Abstract
Superhydrophobic textiles have attracted great interest due to their special functions and wide applications. However, it is still a huge challenge to construct a durable superhydrophobic coating for large-scale applications due to the complicated process and high cost. In this work, a facile two-step method was developed to construct superhydrophobic cotton fabric with fluorine-free treatment. The cotton fabrics were treated with modified nano SiO2 to construct rough surfaces. Then, the silicone oil was introduced into the surface of the cotton fabric to form superhydrophobic cotton fabric. The results showed that the modified nano SiO2 and silicone oil were stably fixed on the fiber surface. The static water contact angle test showed that contact angle of the modified cotton fabric was 158°, indicating excellent superhydrophobic properties. Furthermore, the self-cleaning and anti-pollution test results showed that the superhydrophobic cotton fabric possessed good self-cleaning and antifouling performance. This superhydrophobic fabric avoids the use of fluoropolymers and reduces the harm to humans and environment, showing a wide range of applications.
Introduction
Cotton is one of the most abundant natural fibers in the world owing to its excellent properties such as ease of handling, low cost, easy access to raw materials, high mechanical stability, and degradable regeneration. 1 Although more and more synthetic fibers are produced, cotton fabric is still a popular garment raw material. Natural fiber textiles based on cotton play a very important role in the world. 2 However, cotton fabric is composed of hydrophilic cellulose fibers, which easily stick to dirt and absorb various liquids. The hydrophilicity of fibers has largely limited the application and development of fabrics in many fields. In order to overcome these shortcomings of cotton and expand its application field, functional finishing is needed.
A surface with a water contact angle (WCA; at equilibrium) higher than 150° and a contact angle hysteresis lower than 10° are considered to comprise a superhydrophobic surface. 3 Nowadays, superhydrophobic coatings have gained increasing attention due to their diverse applications. Superhydrophobic textiles have lots of advantages, such as non-adhesion, anti-pollution, and self-cleaning, which can be widely used for a wide range of applications, such as oil/water separation, self-cleaning, and multifunctional materials with UV-shielding, flame-retardant, anti-icing, and photocatalytic properties.4–8 They are more and more favored by people and have become a hot spot of functional textile research.
A superhydrophobic surface inspired by the “lotus leaf effect” in nature has developed rapidly, which has become one of the most interesting areas in both academic and industrial research. The superhydrophobic surface of the lotus leaf is determined by two characteristics: surface roughness and low surface energy. 9 Therefore, superhydrophobic fabrics can be produced through creating appropriate rough structures and subsequently hydrophobic treatment with low-surface-energy additives.10,11 The hydrophilic surfaces’ superhydrophobicity can be obtained by chemically modifying the micro-nano rough surface with low-surface-energy compounds. 12 The increase in surface roughness can be achieved by the development of micro/nanostructures such as micro/nanoparticles, micro/nano flower-like structures, nanowires, and nanorods. A hierarchical structure is formed by combining micro/nanoscale roughness. In this structure, a large number of air pockets are formed, which improves the superhydrophobicity of the hierarchical structure. The micro- and nano-scale hierarchical structures can be created from SiO2, with its non-toxic, tasteless, and pollution-free features. 13
However, the surface of SiO2 is rich in hydroxyl groups, has a small particle size and large specific surface area, and has high surface energy, which makes it prone to agglomeration and poor dispersion in organic matrices. It is difficult to uniformly disperse in the matrix when is used as a functional filler with polymer. Therefore, physical or chemical methods are used to modify the nano SiO2 to eliminate or reduce the amount of –OH on the particle surface to achieve the compatibility of the nano SiO2 with the polymer system. Surface modification mainly includes coupling agent method, surface grafting method, and one-step method. The coupling agent method is a widely used method to modify nano SiO2.14,15 Moreover, organically modified silica hydrophobic thin films have shown great potential for the functionalization of textile materials and other supports due to their good mechanical and thermal stability. 16
Some expensive fluoride polymers have been commonly used to change cotton fibers from hydrophilic to hydrophobic. However, they are toxic and bioaccumulative and thus have negative impacts on the environment. Therefore, some environmentally friendly and fluorine-free polymers with low surface energy are urgently needed for scientific researchers to exploit superhydrophobic cotton fabrics. Silicone oils are widely applied in the textile industry due to their unique properties of low surface energy, low toxicity, and intrinsic hydrophobicity. The use of low molecular weight silicone oil can save energy and reduce the use of fluoropolymers and the harm to humans and the environment.17–19
In this study, superhydrophobic cotton fabric was prepared by combining linear silicone oil and nanoparticles. The surface roughness of cotton fabric was formed by modified nano SiO2, and the surface energy of cotton fabric was reduced by silicone oil. The structure and properties of superhydrophobic cotton fabric were investigated. The hierarchical morphology and chemical composition of cotton fabric surface were explored using scanning electron microscopy (SEM), X-ray diffraction (XRD), Fourier transform infrared spectrometry (FTIR), and energy-dispersive spectrometry (EDS). Superhydrophobicity was examined in terms of static WCA, self-cleaning performance, and anti-pollution performance.
