Abstract
In the previous work by the same authors, optimisation of sequential application of acid protease and transglutaminase was carried out to achieve Woolmark specification AW-1 for the machine washable care claim, that is, less than 3% total shrinkage. As a result of optimisation, the total warp shrinkage reduced to 2.64% with only 2.82% loss in warp tensile strength. In this work, simultaneous application of acid protease and transglutaminase was explored and optimised using Box–Behnken design of experiment. The optimisation was carried out using two criteria. In the first criterion, the model was used to predict treatment parameters for minimum total warp shrinkage with minimum loss in warp tensile strength. Taking these predicted treatment parameters, wool was treated with acid protease (1%, owf) and transglutaminase (2.8%, owf) at pH ∼5.3 and 50°C for 60 min. After treatment, the total warp shrinkage reduced to only 4.32% with 4.65% loss in warp tensile strength. This was not only higher than the optimised sequential application of both the enzymes but also failed to meet the Woolmark specification for machine washable care claim. So, the second criterion was used in which the prediction was taken for treatment parameters to achieve the minimum total warp shrinkage without any constraints on loss in warp strength. Again, taking these predicted treatment parameters, wool was treated with acid protease (1.3%, owf), transglutaminase (1.6%, owf), pH ∼4.7 at 55°C for 85 min. Under these treatment conditions, the total warp shrinkage reduced to 2.89% with 10.1% loss in warp strength. Furthermore, interaction between the enzymes also plays a role in the outcomes of the treatment. It was evident that there was a loss in transglutaminase activity as a result of acid protease digestion. However, there was no loss in the activity of acid protease, indicating that transglutaminase did not have any negative influence on the activity of acid protease.
Keywords
Introduction
The biggest drawback of wool-based products is their tendency to shrink excessively during laundering. This has been attributed to the cuticle cells all pointing in the same direction, giving rise to a ratchet-like action thus interlocking the fibres and not allowing them to return to their original position. Wool has a very complex morphological structure with 10 levels of structure. 1 The cuticle, which makes up 10% of the weight of wool, consists of four layers, namely, the epicuticle, exocuticle A-layer, exocuticle B-layer and the endocuticle. 2 The epicuticle is heavily cross-linked and has an F-layer on the outside. This layer is composed of fatty acids, predominantly 18-methyleicosanoic acid, imparting a hydrophobic character to the wool. 3 The cortex is composed of a compacted mass of spindle-shaped cortical cells embedded in a cell membrane complex. 4
For decades, extensive research has been carried out to develop the methodology for manufacturing machine washable products, and many such processes have been commercialised. Oxidising agents, especially chlorine-based chemicals, were successful in damaging the scales extensively to reduce felting shrinkage. The Chlorine–Hercosett process is one of such most widely used processes. 5 The main disadvantage of these processes was extensive damage to the cortex of the wool, resulting in excessive loss of strength and weight, harshness and yellowing of the wool. Further research led to the introduction of many other chemicals, for example, dichloroisocyanuric acid (DCCA), phenylmethanesulfonic acid (PMS), potassium permanganate (KMnO4) and so on. Many polymers were also used simultaneously with oxidative treatments, for example, Polymer GE (PPT), Basolan SW and MW (BASF). These polymers played a dual role of reducing the felting shrinkage and increasing the softness of the fibre. 6 Another method of reducing felting shrinkage of wool is by causing interfibre cross-linking using polyurethanes, for example, Synthappret LKF and Synthappret BAP, silicones and polyacrylates. However, the use of these chemicals resulted in excessive increase in stiffness and harsh handle of the fibre. 7 Furthermore, these chemical-based processes posed serious environmental concerns in addition to unwanted damage to the wool.
