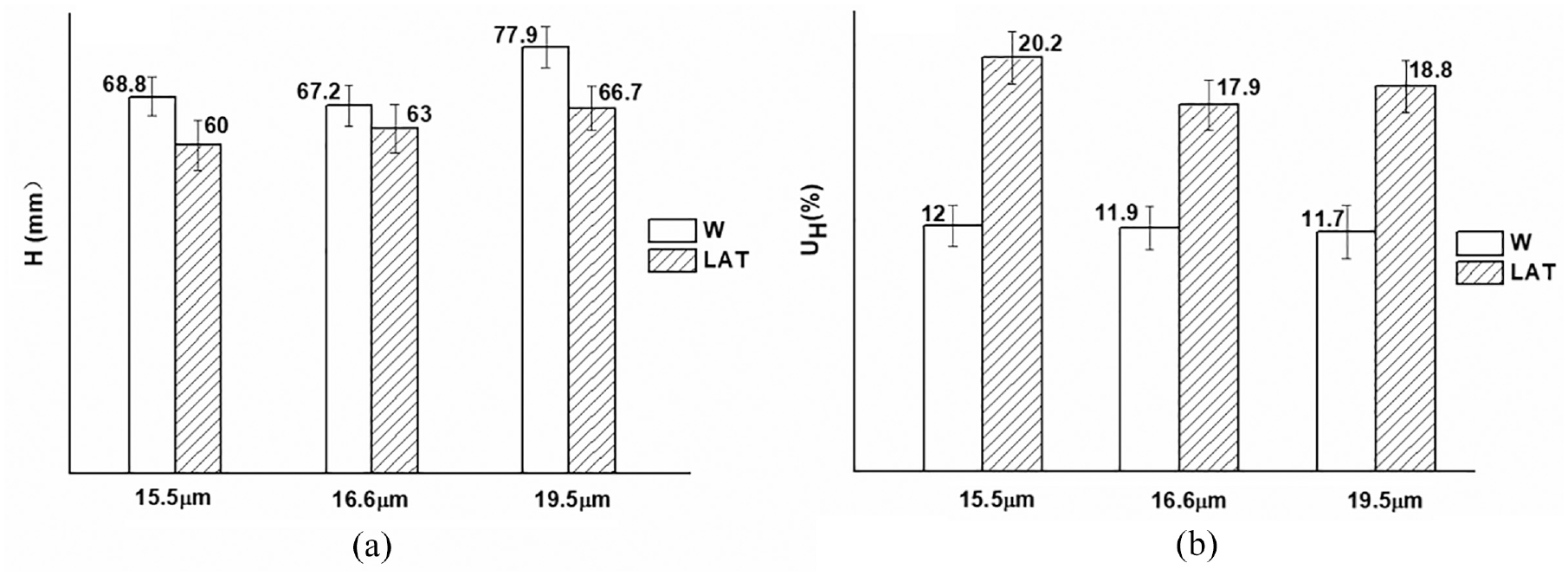
Length variation and its discrete distribution of wool fibers: (a) H (mm) and (b) UH (%) (≤30 mm).
The strength and evenness of yarn are significantly affected by the content of undercoat fibers (≤30 mm). The undercoat contents (≤30 mm) of superfine wool fibers were higher than those of W-19.5 µm, and the content increased significantly after liquid ammonia treatment. Therefore, in order to mitigate the impact of short wool on yarn quality, it is recommended to employ a spinning process for superfine wool fiber characterized by low rotational speed, small roller gauge, and low drafting.
Surface Friction Property
The felting issue should be taken into consideration during processing due to the directional friction effect of wool surface scales. Therefore, study of the surface friction properties of wool fibers is highly significant for enhancing processing technology and improving abrasion resistance and anti-felting properties.36–38 The dynamic friction effect (D.F.Ed) and the static friction effect (D.F.Es) of wool fibers are illustrated in Figure 12.