Experiments
Materials
Nano SiO2 (99.99%, 20 nm) was purchased from Beijing Nachen Technology Development Co. Ltd. Linear silicone oil 0156 was purchased from Dow Corning Co. Ltd. Ethanol (99.7%, 46.07) was purchased from Sinopharm Chemical Reagent Co. Ltd. (3-Aminopropyl) triethoxysilane (98%, 221.37) was purchased from Aladdin Industrial Corporation. All reagents were used without further purification.
Modification of Nano SiO2
Modification of nano SiO2 was conducted according to previous report. 20 One gram of nano SiO2 was mixed with 20 mL of ethanol and dispersed using ultrasonic processing for 10 min and then magnetically stirred at 400 rpm for 1 h. 7% KH550 silane coupling agent was added into nano SiO2 sol and reacted for 90 min at 55°C in the water bath. After the reaction, the modified nano-SiO2 sample was put into the sample bottle for later use.
Fabrication of Superhydrophobic Fabric
The cotton fabric was immersed in the modified nano SiO2 dispersion, two dipping and two rolling, dried at 100°C for 5 min, baked at 120°C for 3 min, and the modified nano SiO2 sol finishing cotton fabric was obtained. The 10% silicone oil was emulsified on the high shear dispersion emulsifier at 12,000 rpm for 10 min and then the modified nano SiO2 sol treated cotton was immersed in the silicone oil homogeneous solution, two dipping and two rolling, dried at 100°C for 5 min, baked at 120°C for 3 min, and the superhydrophobic cotton fabric was obtained. A schematic of the preparation process, SEM, WCA, and anti-fouling images of the superhydrophobic cotton fabric is illustrated in Figure 1.
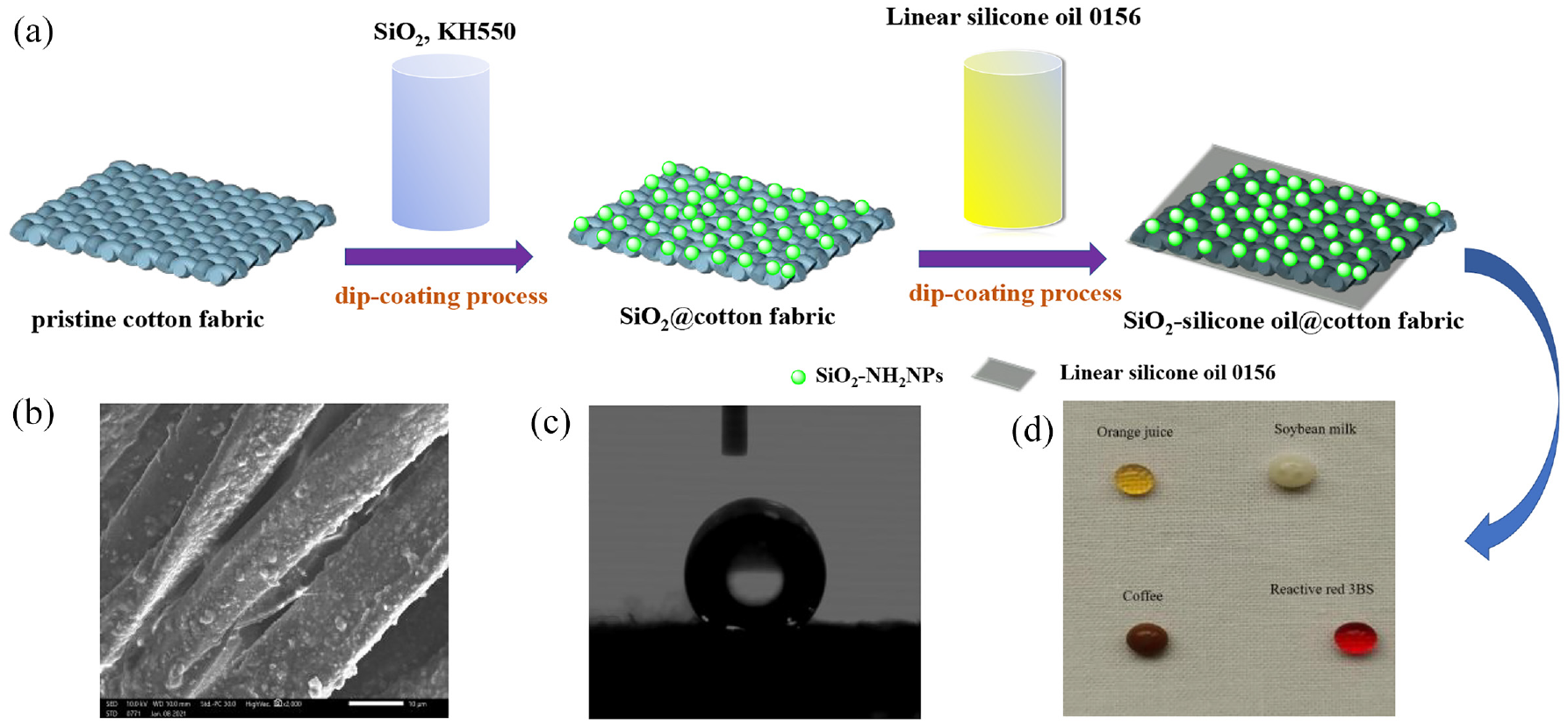
(a) Schematic diagram of the preparation process of the superhydrophobic cotton fabric, (b) SEM of the superhydrophobic cotton fabric, scale bar: 10 μm, (c) WCA of the superhydrophobic cotton fabric, and (d) anti-fouling images of the superhydrophobic cotton fabric.
Characterization
The surface morphologies and EDS were determined by scanning electron microscope (JSM IT500A, Japan) with an acceleration voltage of 10.0 kV. Infrared spectra were performed with FTIR (Tensor 27, Bruker Optik GmbH, Germany) in the range from 400 to 4000 cm−1 with 32 scans at 2.0 cm−1 resolution. The crystal structure of sample was characterized by XRD. Wide-angle X-ray