These concerns led to the introduction of enzyme-based ecofriendly alternatives using proteases. Wool was treated with papain in combination with lipase and sodium monoperoxyphthalate (SMPP), thus reducing felting shrinkage from 32.6% to 1.8% with loss of tensile strength and weight to the tune of 12%–13% and 4.86%, respectively. 8 The effect of alkaline protease and acidic protease treatment to wool in combination with alkaline and acidic hydrogen peroxide bleaching was also compared. It was observed that treatment with alkaline peroxide followed by alkaline protease resulted in the lowest felting shrinkage of 3.64% while treatment with acidic peroxide followed by acidic protease treatment showed higher felting shrinkage of 7.77%. However, the treatment of wool with alkaline peroxide followed by alkaline protease showed the highest loss both in strength (19.04%) and in weight (6.45%). The effect of alkaline peroxide bleaching followed by acid protease treatment was explored and it was observed that the felting shrinkage, loss in strength and weight reduced to 5.23%, 9.34% and 2.34%, respectively. The softness rating was, however, the highest at 4.6 compared to 4.4 for acid peroxide treatment followed by acid protease treatment and 3.4 for alkaline peroxide treatment followed by alkaline protease treatment. 9 Bromelain, a class of protease, was used to treat wool at pH ∼6 in the presence of salt, resulting in less than 6% area shrinkage with 9.53% loss in strength and 7.56% loss in weight. The softness rating was observed to be 3.7. 10 A coupled hydrogen peroxide–Aspergillus usamii L86 (containing an acid protease and carboxymethyl cellulase (CMCase)) treatment was studied and compared with their individual treatments. It was observed that the coupled treatment caused higher weight loss of 8.8% as compared to 7.2% for hydrogen peroxide treatment and 4.5% for enzyme treatment only. A similar trend was observed for bending stiffness and bursting strength too. 11 Different studies have explored the effects of different proteases on strength, frictional properties and bending rigidity of wool. 12 Application of other enzymes such as oxidases, peroxidases, lipases and isomerases was explored with limited success in reducing felting shrinkage of wool. On comparing the performance of 16 different types of proteases, Esperase was found to be the most effective in reducing felting shrinkage of wool. Furthermore, pretreatment with alkaline sodium bisulphite showed a synergistic effect on reducing the shrinkage. 13
Treatment with protease resulted in very high strength loss and negative impact on many other properties of wool. This was attributed to the excessive damage to the cortex as a result of enzyme attack. Many strategies were adopted to minimise this damage, one being with immobilised enzymes. Many studies were carried out on the treatment of wool with immobilised enzymes, achieving better results in terms of lesser loss in tensile strength.14–18 In the second strategy, the diffusion of enzyme in wool was inhibited using a high concentration of salt. 19 The third strategy involves the use of transglutaminases to recover the tensile strength lost as a result of protease treatment. Both Savinase 16L and microbial transglutaminases were sequentially applied to wool. Treatment with Savinase 16L resulted in the reduction of felting shrinkage to 7% with 19.4% loss of tensile strength. Subsequent treatment with transglutaminase resulted in further reduction in area shrinkage to 5.4% accompanied by 80% recovery of tensile strength. 20 Wool on treatment with hydrogen peroxide followed by protease showed 6% felting shrinkage, 25% loss in tensile strength and 23% higher alkali solubility. On follow-up treatment with transglutaminases, there was 12–16% improvement in tensile strength and 2–4% reduction in alkali solubility. 21 On application of both the tissue transglutaminase and microbial transglutaminase on Savinase-treated wool, it was observed that both the enzymes were capable of remediating the damage caused by the protease treatment with the tensile strength almost recovered to the pre-protease treatment levels. 22 The use of air plasma in conjunction with Savinase 16L and microbial transglutaminase reduced the area shrinkage from 17.2% to 2.3% with only 2% loss of tensile strength. 