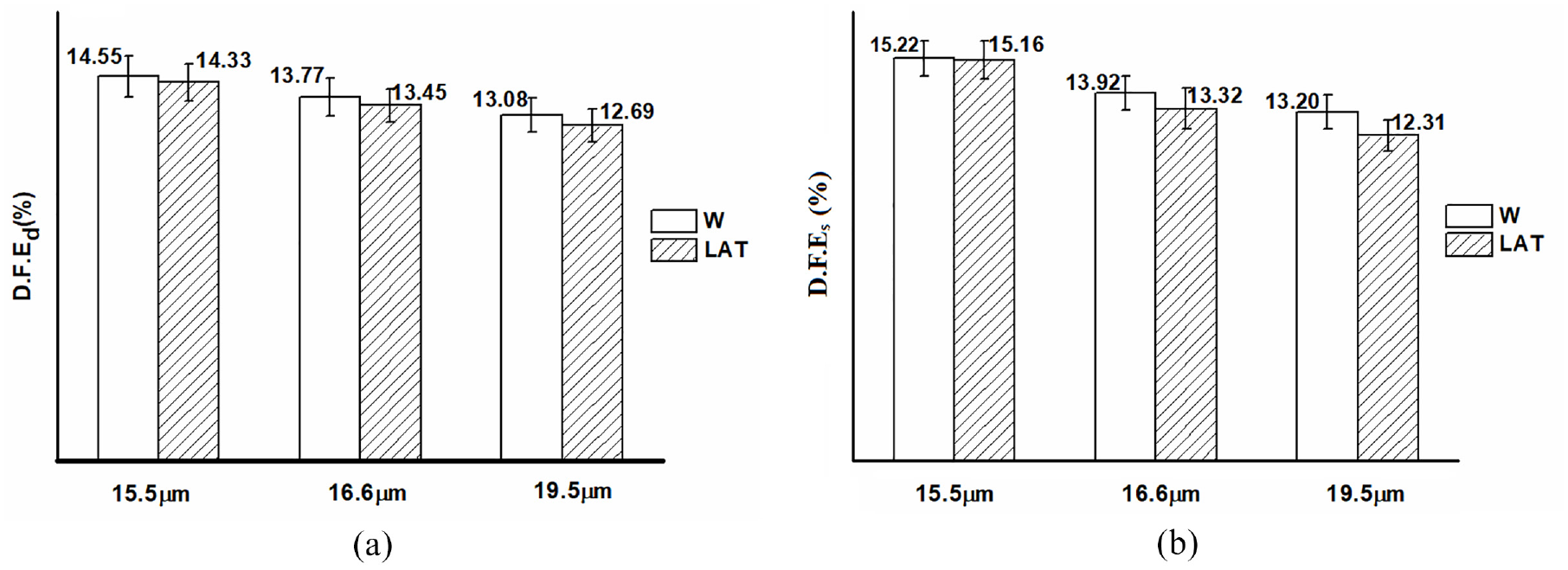
The (a) D.F.Ed and (b) D.F.Es of wool fibers.
The static directional friction effect of W exhibited a higher magnitude compared to its dynamic counterpart. Moreover, both the dynamic and static directional friction effects of superfine wool fiber were higher than those of W-19.5 µm. After liquid ammonia treatment, the D.F.E of wool was decreased. The findings indicate that liquid ammonia can improve the anti-felting performance of wool fibers. However, for superfine wool fibers, the improvement in anti-felting performance was less pronounced compared to LAT-19.5 µm due to the small impact of liquid ammonia.
The dynamic and static friction coefficients of wool fibers along and against the direction of scales are investigated in Figures 13 and 14.