diffractometry (XRD, Rigaku Ultima IV, Japan) was used to investigate the crystallinity of the samples under the conditions of 2θ = 5°–60°, 5°/min, Cu-Ka, 40 kV, and 40 mA. The WCAs on the fabric surface were evaluated with a deionized water droplet of 5 μL at ambient temperature on a professional contact angle measurement instrument (DSA20, KRUSS GmbH Germany). The self-cleaning property was evaluated using reactive red dye liquor as a contaminant. The samples were immersed in reactive red dye liquor for 1 min, taken out, and then dried. The contaminated samples were washed with deionized water and then dried. The laundering durability of the cotton fabric was tested according to AATCC Test Method 61-2006. The abrasion resistance of the cotton fabric was tested according to ISO105-X12:2001 using the Y571N type color fastness tester (Nantong Hongda Experimental Instrument Co., Ltd, China).
Results and Discussion
Surface Morphologies
The surface morphology of the pristine cotton fabric and the modified cotton fabric was investigated using SEM (Figure 2). As shown in Figure 2(a), the surface of the pristine cotton fiber was not completely flat and its surface has a natural texture, but in general, the surface of the pristine cotton fiber was relatively clean and smooth. When the cotton was treated with the silicone oil (designated as silicone oil@cotton), as shown in Figure 2(b), the surface of cotton fiber was covered with a film. When the cotton was treated with nano SiO2 (designated as SiO2@cotton), as shown in Figure 2(c), it was clearly observed that the surface of the cotton fiber was covered with SiO2 nanoparticles, which provided nanoscaled roughness on the cotton. This micro/nano hierarchical structure of the coated fabric was critical for obtaining the superhydrophobicity. When the SiO2@cotton was treated with silicone oil (designated as SiO2-silicone oil@cotton), as shown in Figure 2(d), the surface of cotton fabric did not change significantly compared with the SiO2@cotton. But a film was formed by silicone oil to coat silica nanoparticles on the surface of SiO2-silicone oil@cotton. The SEM results showed that the surface of the superhydrophobic cotton fabric had rough structure and low surface energy substances, which were responsible for the superhydrophobicity.