23 In another strategy, transglutaminase was used to incorporate different proteins onto to the wool surface. Casein incorporated in KMnO4-treated wool by transglutaminase yielded a wool with 4.57% area shrinkage with an increase in tensile strength from 275 to 315 N. 24 The Agricultural Research Service (ARS) process was modified to allow incorporation of keratin hydrolysates (KH) and their lyophilised powders (KP) on wool using transglutaminases. The loss of bursting strength was only 4.7% compared to 18% for the full ARS process with 5.21% felting shrinkage.25,26 An attempt was made to apply Esperase 6.0T protease and microbial transglutaminase simultaneously on wool. The methodology resulted in only 2% loss of strength and weight as compared to 25% strength loss for only protease treatment. Furthermore, this combined treatment showed 9% reduction in fabric shrinkage as compared to 5% reduction in fabric shrinkage for only protease treatment. The interaction between the enzymes in simultaneous application was also evaluated. Smaller fragments of transglutaminase were found, indicating the digestion of transglutaminase by the protease. However, no evidence of cross-linking of the protease enzyme by transglutaminase was found. 27
In the previous work by the same authors, sequential application of acid protease and transglutaminase was optimised using Box–Behnken design of experiment. First, the application of acid protease was optimised, which was found to be at 1% (owf) enzyme, pH ∼5.7, 50°C and 60 min treatment time. When wool is treated with acid protease using these optimised parameters, the total warp shrinkage is reduced from 7.66% to 2.77% with 9.95% loss of warp tensile strength. Subsequently, transglutaminase treatment was carried out on acid protease treated wool and the parameters were optimised as 2.7% (owf) enzyme, pH ∼5.2, 50°C for 65 min. When acid protease treated wool was treated with transglutaminase, the total warp shrinkage reduced to 2.64%. There was approximately 70% recovery in earlier lost tensile strength with only 2.82% net loss in warp tensile strength. 28 In this study, simultaneous application of acid protease and transglutaminase was conducted on wool. Optimisation was carried out keeping in mind the machine washable care claim as specified in Woolmark specification AW-1 by The Woolmark Company which required the total shrinkage to be less than 3%. This simultaneous application technique, if found successful, will allow one-bath application of both the enzymes to achieve the machine washable care claim and overcome the problem of excessive damage to wool caused by only protease treatment.
Material and Methods
Materials
The scoured wool fabric, possessing 2/64s in both warp and weft, EPI 81, PPI 64, GSM 185 with 21.036 Kgf tensile strength (warp) was used in this study.
Enzyme and Chemicals
Acid protease (activity: 100,000 µ/gm) and microbial transglutaminase (activity: 100–120 µ/gm) enzymes were sourced from Rajvi Enterprise, Ahmedabad, India. Citric acid (AR grade, 99%), trisodium citrate dihydrate (98%), dibasic sodium phosphate (AR grade, 99%), monobasic sodium phosphate (AR grade, 99%), glycine (98.5%), acetic acid (glacial, 99.5%) and hydrochloric acid (AR grade, 37%) sourced from SRL were used in the study.
Instruments and Apparatus
Whirlpool, USA makes a top loading washing machine meeting the specifications as per AATCC TM135: Dimensional Changes of Fabrics after Home Laundering. A Testometric M350-5CT Universal Testing Machine by Rochdale was also used. Both the testing machines were used at OCM Woollen Mills, Amritsar.
Methods
Treatment of Wool Fabric with Enzymes
The treatment of wool with enzymes was carried out in a Glycerine Bath Beaker dyeing machine at a liquor ratio of 1:20 as per the experimental design mentioned in Table 1. To deactivate the enzymes and terminate the treatment, specimens were first washed under running water followed by immersion in boiling water for 15 min. Finally, the specimens were washed with cold running water again and allowed to dry at room temperature. Before conducting any tests, the specimens were subjected to standard atmospheric conditions for 8 h.
Experimental setup for optimisation.
Testing Methods
Shrinkage of Specimens
AATCC TM135: Dimensional Changes of Fabrics after Home Laundering was used to evaluate the total shrinkage. The specimens, with a total wash load of 1.8 ± 0.1 kg, were washed in Whirlpool top loading washing machine. Relaxation shrinkage was measured after the first wash cycle and felting shrinkage was measured after five consecutive wash cycles. The total shrinkage in the weft direction for untreated specimens was below 3%. Consequently, only total warp shrinkage was considered for optimisation of enzymatic treatment.
Tensile Strength
The tensile strength of fabric specimens was evaluated in accordance with ASTM D5035–06 (1R-T Ravelled Strip Test Method). An average of three values were used for further analysis. As only total warp shrinkage was considered for the study, consequently, only warp tensile strength was considered for optimisation.