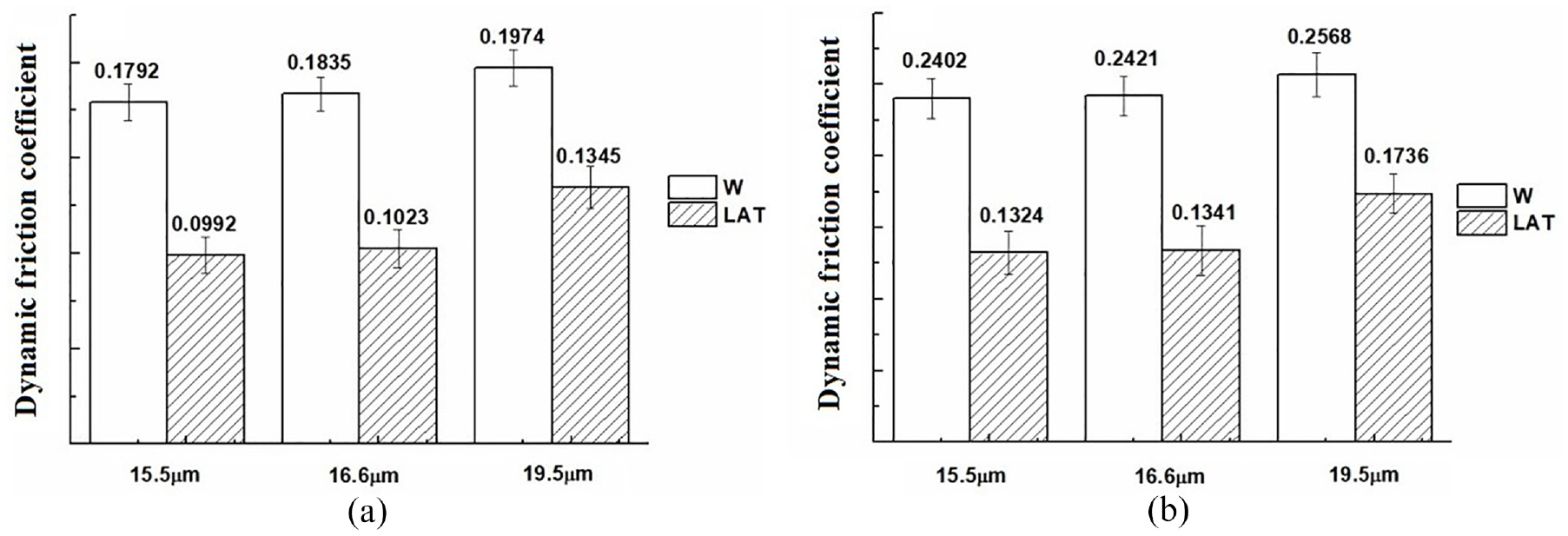
Dynamic friction coefficient (a) µw and (b) µa of wool fibers.
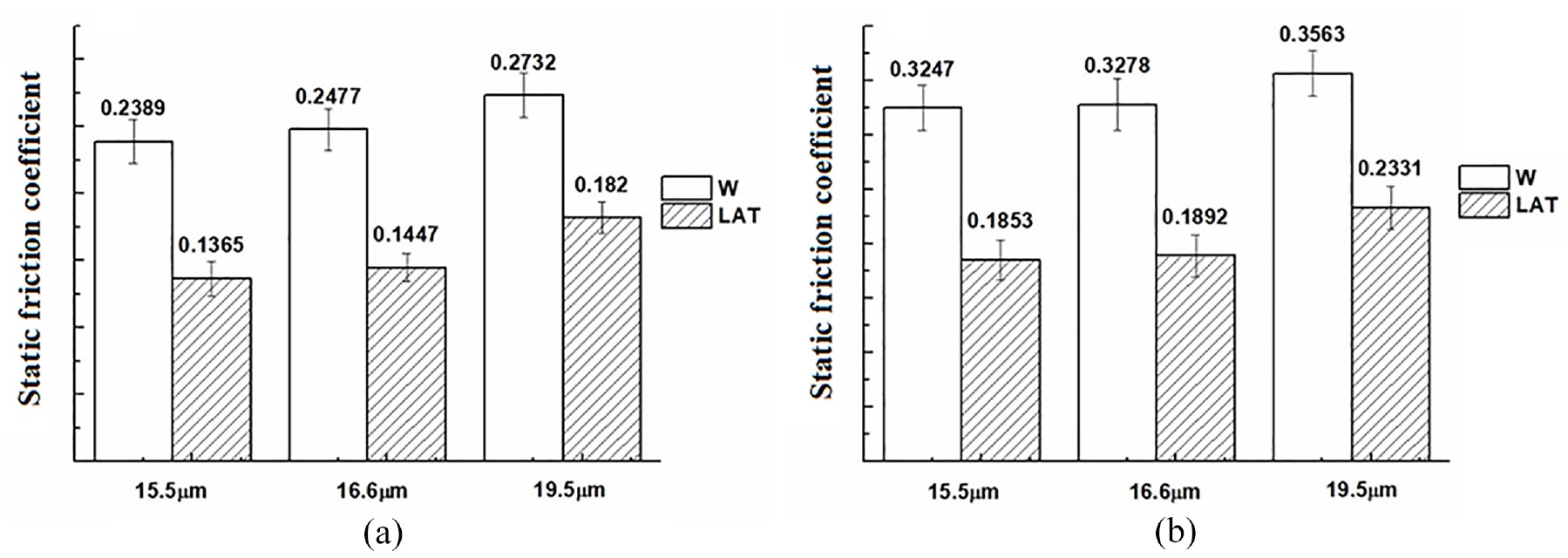
Static friction coefficient (a) µw and (b) µa of wool fibers.