SEM images of (a) pristine cotton, (b) silicone oil@cotton, (c) SiO2@cotton, and (d) SiO2-silicone oil@cotton.
EDS Analysis
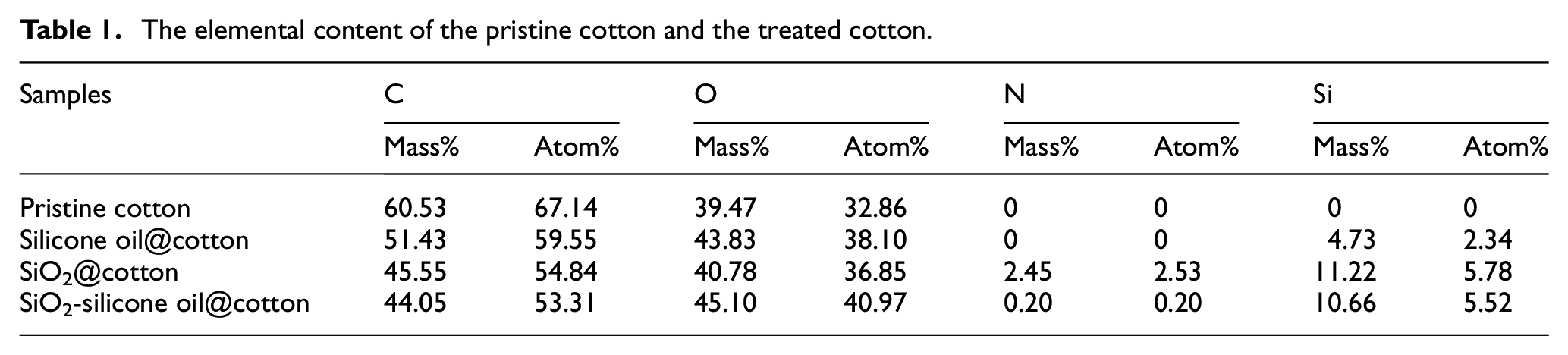
The chemical composition in mass% and atomic% of the pristine cotton, silicone oil@cotton, SiO2@cotton, and SiO2-silicone oil@cotton were investigated by characterizing the element change obtained from the EDS test, as shown in Table 1. Element mapping of the SiO2-silicone oil@cotton is shown in Figure 3. It can be seen from Table 1 and Figure 3 that pristine cotton contained only C, O; silicone oil@cotton contained C, O, Si; and SiO2@cotton and SiO2-silicone oil@cotton contained C, O, N, Si. Si was detected in the silicone oil@cotton, which indicated that the silicone oil was successfully treated onto the cotton. N and Si were detected in SiO2@cotton, because the coupling agent KH550 modified nano SiO2 contained Si and N elements. This indicated that the nano SiO2 was successfully treated onto the cotton. The EDS results showed that the N, Si elements were detected and evenly distributed on the SiO2-silicone oil@cotton. As shown in Figure 2(d), SEM results demonstrated that a thin film was formed after the silicone oil treatment on the fibers. Thus, the SiO2 nanoparticles had been covered by the silicone oil, and the EDS result detected the elemental distribution of the covered film. In other words, the EDS results reflected the elemental distribution of the silicone oil. Therefore, the presence of the N, Si elements indicated that nano SiO2 and silicone oil had been successfully loaded onto the cotton fibers.
The elemental content of the pristine cotton and the treated cotton.

EDS spectrum of the SiO2-silicone oil@cotton: (a) SEM image of the area and (b–f) element mapping of and the corresponding element distribution.