Results and Discussion
The optimisation of the enzyme treatment was carried out for achieving minimum shrinkage with minimal loss in tensile strength using Box–Behnken design of experiment. The factors considered for optimisation are detailed in Table 1 and include concentration of both the enzymes, pH, temperature and duration of treatment. The results of the experiments are shown in Table 2.
Total warp shrinkage and warp strength loss for enzyme-treated wool.
The response surface plots for all the control factors are shown in Figures 1 and 2. It was observed that the total warp shrinkage decreased as the concentration of acid protease was increased. The rate of fall in shrinkage decreased gradually with an increase in concentration and was almost constant beyond 1.3% protease. The concentration of transglutaminase, however, did not have any impact on shrinkage. This indicated that increased cross-linking did not have any beneficial effect on the shrinkage. With an increase in pH, there was no significant change in shrinkage until a pH around 5 and it increased rapidly beyond pH 5.5. Thus, there was a loss in activity of acid protease at higher pH values. The shrinkage decreased as the temperature increased and the minimum shrinkage was observed at 50–55°C. Beyond 55°C, there was a slight increase in the shrinkage. Furthermore, the shrinkage decreased as the time of treatment increased.
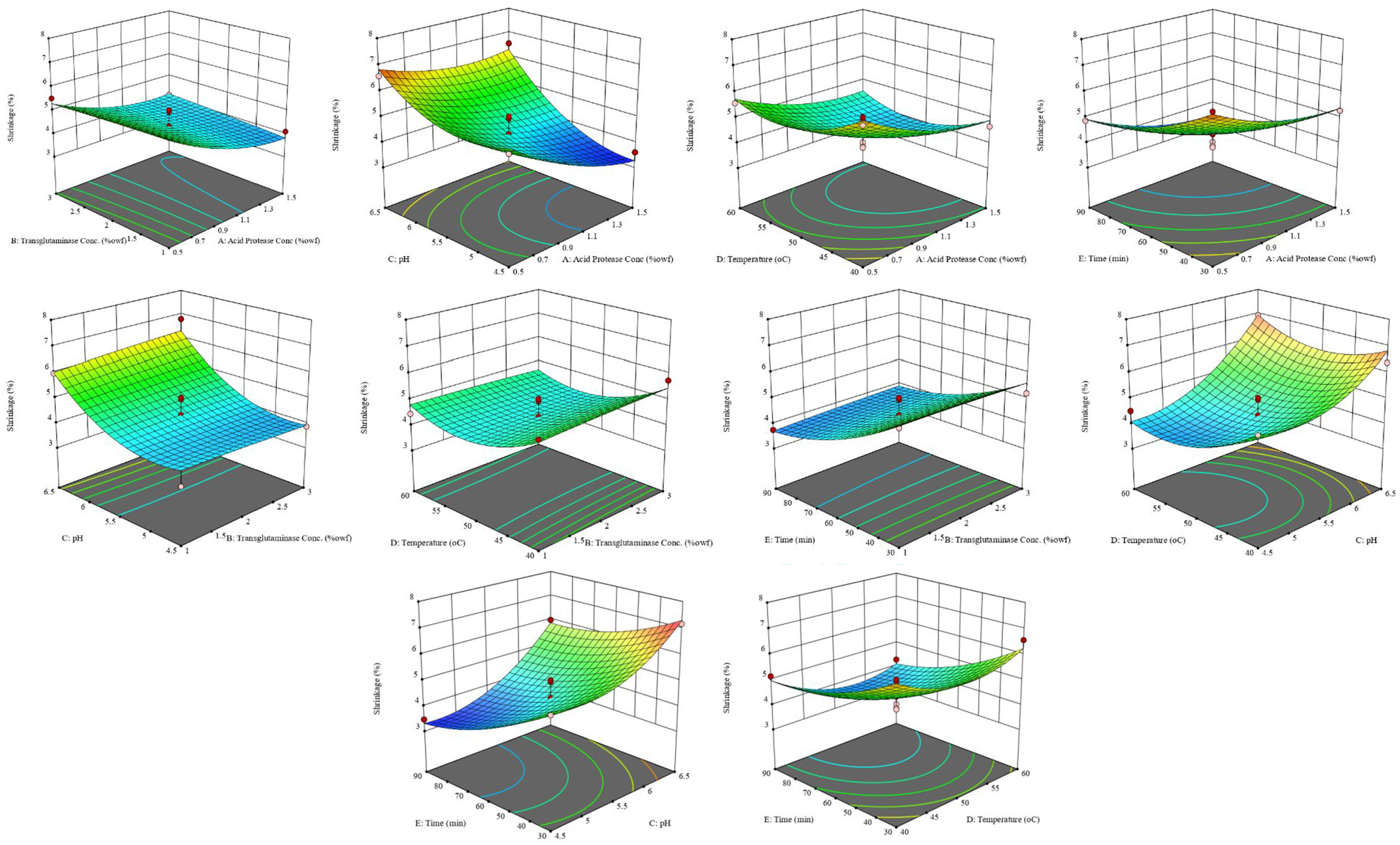
Response surface plots for shrinkage after simultaneous application of acid protease and transglutaminase.