The static friction coefficient of untreated wool is higher than the dynamic friction coefficient, regardless of whether it is against or along the direction of scales. Moreover, µa is higher than µw, which also contributes significantly to the directional friction effect of wool fibers. After liquid ammonia treatment, both the static and dynamic friction coefficients exhibited a decrease, with a significant reduction in µa. The frictional property of wool was attributed by King 39 to the greater frictional force exerted during motion form tip to root compared to that during motion from root to tip. After liquid ammonia treatment, the µa exhibited a greater decreased compared to that of µw, thereby providing further evidence for the enhanced shrink resistance of wool.
Dyeing Properties
Considering the detrimental impact of liquid ammonia on the surface scales and intercellular substance of wool, an investigation was conducted to examine the dyeing properties of wool at a temperature of 85°C. The dyeing exhaustion curve and K/S curve can be observed in Figures 15 and 16.
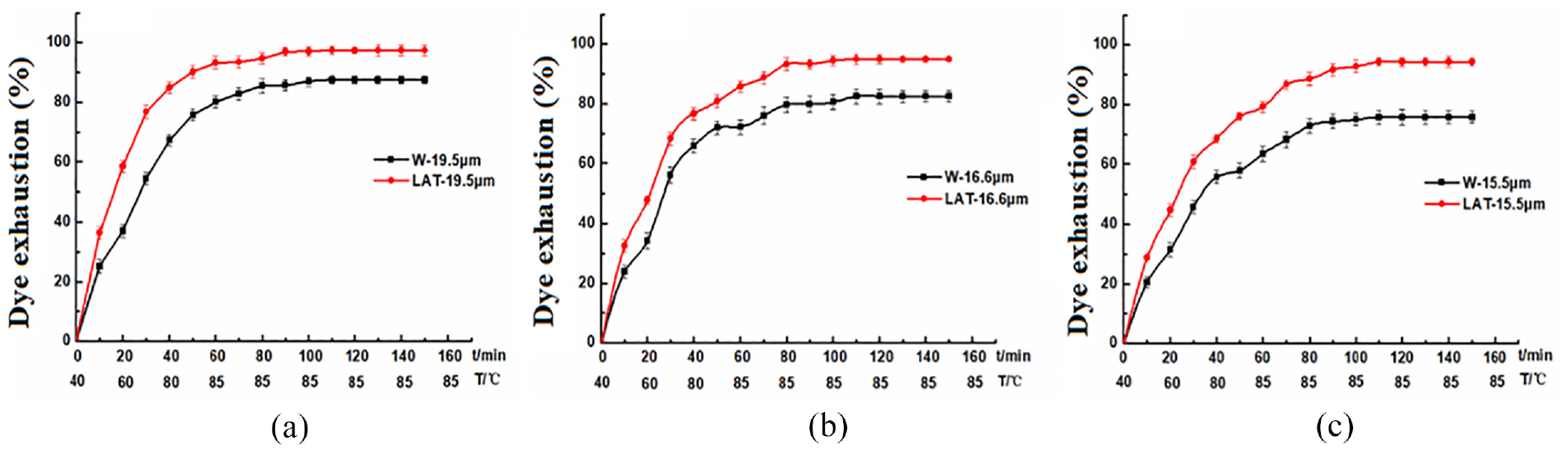
Dyeing exhaustion of (a) 19.5 µm, (b) 16.6 µm, and (c) 15.5 µm wool dyed at 85°C.