FT-IR Analysis
Apart from the EDS analyses describe above, the surface chemical composition of the pristine cotton, SiO2@cotton, and SiO2-silicone oil@cotton were further analyzed by FT-IR. Figure 4(a) shows the FT-IR spectra of the pristine cotton, SiO2@cotton, and SiO2-silicone oil@cotton. For the pristine cotton, the wide peak at 3420 cm−1 was attributed to the O–H stretching vibration peak, while at 2905 cm−1 has a relatively narrow peak for the C–H stretching vibration peak. There were also some other characteristic peaks: 2854 cm−1 (symmetric –CH2 stretching), 1423 cm−1 (C–H wagging in-plan bending), 1312 cm−1 (C–H wagging), 1160 and 1109 cm−1 (asymmetric C–O–C stretching), 1060 cm−1 (asymmetric in-plane ring stretching), and 1030 cm−1 (C–O stretching). The above characteristic peaks were characteristic bands of cellulose. The results showed that the main structure of the treated cotton fabric did not change. After coating treatment, some new absorption peaks appeared. The peaks at 1060 cm−1 corresponded to the Si–O–Si asymmetric stretching vibration absorption peak. This coincided with the C–O–C asymmetric in-plane ring stretching vibration absorption peak of pristine cotton. The peaks at 802 cm−1 corresponded to the Si–O symmetric stretching vibration absorption peak. The peaks at 460 cm−1 corresponded to the Si–O–Si bending vibration absorption peak. This indicated that SiO2 had been finished on the surface of cotton fabric. Moreover, compared to SiO2@cotton, the absorption intensity of Si–O symmetric stretching vibration absorption peak increased significantly, which was attributed to the stretching vibration of Si–O of silicone oil.21–23 These results confirmed that the silica nanoparticles and silicone oil were successfully attached to the pristine cotton.
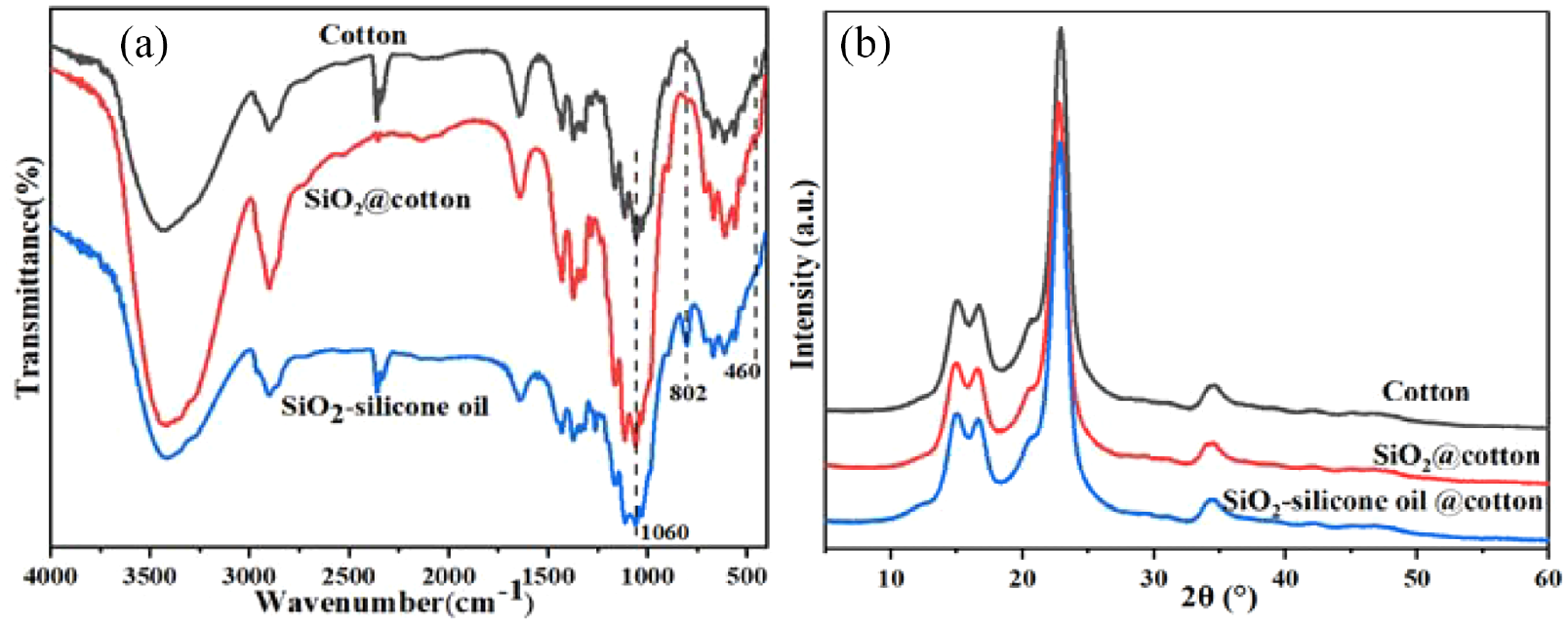
(a) FT-IR spectra of the pristine cotton, SiO2@cotton, and SiO2-silicone oil@cotton and (b) X-ray diffraction spectra of the pristine cotton, SiO2@cotton, and SiO2-silicone oil@cotton.