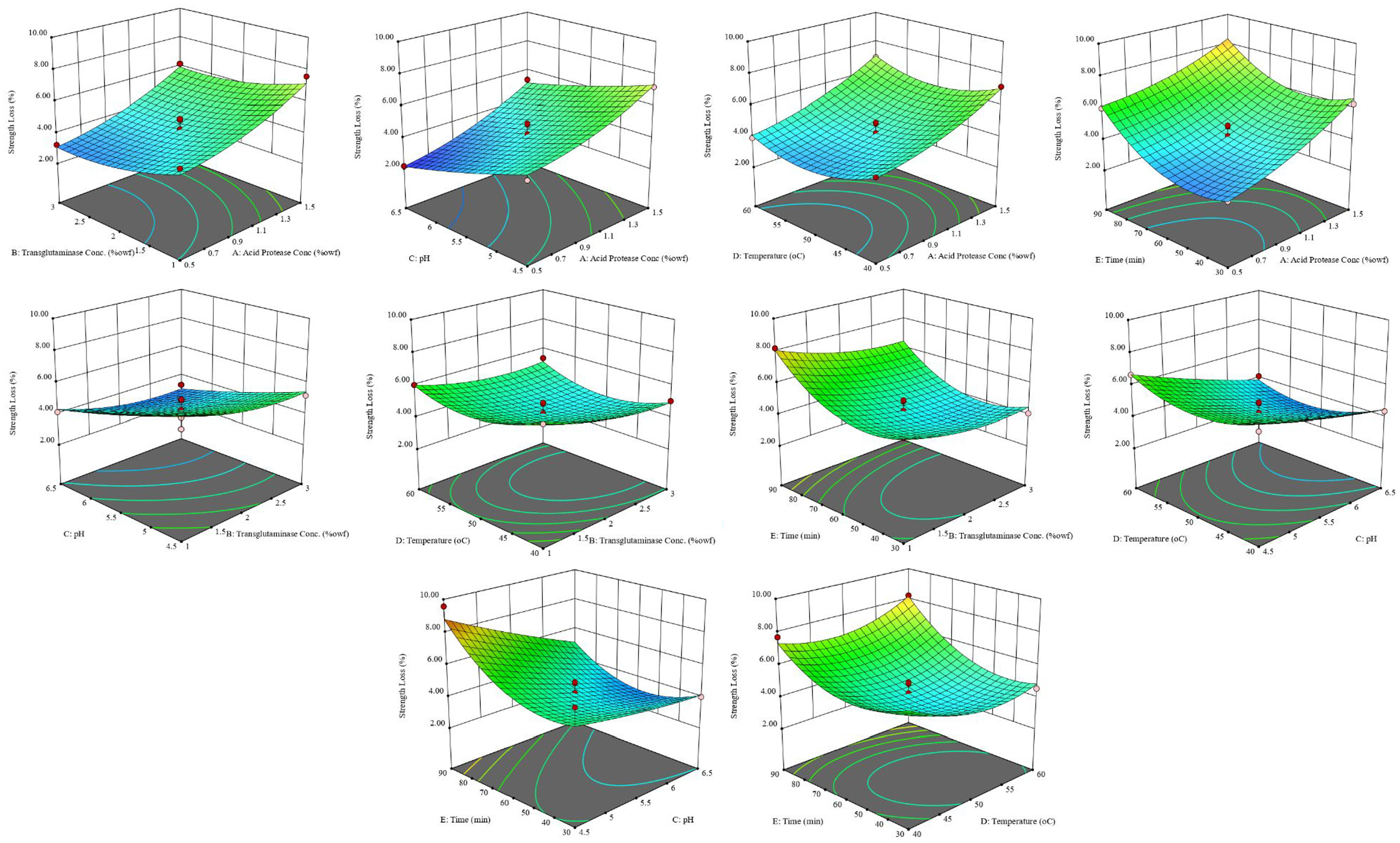
Response surface plots for strength after simultaneous application of acid protease and transglutaminase.