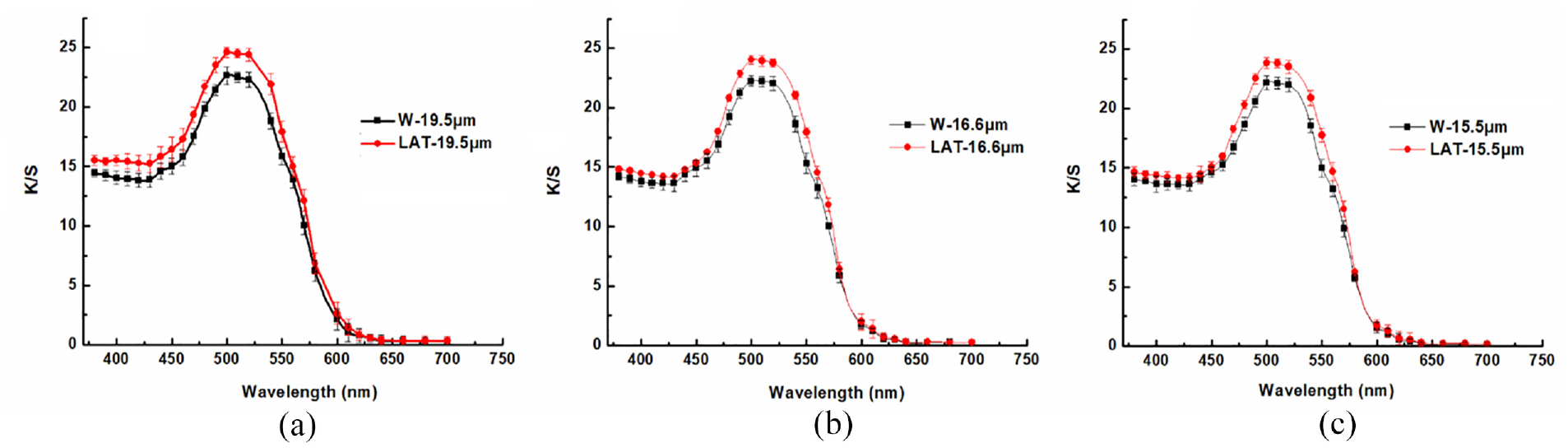
K/S curves of (a) 19.5 µm, (b) 16.6 µm, and (c) 15.5 µm wool dyed at 85°C.
The results presented in Figures 15 and 16 demonstrate that the application of liquid ammonia on wool leads to a significantly enhanced dye exhaustion and K/S value, as compared to untreated wool. The damage effect of liquid ammonia treatment on the wool surface scales reduces barrier properties, facilitating the diffusion of dye molecules into the fiber. Simultaneously, liquid ammonia affects the structure of the hydrophobic lipid layer on scale surfaces, exposing more hydrophilic groups and improving the affinity between dye and wool fiber. Moreover, the intercellular substance continuity is disrupted after liquid ammonia treatment, promoting rapid movement of dye molecules within the cell membrane complex and facilitating their penetration into the interior of fibers. Consequently, this enhances both the dyeing rate and the exhaustion rate. After liquid ammonia treatment, the dyeing exhaustion and K/S values of LAT-19.5 μm were found to be higher compared to those of LAT-16.6 μm and LAT-15.5 μm. The dyeing equilibrium time for LAT-19.5 μm at 85°C was achieved after a holding period of 60 min, while for LAT-16.6 μm and LAT-15.5 μm, it took 80 min.