XRD Analysis
The XRD results showed some important information about the crystallinity of cellulose. The XRD patterns of the pristine cotton, SiO2@cotton, and SiO2-silicone oil@cotton are shown in Figure 4(b). It can be seen that the pristine cotton had 2θ diffraction peaks at 14.98°, 16.62°, 22.88°, and 34.52°, which were typical cellulose I crystalline form. These characteristic peaks also appeared at the XRD patterns of SiO2@cotton and SiO2-silicone oil@cotton, which revealed that the superhydrophobic treatment did not change the main crystalline form of cotton. The modified nano SiO2 had amorphous structure, and only had an amorphous diffraction peak at about 23°, which overlapped with the peak of cellulose. At the same time, silicone oil formed a film on the surface of cotton fabric, without a crystal structure and diffraction peak. Therefore, no peaks were observed for the nano SiO2 and silicone oil. The XRD results showed that the reaction mainly occurred in the amorphous region, and the treatment did not change the crystal structure of the cotton fabric.
Surface Wettability
The wettability is one important property when it comes to superhydrophobicity. This property (dependent on the surface chemical composition and roughness) was evaluated by measuring the static WCAs. Photographic images of water droplets on pristine cotton, silicone oil@cotton, SiO2@cotton, and SiO2-silicone oil@cotton sample surface are shown in Figure 5. The WCA of pristine cotton fabric was 0° because of its hydrophilic property. The cellulose hydroxyl groups of cotton fabric made the water droplets spread instantly when placed on the surface of the substrate. The WCA of silicone oil@cotton was 0°. Although the long carbon chain of alkane siloxane was helpful for shielding hydrophilic groups, which resulted in the formation of siloxane networks thus lowering the surface energy of the coating, the linear bulk silicone oil used in this experiment was the raw material for the synthesis of organic silicon, and its molecular weight was small, which reduced the surface energy of the cotton fabric to a smaller degree, so it could not give the cotton fabric hydrophobicity. The WCA of SiO2@cotton was 0°. It can be seen from the SEM image that nano SiO2 built a micro-nano rough structure on the surface of the cotton fabric, but the surface of the silica contained hydroxyl groups, so SiO2@cotton was hydrophilic. Compared to SiO2@cotton, the WCA of SiO2-silicone oil@cotton was 158°, which showed that it had good superhydrophobic properties. The wetting behavior falls into the Cassie-Baxter model, in which micro/nano hierarchical structures could promote the formation of air pockets at the solid–liquid interface and reduced the contact area between the solid and liquid phases. Meanwhile, a layer of silicone oil film was coated on the surface of nano SiO2, resulting in lower surface energy. Therefore, it could be seen from the result of static WCAs that the fabric had superhydrophobic properties only by constructing rough structure and treating with low-surface-energy material. 24

Photographic images of water droplet on to (a) pristine cotton, (b) silicone oil@cotton, (c) SiO2@cotton, and (d) SiO2-silicone oil@cotton sample surface.