In the case of warp strength loss, it was observed that as the concentration of acid protease increased, there was an increase in strength loss and it was slightly faster as the concentration increased beyond 1%. However, as the concentration of transglutaminase increased, there was a decrease in strength loss as a consequence of internal cross-linking. The extent of change was, however, less for transglutaminase than for acid protease concentration. As the pH increased, the strength loss decreased, indicating that the transglutaminase was getting more active and there was loss of activity of acid protease. The loss of strength was observed to be quite high below or beyond 50°C, at which it was found to be minimal. As the time of the treatment increased, initially there was a decrease in strength loss until around 50 min and beyond that the strength loss increased. Taking into consideration the effect of time on both shrinkage and strength loss, it becomes evident that there is loss in transglutaminase activity but not in acid protease activity as the time of the treatment increases. This can be attributed to the digestion of transglutaminase by acid protease which in turn resulted in loss in of transglutaminase activity and consequently a decrease in the remediation of wool. Furthermore, it is evident that the reverse negative effect of transglutaminase by cross-linking acid protease does not take place.
Statistical analysis of the data was carried out, and insignificant terms were removed from the model. In case of total warp shrinkage, the concentration of the transglutaminase enzyme and its interaction with other control factors that had p-values greater than 0.05 were thus excluded. This again indicated that the concentration of transglutaminase had no significant effect on the total warp shrinkage. The same behaviour was observed in the previous work by the same authors when transglutaminase was applied to acid protease-treated wool. In case of warp strength loss, temperature had a p-value greater than 0.05 but was included in the model to preserve the hierarchy of the model. The quadratic models for both total warp shrinkage and loss in tensile strength so obtained are shown in Table 3.