The dyeing properties of wool, including K/S value, dyeing fastness, and bundle strength were investigated after being dyed at 85°C for 60 min. A comparative analysis was conducted between the treated and untreated wool. The results are presented in Table 1.
Dyeing properties of different wool fibers dyed at 85°C for 60 min.
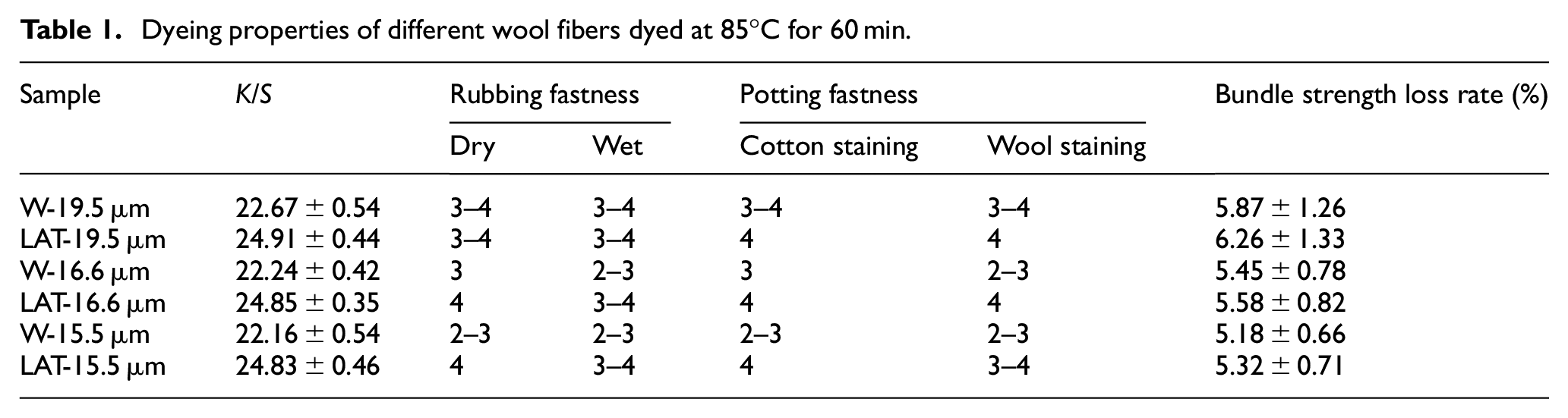
Table 1 shows that the dyeing properties of wool can be effectively improved after liquid ammonia treatment, with higher K/S value and dyeing fastness compared to untreated wool. However, the fiber strength slightly decreases due to the breakage of surface scale caused by the liquid ammonia treatment. The superfine wool fibers also exhibit excellent dyeing properties, particularly with regard to less strength loss compared to LAT-19.5 μm.
Conclusion
The work provided an efficient and sustainable method for superfine wool processing in the textile industry, wherein liquid ammonia is first employed in batch production of wool tops. After liquid ammonia treatment, the surface scales and the intercellular substance between the cuticle and cortex were disrupted, thereby facilitating enhanced penetration of liquid ammonia into the interior of the fiber. Consequently, this led to fiber swelling and a more uniform and compact cross-section morphology of superfine wool fibers. Seen from the physical indicators, it was found that liquid ammonia had little impact on the fineness of superfine wool. However, it significantly affected the length, leading to a reduction in length and an increase in undercoat content (≤30 mm). Therefore, to achieve optimal spinning quality of superfine wool fibers, it is advisable to employ flexible spinning technology, such as utilizing low speed and low drafting techniques. The anti-felting performance and dyeing properties of the superfine wool fiber were improved, and the dyeing temperature could be reduced to 85°C
The work not only provides a sustainable dyeing and finishing method for superfine wool but also establishes a foundation for the combination of liquid ammonia with other finishing techniques. Moreover, it also provides a guideline for the application of liquid ammonia on the other protein fibers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Key R&D Program of China (2021YFC2104000) and Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX18_1832). The authors thank for the support from International Joint Research Laboratory for Eco-Textile Technology at Jiangnan University.