Self-Cleaning Performance
There are two principal ways of self-cleaning materials, namely hydrophobicity and hydrophilicity. Both types of coating clean themselves with the action of water by rolling droplets for hydrophobic and sheeting water for hydrophilic that carries dirt away. The superhydrophobic coatings (Lotus Effect) way was used in this study. The self-cleaning performance of pristine cotton, silicone oil@cotton, SiO2@cotton, and SiO2-silicone oil@cotton was investigated using reactive red dye as a contaminant, and the results are shown in Figure 6. When the samples were immersed in reactive red dye liquor, the surface of pristine cotton, silicone oil@cotton, and SiO2@cotton had accumulated a large number of red stains, but SiO2-silicone oil@cotton had distributed sparse red stains on the surface. After washing with deionized water, it was obvious that the water droplets swiftly rolled down the surface of the SiO2-silicone oil@cotton and took dye away at the same time and then a clean surface was left. This was due to the weak adhesion of the dirt on the superhydrophobic surface, and the dirt on the fabric was easy to be taken away by water drops, leaving a clean surface. This result showed the excellent self-cleaning performance of the superhydrophobic SiO2-silicone oil@cotton.

Photographs of self-cleaning property test for (a1–a3) pristine cotton fabric, (b1–b3) silicone oil@cotton, (c1–c3) SiO2@cotton, and (d1–d3) SiO2-silicone oil@cotton samples. Number 1 represents the fabric before contamination, number 2 represents the fabric after contamination, and number 3 represents the fabric after cleaning.
Wetting and Anti-Pollution Performance
In real life, the external environment is often complex and diverse, often containing pollutants such as orange juice, coffee, dyes, and soybean milk. Therefore, wetting and anti-pollution capabilities are important in practical applications.25–27 We tested the performance of several types of liquids on pristine cotton, silicone oil@cotton, SiO2@cotton, and SiO2-silicone oil@cotton, and the results are shown in Figures 7 and 8. In order to directly show the hydrophobicity of the fabric before and after treatment, reactive red dye liquor was dropped on the fabric and the wetting state of the fabric was recorded within 60 s. The photographs are shown in Figure 7. When the dye liquor drops, the pristine cotton, silicone oil@cotton, and SiO2@cotton were completely permeated at the moment, as shown in Figure 7(a)–(c). The droplet on the SiO2-silicone oil@cotton could maintain a relatively complete state of droplet within 60 s, and the surface of the fabric was not wetted, as shown in Figure 7(d). Therefore, SiO2-silicone oil@cotton could maintain superhydrophobic state for a long time. Figure 8 shows that all the pollutant liquids were round and spherical on the SiO2-silicone oil@cotton surface without wetting. The result showed that the superhydrophobic cotton fabric could not be polluted. Hence, the superhydrophobic treated cotton had good anti-pollution performance.28–30