Quadratic models’ total warp shrinkage and warp strength loss.
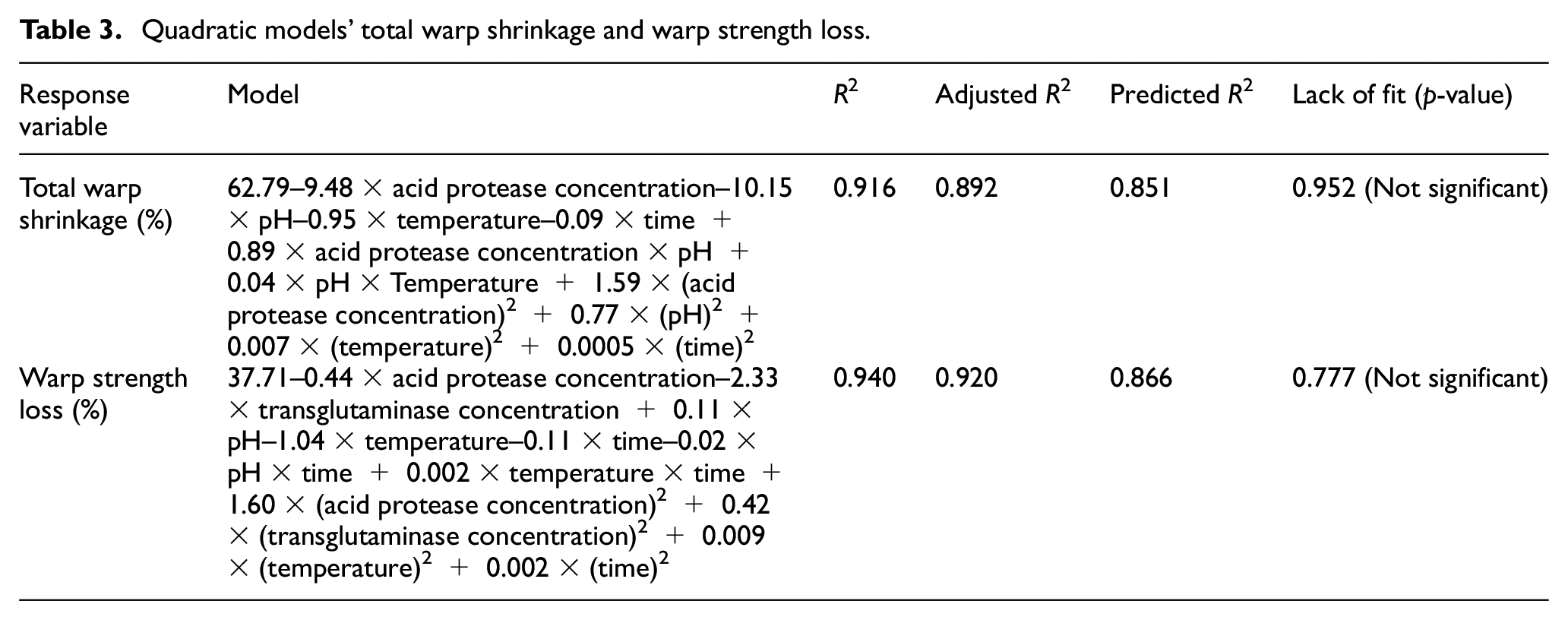
Furthermore, the model was used to optimise the treatment parameters to achieve minimum total warp shrinkage with minimal warp strength loss. Table 4 and Figure 3 show the results of this optimisation process. It was observed that the predicted value of total warp shrinkage is 4.05% with 4.45% loss in tensile strength. Table 4 also shows the observed values of the actual specimen which gave 4.32% total warp shrinkage with 4.65% warp strength loss. This total shrinkage was higher than the 3% total shrinkage required for machine washable care claim as specified by The Woolmark Company. Another set of predictions was taken from the model in which the minimum warp strength loss criteria were removed. Many solutions were predicted as the outcome of this. Out of these iterations, the prediction which resulted in minimum shrinkage was selected and is shown in Table 4. The results of the actual specimen are also shown in the table. It was observed that though the total warp shrinkage was less than 3%, the total warp strength loss was around 10% which is quite high. It can be said that if this high strength loss is acceptable, then the simultaneous treatment can be used. The scanning electron microscope (SEM) images of untreated and treated wool are shown in Figure 4. It can be seen that as compared to untreated wool, the scales in the treated wool seem to be less prominent indicating their damage by acid protease. Furthermore, there is lesser prominence of scales when wool is treated under the optimised conditions of criterion 2 mentioned in Table 4 as compared to when wool is treated with the optimised conditions of criterion 1 mentioned in Table 4. These observations are in agreement with the shrinkage results obtained for the treated specimen.
Predicted and actual results of optimisation for simultaneous treatment.