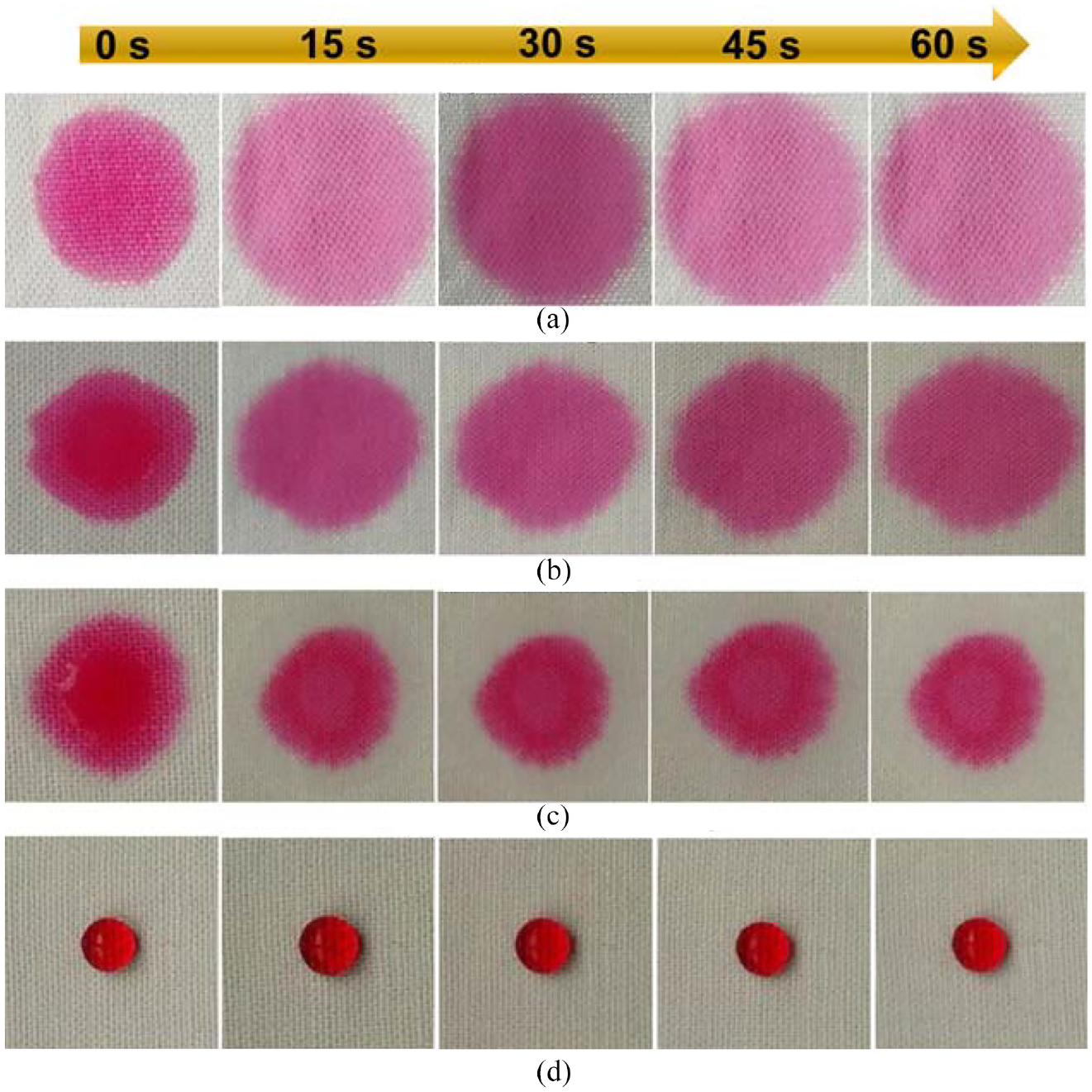
Photographs of the water drop on the surface of different cotton fabrics at different time intervals ranging from 0 to 60 s: (a) pristine cotton, (b) silicon oil@cotton, (c) SiO2@cotton, and (d) SiO2-silicon oil@cotton.

Renderings of different pollutants on SiO2-silicone oil@cotton.
It is well known that the stability of functional fabric properties is critical. Therefore, the laundering and abrasion resistance of the hydrophobic property of the fabric were tested, and the results are shown in Figure 9. After 50 laundering cycles, Figure 9(a) showed that the droplet was spherical and did not wet the surface of superhydrophobic cotton fabric. This indicated that the cotton fabric still maintained good superhydrophobicity. After different intense friction cycles, the photographs of superhydrophobic cotton fabric are shown in Figure 9(b). The photographs of superhydrophobic cotton fabric vividly showed that the droplet did not wet the surface of superhydrophobic cotton fabric after 300 friction cycles. This showed that the cotton fabric still had good hydrophobicity. Therefore, the laundering and abrasion resistance results indicated that modified nano SiO2 and silicone oil were stably loaded on the cotton fabric surface.

Photographs of SiO2-silicone oil@cotton fabric after (a) different laundering cycles and (b) different friction cycles times.
Conclusion
In this study, a simple two-step method was used to prepare superhydrophobic cotton fabric with fluorine-free treatment. The silicone oil modified nano-SiO2 was deposited on the surface of cotton fabric to construct the micro-nano-scale rough structure with low surface energy. The results showed that silicone oil modified nano-SiO2 could be uniformly and stably loaded on the fiber surface. Compared with the pristine cotton with 0°, the static WCA of the modified cotton fabric was increased to 158°, showing an excellent superhydrophobic feature. The further test results showed that the superhydrophobic cotton fabric possessed good self-cleaning and antifouling performance. This facile and environmentally friendly method may be useful for the development of various superhydrophobic fabric products in large-scale production in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Opening Project of Jiangsu Engineering Research Center of Textile Dyeing and Printing for Energy Conservation, Discharge Reduction and Cleaner Production (ERC), Grant number: SDGC2104; Scientific Research Program Guiding Project of Hubei Provincial Department of Education, Grant number: B2021088; Hubei Key Laboratory of Biomass Fibers and Eco-Dyeing & Finishing, Grant number: STRZ202123; and Wuhan research center of eco-dyeing & finishing and functional textile, Wuhan Textile University, Grant number: EDFT2021003.