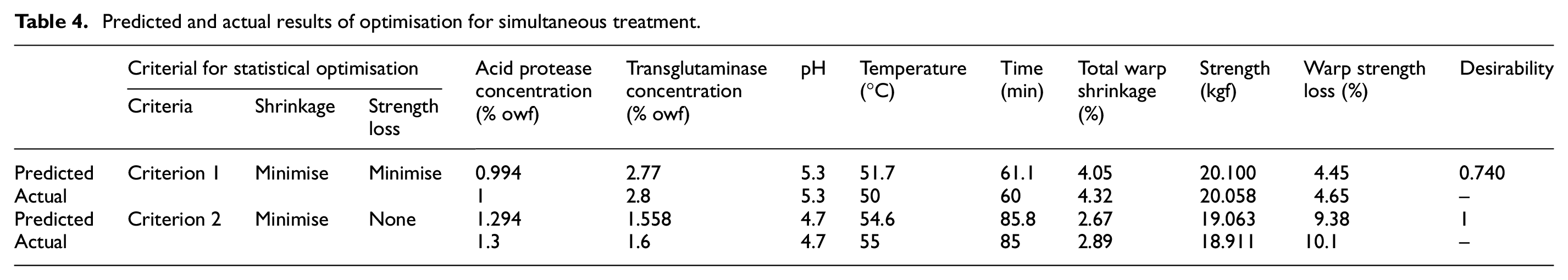
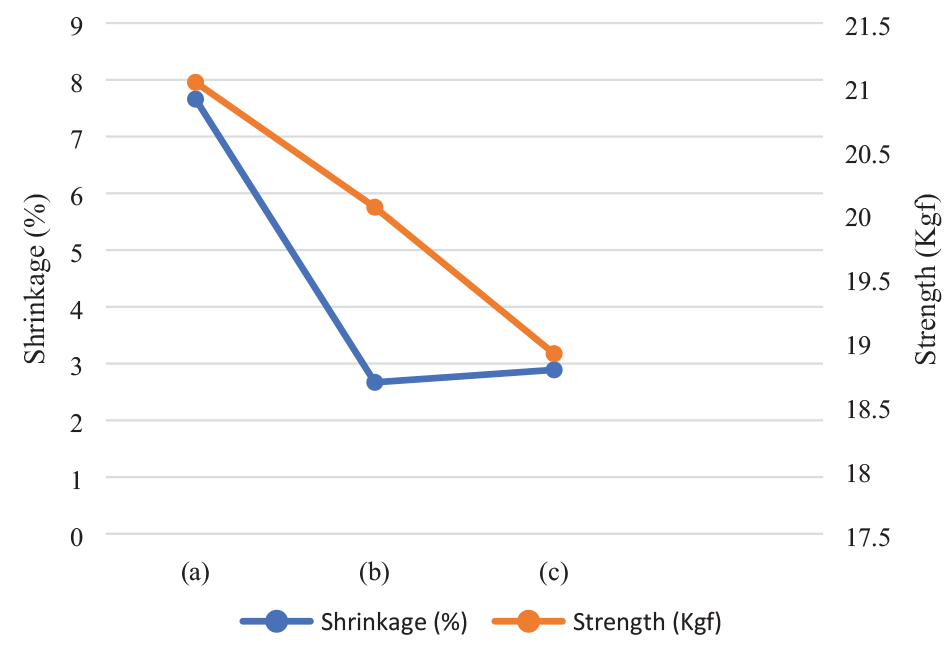
Effect of simultaneous application of acid protease and transglutaminase using optimised parameters: (a) untreated wool, (b) treated wool with optimised parameters using minimum warp shrinkage and minimum warp strength loss criteria, and (c) treated wool with optimised parameters using only minimum warp shrinkage criteria.

Conclusion
In this work, simultaneous application of acid protease and transglutaminase was optimised using two criteria. In the first instance, the model was asked to predict optimised parameters to achieve minimum total warp shrinkage with minimum warp strength loss. As a result of this optimisation, the total warp shrinkage reduced to 4.32% with 4.65% warp strength loss. The optimised treatment conditions were 1% acid protease, 2.8% transglutaminase, pH ∼5.3 at 50°C for 60 min. The obtained shrinkage was higher than the 3% required to achieve the machine washable care claim as per Woolmark specification AW-1. To achieve this specification, a second optimisation method was used. The model was asked to predict optimised parameters to achieve minimum total warp shrinkage without any constraints on warp strength loss. Taking this optimisation method, the total warp shrinkage was reduced to 2.89% with 10.1% warp strength loss. The optimised treatment conditions were 1.3% acid protease, 1.6% transglutaminase, pH ∼4.7 at 55°C for 85 min. If such a high strength loss is acceptable, then a simultaneous application process could be used. Furthermore, the interaction of both the enzymes becomes subtractive in nature to each other. There is loss of the activity of transglutaminase, most probably because of digestion of the enzyme by acid protease. Loss of activity of acid protease was not evident, which indicated that transglutaminase did not have any negative influence on acid protease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
